3-Hydroxyflavones and 3-Hydroxy-4-oxoquinolines as Carbon Monoxide-Releasing Molecules
Abstract
:1. Introduction
2. CO Production from 3-Hydroxyflavones in Enzyme-Catalyzed Reactions and Model Systems
2.1. Fungal Flavonol Dioxygenases
2.2. Bacterial Flavonol Dioxygenases
2.3. Model Systems for Cu(II)-Containing Fungal QDOs
2.4. Model Systems for Bacterial QDOs
2.5. CO-Release Reactivity of Other Metal Flavonolato Complexes
2.6. Summary
3. CO Production from 3-Hydroxy-4-oxoquinolines via Cofactor-Free Enzyme-Catalyzed Reactions and in Metal-Containing Synthetic Systems
3.1. Enzyme-Catalyzed Reactions
3.2. Synthetic Systems
3.3. Summary
4. CO Production from 3-Hydroxyflavones and 3-Hydroxy-4-oxoquinolines via Base-Catalyzed and Non-Redox Metal Assisted Reactions
4.1. 3-Hydroxyflavones
4.2. 3-Hydroxy-4-oxoquinolines
4.3. Summary
5. CO Production from 3-Hydroxyflavones via Photochemical Reactions
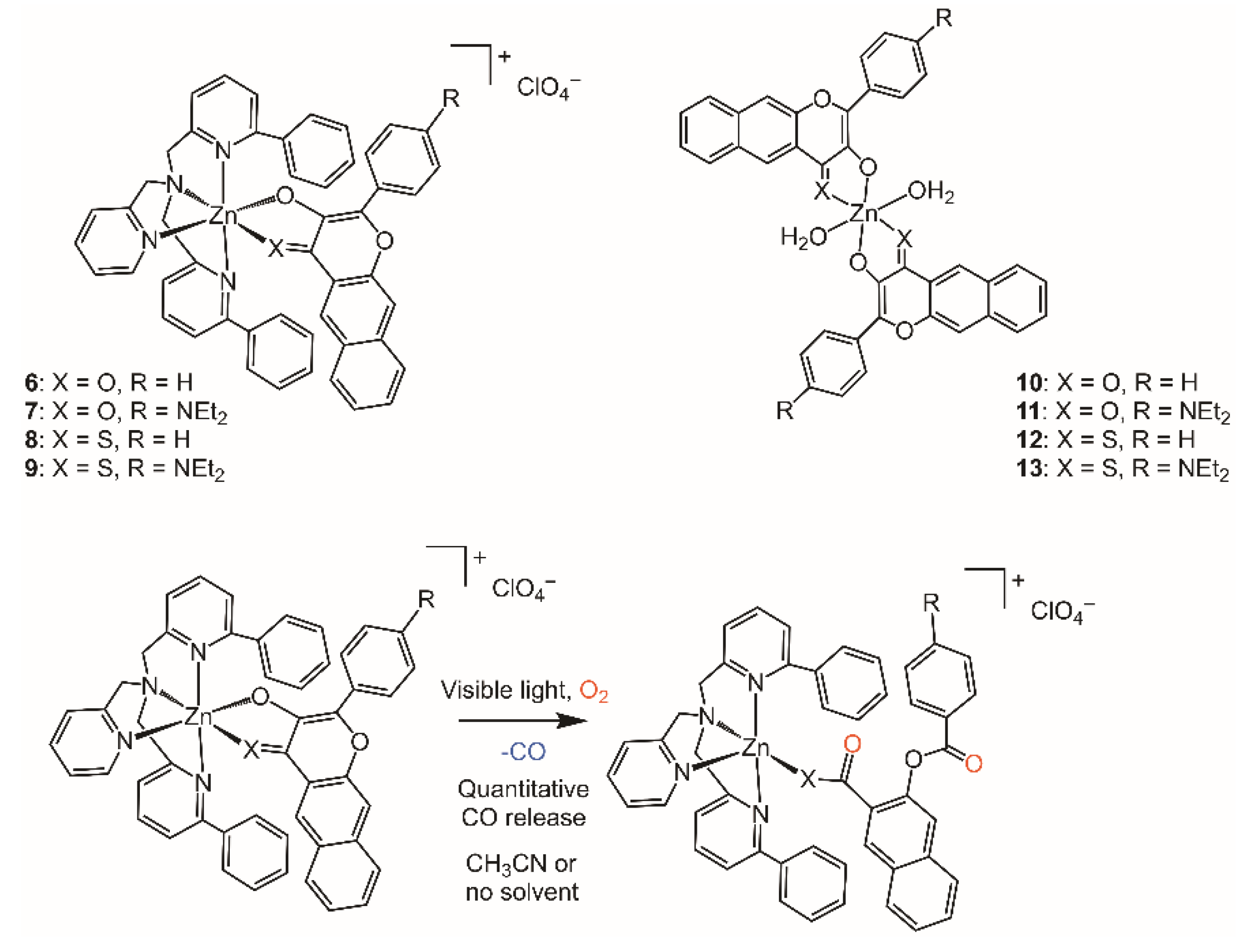
6. Visible Light-Induced CO Release from Extended 3-Hydroxyflavone and 3-Hydroxybenzo[g]quinolone Frameworks
7. Concluding Remarks and Future Directions
Funding
Conflicts of Interest
References
- Motterlini, R.; Foresti, R. Biological signaling by carbon monoxide and carbon monoxide-releasing molecules. Am. J. Physiol. Cell Physiol. 2017, 312, C302–C313. [Google Scholar] [CrossRef] [PubMed]
- Motterlini, R.; Otterbein, L.E. The therapeutic potential of carbon monoxide. Nat. Rev. Drug Discov. 2010, 9, 728–743. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-H.; Choi, S. Therapeutic aspects of carbon monoxide in cardiovascular disease. Int. J. Mol. Sci. 2018, 19, 2381. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.R.; Nilius, B.; Han, J. Gaseous Signaling Molecules in Cardiovascular Function: From Mechanism to Clinical Translation. In Reviews of Physiology, Biochemistry, and Pharmacology; Nilius, B., de Tombe, P., Gundermann, T., Jah, R., Lill, R., Peterson, O.H., Eds.; Springer International Publishing: Berlin/Heidelberg, Germany, 2018; Volume 174, pp. 81–156. [Google Scholar]
- Otterbein, L.E.; Foresti, R.; Motterlini, R. Heme oxygeanse-1 and carbon monoxide in the heart: The balancing act between danger signaling and pro-survival. Circ. Res. 2016, 118, 1940–1959. [Google Scholar] [CrossRef] [PubMed]
- Fredenburgh, L.E.; Merz, A.A.; Cheng, S. Haeme oxygenase signalling pathway: Implications for cardiovascular disease. Eur. Heart J. 2015, 36, 1512–1518. [Google Scholar] [CrossRef] [PubMed]
- Szabo, C. Gasotransmitters in cancer: From pathophysiology to experimental therapy. Nat. Rev. Drug Discov. 2016, 15, 185–203. [Google Scholar] [CrossRef] [PubMed]
- Jin, Q.; Deng, Y.; Jia, F.; Tang, Z.; Ji, J. Gas therapy: An emerging “green” strategy for anticancer therapeutics. Adv. Therap. 2018, 1, 1800084. [Google Scholar] [CrossRef]
- Motterlini, R. Carbon monoxide-releasing molecules (CO-RMs): Vasodilatory, anti-ischaemic and anti-inflammatory activities. Biochem. Soc. Trans. 2007, 35, 1142–1146. [Google Scholar] [CrossRef]
- Crook, S.H.; Mann, B.E.; Meijer, A.J.H.M.; Adams, H.; Sawle, P.; Scapens, D.; Motterlini, R. [Mo(CO)4{S2CNMe(CH2CO2H)}], a new water-soluble CO-releasing molecule. Dalton Trans. 2011, 40, 4230–4235. [Google Scholar] [CrossRef]
- Aki, T.; Unuma, K.; Noritake, K.; Kurahashi, H.; Funakoshi, T.; Uemura, K. Interaction of carbon monoxide-releasing ruthenium carbonyl CORM-3 with plasma fibronectin. Toxicol. In Vitro 2018, 50, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Chaves-Ferreira, M.; Albuquerque, I.S.; Matak-Vinkovic, D.; Coelho, A.C.; Carvalho, S.M.; Saraiva, L.M.; Romão, C.C.; Bernardes, G.J.L. Spontaneous CO release from RuII(CO)2-protein complexes in aqueous solution, cells, and mice. Angew. Chem. Int. Ed. 2015, 54, 1172–1175. [Google Scholar] [CrossRef] [PubMed]
- Seixas, J.D.; Santos, M.F.A.; Mukhopadhyay, A.; Coelho, A.C.; Reis, P.M.; Veiros, L.F.; Marques, A.R.; Penacho, N.; Gonçalves, A.M.; Romão, M.J.; et al. A contribution to the rational design of Ru(CO)3Cl2L complexes for in vivo delivery of CO. Dalton Trans. 2015, 44, 5058–5075. [Google Scholar] [CrossRef] [PubMed]
