Transformation Products of Organic Contaminants and Residues—Overview of Current Simulation Methods
Abstract
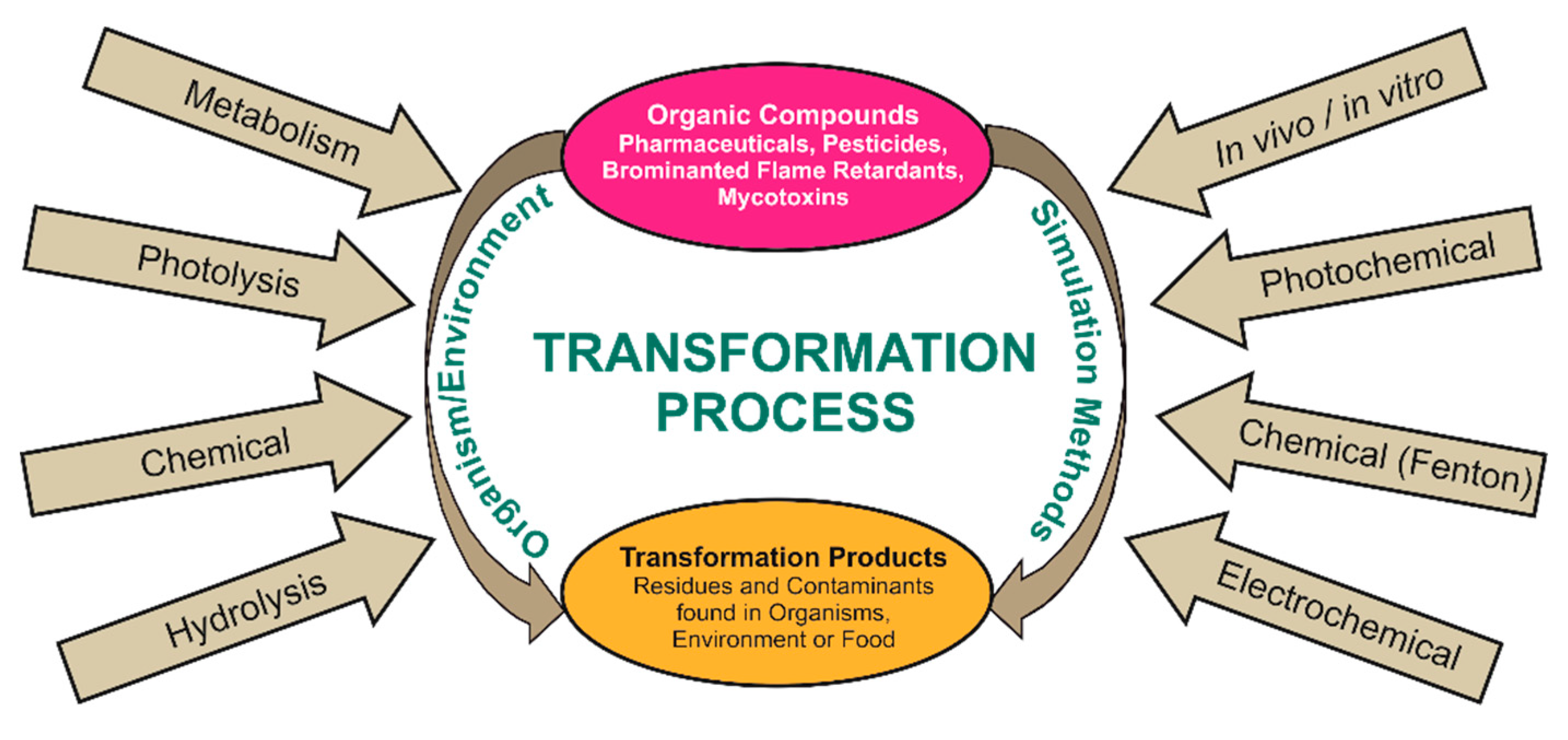
:1. Introduction
2. Methods for Transformation and Analysis
2.1. Metabolism Methods
2.2. Electrochemical-Based Methods
2.3. Photochemical-Based Methods
2.4. Fenton-Based Methods
2.5. Analytical Techniques for TP Identification
3. Transformation Processes of Selected Compound Classes
3.1. Pesticides
3.2. Pharmaceuticals/Drugs
3.3. Brominated Flame Retardants
3.4. Mycotoxins
4. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, Z.; Tang, W. Drug metabolism in drug discovery and development. Acta Pharm. Sin. B 2018, 8, 721–732. [Google Scholar] [CrossRef] [PubMed]
- Bletsou, A.A.; Jeon, J.; Hollender, J.; Archontaki, E.; Thomaidis, N.S. Targeted and non-targeted liquid chromatography-mass spectrometric workflows for identification of transformation products of emerging pollutants in the aquatic environment. TrAC. Trends Anal. Chem. 2015, 66, 32–44. [Google Scholar] [CrossRef]
- Pico, Y.; Barcelo, D. Transformation products of emerging contaminants in the environment and high-resolution mass spectrometry: A new horizon. Anal. Bioanal. Chem. 2015, 407, 6257–6273. [Google Scholar] [CrossRef] [PubMed]
- Fatta-Kassinos, D.; Vasquez, M.I.; Kummerer, K. Transformation products of pharmaceuticals in surface waters and wastewater formed during photolysis and advanced oxidation processes—Degradation, elucidation of byproducts and assessment of their biological potency. Chemosphere 2011, 85, 693–709. [Google Scholar] [CrossRef] [PubMed]
- Petrovic, M.; Barceló, D. LC-MS for identifying photodegradation products of pharmaceuticals in the environment. TrAC. Trends Anal. Chem. 2007, 26, 486–493. [Google Scholar] [CrossRef]
- Speight, J.G. Chemical Transformations in the Environment. In Environmental Organic Chemistry for Engineers; Butterworth-Heinemann: Oxford, UK, 2017; pp. 305–353. [Google Scholar] [CrossRef]
- Lohmann, W.; Karst, U. Electrochemistry meets enzymes: Instrumental on-line simulation of oxidative and conjugative metabolism reactions of toremifene. Anal. Bioanal. Chem. 2009, 394, 1341–1348. [Google Scholar] [CrossRef] [PubMed]
- Hubner, U.; von Gunten, U.; Jekel, M. Evaluation of the persistence of transformation products from ozonation of trace organic compounds—A critical review. Water Res. 2015, 68, 150–170. [Google Scholar] [CrossRef] [PubMed]
- Kosjek, T.; Heath, E. Occurrence, fate and determination of cytostatic pharmaceuticals in the environment. TrAC. Trends Anal. Chem 2011, 30, 1065–1087. [Google Scholar] [CrossRef]
- Karlovsky, P.; Suman, M.; Berthiller, F.; De Meester, J.; Eisenbrand, G.; Perrin, I.; Oswald, I.P.; Speijers, G.; Chiodini, A.; Recker, T.; et al. Impact of food processing and detoxification treatments on mycotoxin contamination. Mycotoxin Res. 2016, 32, 179–205. [Google Scholar] [CrossRef] [PubMed]
- Vanhoutte, I.; Audenaert, K.; De Gelder, L. Biodegradation of Mycotoxins: Tales from Known and Unexplored Worlds. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Sono, M.; Roach, M.P.; Coulter, E.D.; Dawson, J.H. Heme-containing oxygenases. Chem. Rev. 1996, 96, 2841–2887. [Google Scholar] [CrossRef] [PubMed]
- Lynch, T.J.P.; Anita, L. The effect of cytochrome P450 metabolism on drug response, interactions, and adverse effects. Am. Fam. Physician 2007, 76, 391–397. [Google Scholar]
- Prost, F.; Thormann, W. Capillary electrophoresis to assess drug metabolism induced in vitro using single CYP450 enzymes (Supersomes): Application to the chiral metabolism of mephenytoin and methadone. Electrophoresis 2003, 24, 2577–2587. [Google Scholar] [CrossRef] [PubMed]
- Brandon, E.F.A.; Raap, C.D.; Meijerman, I.; Beijnen, J.H.; Schellens, J.H.M. An update on in vitro test methods in human hepatic drug biotransformation research: Pros and cons. Toxicol. Appl. Pharmacol. 2003, 189, 233–246. [Google Scholar] [CrossRef]
- Portychova, L.; Schug, K.A. Instrumentation and applications of electrochemistry coupled to mass spectrometry for studying xenobiotic metabolism: A review. Anal. Chim. Acta. 2017, 993, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Luo, G.; Ding, X.; Lu, C. Preclinical experimental models of drug metabolism and disposition in drug discovery and development. Acta Pharm. Sin. B 2012, 2, 549–561. [Google Scholar] [CrossRef]
- Lecours, M.-A.; Eysseric, E.; Yargeau, V.; Lessard, J.; Brisard, G.; Segura, P. Electrochemistry-High Resolution Mass Spectrometry to Study Oxidation Products of Trimethoprim. Environments 2018, 5, 18. [Google Scholar] [CrossRef]
