A New Propolis Type from Changbai Mountains in North-east China: Chemical Composition, Botanical Origin and Biological Activity
Abstract
:1. Introduction
2. Results and Discussion
2.1. Contents of Total Flavonoids, Total Phenolics and Antioxidant Activity of CBM Propolis
2.2. Profiling of Samples with HPLC-UV and HPLC-ESI/MS
2.3. The Difference between CBM Propolis and other Propolis from China
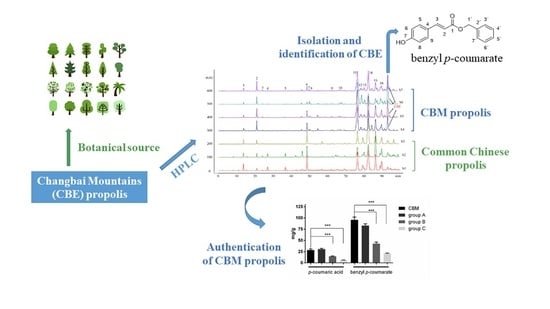
2.4. Isolation and Identification of CBE in CBM Propolis
2.5. Authentication of CBM Propolis
2.6. The Botanical Source of the CBM Propolis
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Collection of Propolis and Plant Material
3.3. HPLC and HPLC-ESI/MS Analysis
3.4. Antioxidant Capacity
3.5. Determinations of Total Flavonoids and Total Phenolics
3.6. Isolation and Identification of the Unknown Compound CBE
3.7. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhang, C.P.; Zheng, H.Q.; Hu, F.L. Extraction, partial characterization, and storage stability of beta-glucosidase from propolis. J. Food Sci. 2011, 76, C75–C79. [Google Scholar] [CrossRef]
- Burdock, G. Review of the biological properties and toxicity of bee propolis (propolis). Food Chem. Toxicol. 1998, 36, 347–363. [Google Scholar] [CrossRef]
- Meyer, W.; Ulrich, W. “Propolis Bees” and Their Activities. Bee World 2015, 37, 25–36. [Google Scholar] [CrossRef]
- Bankova, V.; Popova, M.; Trusheva, B. The phytochemistry of the honeybee. Phytochemistry 2018, 155, 1–11. [Google Scholar] [CrossRef]
- Kuropatnicki, A.K.; Szliszka, E.; Krol, W. Historical aspects of propolis research in modern times. Evid-Based Compl. Alt. Med 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Batista, C.M.; Alves, A.V.F.; Queiroz, L.A.; Lima, B.S.; Filho, R.N.P.; Araujo, A.A.S.; de Albuquerque Junior, R.L.C.; Cardoso, J.C. The photoprotective and anti-inflammatory activity of red propolis extract in rats. J. Photochem. Photobiol. B 2018, 180, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, C.; Prasniewski, A.; Calegari, M.A.; de Lima, V.A.; Oldoni, T.L.C. Determination of Total Phenolic Compounds and Antioxidant Activity of Ethanolic Extracts of Propolis Using ATR–FT-IR Spectroscopy and Chemometrics. Food Anal. Method 2018, 11, 2013–2021. [Google Scholar] [CrossRef]
- Nna, V.U.; Abu Bakar, A.B.; Md Lazin, M.; Mohamed, M. Antioxidant, anti-inflammatory and synergistic anti-hyperglycemic effects of Malaysian propolis and metformin in streptozotocin-induced diabetic rats. Food Chem. Toxicol. 2018, 120, 305–320. [Google Scholar] [CrossRef]
- Rufatto, L.C.; Luchtenberg, P.; Garcia, C.; Thomassigny, C.; Bouttier, S.; Henriques, J.A.P.; Roesch-Ely, M.; Dumas, F.; Moura, S. Brazilian red propolis: Chemical composition and antibacterial activity determined using bioguided fractionation. Microbiol. Res. 2018, 214, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Wu, Y.; Chen, X.; Jiang, X.; Wang, K.; Hu, F. Chinese Propolis Exerts Anti-Proliferation Effects in Human Melanoma Cells by Targeting NLRP1 Inflammatory Pathway, Inducing Apoptosis, Cell Cycle Arrest, and Autophagy. Nutrients 2018, 10, 1170. [Google Scholar] [CrossRef] [PubMed]
