Possible Role of the Ca2+/Mn2+ P-Type ATPase Pmr1p on Artemisinin Toxicity through an Induction of Intracellular Oxidative Stress
Abstract
:1. Introduction
2. Results
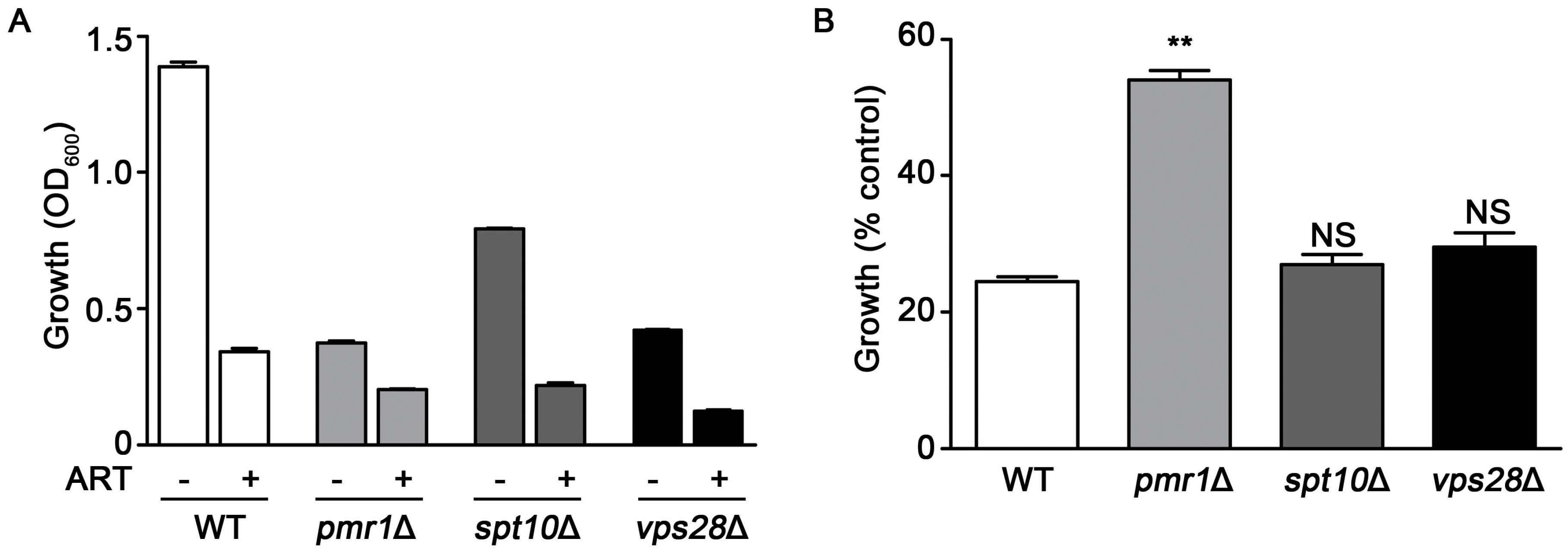
2.1. Yeast Cells Lacking Pmr1p Display Resistance to Artemisinin and Its Derivatives
2.2. The Slower Growth Rate of pmr1∆ Cells Does Not Appear Sufficient to Reduce Artemisinin Sensitivity
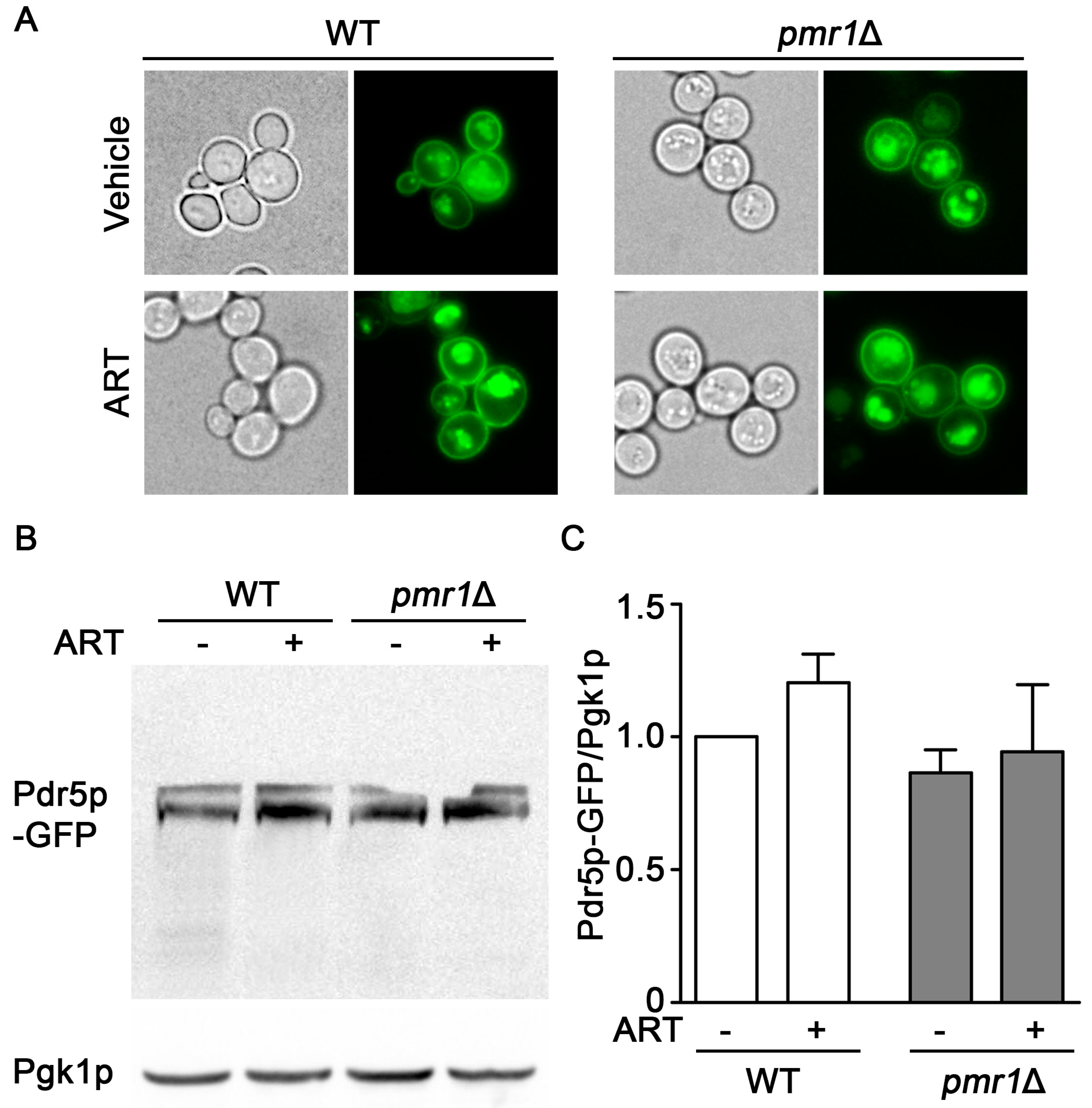
2.3. Localization and Accumulation of the Drug Efflux Transporter pdr5p Is Not Altered in pmr1∆ Cells
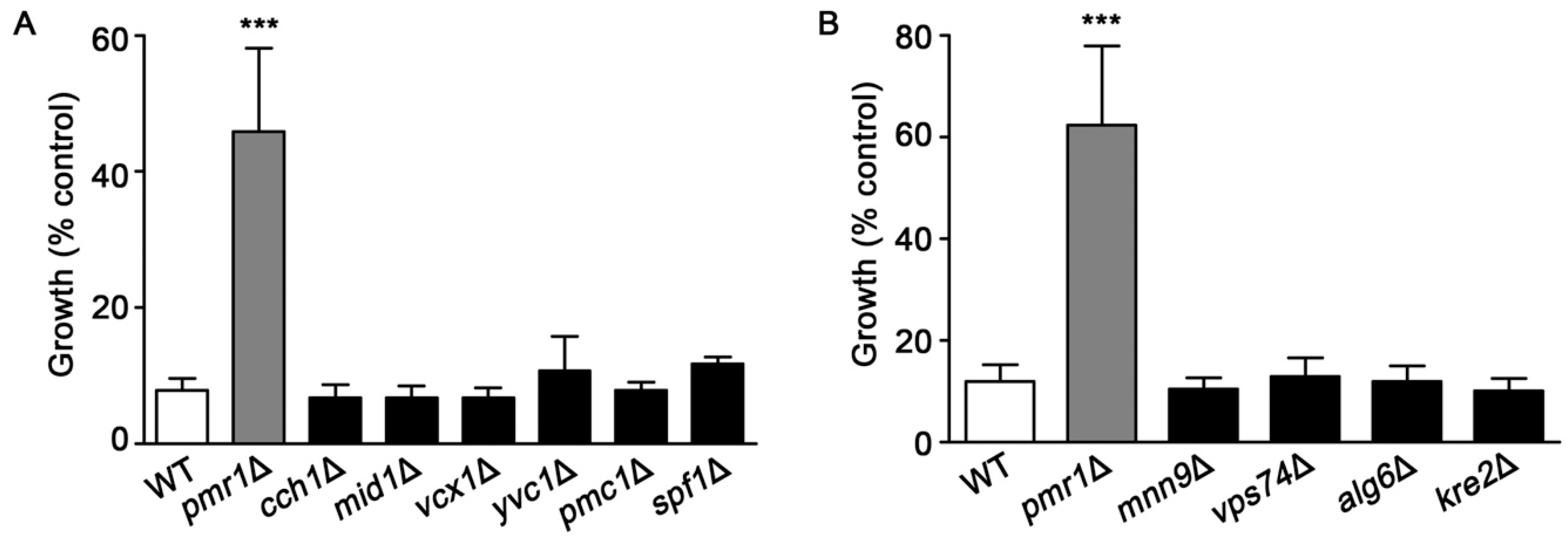
2.4. Deletions of Genes Encoding Proteins Involved in Ca2+ homeostasis or Protein Glycosylation Does Not Promote Artemisinin Resistance
2.5. Increased Intracellular Oxidation Is Not Observed in pmr1∆ Yeast Following Artemisinin Exposure
2.6. Elevated Intracellular Mn Is Not Protective against Artemisinin Toxicity
3. Discussion
4. Materials and Methods
4.1. Yeast Strains, Plasmid, and Culture Conditions
4.2. Fluorescence Microscopy
4.3. Measurement of Intracellular ROS Levels
4.4. Immunoblots and Protein Carbonylation Analysis
4.5. Measurement of Intracellular Manganese
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Klayman, D.L. Qinghaosu (artemisinin): An antimalarial drug from China. Science 1985, 228, 1049–1055. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, H.; Olliaro, P. Artemisinin derivatives for treating uncomplicated malaria. Cochrane Database Syst. Rev. 1999. [Google Scholar] [CrossRef]
- Cui, L.; Su, X.Z. Discovery, mechanisms of action and combination therapy of artemisinin. Expert Rev. Anti Infect. Ther. 2009, 7, 999–1013. [Google Scholar] [CrossRef] [Green Version]
- White, N.J. Artemisinin: Current status. Trans. R. Soc. Trop. Med. Hyg. 1994, 88 (Suppl. 1), S3–S4. [Google Scholar] [CrossRef]
- Hien, T.T.; White, N.J. Qinghaosu. Lancet 1993, 341, 603–608. [Google Scholar] [CrossRef]
- De Vries, P.J.; Dien, T.K. Clinical pharmacology and therapeutic potential of artemisinin and its derivatives in the treatment of malaria. Drugs 1996, 52, 818–836. [Google Scholar] [CrossRef]
- Li, G.Q.; Guo, X.B.; Fu, L.C.; Jian, H.X.; Wang, X.H. Clinical trials of artemisinin and its derivatives in the treatment of malaria in China. Trans. R. Soc. Trop. Med. Hyg. 1994, 88 (Suppl. 1), S5–S6. [Google Scholar] [CrossRef]
- Krishna, S.; Uhlemann, A.C.; Haynes, R.K. Artemisinins: Mechanisms of action and potential for resistance. Drug Resist. Updat. 2004, 7, 233–244. [Google Scholar] [CrossRef] [PubMed]