- Guengerich, F.P. Common and Uncommon Cytochrome P450 Reactions Related to Metabolism and Chemical Toxicity. Chem. Res. Toxicol. 2001, 14, 611–650. [Google Scholar] [CrossRef] [PubMed]
- Jahn, S.; Karst, U. Electrochemistry coupled to (liquid chromatography/) mass spectrometry--current state and future perspectives. J. Chromatogr. A 2012, 1259, 16–49. [Google Scholar] [CrossRef] [PubMed]
- Mandic, Z. Electrochemical methods in drug discovery and development. Bulg. Chem. Commun. 2017, 49, 65–73. [Google Scholar]
- Martin, E.T.; McGuire, C.M.; Mubarak, M.S.; Peters, D.G. Electroreductive Remediation of Halogenated Environmental Pollutants. Chem. Rev. 2016, 116, 15198–15234. [Google Scholar] [CrossRef] [PubMed]
- Bruins, A.P. An overview of electrochemistry combined with mass spectrometry. TrAC. Trends Anal. Chem. 2015, 70, 14–19. [Google Scholar] [CrossRef]
- Santos, M.S.F.; Alves, A.; Madeira, L.M. Chemical and photochemical degradation of polybrominated diphenyl ethers in liquid systems—A review. Water Res. 2016, 88, 39–59. [Google Scholar] [CrossRef] [PubMed]
- Fujishima, A.; Zhang, X.; Tryk, D.A. TiO2 photocatalysis and related surface phenomena. Surf. Sci. Rep. 2008, 63, 515–582. [Google Scholar] [CrossRef]
- Ruokolainen, M.; Gul, T.; Permentier, H.; Sikanen, T.; Kostiainen, R.; Kotiaho, T. Comparison of TiO2 photocatalysis, electrochemically assisted Fenton reaction and direct electrochemistry for simulation of phase I metabolism reactions of drugs. Eur. J. Pharm. Sci. 2016, 83, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Menz, J.; Toolaram, A.P.; Rastogi, T.; Leder, C.; Olsson, O.; Kummerer, K.; Schneider, M. Transformation products in the water cycle and the unsolved problem of their proactive assessment: A combined in vitro/in silico approach. Environ. Int. 2017, 98, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Gligorovski, S.; Strekowski, R.; Barbati, S.; Vione, D. Environmental Implications of Hydroxyl Radicals ((*)OH). Chem. Rev. 2015, 115, 13051–13092. [Google Scholar] [CrossRef] [PubMed]
- Soo-Myung, K.; Alfons, V. Degradation of Organic Pollutants by the Photo-Fenton-Process. Chem. Eng. Technol. 1998, 21, 187–191. [Google Scholar]
- Brillas, E.; Sirés, I.; Oturan, M.A. Electro-Fenton Process and Related Electrochemical Technologies Based on Fenton’s Reaction Chemistry. Chem. Rev. 2009, 109, 6570–6631. [Google Scholar] [CrossRef] [PubMed]
- Ventura, A.; Jacquet, G.; Bermond, A.; Camel, V. Electrochemical generation of the Fenton’s reagent: Application to atrazine degradation. Water Res. 2002, 36, 3517–3522. [Google Scholar] [CrossRef]
- Portoles, T.; Mol, J.G.; Sancho, J.V.; Hernandez, F. Use of electron ionization and atmospheric pressure chemical ionization in gas chromatography coupled to time-of-flight mass spectrometry for screening and identification of organic pollutants in waters. J. Chromatogr. A 2014, 1339, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, A. GC-MS Technique and its Analytical Applications in Science and Technology. J. Anal. Bioanal. Tech. 2014, 5. [Google Scholar] [CrossRef]
- Hao, C.; Zhao, X.; Yang, P. GC-MS and HPLC-MS analysis of bioactive pharmaceuticals and personal-care products in environmental matrices. TrAC. Trends Anal. Chem. 2007, 26, 569–580. [Google Scholar] [CrossRef]
- Churchwell, M.I.; Twaddle, N.C.; Meeker, L.R.; Doerge, D.R. Improving LC-MS sensitivity through increases in chromatographic performance: Comparisons of UPLC-ES/MS/MS to HPLC-ES/MS/MS. J. Chromatog. B 2005, 825, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Gumustas, M.; Kurbanoglu, S.; Uslu, B.; Ozkan, S.A. UPLC versus HPLC on Drug Analysis: Advantageous, Applications and Their Validation Parameters. Chromatographia 2013, 76, 1365–1427. [Google Scholar] [CrossRef]
- Gilbert-López, B.; Valdés, A.; Acunha, T.; García-Cañas, V.; Simó, C.; Cifuentes, A. Foodomics: LC and LC-MS-based omics strategies in food science and nutrition. J. Liq. Chromatogr. 2017, 267–299. [Google Scholar] [CrossRef]
- Awad, H.; Khamis, M.M.; El-Aneed, A. Mass Spectrometry, Review of the Basics: Ionization. Appl. Spectrosc. Rev. 2014, 50, 158–175. [Google Scholar] [CrossRef]
- Marshall, A.G.; Hendrickson, C.L. High-Resolution Mass Spectrometers. Annu. Rev. Anal. Chem. 2008, 1, 579–599. [Google Scholar] [CrossRef] [PubMed]
- Nikolaev, E.N.; Kostyukevich, Y.I.; Vladimirov, G.N. Fourier transform ion cyclotron resonance (FT ICR) mass spectrometry: Theory and simulations. Mass Spectrom. Rev. 2016, 35, 219–258. [Google Scholar] [CrossRef] [PubMed]