- Sawicka, D.; Car, H.; Borawska, M.H.; Nikliński, J. The anticancer activity of propolis. Folia Histochem. Cyto. 2012, 50, 25–37. [Google Scholar] [CrossRef] [Green Version]
- Salatino, A.; Teixeira, E.W.; Negri, G.; Message, D. Origin and Chemical Variation of Brazilian Propolis. Evid-Based Compl. Alt. Med. 2005, 2, 33–38. [Google Scholar] [CrossRef]
- Bankova, V.S.; de Castro, S.L.; Marcucci, M.C. Propolis: recent advances in chemistry and plant origin. Apidologie 2000, 31, 3–15. [Google Scholar] [CrossRef] [Green Version]
- Wilson, M.B.; Spivak, M.; Hegeman, A.D.; Rendahl, A.; Cohen, J.D. Metabolomics reveals the origins of antimicrobial plant resins collected by honey bees. PLoS ONE 2013, 8, e77512. [Google Scholar] [CrossRef]
- Aliboni, A. Propolis from Northern California and Oregon: Chemical Composition, Botanical Origin, and Content of Allergens. Zeitschrift für Naturforschung C 2014, 69, 10–20. [Google Scholar] [CrossRef]
- Christov, R.; Trusheva, B.; Popova, M.; Bankova, V.; Bertrand, M. Chemical composition of propolis from Canada, its antiradical activity and plant origin. Nat. Prod. Res. 2006, 20, 531–536. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Suresh, A.; Yasuhiro, T.; Hiroyasu, E.; Shigetoshi, K. Study on the constituents of Mexican propolis and their cytotoxic activity against PANC-1 human pancreatic cancer cells. J. Nat. Prod. 2010, 73, 623–627. [Google Scholar]
- Popova, M.P.; Bankova, V.S.; Bogdanov, S.; Tsvetkova, I.; Naydenski, C.; Marcazzan, G.L.; Sabatini, A.-G. Chemical characteristics of poplar type propolis of different geographic origin. Apidologie 2007, 38, 306. [Google Scholar] [CrossRef]
- Coelho, J.; Falcão, S.I.; Vale, N.; Almeida-Muradian, L.B.; Vilas-Boas, M. Phenolic composition and antioxidant activity assessment of southeastern and south Brazilian propolis. J. Apicult. Res. 2017, 56, 21–31. [Google Scholar] [CrossRef]
- Cui-ping, Z.; Shuai, H.; Wen-ting, W.; Shun, P.; Xiao-ge, S.; Ya-jing, L.; Fu-liang, H. Development of high-performance liquid chromatographic for quality and authenticity control of Chinese propolis. J. Food Sci. 2014, 79, C1315–C1322. [Google Scholar] [CrossRef]
- Ling-he, J. The Determination of Mineral Elements in Honey by ICP-MS and Its Application to Discriminate Northeast-China Black Honey and Linden Honey. Master’s Thesis, Shandong Agricultural University, Taian, China, 14 June 2016. (In Chinese). [Google Scholar]
- Zheng, D.; Wallin, D.O.; Hao, Z. Rates and patterns of landscape change between 1972 and 1988 in the Changbai Mountain area of China and North Korea. Landscape Ecol. 1997, 12, 241–254. [Google Scholar] [CrossRef]
