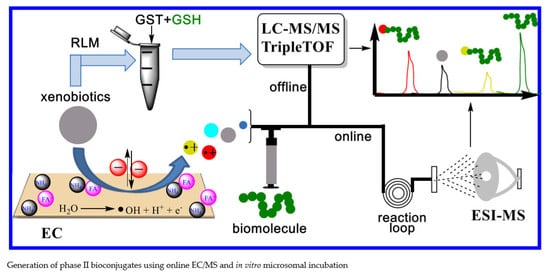
Glucosylation and Glutathione Conjugation of Chlorpyrifos and Fluopyram Metabolites Using Electrochemistry/Mass Spectrometry
Abstract
:1. Introduction
2. Results and Discussion
2.1. Glutathione Conjugation of CPF TPs: Online EC/MS vs. Liver Microsomes
2.2. Glucosylation of CPF TPs in Online EC/MS
2.3. Glutathione Conjugation and Glucosylation of FLP TPs
3. Experimental
3.1. Chemicals and Reagents
3.2. Electrochemical Oxidation and Analysis of TPs
3.3. Adduct Formation with Biomolecules
3.4. Offline LC-MS/MS Analysis of Bioconjugates
3.5. Confirmation by HRMS
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zabaleta, I.; Bizkarguenaga, E.; Nunoo, D.B.O.; Schultes, L.; Leonel, J.; Prieto, A.; Zuloaga, O.; Benskin, J.P. Biodegradation and uptake of the pesticide sulfluramid in a soil-carrot mesocosm. Environ. Sci. Technol. 2018, 52, 2603–2611. [Google Scholar] [CrossRef] [PubMed]
- Von Gunten, U. Oxidation processes in water treatment: Are we on track? Environ. Sci. Technol. 2018, 52, 5062–5075. [Google Scholar] [CrossRef] [PubMed]
- Macherey, A.-C.; Dansette, P.M. Chapter 25—Biotransformations leading to toxic metabolites: Chemical aspects. In The Practice of Medicinal Chemistry, 5th ed.; Academic Press: San Diego, CA, USA, 2015; pp. 585–614. [Google Scholar]
- Masubuchi, Y.; Horie, T. Toxicological significance of mechanism-based inactivation of cytochrome p450 enzymes by drugs. Crit. Rev. Toxicol. 2007, 37, 389–412. [Google Scholar] [CrossRef] [PubMed]
- Zanger, U.M.; Schwab, M. Cytochrome p450 enzymes in drug metabolism: Regulation of gene expression, enzyme activities, and impact of genetic variation. Pharmacol. Ther. 2013, 138, 103–141. [Google Scholar] [CrossRef] [PubMed]
- Testa, B.; Krämer, S.D. The biochemistry of drug metabolism—An introduction. Chem. Biodivers. 2008, 5, 2171–2336. [Google Scholar] [CrossRef] [PubMed]
- Guengerich, F.P. Mechanisms of cytochrome p450 substrate oxidation: Minireview. J. Biochem. Mol. Toxicol. 2007, 21, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Van Bladeren, P.J. Glutathione conjugation as a bioactivation reaction. Chem. Biol. Interact. 2000, 129, 61–76. [Google Scholar] [CrossRef]
- Weber, G.L.; Steenwyk, R.C.; Nelson, S.D.; Pearson, P.G. Identification of n-acetylcysteine conjugates of 1,2-dibromo-3-chloropropane: Evidence for cytochrome p450 and glutathione mediated bioactivation pathways. Chem. Res. Toxicol. 1995, 8, 560–573. [Google Scholar] [CrossRef] [PubMed]
- Monks, T.J.; Anders, M.W.; Dekant, W.; Stevens, J.L.; Lau, S.S.; van Bladeren, P.J. Glutathione conjugate mediated toxicities. Toxicol. Appl. Pharmacol. 1990, 106, 1–19. [Google Scholar] [CrossRef]
- Jurva, U.; Weidolf, L. Electrochemical generation of drug metabolites with applications in drug discovery and development. Trac-Trends Anal. Chem. 2015, 70, 92–99. [Google Scholar] [CrossRef]
- Oberacher, H.; Pitterl, F.; Chervet, J.P. “Omics” applications of electrochemistry coupled to mass spectrometry—A review. LC GC Eur. 2015, 28, 138–150. [Google Scholar]
- Buter, L.; Vogel, M.; Karst, U. Adduct formation of electrochemically generated reactive intermediates with biomolecules. Trac-Trends Anal. Chem. 2015, 70, 74–91. [Google Scholar] [CrossRef]
- The Rapid Alert System for Food and Feed (RASFF). Available online: https://ec.europa.eu/ food/safety/rasff/for_consumers_en (accessed on 1 October 2018).