- Santos-Silva, T.; Mukhopadhyay, A.; Seixas, J.D.; Bernardes, G.J.L.; Romão, C.C.; Romão, M.J. Towards improved therapeutic CORMs: Understanding the reactivity of CORM-3 with proteins. Curr. Med. Chem. 2011, 18, 3361–3366. [Google Scholar] [CrossRef]
- Santos-Silva, T.; Mukhopadhyay, A.; Seixas, J.D.; Bernardes, G.J.L.; Romão, C.C.; Romão, M.J. CORM-3 reactivity toward proteins: The crystal structure of Ru(II) dicarbonyl-lysozyme complex. J. Am. Chem. Soc. 2011, 133, 1192–1195. [Google Scholar] [CrossRef]
- Wang, P.; Liu, H.; Zhao, Q.; Chen, Y.; Liu, B.; Zhang, B.; Zheng, Q. Syntheses and evaluation of drug-like properties of CO-releasing molecules containing ruthenium and group 6 metal. Eur. J. Med. Chem. 2014, 74, 199–215. [Google Scholar] [CrossRef]
- Davidge, K.S.; Motterlini, R.; Mann, B.E.; Wilson, J.L.; Poole, R.K. Carbon monoxide in biology and microbiology: Surprising roles for the “Detroit perfume”. Adv. Microb. Physiol. 2009, 56, 85–167. [Google Scholar]
- Southam, H.M.; Smith, T.W.; Lyon, R.L.; Liao, C.; Trevitt, C.R.; Middlemiss, L.A.; Cox, F.L.; Chapman, J.A.; El-Khamisy, S.F.; Hippler, M.; et al. A thiol-reactive Ru(II) ion, not CO release, underlies the potent antimicrobial and cytotoxic properties of CO-releasing molecule-3. Redox Biol. 2018, 18, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Wareham, L.K.; McLean, S.; Begg, R.; Rana, N.; Ali, S.; Kendall, J.J.; Sanguinetti, G.; Mann, B.E.; Poole, R.K. The broad-spectrum antimicrobial potential of [Mn(CO)4(S2CNCH3(CH2CO2H)], a water-soluble CO-releasing molecule (CORM-401): Intracellular accumulation, transcriptomic and statistical analyses, and membrane polarization. Antioxid. Redox Signal. 2018, 28, 1286–1308. [Google Scholar] [CrossRef] [PubMed]
- Abeyrathna, N.; Washington, K.; Bashur, K.; Liao, Y. Nonmetallic carbon monoxide releasing molecules. Org. Biomol. Chem. 2017, 15, 8692–8699. [Google Scholar] [CrossRef] [PubMed]
- Motterlini, R.; Sawle, P.; Hammad, J.; Bains, S.; Alberto, R.; Foresti, R.; Green, C.J. CORM-A1: A new pharmacologically active carbon monoxide-releasing molecule. FASEB J. 2005, 19, 284–286. [Google Scholar] [CrossRef]
- Ji, X.; Wang, B. Strategies toward carbon monoxide prodrugs. Acc. Chem. Res. 2018, 51, 1377–1385. [Google Scholar] [CrossRef]
- Peng, P.; Wang, C.; Shi, Z.; Johns, V.K.; Ma, L.; Oyer, J.; Copik, A.; Igarashi, R.; Liao, Y. Visible-light activatable organic CO-releasing molecules (PhotoCORMs) that simultaneously generate fluorophores. Org. Biomol. Chem. 2013, 11, 6671–6674. [Google Scholar] [CrossRef] [PubMed]
- Antony, L.A.P.; Slanina, T.; Sebej, P.; Solomek, T.; Klán, P. Fluorescein analogue xanthene-9-carboxylic acid: A transition-metal-free CO releasing molecule activated by green light. Org. Lett. 2013, 15, 4552–4555. [Google Scholar] [CrossRef] [PubMed]
- Palao, E.; Slanina, T.; Muchová, L.; Solomek, T.; Vitek, L.; Klán, P. Transition-metal-free CO-releasing BODIPY derivatives activatable by visible to NIR light as promising bioactive molecules. J. Am. Chem. Soc. 2016, 138, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Sjöstrand, T. The in vitro formation of carbon monoxide in blood. Acta Physiol. Scand. 1952, 24, 314–332. [Google Scholar] [CrossRef] [PubMed]
- Sjöstrand, T. Endogenous formation of carbon monoxide in man under normal and pathological conditions. Scan. J. Clin. Lab. Investig. 1949, 1, 201–214. [Google Scholar] [CrossRef]
- Elbirt, K.K.; Bonkovksy, H.L. Heme oxygenase: Recent advances in understanding its regulation and role. Proc. Assoc. Am. Physicians 1999, 111, 438–447. [Google Scholar] [CrossRef]
- Fetzner, S. Ring-cleaving dioxygenases with a cupin fold. Appl. Environ. Microbiol. 2012, 78, 2505–2514. [Google Scholar] [CrossRef] [PubMed]
- Fetzner, S.; Steiner, R.A. Cofactor-independent oxidases and oxygenases. Appl. Microbiol. Biotechnol. 2010, 86, 791–804. [Google Scholar] [CrossRef]
- Conrad, R.; Seiler, W. Role of microorganisms in the consumption and production of atmospheric carbon monoxide by soil. Appl. Environ. Microbiol. 1980, 40, 437–445. [Google Scholar] [PubMed]