- Falcao, S.I.; Vale, N.; Gomes, P.; Domingues, M.R.; Freire, C.; Cardoso, S.M.; Vilas-Boas, M. Phenolic profiling of Portuguese propolis by LC-MS spectrometry: uncommon propolis rich in flavonoid glycosides. Phytochem. Anal. 2013, 24, 309–318. [Google Scholar] [CrossRef]
- Ahn, M.-R.; Kumazawa, S.; Usui, Y.; Nakamura, J.; Matsuka, M.; Zhu, F.; Nakayama, T. Antioxidant activity and constituents of propolis collected in various areas of China. Food Chem. 2007, 101, 1383–1392. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J. Oxygen toxicity, oxygen radicals, transition metals and disease. Biochem. J. 1984, 219, 1. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.-Q.; Wei, W.-T.; Hu, F.-L. Beekeeping Industry In China. Bee World 2015, 88, 41–44. [Google Scholar] [CrossRef]
- Lee, I.K.; Han, M.S.; Kim, D.W.; Yun, B.S. Phenylpropanoid acid esters from Korean propolis and their antioxidant activities. Bioorg. Med. Chem. Lett. 2014, 24, 3503–3505. [Google Scholar] [CrossRef] [PubMed]
- Savka, M.A.; Dailey, L.; Popova, M.; Mihaylova, R.; Merritt, B.; Masek, M.; Le, P.; Nor, S.R.; Ahmad, M.; Hudson, A.O.; et al. Chemical composition and disruption of quorum sensing signaling in geographically diverse United States propolis. Evid-Based Compl. Alt. Med. 2015, 2015. [Google Scholar] [CrossRef]
- Castro, C.; Mura, F.; Valenzuela, G.; Figueroa, C.; Salinas, R.; Zuniga, M.C.; Torres, J.L.; Fuguet, E.; Delporte, C. Identification of phenolic compounds by HPLC-ESI-MS/MS and antioxidant activity from Chilean propolis. Food Res. Int. 2014, 64, 873–879. [Google Scholar] [CrossRef] [PubMed]
- Elnakady, Y.A.; Rushdi, A.I.; Franke, R.; Abutaha, N.; Ebaid, H.; Baabbad, M.; Omar, M.O.; Al Ghamdi, A.A. Characteristics, chemical compositions and biological activities of propolis from Al-Bahah, Saudi Arabia. Sci. Rep. 2017, 7, 41453. [Google Scholar] [CrossRef] [Green Version]
- Lee, K.M.; Kim, Y.Y.; Hyun, J.O. Genetic variation in populations of Populus davidiana Dode based on microsatellite marker analysis. Genes Genom. 2011, 33, 163–171. [Google Scholar] [CrossRef]
- Wei, Z.; Du, Q.; Zhang, J.; Li, B.; Zhang, D. Genetic diversity and population structure in Chinese indigenous poplar (Populus simonii) populations using microsatellite markers. Plant Mol. Biol. Rep. 2013, 31, 620–632. [Google Scholar] [CrossRef]
- Klaric, I.; Pavic, M.; Miskulin, I.; Blazicevic, V.; Dumic, A.; Miskulin, M. Influence of dietary supplementation of propolis and bee pollen on liver pathology in broiler chickens. Animals 2018, 8, 54. [Google Scholar] [CrossRef]
- Park, Y.K.; Koo, M.H.; Ikegaki, M.; Contado, J.L. Comparison of the flavonoid aglycone contents of Apis mellifera propolis from various regions of Brazil. Arquivos de biologia e tecnologia 1997. [Google Scholar]
- Woisky, R.G.; Salatino, A. Analysis of propolis: some parameters and procedures for chemical quality control. J. Apicult. Res. 2015, 37, 99–105. [Google Scholar] [CrossRef]
