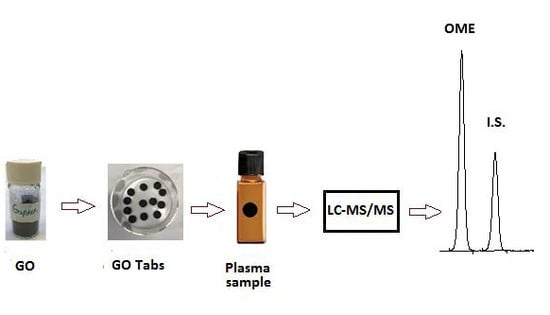
Graphene Oxide Tablets for Sample Preparation of Drugs in Biological Fluids: Determination of Omeprazole in Human Saliva for Liquid Chromatography Tandem Mass Spectrometry
Abstract
:1. Introduction
2. Materials and methods
2.1. Chemicals and Reagents
2.2. Instrumentation
2.3. Preparation and Characterization of GO-Tabs
2.4. Preparation of Stock, Standard and Quality Control Solutions
2.5. Sample Preparation
3. Results and Discussion
3.1. GO-Tab Morphology Analysis
3.2. Optimization of Extraction Protocol
3.2.1. Extraction Time
3.2.2. Desorption Time
3.2.3. Type of Desorption Solvent
3.2.4. Effect of pH
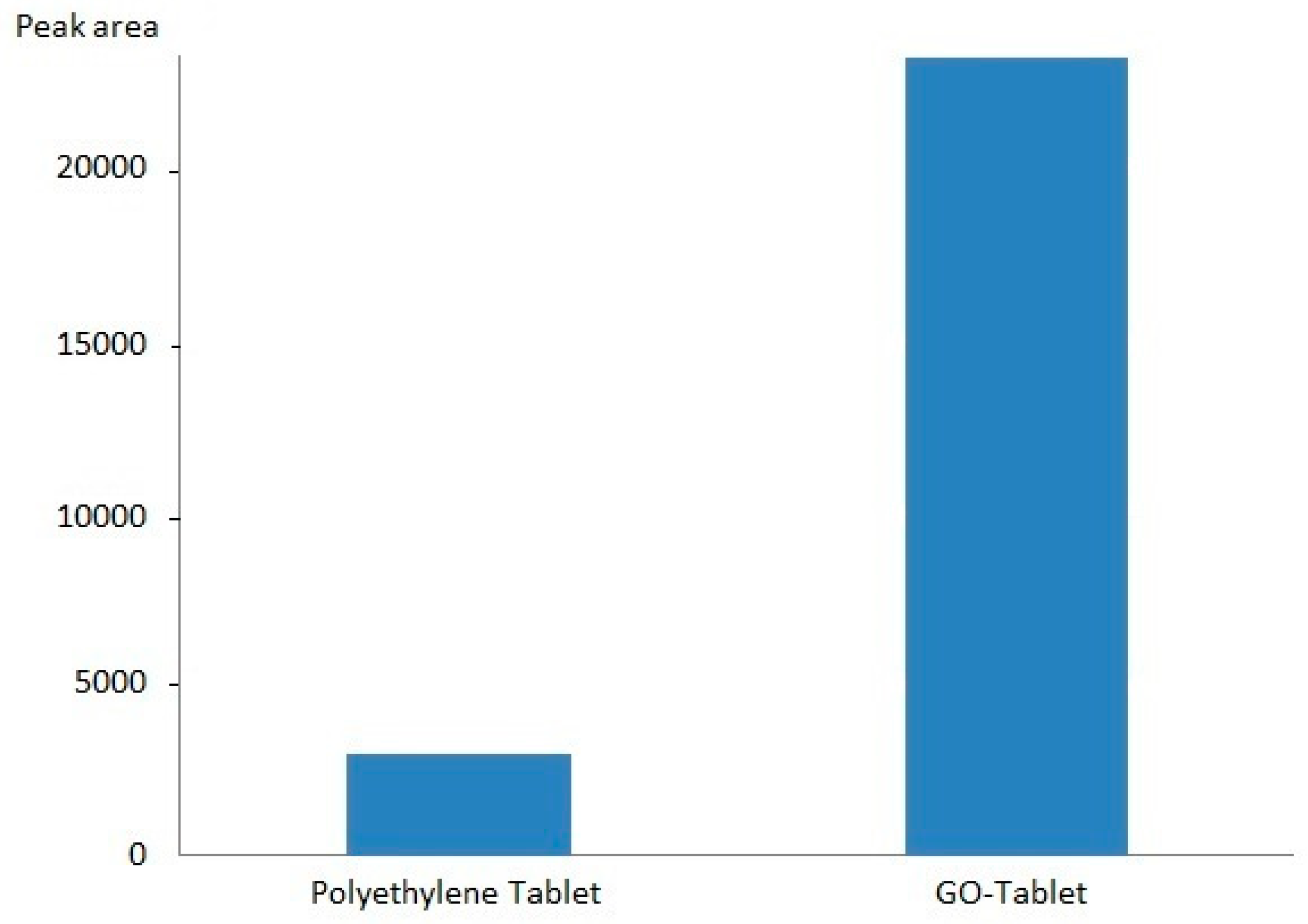
3.3. Adsorption Capacity
3.4. Selectivity of the GO-Tabs
3.5. Method Validation
3.5.1. Calibration, Selectivity and Extraction Efficiency
3.5.2. Accuracy and Precision
3.5.3. Method Selectivity and Matrix Effects
3.5.4. Carry-Over and Reuse of GO-Tabs
4. Analysis of Patient Samples
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Goodman, L.S.; Brunton, L.L.; Gilman, A.; Chabner, B.; Knollmann, B.C. Goodman and Gilman′s: The Pharmacological Basis of Therapeutics; McGraw-Hill Medical: New York, NY, USA, 2011. [Google Scholar]
- World Health Organization. WHO Model Lists of Essential Medicines; World Health Organization: Geneva, Switzerland, April 2015. [Google Scholar]
- Cairns, A.M.; Chiou, R.H.Y.; Rogers, J.D.; Demetriades, J.L.J. Enantioselective high-performance liquid chromatographic determination of omeprazole in human plasma. Chromatogr. B: Biomed. Sci. Appl. 1995, 666, 323–328. [Google Scholar] [CrossRef]
- Ishii, M.; Sato, M.; Ogawa, M.; Takubo, T.; Hara, K.i.; Ishii, Y. Simultaneous Determination of Omeprazole and its Metabolites (5′-Hydroxyomeprazole and Omeprazole Sulfone) in Human Plasma by Liquid Chromatography-Tandem Mass Spectrometry. J. Liquid Chromatogr. Relat. Technol. 2007, 30, 1797–1810. [Google Scholar] [CrossRef]
- Pfaffe, T.; Cooper-White, J.; Beyerlein, P.; Kostner, K.; Punyadeera, C. Diagnostic potential of saliva: Current state and future applications. Clinic. Chem. 2011, 57, 675–687. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Rehim, A.; Abdel-Rehim, M. Screening and determination of drugs in human saliva utilizing microextraction by packed sorbent and liquid chromatography–tandem mass spectrometry. Biomed. Chromatogr. 2013, 27, 1188–1191. [Google Scholar] [CrossRef]
- Liu, J.; Duan, Y. Saliva: A potential media for disease diagnostics and monitoring. Oral Oncol. 2012, 48, 569–577. [Google Scholar] [CrossRef]
- Tamminga, W.J. Polymorphic Drug Metabolising Enzymes: Assessment of Activities by Phenotyping and Genotyping in Clinical Pharmacology. Ph.D. Thesis, University of Groningen, Groningen, The Netherlands, 2001. [Google Scholar]