- Shaw, J.B.; Lin, T.-Y.; Leach, F.E.; Tolmachev, A.V.; Tolić, N.; Robinson, E.W.; Koppenaal, D.W.; Paša-Tolić, L. 21 Tesla Fourier Transform Ion Cyclotron Resonance Mass Spectrometer Greatly Expands Mass Spectrometry Toolbox. J. Am. Soc. Mass. Spectrom. 2016, 27, 1929–1936. [Google Scholar] [CrossRef] [PubMed]
- Schymanski, E.L.; Singer, H.P.; Slobodnik, J.; Ipolyi, I.M.; Oswald, P.; Krauss, M.; Schulze, T.; Haglund, P.; Letzel, T.; Grosse, S.; et al. Non-target screening with high-resolution mass spectrometry: Critical review using a collaborative trial on water analysis. Anal. Bioanal. Chem. 2015, 407, 6237–6255. [Google Scholar] [CrossRef] [PubMed]
- Zedda, M.; Zwiener, C. Is nontarget screening of emerging contaminants by LC-HRMS successful? A plea for compound libraries and computer tools. Anal. Bioanal. Chem. 2012, 403, 2493–2502. [Google Scholar] [CrossRef] [PubMed]
- Du, B.; Lofton, J.M.; Peter, K.T.; Gipe, A.D.; James, C.A.; McIntyre, J.K.; Scholz, N.L.; Baker, J.E.; Kolodziej, E.P. Development of suspect and non-target screening methods for detection of organic contaminants in highway runoff and fish tissue with high-resolution time-of-flight mass spectrometry. Environ. Sci. Process Impacts 2017, 19, 1185–1196. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, D.; Wang, Y.; Zhang, P.; Zhou, Z.; Zhu, W. A combined non-targeted and targeted metabolomics approach to study the stereoselective metabolism of benalaxyl enantiomers in mouse hepatic microsomes. Environ. Pollut. 2016, 212, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Maggio, R.M.; Calvo, N.L.; Vignaduzzo, S.E.; Kaufman, T.S. Pharmaceutical impurities and degradation products: Uses and applications of NMR techniques. J. Pharm. Biomed. Anal. 2014, 101, 102–122. [Google Scholar] [CrossRef] [PubMed]
- Bauer, A.; Luetjohann, J.; Hanschen, F.S.; Schreiner, M.; Kuballa, J.; Jantzen, E.; Rohn, S. Identification and characterization of pesticide metabolites in Brassica species by liquid chromatography travelling wave ion mobility quadrupole time-of-flight mass spectrometry (UPLC-TWIMS-QTOF-MS). Food Chem. 2018, 244, 292–303. [Google Scholar] [CrossRef] [PubMed]
- Mekonnen, T.F.; Byrne, L.; Panne, U.; Koch, M. Investigation of Chlorpyrifos and Its Transformation Products in Fruits and Spices by Combining Electrochemistry and Liquid Chromatography Coupled to Tandem Mass Spectrometry. Food Anal. Methods 2018, 11, 2657–2665. [Google Scholar] [CrossRef]
- Rousis, N.I.; Bade, R.; Bijlsma, L.; Zuccato, E.; Sancho, J.V.; Hernandez, F.; Castiglioni, S. Monitoring a large number of pesticides and transformation products in water samples from Spain and Italy. Environ. Res. 2017, 156, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Fine, J.D.; Mullin, C.A. Metabolism of N-Methyl-2-Pyrrolidone in Honey Bee Adults and Larvae: Exploring Age Related Differences in Toxic Effects. Environ. Sci. Technol. 2017, 51, 11412–11422. [Google Scholar] [CrossRef] [PubMed]
- Negreira, N.; Regueiro, J.; Valdersnes, S.; Berntssen, M.H.G.; Ornsrud, R. Comprehensive characterization of ethoxyquin transformation products in fish feed by traveling-wave ion mobility spectrometry coupled to quadrupole time-of-flight mass spectrometry. Anal. Chim. Acta. 2017, 965, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Gao, Z.; Wang, Y.; Yuan, Y.; Dong, J.; Yue, T. Transformation products elucidation of forchlorfenuron in postharvest kiwifruit by time-of-flight mass spectrometry. PLoS ONE 2017, 12, e0184021. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, P.P. Leaf cuticle-assisted phototransformation of isoproturon. Acta Physiol. Plant 2017, 39. [Google Scholar] [CrossRef]
- Jurva, U.; Weidolf, L. Electrochemical generation of drug metabolites with applications in drug discovery and development. Trac-Trend Anal. Chem. 2015, 70, 92–99. [Google Scholar] [CrossRef]
- Kimura, H.; Sakai, Y.; Fujii, T. Organ/body-on-a-chip based on microfluidic technology for drug discovery. Drug Metab. Pharmacokinet 2018, 33, 43–48. [Google Scholar] [CrossRef] [PubMed]
- von Gunten, U. Oxidation Processes in Water Treatment: Are We on Track? Environ. Sci. Technol. 2018, 52, 5062–5075. [Google Scholar] [CrossRef] [PubMed]
- Berberidou, C.; Kitsiou, V.; Kazala, E.; Lambropoulou, D.A.; Kouras, A.; Kosma, C.I.; Albanis, T.A.; Poulios, I. Study of the decomposition and detoxification of the herbicide bentazon by heterogeneous photocatalysis: Kinetics, intermediates and transformation pathways. Appl. Catal. B 2017, 200, 150–163. [Google Scholar] [CrossRef]