- White, N.J.; Olliaro, P.L. Strategies for the prevention of antimalarial drug resistance: Rationale for combination chemotherapy for malaria. Parasitol. Today 1996, 12, 399–401. [Google Scholar] [CrossRef]
- Nosten, F.; White, N.J. Artemisinin-based combination treatment of falciparum malaria. Am. J. Trop. Med. Hyg. 2007, 77 (Suppl. 6), 181–192. [Google Scholar] [CrossRef] [PubMed]
- Kremsner, P.G.; Krishna, S. Antimalarial combinations. Lancet 2004, 364, 285–294. [Google Scholar] [CrossRef]
- Yang, H.; Liu, D.; Yang, Y.; Fan, B.; Yang, P.; Li, X.; Li, C.; Dong, Y.; Yang, C. Changes in susceptibility of Plasmodium falciparum to artesunate in vitro in Yunnan Province, China. Trans. R. Soc. Trop. Med. Hyg. 2003, 97, 226–228. [Google Scholar] [PubMed]
- Jambou, R.; Legrand, E.; Niang, M.; Khim, N.; Lim, P.; Volney, B.; Ekala, M.T.; Bouchier, C.; Esterre, P.; Fandeur, T.; et al. Resistance of Plasmodium falciparum field isolates to in-vitro artemether and point mutations of the SERCA-type PfATPase6. Lancet 2005, 366, 1960–1963. [Google Scholar] [CrossRef]
- Huong, N.M.; Hewitt, S.; Davis, T.M.; Dao, L.D.; Toan, T.Q.; Kim, T.B.; Hanh, N.T.; Phuong, V.N.; Nhan, D.H.; Cong, L.D. Resistance of Plasmodium falciparum to antimalarial drugs in a highly endemic area of southern Viet Nam: A study in vivo and in vitro. Trans. R. Soc. Trop. Med. Hyg. 2001, 95, 325–329. [Google Scholar] [CrossRef]
- Noedl, H.; Se, Y.; Schaecher, K.; Smith, B.L.; Socheat, D.; Fukuda, M.M.; Artemisinin Resistance in Cambodia 1 (ARC1) Study Consortium. Evidence of artemisinin-resistant malaria in western Cambodia. N. Engl. J. Med. 2008, 359, 2619–2620. [Google Scholar] [CrossRef] [PubMed]
- Dondorp, A.M.; Nosten, F.; Yi, P.; Das, D.; Phyo, A.P.; Tarning, J.; Lwin, K.M.; Ariey, F.; Hanpithakpong, W.; Lee, S.J.; et al. Artemisinin resistance in Plasmodium falciparum malaria. N. Engl. J. Med. 2009, 361, 455–467. [Google Scholar] [CrossRef]
- O’Neill, P.M.; Posner, G.H. A medicinal chemistry perspective on artemisinin and related endoperoxides. J. Med. Chem. 2004, 47, 2945–2964. [Google Scholar] [CrossRef]
- Mercer, A.E. The role of bioactivation in the pharmacology and toxicology of the artemisinin-based antimalarials. Curr. Opin. Drug Discov. Dev. 2009, 12, 125–132. [Google Scholar]