- Patel, R.V.; Mistry, B.M.; Shinde, S.K.; Syed, R.; Singh, V.; Shin, H.S. Therapeutic potential of quercetin as a cardiovascular agent. Eur. J. Med. Chem. 2018, 155, 889–904. [Google Scholar] [CrossRef]
- Rauf, A.; Imran, M.; Khan, I.A.; Ur-Rehman, M.; Gilani, S.A.; Mehmood, Z.; Mubarak, M.S. Anticancer potential of quercetin: A comprehensive review. Phytother. Res. 2018, 32, 2109–2130. [Google Scholar] [CrossRef]
- Sharma, A.; Kashyap, D.; Sak, K.; Tuli, H.S.; Sharma, A.K. Therapeutic charm of quercetin and its derivatives: A review of research and patents. Pharm. Pat. Anal. 2018, 7, 15–32. [Google Scholar] [CrossRef]
- Steiger, C.; Hermann, C.; Meinel, L. Localized delivery of carbon monoxide. Eur. J. Pharm. Biopharm. 2017, 118, 3–12. [Google Scholar] [CrossRef]
- Fusetti, F.; Schröter, K.H.; Steiner, R.A.; van Noort, P.I.; Pijning, T.; Rozeboom, H.J.; Kalk, K.H.; Egmond, M.R.; Dijkstra, B.W. Crystal structure of the copper-containing quercetin 2,3-dioxygenase from Aspergillus japonicus. Structure 2002, 10, 259–268. [Google Scholar] [CrossRef]
- Steiner, R.A.; Meyer-Klaucke, W.; Dijkstra, B.W. Functional analysis of the copper-dependent quercetin 2,3-dioxygenase. 2. X-ray absorption studies of native enzyme and anaerobic complexes with the substrates quercetin and myricetin. Biochemistry 2002, 41, 7963–7968. [Google Scholar] [CrossRef] [PubMed]
- Steiner, R.A.; Kalk, K.H.; Dijkstra, B.W. Anaerobic enzyme-substrate structures provide insight into the reaction mechanism of the copper-dependent quercetin 2,3-dioxygenase. Proc. Natl. Acad. Sci. USA 2002, 99, 16625–16630. [Google Scholar] [CrossRef] [PubMed]
- Siegbahn, P.E.M. Hybrid DFT study of the mechanism of quercetin 2,3-dioxygenase. Inorg. Chem. 2004, 43, 5944–5953. [Google Scholar] [CrossRef] [PubMed]
- Fiorucci, S.; Golebiowski, J.; Carbol-Bass, D.; Antonczak, S. Oxygenolysis of flavonoid compounds: DFT description of the mechanism for quercetin. ChemPhysChem 2004, 5, 1726–1733. [Google Scholar] [CrossRef] [PubMed]
- Oka, T.; Simpson, F.J.; Krishnamurty, H.G. Degradation of rutin by Aspergillus flavus. Studies on specificity, inhibition, and possible reaction mechanism of quercetinase. Can. J. Microbiol. 1972, 18, 493–508. [Google Scholar] [CrossRef]
- Fiorucci, S.; Golebiowski, J.; Carbrol-Bass, D.; Antonczak, S. Molecular simulations bring new insights into flavonoid/quercetinase interaction modes. Proteins 2007, 67, 961–970. [Google Scholar] [CrossRef] [PubMed]
- Barney, B.M.; Schaab, M.R.; LoBrutto, R.; Francisco, W.A. Evidence for a new metal in a known active site: Purification and characterization of an iron-containing quercetin 2,3-dioxygenase from Bacillus subtilis. Protein Expr. Purif. 2004, 35, 131–141. [Google Scholar] [CrossRef]
- Bowater, L.; Fairhurst, S.A.; Just, V.J.; Bornemann, S. Bacillus subtilis YxaG is a novel Fe-containing quercetin 2,3-dioxygenase. FEBS Lett. 2004, 557, 45–48. [Google Scholar] [CrossRef]
- Gopal, B.; Madan, L.L.; Betz, S.F.; Kossiakoff, A.A. The crystal structure of a quercetin 2,3-dioxygenase from Bacillus subtilis suggests modulation of enzyme activity by a change in the metal ion at the active site(s). Biochemistry 2005, 44, 193–201. [Google Scholar] [CrossRef]
- Schaab, M.R.; Barney, B.M.; Francisco, W.A. Kinetic and spectroscopic studies on the quercetin 2,3-dioxygenase from Bacillus subtilis. Biochemistry 2006, 45, 1009–1016. [Google Scholar] [CrossRef]
- Merkens, H.; Sielker, S.; Rose, K.; Fetzner, S. A new monocupin quercetinase of Streptomyces sp. FLA: Identification and heterologous expression of the queD gene and activity of the recombinant enzyme towards different flavonols. Arch. Microbiol. 2007, 187, 475–487. [Google Scholar] [CrossRef]
- Merkens, H.; Kappl, R.; Jakob, R.P.; Schmid, F.X.; Fetzner, S. Quercetinase QueD of Streptomyces sp. FLA, a monocupin dioxygenase with a preference for nickel and cobalt. Biochemistry 2008, 47, 12185–12196. [Google Scholar] [CrossRef]
- Nianios, D.; Thierbach, S.; Steimer, L.; Lulchev, P.; Klostermeier, D.; Fetzner, S. Nickel quercetinase, a “promiscuous” metalloenzyme: Metal incorporation and metal ligand substitution studies. BMC Biochem. 2015, 16, 10. [Google Scholar] [CrossRef] [PubMed]