- Nicol, E.; Chayata, H.; Genty, C.; Bouchonnet, S.; Bourcier, S. Photodegradation of cyprodinil under UV-visible irradiation—Chemical and toxicological approaches. Rapid Commun. Mass Spectrom. 2016, 30, 2201–2211. [Google Scholar] [CrossRef] [PubMed]
- Liao, X.; Zhang, C.; Liu, Y.; Luo, Y.; Wu, S.; Yuan, S.; Zhu, Z. Abiotic degradation of methyl parathion by manganese dioxide: Kinetics and transformation pathway. Chemosphere 2016, 150, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Dong, B.; Hu, J. Photodegradation of the novel fungicide fluopyram in aqueous solution: Kinetics, transformation products, and toxicity evolvement. Environ. Sci. Pollut. Res. Int. 2016, 23, 19096–19106. [Google Scholar] [CrossRef] [PubMed]
- Lohmann, W.; Dotzer, R.; Gutter, G.; Van Leeuwen, S.M.; Karst, U. On-line electrochemistry/liquid chromatography/mass spectrometry for the simulation of pesticide metabolism. J. Am. Soc. Mass. Spectrom. 2009, 20, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Mekonnen, T.F.; Panne, U.; Koch, M. Prediction of biotransformation products of the fungicide fluopyram by electrochemistry coupled online to liquid chromatography-mass spectrometry and comparison with in vitro microsomal assays. Anal. Bioanal.Chem. 2018, 410, 2607–2617. [Google Scholar] [CrossRef] [PubMed]
- Mekonnen, T.F.; Panne, U.; Koch, M. Electrochemistry coupled online to liquid chromatography-mass spectrometry for fast simulation of biotransformation reactions of the insecticide chlorpyrifos. Anal. Bioanal.Chem. 2017, 409, 3359–3368. [Google Scholar] [CrossRef] [PubMed]
- Mohle, S.; Zirbes, M.; Rodrigo, E.; Gieshoff, T.; Wiebe, A.; Waldvogel, S.R. Modern Electrochemical Aspects for the Synthesis of Value-Added Organic Products. Angew. Chem. Int. Ed. Engl. 2018, 57, 6018–6041. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.L.; Zhao, Z.D.; Zhang, X.L.; Tang, L.L.; Zhang, Y.; Zhang, C.H. Simultaneous analysis of herbicide metribuzin and its transformation products in tomato using QuEChERS-based gas chromatography coupled to a triple quadrupole mass analyzer. Microchem. J. 2017, 133, 468–473. [Google Scholar] [CrossRef]
- Lopez-Ruiz, R.; Ruiz-Muelle, A.B.; Romero-Gonzalez, R.; Fernandez, I.; Vidal, J.L.M.; Frenich, A.G. The metabolic pathway of flonicamid in oranges using an orthogonal approach based on high-resolution mass spectrometry and nuclear magnetic resonance. Anal. Methods-UK 2017, 9, 1718–1726. [Google Scholar] [CrossRef]
- Ferri, P.; Ramil, M.; Rodriguez, I.; Bergamasco, R.; Vieira, A.M.; Cela, R. Assessment of quinoxyfen phototransformation pathways by liquid chromatography coupled to accurate mass spectrometry. Anal. Bioanal. Chem. 2017, 409, 2981–2991. [Google Scholar] [CrossRef] [PubMed]
- Mackintosh, S.A.; Dodder, N.G.; Shaul, N.J.; Aluwihare, L.I.; Maruya, K.A.; Chivers, S.J.; Danil, K.; Weller, D.W.; Hoh, E. Newly Identified DDT-Related Compounds Accumulating in Southern California Bottlenose Dolphins. Environ. Sci. Technol. 2016, 50, 12129–12137. [Google Scholar] [CrossRef] [PubMed]
- Szultka-Mlynska, M.; Buszewski, B. Electrochemistry-mass spectrometry for in-vitro determination of selected chemotherapeutics and their electrochemical products in comparison to in-vivo approach. Talanta 2016, 160, 694–703. [Google Scholar] [CrossRef] [PubMed]
- Gawlik, M.; Skibinski, R. Simulation of phase I metabolism reactions of clozapine by HLM and photocatalytic methods with the use of UHPLC-ESI-MS/MS. Biomed. Chromatogr. 2018, 32, e4297. [Google Scholar] [CrossRef] [PubMed]
- Zwiener, C. Occurrence and analysis of pharmaceuticals and their transformation products in drinking water treatment. Anal. Bioanal. Chem. 2007, 387, 1159–1162. [Google Scholar] [CrossRef] [PubMed]
- Szultka-Mlynska, M.; Bajkacz, S.; Baranowska, I.; Buszewski, B. Structural characterization of electrochemically and in vivo generated potential metabolites of selected cardiovascular drugs by EC-UHPLC/ESI-MS using an experimental design approach. Talanta 2018, 176, 262–276. [Google Scholar] [CrossRef] [PubMed]
- Serna-Galvis, E.A.; Silva-Agredo, J.; Giraldo, A.L.; Flórez, O.A.; Torres-Palma, R.A. Comparison of route, mechanism and extent of treatment for the degradation of a β-lactam antibiotic by TiO2 photocatalysis, sonochemistry, electrochemistry and the photo-Fenton system. Chem. Eng. J. 2016, 284, 953–962. [Google Scholar] [CrossRef]