- Eckstein-Ludwig, U.; Webb, R.J.; Van Goethem, I.D.; East, J.M.; Lee, A.G.; Kimura, M.; O’Neill, P.M.; Bray, P.G.; Ward, S.A.; Krishna, S. Artemisinins target the SERCA of Plasmodium falciparum. Nature 2003, 424, 957–961. [Google Scholar] [CrossRef]
- Mishina, Y.V.; Krishna, S.; Haynes, R.K.; Meade, J.C. Artemisinins inhibit Trypanosoma cruzi and Trypanosoma brucei rhodesiense in vitro growth. Antimicrob. Agents Chemother. 2007, 51, 1852–1854. [Google Scholar] [CrossRef]
- Cardi, D.; Pozza, A.; Arnou, B.; Marchal, E.; Clausen, J.D.; Andersen, J.P.; Krishna, S.; Moller, J.V.; le Maire, M.; Jaxel, C. Purified E255L mutant SERCA1a and purified PfATP6 are sensitive to SERCA-type inhibitors but insensitive to artemisinins. J. Biol. Chem. 2010, 285, 26406–26416. [Google Scholar] [CrossRef]
- Jambou, R.; Martinelli, A.; Pinto, J.; Gribaldo, S.; Legrand, E.; Niang, M.; Kim, N.; Pharath, L.; Volnay, B.; Ekala, M.T.; et al. Geographic Structuring of the Plasmodium falciparum Sarco(endo)plasmic Reticulum Ca2+ ATPase (PfSERCA) Gene Diversity. PLoS ONE 2010, 5, e9424. [Google Scholar] [CrossRef]
- Menacho-Marquez, M.; Murguia, J.R. Yeast on drugs: Saccharomyces cerevisiae as a tool for anticancer drug research. Clin. Transl. Oncol. 2007, 9, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Sturgeon, C.M.; Kemmer, D.; Anderson, H.J.; Roberge, M. Yeast as a tool to uncover the cellular targets of drugs. Biotechnol. J. 2006, 1, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Hughes, T.R. Yeast and drug discovery. Funct. Integr. Genom. 2002, 2, 199–211. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Mo, W.; Shen, D.; Sun, L.; Wang, J.; Lu, S.; Gitschier, J.M.; Zhou, B. Yeast model uncovers dual roles of mitochondria in action of artemisinin. PLoS Genet. 2005, 1, e36. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, K.W.; Fink, G.R. Ca2+ transport in Saccharomyces cerevisiae. J. Exp. Biol. 1994, 196, 157. [Google Scholar]
- Moore, C.M.; Hoey, E.M.; Trudgett, A.; Timson, D.J. Artemisinins act through at least two targets in a yeast model. Fems Yeast Res. 2011, 11, 233–237. [Google Scholar] [CrossRef] [PubMed]
- Pulcini, S.; Staines, H.M.; Pittman, J.K.; Slavic, K.; Doerig, C.; Halbert, J.; Tewari, R.; Shah, F.; Avery, M.A.; Haynes, R.K.; et al. Expression in yeast links field polymorphisms in PfATP6 to in vitro artemisinin resistance and identifies new inhibitor classes. J. Infect. Dis. 2013, 208, 468–478. [Google Scholar] [CrossRef]