- Jeoung, J.H.; Nianios, D.; Fetzner, S.; Dobbek, H. Quercetin 2,4-dioxygenase activates dioxygen in a side-on O2-Ni Complex. Angew. Chem. Int. Ed. Engl. 2016, 55, 3281–3284. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wang, X.; Tian, G.; Liu, Y. Insights into the dioxygen activation and catalytic mechanism of the nickel-containing quercetinase. Catal. Sci. Technol. 2018, 8, 2340–2351. [Google Scholar] [CrossRef]
- Wang, W.-J.; Wei, W.-J.; Liao, R.-Z. Deciphering the chemoselectivity of nickel-dependent quercetin 2,4-dioxygenase. Phys. Chem. Chem. Phys. 2018, 20, 15784–15794. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.R.; Zapata, A.; Ramirez, A.J.; Bowen, S.K.; Francisco, W.A.; Farmer, P.J. Nitrosyl hydride (HNO) replaces dioxygen in nitroxygenase activity of manganese quercetin dioxygenase. Proc. Natl. Acad. Sci. USA 2011, 108, 18926–18931. [Google Scholar] [CrossRef] [Green Version]
- Shafirovich, V.; Lymar, S.V. Spin-forbidden deprotonation of aqueous nitroxyl (HNO). J. Am. Chem. Soc. 2003, 125, 6547–6552. [Google Scholar] [CrossRef] [PubMed]
- Wojdyla, Z.; Borowski, T. DFT study of the mechanism of manganese quercetin 2,3-dioxygenase: Quest for origins of enzyme unique nitroxygenase activity and regioselectivity. J. Biol. Inorg. Chem. 2016, 21, 475–489. [Google Scholar] [CrossRef]
- Kaizer, J.; Balogh-Hergovich, E.; Czaun, M.; Csay, T.; Speier, G. Redox and nonredox metal assisted model systems with relevance to flavonol and 3-hydroxyquinolin-4(1H)-one 2,4-dioxygenase. Coord. Chem. Rev. 2006, 250, 2222–2233. [Google Scholar] [CrossRef]
- Pap, J.S.; Kaizer, J.; Speier, G. Model systems for the CO-releasing flavonol 2,4-dioxygenase enzyme. Coord. Chem. Rev. 2010, 254, 781–793. [Google Scholar] [CrossRef]
- Kaizer, J.; Pap, J.S.; Speier, G. Copper Dioxygenases. In Copper-Oxygen Chemistry; Karlin, K.D., Itoh, S., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2011; pp. 23–52. [Google Scholar]
- Barhács, L.; Kaizer, J.; Pap, J.; Speier, G. Kinetics and mechanism of the stoichiometric oxygenation of [CuII(fla)(idpa)ClO4 [fla = flavonolate, idpa = 3,3’-imino-bis(N,N-dimethylpropylamine)] and the [CuII(fla)(idpa]ClO4-catalyzed oxygenation of flavonol. Inorg. Chim. Acta 2001, 320, 83–91. [Google Scholar] [CrossRef]
- Balogh-Hergovich, É.; Kaizer, J.; Speier, G. Carboxylate-enhanced reactivity in the oxygenation of copper flavonolate complexes. J. Mol. Catal. A Chem. 2003, 206, 83–87. [Google Scholar] [CrossRef]
- Balogh-Hergovich, É.; Kaizer, J.; Pap, J.; Speier, G.; Huttner, G.; Zsolnai, L. Copper-mediated oxygenolysis of flavonols via endoperoxide and dioxetan intermediates. Synthesis and oxygenation of [CuII(phen)2(fla)]ClO4 and [CuII(L)(fla)2] [flaH = flavonol; L = 1,10-phenanthroline (phen), 2,2’-bipyridine (bpy), N,N,N’,N’-tetramethylethylenediamine (tmeda)] complexes. Eur. J. Inorg. Chem. 2002, 2002, 2287–2295. [Google Scholar]
- Hiatt, R. Organic Peroxides; Swern, D., Ed.; Wiley Interscience: New York, NY, USA, 1971; Volume III, p. 70. [Google Scholar]
- Grubel, K.; Rudzka, K.; Arif, A.M.; Klotz, K.L.; Halfen, J.A.; Berreau, L.M. Synthesis, characterization, and ligand exchange reactivity of a series of first row divalent metal 3-hydroxyflavonolate complexes. Inorg. Chem. 2010, 49, 82–96. [Google Scholar] [CrossRef]
- Sun, Y.-J.; Huang, Q.-Q.; Tano, T.; Itoh, S. Flavonolate complexes of MII (M = Mn, Fe, Co, Ni, Cu, and Zn). Structural and functional models for the ES (enzyme–substrate) complex of quercetin 2,3-dioxygenase. Inorg. Chem. 2013, 52, 10936–10948. [Google Scholar] [CrossRef] [PubMed]
- Matuz, A.; Giorgi, M.; Speier, G.; Kaizer, J. Structural and functional comparison of manganese-, iron-, cobalt-, nickel-, and copper-containing biomimic quercetinase models. Polyhedron 2013, 63, 41–49. [Google Scholar] [CrossRef]
- Sun, Y.-J.; Huang, Q.-Q.; Zhang, J.-J. Series of structural and functional models for the ES (enzyme–substrate) complex of the Co(II)-containing quercetin 2,3-dioxygenase. Inorg. Chem. 2014, 53, 2932–2942. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.-J.; Huang, Q.-Q.; Zhang, J.-J. A series of NiII-flavonolate complexes as structural and functional ES (enzyme-substrate) models of the NiII-containing quercetin 2,3-dioxygenase. Dalton Trans. 2014, 43, 6480–6489. [Google Scholar] [CrossRef]