- Gawlik, M.; Trawinski, J.; Skibinski, R. Imitation of phase I metabolism reactions of MAO-A inhibitors by titanium dioxide photocatalysis. Eur. J. Pharm. Sci. 2018, 114, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Trawinski, J.; Skibinski, R. Photolytic and photocatalytic degradation of the antipsychotic agent tiapride: Kinetics, transformation pathways and computational toxicity assessment. J. Hazard. Mater. 2017, 321, 841–858. [Google Scholar] [CrossRef] [PubMed]
- Kosjek, T.; Negreira, N.; Heath, E.; Lopez de Alda, M.; Barcelo, D. Aerobic activated sludge transformation of vincristine and identification of the transformation products. Sci. Total Environ. 2018, 610–611, 892–904. [Google Scholar] [CrossRef] [PubMed]
- Lambropoulou, D.; Evgenidou, E.; Saliverou, V.; Kosma, C.; Konstantinou, I. Degradation of venlafaxine using TiO2/UV process: Kinetic studies, RSM optimization, identification of transformation products and toxicity evaluation. J. Hazard. Mater. 2017, 323, 513–526. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Shao, Y.; Xiao, H.; Santiago-Schubel, B.; Meyer-Alert, H.; Schiwy, S.; Yin, D.; Hollert, H.; Kuppers, S. Electrochemical simulation of triclosan metabolism and toxicological evaluation. Sci. Total Environ. 2018, 622–623, 1193–1201. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Y.; Fang, J.; Shang, C. Kinetics and pathways of ibuprofen degradation by the UV/chlorine advanced oxidation process. Water Res. 2016, 90, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Kosma, C.I.; Lambropoulou, D.A.; Albanis, T.A. Photochemical transformation and wastewater fate and occurrence of omeprazole: HRMS for elucidation of transformation products and target and suspect screening analysis in wastewaters. Sci. Total Environ. 2017, 590–591, 592–601. [Google Scholar] [CrossRef] [PubMed]
- Ofiarska, A.; Pieczyńska, A.; Fiszka Borzyszkowska, A.; Stepnowski, P.; Siedlecka, E.M. Pt–TiO2 -assisted photocatalytic degradation of the cytostatic drugs ifosfamide and cyclophosphamide under artificial sunlight. Chem. Eng. J. 2016, 285, 417–427. [Google Scholar] [CrossRef]
- Arsand, J.B.; Hoff, R.B.; Jank, L.; Meirelles, L.N.; Silvia Diaz-Cruz, M.; Pizzolato, T.M.; Barcelo, D. Transformation products of amoxicillin and ampicillin after photolysis in aqueous matrices: Identification and kinetics. Sci. Total Environ. 2018, 642, 954–967. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Cheng, S.; Yang, X.; Ren, J.; Fang, J.; Shang, C.; Song, W.; Lian, L.; Zhang, X. UV/chlorine treatment of carbamazepine: Transformation products and their formation kinetics. Water Res. 2017, 116, 254–265. [Google Scholar] [CrossRef] [PubMed]
- Jallouli, N.; Elghniji, K.; Hentati, O.; Ribeiro, A.R.; Silva, A.M.; Ksibi, M. UV and solar photo-degradation of naproxen: TiO(2) catalyst effect, reaction kinetics, products identification and toxicity assessment. J. Hazard. Mater. 2016, 304, 329–336. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Contaminants in the Food Chain. Scientific Opinion on Emerging and Novel Brominated Flame Retardants (BFRs) in Food. EFSA J. 2012. [Google Scholar] [CrossRef]
- Bergman, Å.; Rydén, A.; Law, R.J.; de Boer, J.; Covaci, A.; Alaee, M.; Birnbaum, L.; Petreas, M.; Rose, M.; Sakai, S.; et al. A novel abbreviation standard for organobromine, organochlorine and organophosphorus flame retardants and some characteristics of the chemicals. Environ. Int. 2012, 49, 57–82. [Google Scholar] [CrossRef] [PubMed]
- Su, G.; Saunders, D.; Yu, Y.; Yu, H.; Zhang, X.; Liu, H.; Giesy, J.P. Occurrence of additive brominated flame retardants in aquatic organisms from Tai Lake and Yangtze River in Eastern China, 2009–2012. Chemosphere 2014, 114, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Sühring, R.; Barber, J.L.; Wolschke, H.; Kötke, D.; Ebinghaus, R. Fingerprint analysis of brominated flame retardants and Dechloranes in North Sea sediments. Environ. Res. 2015, 140, 569–578. [Google Scholar] [CrossRef] [PubMed]
- Hendriks, H.S.; Westerink, R.H. Neurotoxicity and risk assessment of brominated and alternative flame retardants. Neurotoxicol. Teratol. 2015, 52, 248–269. [Google Scholar] [CrossRef] [PubMed]
- Leonetti, C.; Butt, C.M.; Hoffman, K.; Miranda, M.L.; Stapleton, H.M. Concentrations of polybrominated diphenyl ethers (PBDEs) and 2, 4, 6-tribromophenol in human placental tissues. Environ. Int. 2016, 88, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Heberle, A.N.A.; da Silva, S.W.; Klauck, C.R.; Ferreira, J.Z.; Rodrigues, M.A.S.; Bernardes, A.M. Electrochemical enhanced photocatalysis to the 2, 4, 6 Tribromophenol flame retardant degradation. J. Catal. 2017, 351, 136–145. [Google Scholar] [CrossRef]