- Brown, M.R.W.; Williams, P. Influence of substrate limitation and growth phase on sensitivity to antimicrobial agents. J. Antimicrob. Chemother. 1985, 15 (Suppl. A), 7–14. [Google Scholar] [CrossRef]
- Natsoulis, G.; Dollard, C.; Winston, F.; Boeke, J.D. The products of the SPT10 and SPT21 genes of Saccharomyces cerevisiae increase the amplitude of transcriptional regulation at a large number of unlinked loci. New Biol. 1991, 3, 1249–1259. [Google Scholar] [PubMed]
- Rothman, J.H.; Howald, I.; Stevens, T.H. Characterization of genes required for protein sorting and vacuolar function in the yeast Saccharomyces cerevisiae. Embo J. 1989, 8, 2057–2065. [Google Scholar] [CrossRef] [PubMed]
- Kolaczkowski, M.; van der Rest, M.; Cybularz-Kolaczkowska, A.; Soumillion, J.P.; Konings, W.N.; Goffeau, A. Anticancer drugs, ionophoric peptides, and steroids as substrates of the yeast multidrug transporter Pdr5p. J. Biol. Chem. 1996, 271, 31543–31548. [Google Scholar] [CrossRef]
- Alenquer, M.; Tenreiro, S.; Sa-Correia, I. Adaptive response to the antimalarial drug artesunate in yeast involves Pdr1p/Pdr3p-mediated transcriptional activation of the resistance determinants TPO1 and PDR5. Fems Yeast Res. 2006, 6, 1130–1139. [Google Scholar] [CrossRef] [Green Version]
- Egner, R.; Mahe, Y.; Pandjaitan, R.; Kuchler, K. Endocytosis and vacuolar degradation of the plasma membrane-localized Pdr5 ATP-binding cassette multidrug transporter in Saccharomyces cerevisiae. Mol. Cell Biol. 1995, 15, 5879–5887. [Google Scholar] [CrossRef]
- Rudolph, H.K.; Antebi, A.; Fink, G.R.; Buckley, C.M.; Dorman, T.E.; LeVitre, J.; Davidow, L.S.; Mao, J.I.; Moir, D.T. The yeast secretory pathway is perturbed by mutations in PMR1, a member of a Ca2+ ATPase family. Cell 1989, 58, 133–145. [Google Scholar] [CrossRef]
- Dürr, G.; Strayle, J.; Plemper, R.; Elbs, S.; Klee, S.K.; Catty, P.; Wolf, D.H.; Rudolph, H.K. The medial-Golgi Ion Pump Pmr1 Supplies the Yeast Secretory Pathway with Ca(2+) and Mn(2+) Required for Glycosylation, Sorting, and Endoplasmic Reticulum-Associated Protein Degradation. Mol. Biol. Cell 1998, 9, 1149–1162. [Google Scholar] [CrossRef] [PubMed]
- Antebi, A.; Fink, G.R. The yeast Ca(2+)-ATPase homologue, PMR1, is required for normal Golgi function and localizes in a novel Golgi-like distribution. Mol. Biol. Cell 1992, 3, 633–654. [Google Scholar] [CrossRef]
- Sorin, A.; Rosas, G.; Rao, R. PMR1, a Ca2+-ATPase in yeast Golgi, has properties distinct from sarco/endoplasmic reticulum and plasma membrane calcium pumps. J. Biol. Chem. 1997, 272, 9895–9901. [Google Scholar] [CrossRef] [PubMed]