- Donzelli, M. Development, Validation and Application of the Basel Phenotyping Cocktail. Ph.D. Thesis, University of Basel, Basel, Switzerland, 2015. [Google Scholar]
- Ashri, N.; Abdel-Rehim, M. Sample treatment based on extraction techniques in biological matrices. Bioanalysis 2011, 3, 1993–2008. [Google Scholar] [CrossRef]
- Vita, M.; Abdel-Rehim, M.; Nilsson, C.; Hassan, Z.; Skansen, P.; Wan, H.; Meurling, L.; Hassan, M.J. Stability, pKa and plasma protein binding of roscovitine. Chromatogr. B 2005, 821, 75–80. [Google Scholar] [CrossRef]
- Klimowska, A.; Wielgomas, B. Off-line microextraction by packed sorbent combined with on solid support derivatization and GC–MS: Application for the analysis of five pyrethroid metabolites in urine samples. Talanta 2018, 176, 165–171. [Google Scholar] [CrossRef]
- Said, R.; Kamel, M.; El Beqqali, A.; Abdel-Rehim, M. Microextraction by packed sorbent for LC–MS/MS determination of drugs in whole blood samples. Bioanalysis 2011, 29, 197–205. [Google Scholar] [CrossRef]
- Noche, G.G.; Laespada, M.E.F.; Pavón, J.L.P.; Cordero, B.M.; Lorenzo, S.M.; Noche, G.G.; Laespada, M.E.F.; Pavón, J.L.P.; Cordero, B.M.; Lorenzo, S.M. Determination of chlorobenzenes in water samples based on fully automated microextraction by packed sorbent coupled with programmed temperature vaporization–gas chromatography–mass spectrometry. Anal. Bioanal. Chem. 2013, 405, 6739–6748. [Google Scholar] [CrossRef] [PubMed]
- Daryanavard, S.M.; Jeppsson-Dadoun, A.; Andersson, L.; Hashemi, M.; Colmsjö, A.; Abdel-Rehim, M. Molecularly imprinted polymer in microextraction by packed sorbent for the simultaneous determination of local anesthetics: lidocaine, ropivacaine, mepivacaine and bupivacaine in plasma and urine samples. Biomed. Chromatogr. 2013, 27, 1481–1488. [Google Scholar] [CrossRef] [PubMed]
- El-Beqqali, A.; Abdel-Rehim, M. Molecularly imprinted polymer-sol-gel tablet toward micro-solid phase extraction: I. Determination of methadone in human plasma utilizing liquid chromatography–tandem mass spectrometry. Anal. Chim. Acta 2016, 936, 116–122. [Google Scholar] [CrossRef] [PubMed]
- El-Beqqali, A.; Andersson, L.; Dadoun Jeppsson, A.; Abdel-Rehim, M.J. Molecularly imprinted polymer-sol-gel tablet toward micro-solid phase extraction: II. Determination of amphetamine in human urine samples by liquid chromatography–tandem mass spectrometry. Chromatogr. B 2017, 1063, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, M.; Moein, M.M.; Madrakian, T.; Afkhami, A.; Bahar, S.; Abdel-Rehim, M.J. Reduced graphene oxide as an efficient sorbent in microextraction by packed sorbent: Determination of local anesthetics in human plasma and saliva samples utilizing liquid chromatography-tandem mass spectrometry. Chromatogr. B 2018, 1095, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, M.; Madrakian, T.; Abdel Rehim, M. Nanomaterials as sorbents for sample preparation in bioanalysis: A review. Anal. Chim. Acta. 2017, 958, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Seabra, A.B.; Paula, A.J.; Lima, S.S.; Alves, O.L.; Durán, N. Nanotoxicity of graphene and graphene oxide. Chem. Res. Toxicol. 