- Marková, E.; Kučerová, P.; Skopalová, J.; Barták, P. Electrochemical Oxidation of 2, 4, 6-Tribromophenol in Aqueous-Alcoholic Media. Electroanalysis 2015, 27, 156–165. [Google Scholar] [CrossRef]
- Wagoner, E.R.; Baumberger, C.P.; Peverly, A.A.; Peters, D.G. Electrochemical reduction of 1, 2, 5, 6, 9, 10-hexabromocyclododecane at carbon and silver cathodes in dimethylformamide. J. Electroanal. Chem. 2014, 713, 136–142. [Google Scholar] [CrossRef]
- Xu, D.; Song, X.; Qi, W.; Wang, H.; Bian, Z. Degradation mechanism, kinetics, and toxicity investigation of 4-bromophenol by electrochemical reduction and oxidation with Pd–Fe/graphene catalytic cathodes. Chem. Eng. J. 2018, 333, 477–485. [Google Scholar] [CrossRef]
- Skopalová, J.; Barták, P.; Bednář, P.; Tomková, H.; Ingr, T.; Lorencová, I.; Kučerová, P.; Papoušek, R.; Borovcová, L.; Lemr, K. Carbon fiber brush electrode as a novel substrate for atmospheric solids analysis probe (ASAP) mass spectrometry: Electrochemical oxidation of brominated phenols. Anal. Chim. Acta 2018, 999, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Tang, T.; Lu, G.; Huang, K.; Chen, M.; Tao, X.; Yin, H.; Dang, Z. Formation and degradation of polybrominated dibenzofurans (PBDFs) in the UV photolysis of polybrominated diphenyl ethers (PBDEs) in various solutions. Chem. Eng. J. 2017. [Google Scholar] [CrossRef]
- Wang, R.; Tang, T.; Xie, J.; Tao, X.; Huang, K.; Zou, M.; Yin, H.; Dang, Z.; Lu, G. Debromination of polybrominated diphenyl ethers (PBDEs) and their conversion to polybrominated dibenzofurans (PBDFs) by UV light: Mechanisms and pathways. J. Hazard. Mater. 2018. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Zhao, H.; Quan, X.; He, X.; Chen, S. Photochemical Formation of Hydroxylated Polybrominated Diphenyl Ethers (OH-PBDEs) from Polybrominated Diphenyl Ethers (PBDEs) in Aqueous Solution under Simulated Solar Light Irradiation. Environ. Sci. Technol. 2015, 49, 9092–9099. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Hu, X.; Zhang, H.; Chang, F.; Luo, Y. Photolysis kinetics, mechanisms, and pathways of tetrabromobisphenol A in water under simulated solar light irradiation. Environ. Sci. Technol. 2015, 49, 6683–6690. [Google Scholar] [CrossRef] [PubMed]
- Bao, Y.; Niu, J. Photochemical transformation of tetrabromobisphenol A under simulated sunlight irradiation: Kinetics, mechanism and influencing factors. Chemosphere 2015, 134, 550–556. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.; Wang, C.; Sun, J.; Li, S.P. Photolytic degradation of tris-(2,3-dibromopropyl) isocyanurate (TBC) in aqueous systems. Environ. Technol. 2016, 37, 2292–2297. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-n.; Chen, J.; Xie, Q.; Li, Y.; Zhou, C. Photochemical transformation of five novel brominated flame retardants: Kinetics and photoproducts. Chemosphere 2016, 150, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, K.-H.; Abou-Elwafa Abdallah, M.; Moehring, T.; Harrad, S. Biotransformation of the Flame Retardant 1,2-Dibromo-4-(1,2-dibromoethyl)cyclohexane (TBECH) in vitro by Human Liver Microsomes. Environ. Sci. Technol. 2017, 51, 10511–10518. [Google Scholar] [CrossRef] [PubMed]
- Su, G.; Greaves, A.K.; Teclechiel, D.; Letcher, R.J. In vitro metabolism of photolytic breakdown products of tetradecabromo-1, 4-diphenoxybenzene flame retardant in herring gull and rat liver microsomal assays. Environ. Sci. Technol. 2016, 50, 8335–8343. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, Y.; Du, Z.; Peng, J.; Mao, L.; Gao, S. Biotransformation of OH-PBDEs by pig liver microsomes: Investigating kinetics, identifying metabolites, and examining the role of different CYP isoforms. Chemosphere 2016, 148, 354–360. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Erratico, C.; Abdallah, M.A.-E.; Negreira, N.; Luo, X.; Mai, B.; Covaci, A. In vitro metabolism of BDE-47, BDE-99, and α-, β-, γ-HBCD isomers by chicken liver microsomes. Environ. Res. 2015, 143, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Meca, G.; Zhou, T.; Li, X.; Ritieni, A.; Mañes, J. Ciclohexadespipeptide beauvericin degradation by different strains of Saccharomyces cerevisiae. Food Chem. Toxicol. 2013, 59, 334–338. [Google Scholar] [CrossRef] [PubMed]
- Meca, G.; Luciano, F.; Zhou, T.; Tsao, R.; Mañes, J. Chemical reduction of the mycotoxin beauvericin using allyl isothiocyanate. Food Chem. Toxicol. 2012, 50, 1755–1762. [Google Scholar] [CrossRef] [PubMed]