- Iida, H.; Nakamura, H.; Ono, T.; Okumura, M.S.; Anraku, Y. MID1, a novel Saccharomyces cerevisiae gene encoding a plasma membrane protein, is required for Ca2+ influx and mating. Mol. Cell Biol. 1994, 14, 8259–8271. [Google Scholar] [CrossRef] [PubMed]
- Fischer, M.; Schnell, N.; Chattaway, J.; Davies, P.; Dixon, G.; Sanders, D. The Saccharomyces cerevisiae CCH1 gene is involved in calcium influx and mating. Febs Lett. 1997, 419, 259–262. [Google Scholar] [CrossRef] [Green Version]
- Pozos, T.C.; Sekler, I.; Cyert, M.S. The product of HUM1, a novel yeast gene, is required for vacuolar Ca2+/H+ exchange and is related to mammalian Na+/Ca2+ exchangers. Mol. Cell. Biol. 1996, 16, 3730–3741. [Google Scholar] [CrossRef]
- Cunningham, K.W.; Fink, G.R. Calcineurin inhibits VCX1-dependent H+/Ca2+ exchange and induces Ca2+ ATPases in Saccharomyces cerevisiae. Mol. Cell. Biol. 1996, 16, 2226–2237. [Google Scholar] [CrossRef] [PubMed]
- Denis, V.; Cyert, M.S. Internal Ca(2+) release in yeast is triggered by hypertonic shock and mediated by a TRP channel homologue. J. Cell Biol. 2002, 156, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Palmer, C.P.; Zhou, X.L.; Lin, J.; Loukin, S.H.; Kung, C.; Saimi, Y. A TRP homolog in Saccharomyces cerevisiae forms an intracellular Ca(2+)-permeable channel in the yeast vacuolar membrane. Proc. Natl. Acad. Sci. USA 2001, 98, 7801–7805. [Google Scholar] [CrossRef] [PubMed]
- Cronin, S.R.; Rao, R.; Hampton, R.Y. Cod1p/Spf1p is a P-type ATPase involved in ER function and Ca2+ homeostasis. J. Cell Biol. 2002, 157, 1017–1028. [Google Scholar] [CrossRef] [Green Version]
- Häusler, A.; Ballou, L.; Ballou, C.E.; Robbins, P.W. Yeast glycoprotein biosynthesis: MNT1 encodes an alpha-1,2-mannosyltransferase involved in O-glycosylation. Proc. Natl. Acad. Sci. USA 1992, 89, 6846–6850. [Google Scholar] [CrossRef] [PubMed]
- Corbacho, I.; Olivero, I.; Hernández, L.M. Identification of the MNN3 gene of Saccharomyces cerevisiae. Glycobiology 2010, 20, 1336–1340. [Google Scholar] [CrossRef] [Green Version]
- Huffaker, T.C.; Robbins, P.W. Yeast mutants deficient in protein glycosylation. Proc. Natl. Acad. Sci. USA 1983, 80, 7466–7470. [Google Scholar] [CrossRef] [PubMed]
- Hill, K.; Boone, C.; Goebl, M.; Puccia, R.; Sdicu, A.M.; Bussey, H. Yeast Kre2 Defines a New Gene Family Encoding Probable Secretory Proteins, and Is Required for the Correct N-Glycosylation of Proteins. Genetics 1992, 130, 273–283. [Google Scholar] [PubMed]
