Proteinaceous Residue Removal from Oat β-Glucan Extracts Obtained by Alkaline Water Extraction
Abstract
:1. Introduction
2. Results
2.1. Proteinaceous Residuals Removal Characteristic Description
2.2. Starch and Glucose Content
2.3. Protein Content
2.4. Beta-glucan Content and Molar Mass
2.5. Possible Interactions between Compounds During Purification Process
3. Discussion
4. Materials and Methods
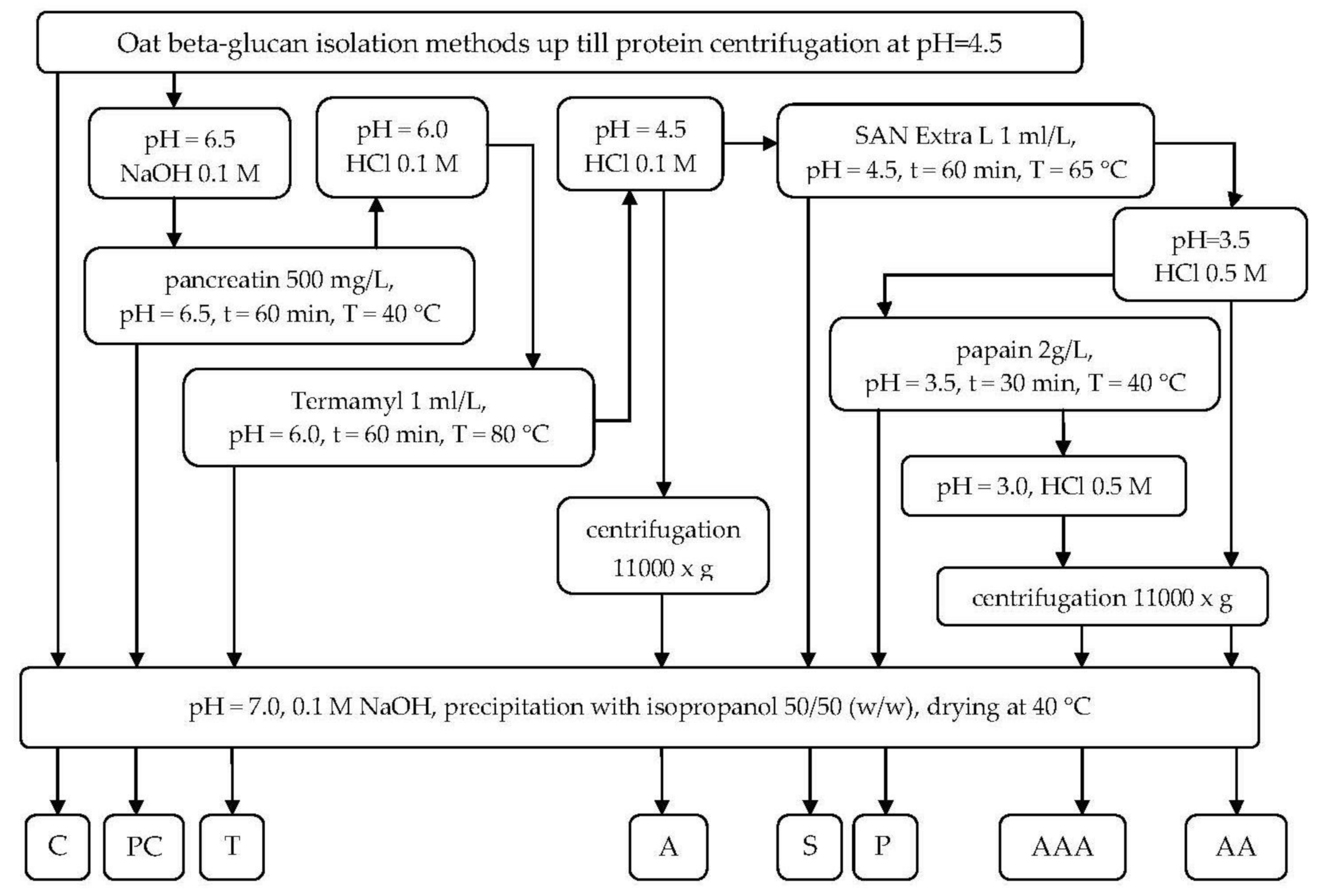
4.1. Oat Beta-Glucan Isolation
4.2. Enzymatic Treatment for Residuals Removal
4.2.1. Pancreatin Digestion
4.2.2. Termamyl Digestion
4.2.3. pH 4.5 Precipitation
4.2.4. San Extra L Hydrolysis
4.2.5. pH 3.5 Precipitation
4.2.6. Papain Digestion
4.2.7. pH 3.0 Precipitation
4.3. Analytical Methods
4.3.1. Beta-glucan Determination
4.3.2. Starch Content Determination
4.3.3. Glucose Determination
4.3.4. Protein Content
4.3.5. Molar Mass Determination
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Błaszczyk, K.; Wilczak, J.; Harasym, J.; Gudej, S.; Suchecka, D.; Królikowski, T.; Lange, E.; Gromadzka-Ostrowska, J. Impact of low and high molecular weight oat beta-glucan on oxidative stress and antioxidant defense in spleen of rats with LPS induced enteritis. Food Hydrocoll. 2015, 51, 272–280. [Google Scholar] [CrossRef]
- Wilczak, J.; Błaszczyk, K.; Kamola, D.; Gajewska, M.; Harasym, J.P.; Jałosińska, M.; Gudej, S.; Suchecka, D.; Oczkowski, M.; Gromadzka-Ostrowska, J. The effect of low or high molecular weight oat beta-glucans on the inflammatory and oxidative stress status in the colon of rats with LPS-induced enteritis. Food Funct. 2015, 6, 590–603. [Google Scholar] [CrossRef]
- Parzonko, A.; Makarewicz-Wujec, M.; Jaszewska, E.; Harasym, J.; Kozłowska-Wojciechowska, M. Pro-apoptotic properties of (1,3)(1,4)-β-d-glucan from avena sativa on human melanoma HTB-140 cells in vitro. Int. J. Biol. Macromol. 2015, 72, 757–763. [Google Scholar] [CrossRef] [PubMed]
- Suchecka, D.; Gromadzka-Ostrowska, J.; Żyła, E.; Harasym, J.P.; Oczkowski, M. Selected physiological activities and health promoting properties of cereal beta-glucans. A review. J. Anim. Feed Sci. 2017, 26, 183–191. [Google Scholar] [CrossRef]
- Choromanska, A.; Kulbacka, J.; Rembialkowska, N.; Pilat, J.; Oledzki, R.; Harasym, J.; Saczko, J. Anticancer properties of low molecular weight oat beta-glucan - an in vitro study. Int. J. Biol. Macromol. 2015, 80, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Suchecka, D.; Harasym, J.P.; Wilczak, J.; Gajewska, M.; Oczkowski, M.; Gudej, S.; Błaszczyk, K.; Kamola, D.; Filip, R.; Gromadzka-Ostrowska, J. Antioxidative and anti-inflammatory effects of high beta-glucan concentration purified aqueous extract from oat in experimental model of LPS-induced chronic enteritis. J. Funct. Foods 2015, 14, 244–254. [Google Scholar] [CrossRef]