- Bordin, K.; Saladino, F.; Fernandez-Blanco, C.; Ruiz, M.J.; Manes, J.; Fernandez-Franzon, M.; Meca, G.; Luciano, F.B. Reaction of zearalenone and alpha-zearalenol with allyl isothiocyanate, characterization of reaction products, their bioaccessibility and bioavailability in vitro. Food Chem. 2017, 217, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Heydt, M.; Cramer, B.; Graf, I.; Lerch, S.; Humpf, H.-U.; Geisen, R. Wavelength-dependent degradation of ochratoxin and citrinin by light in vitro and in vivo and its implications on Penicillium. Toxins 2012, 4, 1535–1551. [Google Scholar] [CrossRef] [PubMed]
- Moreau, M.; Lescure, G.; Agoulon, A.; Svinareff, P.; Orange, N.; Feuilloley, M. Application of the pulsed light technology to mycotoxin degradation and inactivation. J. Appl. Toxicol. 2013, 33, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Kabak, B.; Dobson, A.D.; Var, I.l. Strategies to prevent mycotoxin contamination of food and animal feed: A review. Crit. Rev. Food Sci. Nutr. 2006, 46, 593–619. [Google Scholar] [CrossRef] [PubMed]
- McCormick, S.P. Microbial detoxification of mycotoxins. J. Chem. Ecol. 2013, 39, 907–918. [Google Scholar] [CrossRef] [PubMed]
- Chanique, G.D.; Arévalo, A.H.; Zon, M.A.; Fernández, H. Eletrochemical reduction of patulin and 5-hydroxymethylfurfural in both neutral and acid non-aqueous media. Their electroanalytical determination in apple juices. Talanta 2013, 111, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Calcutt, M.W.; Gillman, I.G.; Noftle, R.E.; Manderville, R.A. Electrochemical oxidation of ochratoxin A: Correlation with 4-chlorophenol. Chem. Res. Toxicol. 2001, 14, 1266–1272. [Google Scholar] [CrossRef] [PubMed]
- Vidal, J.C.; Bonel, L.; Ezquerra, A.; Hernandez, S.; Bertolin, J.R.; Cubel, C.; Castillo, J.R. Electrochemical affinity biosensors for detection of mycotoxins: A review. Biosens. Bioelectron. 2013, 49, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Catanante, G.; Rhouati, A.; Hayat, A.; Marty, J.L. An overview of recent electrochemical immunosensing strategies for mycotoxins detection. Electroanalysis 2016, 28, 1750–1763. [Google Scholar] [CrossRef]
- Simon, H.; Hoffmann, G.; Hubner, F.; Humpf, H.U.; Karst, U. Electrochemical simulation of metabolic reactions of the secondary fungal metabolites alternariol and alternariol methyl ether. Anal. Bioanal. Chem. 2016, 408, 2471–2483. [Google Scholar] [CrossRef] [PubMed]
- Keller, J.; Haase, H.; Koch, M. Electrochemical simulation of biotransformation reactions of citrinin and dihydroergocristine compared to UV irradiation and Fenton-like reaction. Anal. Bioanal. Chem. 2017, 409, 4037–4045. [Google Scholar] [CrossRef] [PubMed]
- Keller, J.; Haase, H.; Koch, M. Hydroxylation and dimerization of zearalenone: Comparison of chemical, enzymatic and electrochemical oxidation methods. World Mycotoxin J. 2017, 10, 297–307. [Google Scholar] [CrossRef]
- Toro, P.C.D.; Arévalo, F.J.; Zon, M.A.; Fernández, H. Studies of the electrochemical behavior of moniliformin mycotoxin and its sensitive determination at pretreated glassy carbon electrodes in a non-aqueous medium. J. Electroanal. Chem. 2015, 738, 40–46. [Google Scholar] [CrossRef]
- Nieto, C.H.D.; Granero, A.M.; Zon, M.A.; Fernández, H. Novel electrochemical properties of an emergent mycotoxin: Sterigmatocystin. J. Electroanal. Chem. 2016, 765, 155–167. [Google Scholar] [CrossRef]

| Name of Compound/s | Type of Pesticides | Matrices | TPs Prediction/Authentication Methods | Simulated Transformation Mechanisms | Ref |
|---|---|---|---|---|---|
| Azoxystrobin Difenoconazole Thiacloprid | fungicide fungicide insecticide | brassica species vegetables (pak choi and broccoli) | Suspect screening, in silico fragmentation and authentic standards | Hydrolysis, hydroxylation, dealkylation (O-, N- & S-), dehydrogenation, glucuronidation, esterification | [47] |
| Chlorpyrifos | insecticide | fruits and spices | Synthesis by EC and UV and used for targeted screening | P-oxidation, O-dealkylation, hydrolysis, −Cl +H | [48] |
| Ethoxyquin | growth regulator | fish feed | Synthesis by chemical (H2O2 and DPPH) and used for authentication | N-oxide, hydroxylation, O-dealkylation, dimerization, epoxidation, quinone formation | [51] |
| Forchlorfenuron | growth regulator | kiwifruit | Comparison with authentic standards | +OH, −Cl +H, N-dealkylation, −Cl +OH, glucuronidation | [52] |
| Isoproturon | herbicide | plant leaf | UV and sunlight (untargeted) | Hydroxylation, N-dealkylation | [53] |
| Bentazon | herbicide | - | Photocatalysis (TiO2, ZnO) | N-dealkylation, hydroxylation, dimerization | [57] |