- Mekonnen, T.F.; Panne, U.; Koch, M. Prediction of biotransformation products of the fungicide fluopyram by electrochemistry coupled online to liquid chromatography-mass spectrometry and comparison with in vitro microsomal assays. Anal. Bioanal. Chem. 2018, 410, 2607–2617. [Google Scholar] [CrossRef] [PubMed]
- Mekonnen, T.F.; Panne, U.; Koch, M. Electrochemistry coupled online to liquid chromatography-mass spectrometry for fast simulation of biotransformation reactions of the insecticide chlorpyrifos. Anal. Bioanal. Chem. 2017, 409, 3359–3368. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.; Joo, H.; Rose, R.L.; Hodgson, E. Metabolism of chlorpyrifos and chlorpyrifos oxon by human hepatocytes. J. Biochem. Mol. Toxicol. 2006, 20, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Bicker, W.; Lammerhofer, M.; Lindner, W. Determination of chlorpyrifos metabolites in human urine by reversed-phase/weak anion exchange liquid chromatography-electrospray ionisation-tandem mass spectrometry. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2005, 822, 160–169. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Cao, Y.; Rose, R.L.; Brimfield, A.A.; Dai, D.; Goldstein, J.A.; Hodgson, E. Metabolism of chlorpyrifos by human cytochrome p450 isoforms and human, mouse, and rat liver microsomes. Drug Metab. Dispos. 2001, 29, 1201–1204. [Google Scholar] [PubMed]
- Sancho, J.V.; Pozo, O.J.; Hernandez, F. Direct determination of chlorpyrifos and its main metabolite 3,5, 6-trichloro-2-pyridinol in human serum and urine by coupled-column liquid chromatography/electrospray-tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2000, 14, 1485–1490. [Google Scholar] [CrossRef]
- Fujioka, K.; Casida, J.E. Glutathione s-transferase conjugation of organophosphorus pesticides yields s-phospho-, s-aryl-, and s-alkylglutathione derivatives. Chem. Res. Toxicol. 2007, 20, 1211–1217. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (efsa), conclusion on the peer review of the pesticide risk assessment of the active substance fluopyram. EFSA J. 2013, 11, 3052–3128. [CrossRef]
- Giustarini, D.; Fanti, P.; Matteucci, E.; Rossi, R. Micro-method for the determination of glutathione in human blood. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2014, 964, 191–194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Australian Pesticides and Veterinary Medicines Authority (APVMA). Available online: https://apvma.gov.au/sites/.../publication/14166-prs-fluopyram.pdf (accessed on 25 July2017).