- Sun, Y.-J.; Huang, Q.-Q.; Zhang, J.-J. Set of Fe(II)-3-hydroxyflavonolate enzyme–substrate model complexes of atypically coordinated mononuclear non-heme Fe(II)-dependent quercetin 2,4-dioxygenase. ACS Omega 2017, 2, 5850–5860. [Google Scholar] [CrossRef]
- Kaizer, J.; Baráth, G.; Pap, J.; Speier, G.; Giorgi, M.; Réglier, M. Manganese and iron flavonolates as flavonol 2,4-dioxygenase mimics. Chem. Commun. 2007, 5235–5237. [Google Scholar] [CrossRef] [PubMed]
- Balogh-Hergovich, É; Kaizer, J.; Speier, G.; Argay, G.; Párkányi, L. Kinetic studies on the copper(II)-mediated oxygenolysis of the flavonolate ligand. Crystal structures of [Cu(fla)2] (fla = flavonolate) and [Cu(O-bs)2(py)3] (O-bs = O-benzoylsalicylate). J. Chem. Soc. Dalton Trans. 1999, 3847–3854. [Google Scholar] [CrossRef]
- Balogh-Hergovich, É.; Kaizer, J.; Speier, G.; Fülöp, V.; Párkányi, L. Quercetin 2,3-dioxygenase mimicking ring cleavage of the flavonolate ligand assisted by copper. Synthesis and characterization of copper(I) complexes [Cu(PPh3)2(fla)] (fla = flavonolate) and [Cu(PPh3)2(O-bs)] (O-bs = O-benzoylsalicylate). Inorg. Chem. 1999, 38, 3787–3795. [Google Scholar] [CrossRef]
- Baráth, G.; Kaizer, J.; Speier, G.; Párkányi, L.; Kuzmann, E.; Vértes, A. One metal–two pathways to the carboxylate-enhanced, iron-containing quercetinase mimics. Chem. Commun. 2009, 3630–3632. [Google Scholar] [CrossRef] [PubMed]
- Frerichs-Deeken, U.; Ranguelova, K.; Kappl, R.; Hüttermann, J.; Fetzner, S. Dioxygenases without requirement for cofactors and their chemical model reaction: Compulsory order ternary complex mechanism of 1H-3-hydroxy-4-oxoquinaldine 2,4-dioxygenase involving general base catalysis by histidine 251 and single-electron oxidation of the substrate dianion. Biochemistry 2004, 43, 14485–14499. [Google Scholar]
- Steiner, R.A.; Janssen, H.J.; Roversi, P.; Oakley, A.J.; Fetzner, S. Structural basis for cofactor-independent dioxygenation of N-heteroaromatic compounds at the alpha/beta-hydrolase fold. Proc. Natl. Acad. Sci. USA 2010, 107, 657–662. [Google Scholar] [CrossRef]
- Thierbach, S.; Bui, N.; Zapp, J.; Chhabra, S.R.; Kappl, R.; Fetzner, S. Substrate-assisted O2 activation in a cofactor-independent dioxygenase. Chem. Biol. 2014, 21, 217–225. [Google Scholar] [CrossRef]
- Hernández-Ortega, A.; Quesne, M.G.; Bui, S.; Heyes, D.J.; Steiner, R.A.; Scrutton, N.S.; de Visser, S.P. Catalytic mechanism of cofactor-free dioxygenases and how they circumvent spin-forbidden oxygenation of their substrates. J. Am. Chem. Soc. 2015, 137, 7474–7487. [Google Scholar] [CrossRef] [PubMed]
- Bui, S.; Steiner, R.A. New insight into cofactor-free oxygenation from combined experimental and computational approaches. Curr. Opin. Struct. Biol. 2016, 41, 109–118. [Google Scholar] [CrossRef] [Green Version]
- Czaun, M.; Speier, G.; Párkányi, L. Facile copper-mediated activation of the N-H bond and the oxidative cleavage of the C2-C3 bond in 1H-2-phenyl-3-hydroxy-4-oxoquinoline. Chem. Commun. 2004, 1004–1005. [Google Scholar] [CrossRef] [PubMed]
- Pap, J.S.; Matuz, A.; Baráth, G.; Kripli, B.; Giorgi, M.; Speier, G.; Kaizer, J. Bio-inspired flavonol and quinolone dioxygenation by a non-heme iron catalyst modeling the action of flavonol and 3-hydroxy-4(1H)—quinolone 2,4-dioxygenases. J. Inorg. Biochem. 2012, 108, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Nishinaga, A.; Tojo, T.; Tomita, H.; Matsuura, T. Base-catalysed oxygenolysis of 3-hydroxyflavones. J. Chem. Soc. Perkin Trans. 1 1979, 2511–2516. [Google Scholar] [CrossRef]
- Barhács, L.; Kaizer, J.; Speier, G. Kinetics and mechanism of the oxygenation of potassium flavonolate. Evidence for an electron transfer mechanism. J. Org. Chem. 2000, 65, 3449–3452. [Google Scholar] [CrossRef]
- Balogh-Hergovich, É.; Speier, G. Kinetics and mechanism of the base-catalyzed oxygenation of flavonol in DMSO−H2O solution. J. Org. Chem. 2001, 66, 7974–7978. [Google Scholar] [CrossRef]