| Cyprodinil | fungicide | - | UV–Vis | Isomerization, cyclization, hydroxylation | [58] |
| Methyl parathion | insecticide | - | MnO2 (chemical) | P-oxidation, hydrolysis, dealkylation | [59] |
| Fluopyram | herbicide | - | UV and sunlight (untargeted) | −Cl +H, −Cl +OH, cyclization via −HCl | [60] |
| Fluopyram | herbicide | commercially available microsomes | Liver microsomes and EC | Hydroxylation, N-dealkylation, −Cl +H, −Cl +OH, dehydrogenation, epoxidation | [62] |
| Chlorpyrifos | insecticide | commercially available microsomes | Liver microsomes and EC | P-oxidation, O-dealkylation, hydrolysis, −Cl +H | [63] |
| Metribuzin | herbicide | tomato | Comparison with authentic standards | Deamination, oxidative desulfuration | [65] |
| Flonicamid | insecticide | orange | Comparison with authentic standards | Hydrolysis of amides, deamination by hydrolysis | [66] |
| Quinoxyfen | fungicide | water | UV and untargeted | −Cl +OH, hydroxylation, cyclization (−H2O, −HCl), hydrolysis | [67] |
| DDT and DDT-related cpds | insecticide | dolphin serum | Comparison with authentic standards | Olefin via −HCl, −Cl +OH | [68] |
| Drugs | Group of Drugs | TPs Prediction/Authentication Methods | Simulated Transformation Mechanisms * | Toxicity Test of TP | Degra Dation Kinetic | Ref |
|---|---|---|---|---|---|---|
| Amoxicillin, Cefotaxime, Linezolid, Moxifloxacin, Metronidazole, Fluconazole | Antibiotic Antifungal | EC-reactor RLM In vivo (urine samples) | Red, Deh, Dealk, OH, X-ox, Dehal, GSH Red, Deh, Dealk, OH, X-ox, Dehal, Red, X-ox, OH, Alkyl | [69] | ||
| Acebutolol, Atenolol, Propranolol, Pindolol, Oxprenolol, Cicloprolol, Pirbuterol, Mexiletine, Propafenone | β-blocker β2-adrenoceptor Antiarrhythmic | EC-reactor In vivo (urine and plasma samples) | OH, X-ox, Dealk, Alkyl, Red, GSH OH, X-ox, Dealk | [72] | ||
| Buspirone Promazine Testosterone 7-ethoxycoumarin | Anxiolytic Antipsychotics Anabolic steroid Test substance (CYP450) | HLM Photocatalytic (TiO2) EC-Fenton EC-Reactor | OH, Deh, X-ox, Dealk, Hyd, OH, Deh, X-ox, Dealk, Hyd OH, Deh, X-ox, Hyd Deh, X-ox, Dealk | [26] | ||
| Oxacillin | β-lactam antibiotic | Photocatalytic (TiO2) Sonochemisty EC-reactor Photo-Fenton | X, ox, rCleav, OH X-ox, rCleav X-ox, rCleav X-ox, rCleav, OH | [73] | ||
| Trimethoprim | Anti-infective | Electrolysis EC-Fenton EC reactor | OH OH, Dealk OH | [18] | ||
| Moclobemide, Toloxatone | Antidepressant | Photocatalytic (TiO2) HLM | X-ox, OH, Dealk, Deh, Dehal, rCleav X-ox, OH, Dealk, Deh | X | [74] | |
| Clozapine | Antipsychotic | Photocatalytic (TiO2, ZnO) HLM | X-ox, OH, Hyd, Dealk, Dehal, rCleav X-ox, OH, Hyd, Dealk, Dehal, rCleav | X | [70] | |
| Tiapride | Antipsychotic | Photolytic Photocatalytic (TiO2, H2O2) | Dealk, OH, Red, Desulf, X-ox Dealk, OH, Red, Desulf, X-ox, rCleav, Deh | X | X | [75] |
| Propranolol | β-blocker | Photolysis | OH, rCleav, X-ox, Ox | X | X | [27] |
| Vincristine | Anticancer | Aerobic activated sludge | rCleav, Ox, Deca, Dealk, Deh, | X | [76] | |
| Venlafaxine | Antidepressant | Photocatalytic (UV/TiO2) | OH, Alkyl, Deh, X-ox, | X | X | [77] |
| Triclosan | Antimicrobial | Electrochemical reactor | OH, Ox, Dehal, Alky | X | [78] | |
| Ibuprofen | Nonsteroidal anti-inflammatory | UV/chlorine UV/H2O2 | OH, Decar, Dealk, +Cl, rCleav | X | X | [79] |
| Omeprazole | Proton pump inhibitors | Photolysis | Red, Dehyd, OH, Ox | X | X | [80] |
| Ifosamine, Cyclophosphamide | Cytostatic | Photocatalytic (TiO2, Pt-TiO2) | OH, Dealk, Ox | X | [81] | |
| Amoxicillin, Ampicillin | Antibiotic | Photolysis | OH, Deh, Dealk | X | [82] | |
| Carbamazepine | Antiepileptic | UV/chlorine | Red, OH, +Cl, Hyd, Deh | X | X | [83] |
| Naproxen | Nonsteroidal anti-inflammatory | UV and solar Photolysis | Dealk, Hydr, OH | X | X | [84] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kotthoff, L.; Keller, J.; Lörchner, D.; Mekonnen, T.F.; Koch, M. Transformation Products of Organic Contaminants and Residues—Overview of Current Simulation Methods. Molecules 2019, 24, 753. https://doi.org/10.3390/molecules24040753
Kotthoff L, Keller J, Lörchner D, Mekonnen TF, Koch M. Transformation Products of Organic Contaminants and Residues—Overview of Current Simulation Methods. Molecules. 2019; 24(4):753. https://doi.org/10.3390/molecules24040753
Chicago/Turabian StyleKotthoff, Lisa, Julia Keller, Dominique Lörchner, Tessema F. Mekonnen, and Matthias Koch. 2019. "Transformation Products of Organic Contaminants and Residues—Overview of Current Simulation Methods" Molecules 24, no. 4: 753. https://doi.org/10.3390/molecules24040753