- Reiss, G.; Heesen, S. t.; Zimmerman, J.; Robbins, P.; Aebi, M. Isolation of the ALG6 locus of Saccharomyces cerevisiae required for glucosylation in the N-linked glycosylation pathway. Glycobiology 1996, 6, 493–498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haynes, R.K.; Chan, W.C.; Lung, C.M.; Uhlemann, A.C.; Eckstein, U.; Taramelli, D.; Parapini, S.; Monti, D.; Krishna, S. The Fe2+-mediated decomposition, PfATP6 binding, and antimalarial activities of artemisone and other artemisinins: The unlikelihood of C-centered radicals as bioactive intermediates. Chem. Med. Chem. 2007, 2, 1480–1497. [Google Scholar] [CrossRef] [PubMed]
- Lapinskas, P.J.; Cunningham, K.W.; Liu, X.F.; Fink, G.R.; Culotta, V.C. Mutations in PMR1 suppress oxidative damage in yeast cells lacking superoxide dismutase. Mol. Cell. Biol. 1995, 15, 1382–1388. [Google Scholar] [CrossRef]
- Archibald, F.S.; Fridovich, I. Manganese and defenses against oxygen toxicity in Lactobacillus plantarum. J. Bacteriol. 1981, 145, 442–451. [Google Scholar]
- Archibald, F.S.; Fridovich, I. The scavenging of superoxide radical by manganous complexes: In vitro. Arch. Biochem. Biophys. 1982, 214, 452–463. [Google Scholar] [CrossRef]
- Reddi, A.R.; Jensen, L.T.; Naranuntarat, A.; Rosenfeld, L.; Leung, E.; Shah, R.; Culotta, V.C. The overlapping roles of manganese and Cu/Zn SOD in oxidative stress protection. Free Radic. Biol. Med. 2009, 46, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Devasahayam, G.; Ritz, D.; Helliwell, S.B.; Burke, D.J.; Sturgill, T.W. Pmr1, a Golgi Ca2+/Mn2+-ATPase, is a regulator of the target of rapamycin (TOR) signaling pathway in yeast. Proc. Natl. Acad. Sci. USA 2006, 103, 17840–17845. [Google Scholar] [CrossRef] [Green Version]
- Jensen, A.N.; Chindaudomsate, W.; Thitiananpakorn, K.; Mongkolsuk, S.; Jensen, L.T. Improper protein trafficking contributes to artemisinin sensitivity in cells lacking the KDAC Rpd3p. Febs Lett. 2014, 588, 4018–4025. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wen, L.; Liang, C.; Chen, E.; Chen, W.; Liang, F.; Zhi, X.; Wei, T.; Xue, F.; Li, G.; Yang, Q.; et al. Regulation of Multi-drug Resistance in hepatocellular carcinoma cells is TRPC6/Calcium Dependent. Sci. Rep. 2016, 6, 23269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Locke, E.G.; Bonilla, M.; Liang, L.; Takita, Y.; Cunningham, K.W. A homolog of voltage-gated Ca(2+) channels stimulated by depletion of secretory Ca(2+) in yeast. Mol. Cell Biol. 2000, 20, 6686–6694. [Google Scholar] [CrossRef]