- Han, X.; Kumar, M.R.; Farmer, P.J. Nitroxygenation of quercetin by HNO. Tetrahedron Lett. 2016, 57, 399–402. [Google Scholar] [CrossRef] [Green Version]
- Czaun, M.; Speier, G. The base-catalyzed oxygenation of quinolone derivatives. Tetrahedron Lett. 2002, 43, 5961–5963. [Google Scholar] [CrossRef]
- Matsuura, T.; Matsushima, H.; Nakashima, R. Photoinduced reactions—XXXVI: Photosensitized oxygenation of 3-hydroxyflavones as a nonenzymatic model for quercetinase. Tetrahedron 1970, 26, 435–443. [Google Scholar] [CrossRef]
- Studer, S.L.; Brewer, W.E.; Martinez, M.L.; Chou, P.T. Time-resolved study of the photooxygenation of 3-hydroxyflavone. J. Am. Chem. Soc. 1989, 111, 7643–7644. [Google Scholar] [CrossRef]
- Brewer, W.E.; Studer, S.L.; Standiford, M.; Chou, P.T. Dynamics of the triplet state and the reverse proton transfer of 3-hydroxyflavone. J. Phys. Chem. 1989, 93, 6088–6094. [Google Scholar] [CrossRef]
- Martinez, M.L.; Studer, S.L.; Chou, P.T. Direct evidence of the triplet-state origin of the slow reverse proton transfer reaction of 3-hydroxyflavone. J. Am. Chem. Soc. 1990, 112, 2427–2429. [Google Scholar] [CrossRef]
- Sisa, M.; Bonnet, S.L.; Ferreira, D.; Van der Westhuizen, J.H. Photochemistry of flavonoids. Molecules 2010, 15, 5196–5245. [Google Scholar] [CrossRef] [PubMed]
- Szakács, Z.; Bojtár, M.; Drahos, L.; Hessz, D.; Kállay, M.; Vidóczy, T.; Bitter, I.; Kubinyi, M. The kinetics and mechanism of photooxygenation of 4′-diethylamino-3-hydroxyflavone. Photochem. Photobiol. Sci. 2016, 15, 219–227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szakács, Z.; Kállay, M.; Kubinyi, M. Theoretical study on the photooxygenation and photorearrangement reactions of 3-hydroxyflavone. RSC Adv. 2017, 7, 32185–32192. [Google Scholar] [CrossRef] [Green Version]
- Matsuura, T.; Takemoto, T.; Nakashima, R. Photoinduced reactions. LII. Photorearragement of 3-hydroxyflavones. Tetrahedron 1971, 12, 1539–1540. [Google Scholar] [CrossRef]
- Matsuura, T.; Takemoto, T.; Nakashima, R. Photoinduced reactions—LXXI: Photorearragement of 3-hydroxyflavones to 3-aryl-3-hydroxy-1,2-indandiones. Tetrahedron 1973, 29, 3337–3340. [Google Scholar] [CrossRef]
- Yokoe, I.; Higuchi, K.; Shirataki, Y.; Komatsu, M. Photochemistry of flavonoids. III. Photorearrangement of flavonols. Chem. Pharm. Bull. 1981, 29, 894–898. [Google Scholar] [CrossRef]
- Grubel, K.; Laughlin, B.J.; Maltais, T.R.; Smith, R.C.; Arif, A.M.; Berreau, L.M. Photochemically-induced dioxygenase-type CO-release reactivity of group 12 metal flavonolate complexes. Chem. Commun. 2011, 47, 10431–10433. [Google Scholar] [CrossRef] [PubMed]
- Grubel, K.; Marts, A.R.; Greer, S.M.; Tierney, D.L.; Allpress, C.J.; Anderson, S.N.; Laughlin, B.J.; Smith, R.C.; Arif, A.M.; Berreau, L.M. Photoinitiated dioxygenase-type reactivity of open-shell 3d divalent metal flavonolato complexes. Eur. J. Inorg. Chem. 2012, 2012, 4750–4757. [Google Scholar] [CrossRef]
- Grubel, K.; Saraf, S.L.; Anderson, S.N.; Laughlin, B.J.; Smith, R.C.; Arif, A.M.; Berreau, L.M. Synthesis, characterization, and photoinduced CO-release reactivity of a Pb(II) flavonolate complex: Comparisons to Group 12 analogs. Inorg. Chim. Acta 2013, 407, 91–97. [Google Scholar] [CrossRef]
- Anderson, S.N.; Noble, M.; Grubel, K.; Marshall, B.; Arif, A.M.; Berreau, L.M. Influence of supporting ligand microenvironment on the aqueous stability and visible light-induced CO-release reactivity of zinc flavonolato species. J. Coord. Chem. 2014, 67, 4061–4075. [Google Scholar] [CrossRef]
- Sorenson, S.; Popova, M.; Arif, A.M.; Berreau, L.M. A bipyridine-ligated zinc(II) complex with bridging flavonolate ligation: Synthesis, characterization and visible-light-induced CO release reactivity. Acta Cryst. 2017, C73, 703–709. [Google Scholar] [CrossRef]
- Su, Y.; Yang, W.; Yang, X.; Zhang, R.; Zhao, J. Visible light-induced CO-release reactivity of a series of ZnII-flavonolate complexes. Aust. J. Chem. 2018, 71, 549–558. [Google Scholar] [CrossRef]
- Protti, S.; Mezzetti, A.; Lapouge, C.; Cornard, J.-P. Photochemistry of metal complexes of 3-hydroxyflavone: Towards a better understanding of the influence of solar light on the metal-soil organic matter interactions. Photochem. Photobiol. Sci. 2008, 7, 109–119. [Google Scholar] [CrossRef]