2014, 27, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Faria, A.F.; Martinez, D.S.T.; Moraes, A.C.M.; Maia da Costa, M.E.H.; Barros, E.B.; Souza Filho, A.G.; Paula, A.J.; Alvez, O.L. Unveiling the role of oxidation debris on the surface chemistry of graphene through the anchoring of Ag nanoparticles. Chem. Mater. 2012, 24, 4080–4087. [Google Scholar] [CrossRef]
- Guidance for Industry: Bioanalytical Method Validation; U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research, Center for Veterinary Medicine: Rockville, MD, USA, 2001.
- Moein, M.; El Beqqali, A.; Abdel-Rehim, M. Bioanalytical method development and validation: Critical concepts and strategies. J. Chromatogr. B 2017, 1043, 3–11. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |







| Compound | Sample (Conc.) | Accuracy (%) (n = 18) | Precision (RSD%) | |
|---|---|---|---|---|
| Intra-day (n = 6) | Inter-day (n = 18) | |||
| Omeprazole | QCL (6.0 nM) | 105.5 | 5.4 | 7.8 |
| QCM (800 nM) | 106.4 | 5.1 | 7.5 | |
| QCH (1600 nM) | 87.8 | 4.1 | 5.6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zohdi, Z.; Hashemi, M.; Uheida, A.; Moein, M.M.; Abdel-Rehim, M. Graphene Oxide Tablets for Sample Preparation of Drugs in Biological Fluids: Determination of Omeprazole in Human Saliva for Liquid Chromatography Tandem Mass Spectrometry. Molecules 2019, 24, 1191. https://doi.org/10.3390/molecules24071191
Zohdi Z, Hashemi M, Uheida A, Moein MM, Abdel-Rehim M. Graphene Oxide Tablets for Sample Preparation of Drugs in Biological Fluids: Determination of Omeprazole in Human Saliva for Liquid Chromatography Tandem Mass Spectrometry. Molecules. 2019; 24(7):1191. https://doi.org/10.3390/molecules24071191
Chicago/Turabian StyleZohdi, Zeynab, Mahdi Hashemi, Abdusalam Uheida, Mohammad Mahdi Moein, and Mohamed Abdel-Rehim. 2019. "Graphene Oxide Tablets for Sample Preparation of Drugs in Biological Fluids: Determination of Omeprazole in Human Saliva for Liquid Chromatography Tandem Mass Spectrometry" Molecules 24, no. 7: 1191. https://doi.org/10.3390/molecules24071191
APA StyleZohdi, Z., Hashemi, M., Uheida, A., Moein, M. M., & Abdel-Rehim, M. (2019). Graphene Oxide Tablets for Sample Preparation of Drugs in Biological Fluids: Determination of Omeprazole in Human Saliva for Liquid Chromatography Tandem Mass Spectrometry. Molecules, 24(7), 1191. https://doi.org/10.3390/molecules24071191