- Suchecka, D.; Harasym, J.; Wilczak, J.; Gromadzka-Ostrowska, J. Hepato- and gastro- protective activity of purified oat 1–3, 1–4-β-d-glucans of different molecular weight. Int. J. Biol. Macromol. 2016, 91, 1177–1185. [Google Scholar] [CrossRef]
- Choromańska, A.; Kulbacka, J.; Harasym, J.; Dubińska-Magiera, M.; Saczko, J. Anticancer activity of oat β-glucan in combination with electroporation on human cancer cells. Acta Pol. Pharm. Drug Res. 2017, 74, 616–623. [Google Scholar]
- Suchecka, D.; Błaszczyk, K.; Harasym, J.; Gudej, S.; Wilczak, J.; Gromadzka-Ostrowska, J. Impact of purified oat 1-3,1-4-β-d-glucan of different molecular weight on alleviation of inflammation parameters during gastritis. J. Funct. Foods 2017, 28, 11–18. [Google Scholar] [CrossRef]
- Choromanska, A.; Kulbacka, J.; Harasym, J.; Oledzki, R.; Szewczyk, A.; Saczko, J. High- and low-molecular weight oat beta-glucan reveals antitumor activity in human epithelial lung cancer. Pathol. Oncol. Res. 2018, 24, 583–592. [Google Scholar] [CrossRef] [PubMed]
- Błaszczyk, K.; Gajewska, M.; Wilczak, J.; Kamola, D.; Majewska, A.; Harasym, J.; Gromadzka-Ostrowska, J. Oral administration of oat beta-glucan preparations of different molecular weight results in regulation of genes connected with immune response in peripheral blood of rats with LPS-induced enteritis. Eur. J. Nutr. 2018. [Google Scholar] [CrossRef]
- Gillisen, L.J.W.J.; van der Meer, O.M.; Smulders, M.J.M. Why oats are safe and healthy for celiac disease patients. Med. Sci. 2016, 4, 21. [Google Scholar]
- Tsopmo, A. Processing Oats and Bioactive Components. In Processing and Impact on Active Components in Food, 1st ed.; Preedy, V., Ed.; Academic Press Elsevier Inc.: San Diego, CA, USA, 2015; pp. 361–368. [Google Scholar]
- Maheshwari, G.; Sowrirajan, S.; Joseph, B. Extraction and isolation of β-glucan from grain sources—A review. J. Food Sci. 2017, 82, 1535–1545. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Anjum, F.M.; Zahoor, T.; Nawaz, H.; Din, A. Physicochemical and functional properties of barley β-glucan as affected by different extraction procedures. Int. J. Food Sci. Tech. 2009, 44, 181–187. [Google Scholar] [CrossRef]
- Babu, L.R. Green extraction techniques, structural analysis and antioxidant activites of β-glucan present in oats. Intl. J. Latest Trends Eng. Technol. 2015, 5, 125–135. [Google Scholar]
- Skendi, A.; Biliaderis, C.G.; Lazaridou, A.; Izydorczyk, M.S. Structure and rheological properties of water soluble β-glucans from oat cultivars of Avena sativa and Avena bysantina. J. Cereal Sci. 2003, 38, 15–31. [Google Scholar] [CrossRef]
- Lazaridou, A.; Biliaderis, C.G.; Micha-Screttas, M.; Steele, B.R. A comparative study on structure-function relations of mixed-linkage (1→3), (1→4) linear β-d-glucans. Food Hydrocoll. 2004, 18, 837–855. [Google Scholar] [CrossRef]
- Papageorgiou, M.; Lakhdara, N.; Lazaridou, A.; Biliaderis, C.G.; Izydorczyk, M.S. Water extractable (1 → 3,1 → 4)-β-d-glucans from barley and oats: An intervarietal study on their structural features and rheological behaviour. J. Cereal Sci. 2005, 42, 213–224. [Google Scholar] [CrossRef]