- Sun, Y.-J.; Huang, Q.-Q.; Li, P.; Zhang, J.-J. Catalytic dioxygenation of flavonol by MII-complexes (M = Mn, Fe, Co, Ni, Cu, and Zn)—Mimicking the MII-substituted quercetin 2,3-dioxygenase. Dalton Trans. 2015, 44, 13926–13938. [Google Scholar] [CrossRef]
- Sun, Y.-J.; Li, P.; Huang, Q.-Q.; Zhang, J.-J.; Itoh, S. Dioxygenation of flavonol catalyzed by copper(II) complexes supported by carboxylate-containing ligands: Structural and functional models of quercetin 2,4-dioxygenase. Eur. J. Inorg. Chem. 2017, 2017, 1845–1854. [Google Scholar] [CrossRef]
- Vogler, A.; Hischa, B. Synthesis, crystal structure, photoluminescence and photochemistry of bis(triphenylphosphine)siliver(I) flavonolate. Z. Naturforsch. B 2018, 74. [Google Scholar] [CrossRef]
- Saraf, S.L.; Fish, T.J.; Benninghoff, A.D.; Buelt, A.A.; Smith, R.C.; Berreau, L.M. Photochemical reactivity of RuII(η6-p-cymene) flavonolato compounds. Organometallics 2014, 33, 6341–6351. [Google Scholar] [CrossRef]
- Kurzwernhart, A.; Kandioller, W.; Bartel, C.; Bächler, S.; Trondl, R.; Mühlgassner, G.; Jakupec, M.A.; Arion, V.B.; Marko, D.; Keppler, B.K.; et al. Targeting the DNA-topoisomerase complex in a double-strike approach with a topoisomerase inhibiting moiety and covalent DNA binder. Chem. Commun. 2012, 48, 4839–4841. [Google Scholar] [CrossRef]
- Kurzwernhart, A.; Kandioller, W.; Bächler, S.; Bartel, C.; Martic, S.; Buczkowska, M.; Mühlgassner, G.; Jakupec, M.A.; Kraatz, H.-B.; Bednarski, P.J.; et al. Structure-activity relationships of targeted RuII(η6-p-cymene) anticancer complexes with flavonol-derived ligands. J. Med. Chem. 2012, 55, 10512–10522. [Google Scholar] [CrossRef]
- Kurzwernhart, A.; Kandioller, W.; Enyedy, É.A.; Novak, M.; Jakupec, M.A.; Keppler, B.K.; Hartinger, C.G. 3-hydroxyflavones vs. 3-hydroxyquinolinones: Structure-activity relationships and stability studies on RuII(arene) anticancer complexes with biologically active ligands. Dalton Trans. 2013, 42, 6193–6202. [Google Scholar] [CrossRef]
- Han, X.; Klausmeyer, K.K.; Farmer, P.J. Characterization of the initial intermediate formed during photoinduced oxygenation of the ruthenium(II) bis(bipyridyl)flavonolate complex. Inorg. Chem. 2016, 55, 7320–7322. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Kumar, M.R.; Hoogerbrugge, A.; Klausmeyer, K.K.; Ghimire, M.M.; Harris, L.M.; Omary, M.A.; Farmer, P.J. Mechanistic investigations of photoinduced oxygenation of Ru(II) bis-bipyridyl flavonolate complexes. Inorg. Chem. 2018, 57, 2416–2424. [Google Scholar] [CrossRef]
- Anderson, S.N.; Richards, J.M.; Esquer, H.J.; Benninghoff, A.D.; Arif, A.M.; Berreau, L.M. A Structurally-tunable 3-hydroxyflavone motif for visible light-induced carbon monoxide-releasing molecules (CORMs). ChemistryOpen 2015, 4, 590–594. [Google Scholar] [CrossRef]
- Popova, M.; Soboleva, T.; Arif, A.M.; Berreau, L.M. Properties of a flavonol-based photoCORM in aqueous buffered solutions: Influence of metal ions, surfactants and proteins on visible light-induced CO release. RSC Adv. 2017, 7, 21997–22007. [Google Scholar] [CrossRef]
- Li, Y.; Shu, Y.; Liang, M.; Xie, X.; Jiao, X.; Wang, X.; Tang, B. A two-photon H2O2 -activated CO photoreleaser. Angew. Chem. Int. Ed. Engl. 2018, 57, 12415–12419. [Google Scholar] [CrossRef] [PubMed]
- Dick, B.; Ernsting, N.P. Excited-state intramolecular proton transfer in 3-hydroflavone isolated in solid argon: Fluorescence and fluorescence-excitation spectra and tautomer fluorescence rise time. J. Phys. Chem. 1987, 91, 4261–4265. [Google Scholar] [CrossRef]
- Soboleva, T.; Esquer, H.J.; Anderson, S.N.; Berreau, L.M.; Benninghoff, A.D. Mitochondrial-localized versus cytosolic intracellular CO-releasing organic photoCORMs: Evaluation of CO effects using bioenergetics. ACS Chem. Biol. 2018, 13, 2220–2228. [Google Scholar] [CrossRef]
- Popova, M.; Soboleva, T.; Ayad, S.; Benninghoff, A.D.; Berreau, L.M. Visible-light-activated quinolone carbon-monoxide-releasing molecule: Prodrug and albumin-assisted delivery enables anticancer and potent anti-inflammatory effects. J. Am. Chem. Soc. 2018, 140, 9721–9729. [Google Scholar] [CrossRef]