- Zhang, C.; Shen, X.; Chen, J.; Jiang, X.; Hu, F. Identification of Free Radical Scavengers from Brazilian Green Propolis Using Off-Line HPLC-DPPH Assay and LC-MS. J. Food Sci. 2017, 82, 1602–1607. [Google Scholar] [CrossRef]
- Zhou, W.; Xie, M.F.; Zhang, X.Y.; Liu, T.T.; Yu, Y.J.; Duan, G.L. Improved liquid chromatography fingerprint of fat-soluble Radix isatidis extract using multi-wavelength combination technique. J. Sep. Sci. 2011, 34, 1123–1132. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds p-coumaric acid and benzyl p-coumarate are available from the authors. |





| Sample No. | Origin (City/Province) | Collection Date | Total Flavonoids (mg/g, QE) | Total Phenolics (mg/g, GAE) | DPPH Scavenging Activity (IC50,μg/mL) | Correlation Coefficients |
|---|---|---|---|---|---|---|
| 1 | Jixi, Heilongjiang | May 2015 | 117.8 ± 5.9 | 284.2 ± 0.4 | 231.1 ± 4.2 | 0.957 |
| 2 | Jixi, Heilongjiang | May 2015 | 105.2 ± 5.7 | 253.0 ± 6.8 | 222.5 ± 9.2 | 0.982 |
| 3 | Mudanjiang, Heilongjiang | May 2015 | 95.5 ± 6.0 | 243.4 ± 3.3 | 256.7 ± 9.0 | 0.968 |
| 4 | Jixi, Heilongjiang | May 2015 | 113.6 ± 2.1 | 285.4 ± 3.7 | 233.2 ± 4.2 | 0.986 |
| 5 | Jixi, Heilongjiang | May 2015 | 121.7 ± 1.4 | 263.2 ± 1.5 | 231.2 ± 10.5 | 0.983 |
| 6 | Shuangyashan, Heilongjiang | May 2016 | 114.3 ± 5.4 | 262.7 ± 1.9 | 267.5 ± 6.3 | 0.904 |
| 7 | Qiqihaer, Heilongjiang | May 2016 | 111.4 ± 4.4 | 286.9 ± 0.4 | 221.2 ± 9.4 | 0.984 |
| 8 | Qitaihe, Heilongjiang | May 2016 | 105.4 ± 4.5 | 316.8 ± 1.2 | 204.5 ± 1.1 | 0.939 |
| 9 | Jixi, Heilongjiang | May 2016 | 98.3 ± 4.8 | 215.6 ± 0.4 | 278.5 ± 2.9 | 0.982 |
| 10 | Jixi, Heilongjiang | May 2016 | 123.1 ± 2.8 | 298.1 ± 1.6 | 193.6 ± 8.1 | 0.966 |
| 11 | Shuangyashan, Heilongjiang | May 2016 | 106.8 ± 6.0 | 273.7 ± 3.7 | 215.5 ± 2.8 | 0.879 |
| 12 | Shuangyashan, Heilongjiang | May 2016 | 108.3 ± 2.7 | 255.8 ± 1.6 | 184.3 ± 4.7 | 0.928 |
| 13 | Qitaihe, Heilongjiang | May 2016 | 114.1 ± 5.2 | 299.6 ± 0.8 | 188.8 ± 2.4 | 0.721 |
| 14 | Mudanjiang, Heilongjiang | May 2016 | 105.5 ± 2.8 | 275.6 ± 2.9 | 170.4 ± 2.5 | 0.955 |
| 15 | Jixi, Heilongjiang | May 2016 | 106.9 ± 1.9 | 268.7 ± 4.2 | 183.9 ± 7.4 | 0.721 |
| 16 | Haerbin, Heilongjiang | July 2016 | 90.5 ± 2.9 | 265.4 ± 3.9 | 188.3 ± 3.5 | 0.949 |
| 17 | Jiilin, Jilin | July 2016 | 123.1 ± 7.5 | 300.6 ± 1.6 | 171.9 ± 2.0 | 0.902 |
| 18 | Jiilin, Jilin | July 2016 | 97.7 ± 3.7 | 257.1 ± 4.0 | 234.4 ± 0.7 | 0.986 |
| 19 | Dunhua, Jilin | July 2016 | 101.1 ± 2.1 | 247.5 ± 1.6 | 263.1 ± 3.3 | 0.983 |
| 20 | Jiilin, Jilin | July 2016 | 106.7 ± 5.4 | 250.3 ± 1.5 | 248.6 ± 2.9 | 0.966 |
| 21 | Jiilin, Jilin | July 2016 | 108.9 ± 1.6 | 245.2 ± 2.4 | 262.9 ± 3.0 | 0.778 |
| Peak | Compounds | MW | [M − H]− | Retention Time (min) |
|---|---|---|---|---|
| 1 | caffeic acid | 180 | 179.1 | 13.694 |
| 2 | p-coumaric acid | 164 | 163.1 | 20.693 |
| 3 | ferulic acid | 194 | 193.1 | 23.646 |
| 4 | isoferulic acid | 194 | 193.1 | 26.474 |