- Cunningham, K.W.; Fink, G.R. Calcineurin-dependent growth control in Saccharomyces cerevisiae mutants lacking PMC1, a homolog of plasma membrane Ca2+ ATPases. J. Cell Biol. 1994, 124, 351–363. [Google Scholar] [CrossRef] [Green Version]
- Meshnick, S.R.; Yang, Y.Z.; Lima, V.; Kuypers, F.; Kamchonwongpaisan, S.; Yuthavong, Y. Iron-dependent free radical generation from the antimalarial agent artemisinin (qinghaosu). Antimicrob. Agents Chemother. 1993, 37, 1108–1114. [Google Scholar] [CrossRef] [PubMed]
- Asawamahasakda, W.; Ittarat, I.; Pu, Y.M.; Ziffer, H.; Meshnick, S.R. Reaction of antimalarial endoperoxides with specific parasite proteins. Antimicrob. Agents Chemother. 1994, 38, 1854–1858. [Google Scholar] [CrossRef]
- Berman, P.A.; Adams, P.A. Artemisinin enhances heme-catalysed oxidation of lipid membranes. Free Radic. Biol. Med. 1997, 22, 1283–1288. [Google Scholar] [CrossRef]
- Gorlach, A.; Bertram, K.; Hudecova, S.; Krizanova, O. Calcium and ROS: A mutual interplay. Redox Biol. 2015, 6, 260–271. [Google Scholar] [CrossRef] [Green Version]
- Cho, Y.S.; Kwon, H.J. Identification and validation of bioactive small molecule target through phenotypic screening. Bioorg. Med. Chem. 2012, 20, 1922–1928. [Google Scholar] [CrossRef]
- Luk, E.E.-C.; Culotta, V.C. Manganese Superoxide Dismutase in Saccharomyces cerevisiae Acquires Its Metal Co-factor through a Pathway Involving the Nramp Metal Transporter, Smf2p. J. Biol. Chem. 2001, 276, 47556–47562. [Google Scholar] [CrossRef] [Green Version]
- Jamieson, D.J. Saccharomyces cerevisiae has distinct adaptive responses to both hydrogen peroxide and menadione. J. Bacteriol. 1992, 174, 6678–6681. [Google Scholar] [CrossRef] [PubMed]
- Collinson, L.P.; Dawes, I.W. Inducibility of the response of yeast cells to peroxide stress. J. Gen. Microbiol. 1992, 138, 329–335. [Google Scholar] [CrossRef] [Green Version]
- Marini, M.; Frabetti, F.; Musiani, D.; Franceschi, C. Oxygen radicals induce stress proteins and tolerance to oxidative stress in human lymphocytes. Int. J. Radiat. Biol. 1996, 70, 337–350. [Google Scholar] [CrossRef] [PubMed]