- Soboleva, T.; Esquer, H.J.; Benninghoff, A.D.; Berreau, L.M. Sense and release: A thiol-responsible flavonol-based photonically driven carbon monoxide-releasing molecule that operates via a multiple-input AND logic gate. J. Am. Chem. Soc. 2017, 139, 9435–9438. [Google Scholar] [CrossRef]
- Soboleva, T.; Benninghoff, A.D.; Berreau, L.M. An H2S-sensing/CO-releasing flavonol that operates via logic gates. ChemPlusChem 2017, 82, 1408–1412. [Google Scholar] [CrossRef] [PubMed]
- Anderson, S.N.; Larson, M.T.; Berreau, L.M. Solution or solid—It doesn’t matter: Visible light-induced CO release reactivity of zinc flavonolato complexes. Dalton Trans. 2016, 45, 14570–14580. [Google Scholar] [CrossRef] [PubMed]
- Bilokin, M.D.; Shvadchak, V.V.; Yushchenko, D.A.; Klymchenko, A.S.; Duportail, G.; Mely, Y.; Pivovarenko, V.G. 3-hydroxybenzo[g]quinolones: Dyes with red-shifted absorption and highly resolved dual emission. Tetrahedron Lett. 2009, 50, 4714–4719. [Google Scholar] [CrossRef]
- Sleep, D. Albumin and its application in drug delivery. Expert Opin. Drug Deliv. 2015, 12, 793–812. [Google Scholar] [CrossRef]
- Hopper, C. Keto Acids in Carbon Monoxide Therapeutics, 2nd ed.; Independently published via Amazon: Seattle, WA, USA, 2018. [Google Scholar]
- Slanina, T.; Sebej, P. Visible-light-activated photoCORMs: Rational design of CO-releasing organic molecules absorbing in the tissue-transparent window. Photochem. Photobiol. Sci. 2018, 17, 692–710. [Google Scholar] [CrossRef]
- Ramão, C.C.; Blättler, W.A.; Seixas, J.D.; Bernardes, G.J.L. Developing drug molecules for therapy with carbon monoxide. Chem. Soc. Rev. 2012, 41, 3571–3583. [Google Scholar] [CrossRef] [PubMed]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef] [PubMed]
- Butun, B.; Topcu, G.; Ozturk, T. Recent advances on 3-hydroxyflavone derivatives: Structures and properties. Mini Rev. Med. Chem. 2018, 18, 98–103. [Google Scholar] [CrossRef] [PubMed]






















| Compound | Absorption Max. (nm) | Quantum Yield for CO Release | Reference |
|---|---|---|---|
| [(6-Ph2TPA)Zn(3-Hfl)]ClO4 | 420 | 0.09(1) a,c | [63,95,97] |
| [(6-Ph2TPA)Zn(3-Hfl)]ClO4 | 420 | 0.012(2) b,c; 0.006(1) b,d | [98] |
| [(6-Ph2TPA)Cd(3-Hfl)]ClO4 | 430 | 0.28(2) a,c | [95,97] |
| [(6-Ph2TPA)Hg(3-Hfl)]ClO4 | 415 | 0.31(2) a,c | [95,97] |
| [(6-Ph2TPA)Mn(3-Hfl)]ClO4 | 415 | 0.005 a,c | [96] |
| [(6-Ph2TPA)Co(3-Hfl)]ClO4 | 430 | 0.005 a,c | [96] |
| [(6-Ph2TPA)Cu(3-Hfl)]ClO4 | 428 | 0.005 a,c | [96] |
| [(6-Ph2TPA)Ni(3-Hfl)]ClO4 | 415 | 0.008 a,c | [96] |
| [(6-Ph2TPA)Pb(3-Hfl)]ClO4 | 406 | 0.21(6) a,c | [97] |
| [(TPA)Zn(3-Hfl)]ClO4 | 415 | 0.006(1)b,c | [98] |
| [(bnpapa)Zn(3-Hfl)]ClO4 | 401 | 0.00027(1) b,c | [98] |
| {[(bpy)Zn(3-Hfl)]2}(ClO4)2 | 414 | 0.004(1) b,c | [99] |
| [Ru(η6-p-cymene)Cl(3-Hfl)] | 472 | 0.001(1) b,c | [105] |
| Compound | Absorption Maximum (nm) | Quantum Yield for CO Release | Reference |
|---|---|---|---|
| 1 | 409 b | 0.007(3) a,c | [111,112] |
| 410 c | 0.006(3) a,c | [112] | |
| 410 d | 0.010(3) a,d | [112] | |
| 410 e | 0.0063(1) a,e | [112] | |
| 410 F | 0.0006(1) a,f | [112] | |
| 2 | 442 b | 0.006(1) a,b | [111] |
| 3 | 478 b | 0.426(3) a,b | [111] |
| 4 | Not determined | [111] | |
| 6 | 480 b | 0.651(2) a,b | [119] |
| 7 | 524 b | 0.583(4) b,g | [119] |
| 8 | 550 b | 0.951(4) b,g | [119] |
| 9 | 600 b | 0.947(7) b,g | [119] |
| 14 | 445 b | 0.0045(1) a,b | [116] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soboleva, T.; Berreau, L.M. 3-Hydroxyflavones and 3-Hydroxy-4-oxoquinolines as Carbon Monoxide-Releasing Molecules. Molecules 2019, 24, 1252. https://doi.org/10.3390/molecules24071252
Soboleva T, Berreau LM. 3-Hydroxyflavones and 3-Hydroxy-4-oxoquinolines as Carbon Monoxide-Releasing Molecules. Molecules. 2019; 24(7):1252. https://doi.org/10.3390/molecules24071252
Chicago/Turabian StyleSoboleva, Tatiana, and Lisa M. Berreau. 2019. "3-Hydroxyflavones and 3-Hydroxy-4-oxoquinolines as Carbon Monoxide-Releasing Molecules" Molecules 24, no. 7: 1252. https://doi.org/10.3390/molecules24071252