| 5 | 3,4-dimethoxycinnamic acid | 208 | 207.1 | 35.903 |
| 6 | pinobanksin | 272 | 271.1 | 46.894 |
| 7 | naringenin | 272 | 271.1 | 48.358 |
| 8 | quercetin | 302 | 301.1 | 52.910 |
| 9 | kaempferol | 286 | 285.1 | 60.956 |
| 10 | apigenin | 270 | 269.1 | 64.924 |
| 11 | pinocembrin | 256 | 255.1 | 73.517 |
| 12 | benzyl caffeate | 270 | 269.1 | 76.338 |
| 13 | 3-O-acetylpinobanksin | 314 | 313.1 | 79.337 |
| 14 | chrysin | 254 | 253.1 | 84.079 |
| 15 | CAPE | 284 | 283.1 | 86.048 |
| 16 | galangin | 270 | 269.1 | 87.329 |
| Sample No. | Correlation Coefficients | Samples No. | Correlation Coefficients | Samples No. | Correlation Coefficients | Samples No. | Correlation Coefficients | Samples No. | Correlation Coefficients |
|---|---|---|---|---|---|---|---|---|---|
| Anhui1 | 0.566 | Shandong2 | 0.566 | Liaoning1 | 0.586 | Henan2 | 0.596 | Hubei4 | 0.618 |
| Anhui2 | 0.427 | Shandong3 | 0.551 | Liaoning2 | 0.596 | Henan3 | 0.663 | Zhejiang1 | 0.848 |
| Anhui3 | 0.415 | Shandong4 | 0.54 | Liaoning3 | 0.574 | Henan4 | 0.591 | Zhejiang2 | 0.612 |
| Anhui4 | 0.522 | Shandong5 | 0.505 | Heilongjiang1 | 0.533 | Henan5 | 0.599 | Ningxia | 0.503 |
| Anhui5 | 0.509 | Shandong6 | 0.505 | Heilongjiang2 | 0.758 | Henan6 | 0.631 | Xinjiang | 0.664 |
| Anhui6 | 0.563 | Shaanxi1 | 0.51 | Heilongjiang3 | 0.798 | Jiangsu1 | 0.596 | Beijing | 0.668 |
| Guizhou | 0.598 | Shaanxi2 | 0.491 | Heilongjiang4 | 0.907 | Jiangsu2 | 0.538 | Inner Mongolia | 0.577 |
| Sichuan1 | 0.651 | Jilin1 | 0.672 | Heilongjiang5 | 0.496 | Hubei1 | 0.684 | Hebei1 | 0.559 |
| Sichuan2 | 0.643 | Jilin2 | 0.745 | Heilongjiang6 | 0.819 | Hubei2 | 0.696 | Hebei2 | 0.469 |
| Shandong1 | 0.531 | Jilin3 | 0.741 | Henan1 | 0.545 | Hubei3 | 0.486 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, X.; Tian, J.; Zheng, Y.; Zhang, Y.; Wu, Y.; Zhang, C.; Zheng, H.; Hu, F. A New Propolis Type from Changbai Mountains in North-east China: Chemical Composition, Botanical Origin and Biological Activity. Molecules 2019, 24, 1369. https://doi.org/10.3390/molecules24071369
Jiang X, Tian J, Zheng Y, Zhang Y, Wu Y, Zhang C, Zheng H, Hu F. A New Propolis Type from Changbai Mountains in North-east China: Chemical Composition, Botanical Origin and Biological Activity. Molecules. 2019; 24(7):1369. https://doi.org/10.3390/molecules24071369
Chicago/Turabian StyleJiang, Xiasen, Jing Tian, Yufei Zheng, Yanzheng Zhang, Yuqi Wu, Cuiping Zhang, Huoqing Zheng, and Fuliang Hu. 2019. "A New Propolis Type from Changbai Mountains in North-east China: Chemical Composition, Botanical Origin and Biological Activity" Molecules 24, no. 7: 1369. https://doi.org/10.3390/molecules24071369
APA StyleJiang, X., Tian, J., Zheng, Y., Zhang, Y., Wu, Y., Zhang, C., Zheng, H., & Hu, F. (2019). A New Propolis Type from Changbai Mountains in North-east China: Chemical Composition, Botanical Origin and Biological Activity. Molecules, 24(7), 1369. https://doi.org/10.3390/molecules24071369