Sample Availability: Not available. |






| Abbreviation/Symbol | tR (min) | Calculated [M + H]+ | Product Ions by QTRAP-MS/MS | Measured [M + H]+ | δm/m (ppm) | Type of Conjugate |
|---|---|---|---|---|---|---|
| C1 | 19.5 | 487.0007 | 308, 291, 180, 179, 205, 233 | 487.0030 | 4.7 | TCP + GSH – H2O |
| C2 | 20.2 | 469.0346 | 469, 451/453, 433, 437, 405 | 469.0312 | 1.3 | TCP + GSH – HCl |
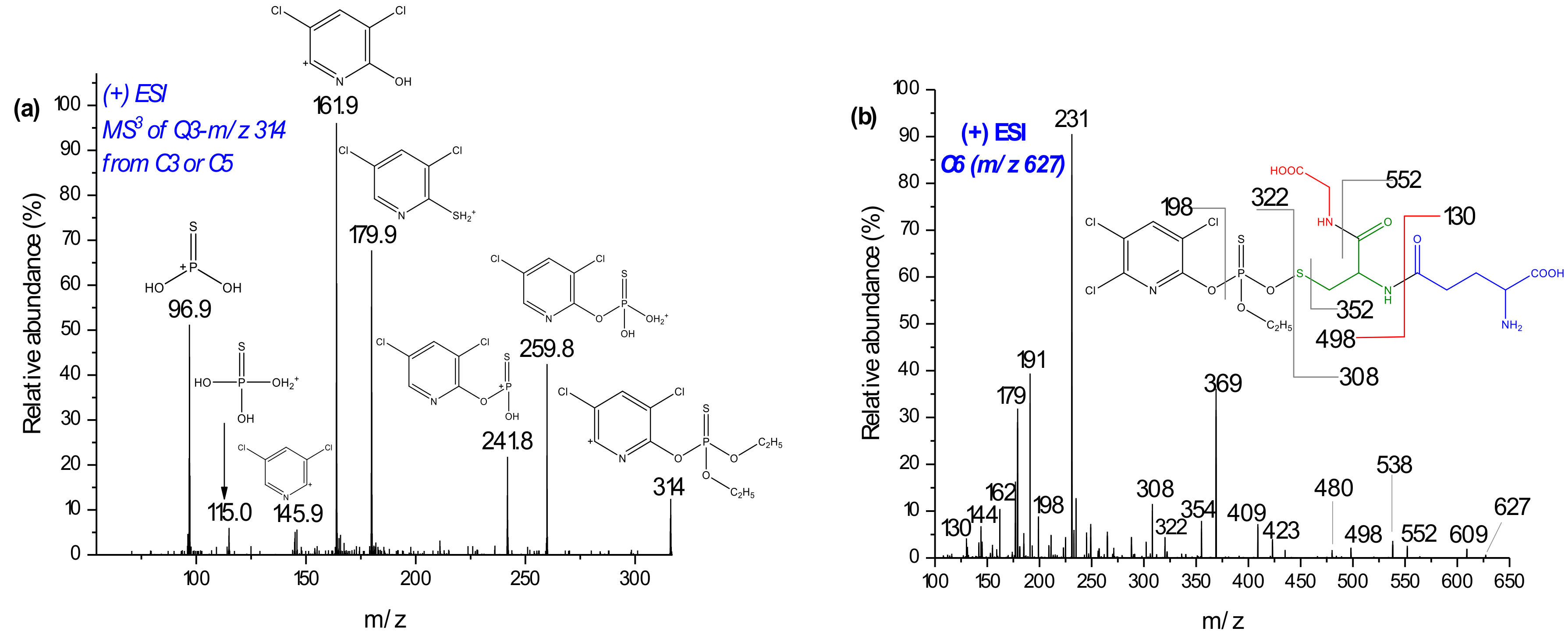
| C3C5 | 21.530.4 | 621.0407 | 621, 603/605, 564, 593, 541, 469, 314, 288, 171, 154 | 621.0534 | 4.8 | CPF + GSH – HCl |
| C4 | 23.3 | 605.0635 | 605, 564, 587, 573, 569, 555, 540, 339, 327, 298, 251, 154 | 605.0520 | 2.2 | Oxon +GSH – HCl |
| C6 | 29.6 | 626.9704 | 609, 538, 369, 322, 308, 231 | 627.0068 | −7.2 | U |
| C7 | 15.6 | 359.9803 | 360, 362, 364, 324, 198, 180 | 359.9891 | −5.2 | TCP + Glc – H2O |
| C8 | 18.4 | 478.0431 | 316, 163, 153, 137, 162 | 478.0410 | 6.5 | Oxon + Glc – HCl |
| C9 | 20.5 | 494.0203 | 515, 314, 171, 163 | 494.0299 | −3.2 | CPF + Glc – HCl |
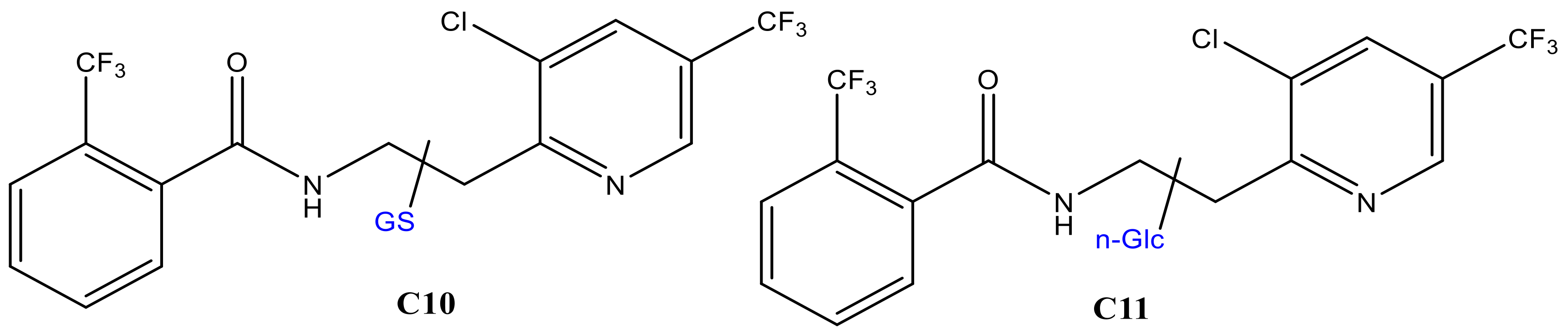
| C10 * | 23.84 | 702.1280 | 705, 513, 429, 395, 308, 173, 145, 129, 291, 179, 162 | 702.1832 | −5.1 | Hydroxyl FLP + GSH – H2O |
| C11 * | 28.9 | 701.2423 | 703, 413, 395, 307, 289, 573, 173, 145, 129 | 701.2093 | −4.3 | Hydroxyl FLP + n-Glc – H2O |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mekonnen, T.F.; Panne, U.; Koch, M. Glucosylation and Glutathione Conjugation of Chlorpyrifos and Fluopyram Metabolites Using Electrochemistry/Mass Spectrometry. Molecules 2019, 24, 898. https://doi.org/10.3390/molecules24050898
Mekonnen TF, Panne U, Koch M. Glucosylation and Glutathione Conjugation of Chlorpyrifos and Fluopyram Metabolites Using Electrochemistry/Mass Spectrometry. Molecules. 2019; 24(5):898. https://doi.org/10.3390/molecules24050898
Chicago/Turabian StyleMekonnen, Tessema Fenta, Ulrich Panne, and Matthias Koch. 2019. "Glucosylation and Glutathione Conjugation of Chlorpyrifos and Fluopyram Metabolites Using Electrochemistry/Mass Spectrometry" Molecules 24, no. 5: 898. https://doi.org/10.3390/molecules24050898