- Gietz, R.D.; Schiestl, R.H. Applications of high efficiency lithium acetate transformation of intact yeast cells using single-stranded nucleic acids as carrier. Yeast 1991, 7, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Jensen, L.T.; Carroll, M.C.; Hall, M.D.; Harvey, C.J.; Beese, S.E.; Culotta, V.C. Down-regulation of a manganese transporter in the face of metal toxicity. Mol. Biol. Cell 2009, 20, 2810–2819. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.Y.; Bird, A.J.; Winge, D.R.; Eide, D.J. Regulation of the Yeast TSA1 Peroxiredoxin by ZAP1 Is an Adaptive Response to the Oxidative Stress of Zinc Deficiency. J. Biol. Chem. 2006, 282, 2184–2195. [Google Scholar] [CrossRef] [Green Version]
- James, J.; Fiji, N.; Roy, D.; Andrew MG, D.; Shihabudeen, M.S.; Chattopadhyay, D.; Thirumurugan, K. A rapid method to assess reactive oxygen species in yeast using H2DCF-DA. Anal. Methods 2015, 7, 8572–8575. [Google Scholar] [CrossRef]
- Levine, R.L.; Garland, D.; Oliver, C.N.; Amici, A.; Climent, I.; Lenz, A.; Ahn, B.; Shaltiel, S.; Stadtman, E.R. Determination of carbonyl content in oxidatively modified proteins. In Methods in Enzymology; Packer, L., Glazer, A.N., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 1990; Volume 186, pp. 464–478. [Google Scholar]
- Kanprasoet, W.; Jensen, L.T.; Sriprach, S.; Thitiananpakorn, K.; Rattanapornsompong, K.; Jensen, A.N. Deletion of Mitochondrial Porin Alleviates Stress Sensitivity in the Yeast Model of Shwachman-Diamond Syndrome. J. Genet. Genom. 2015, 42, 671–684. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Sample Availability: Samples of artemisinin and derivatives are available from commercial chemical suppliers. |




© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pongwattanakewin, O.; Phyu, T.; Suesattayapirom, S.; Jensen, L.T.; Jensen, A.N. Possible Role of the Ca2+/Mn2+ P-Type ATPase Pmr1p on Artemisinin Toxicity through an Induction of Intracellular Oxidative Stress. Molecules 2019, 24, 1233. https://doi.org/10.3390/molecules24071233
Pongwattanakewin O, Phyu T, Suesattayapirom S, Jensen LT, Jensen AN. Possible Role of the Ca2+/Mn2+ P-Type ATPase Pmr1p on Artemisinin Toxicity through an Induction of Intracellular Oxidative Stress. Molecules. 2019; 24(7):1233. https://doi.org/10.3390/molecules24071233
Chicago/Turabian StylePongwattanakewin, Onnicha, The Phyu, Suchanya Suesattayapirom, Laran T. Jensen, and Amornrat N. Jensen. 2019. "Possible Role of the Ca2+/Mn2+ P-Type ATPase Pmr1p on Artemisinin Toxicity through an Induction of Intracellular Oxidative Stress" Molecules 24, no. 7: 1233. https://doi.org/10.3390/molecules24071233