- Wood, P.J.; Weisz, J.; Fedec, P.; Burrows, V.D. Large-scale preparation and properties of oat fractions enriched in (1→3)(1→4)-β-d-glucan. Cereal Chem. 1989, 66, 97–103. [Google Scholar]
- Beer, M.U.; Arrigoni, E.; Amadò, R. Extraction of oat gum from oat bran: Effects of process on yield, molecular weight distribution, viscosity and (1→3)(1→4)-β-d-glucan content of the gum. Cereal Chem. 1996, 73, 58–62. [Google Scholar]
- Bhatty, R.S. Laboratory and pilot plant extraction and purification of β-glucans from hull-less barley and oat brans. J. Cereal Sci. 1995, 22, 163–170. [Google Scholar] [CrossRef]
- Harasym, J.; Suchecka, D.; Gromadzka-Ostrowska, J. Effect of size reduction by freeze-milling on processing properties of beta-glucan oat bran. J. Cereal Sci. 2015, 61, 119–125. [Google Scholar] [CrossRef]
- Korompokis, K.; Nilsson, L.; Zielke, C. The effect of in vitro gastrointestinal conditions on the structure and conformation of oat β-glucan. Food Hydrocoll. 2018, 77, 659–668. [Google Scholar] [CrossRef]
- Aktas-Akyildiz, E.; Sibakov, J.; Nappa, M.; Hytönen, E.; Koksel, H.; Poutanen, K. Extraction of soluble β-glucan from oat and barley fractions: Process efficiency and dispersion stability. J. Cereal Sci. 2018, 81, 60–68. [Google Scholar] [CrossRef]
- Konak, T.T.; Ercili-Cura, D.; Sibakov, J.; Sontag-Strohm, T.; Certel, M.; Loponen, J. CO2-defatted oats: Solubility, emulsification and foaming properties. J. Cereal Sci. 2014, 60, 37–41. [Google Scholar] [CrossRef]
- Shotwell, M.A. Oat Globulins. In Seed Proteins; Shewry, P.R., Casey, R., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1999; pp. 389–400. [Google Scholar]
- Colyer, T.E.; Luthe, D.S. Quantitation of oat globulin by radioimmunoassay. Plant Physiol. 1984, 74, 455–456. [Google Scholar] [CrossRef]
- Frey, K.J. The relation between alcohol-soluble and total nitrogen content of oats. Cereal Chem. 1951, 28, 506–509. [Google Scholar]
- Peterson, D.M.; Smith, D. Changes in nitrogen and carbohydrate fractions in developing oat groats. Crop Sci. 1976, 16, 67–71. [Google Scholar] [CrossRef]
- Cavazos, A.; Gonzalez de Mejia, E. Identification of bioactive peptides from cereal storage proteins and their potential role in prevention of chronic diseases. Compr. Rev. Food Sci. F. 2013, 12, 364–380. [Google Scholar] [CrossRef]
- Cheung, I.W.; Nakayama, S.; Hsu, M.N.; Samaranayaka, A.G.; Li-Chan, E.C. Angiotensin-I converting enzyme inhibitory activity of hydrolysates from oat (Avena sativa) proteins by in silico and in vitro analyses. J. Agric. Food Chem. 2009, 57, 9234–9242. [Google Scholar] [CrossRef]
- Gangopadhyay, N.; Hossain, M.B.; Rai, D.K.; Brunton, N.P. A review of extraction and analysis of bioactives in oat and barley and scope for use of novel food processing technologies. Molecules 2015, 20, 10884–10909. [Google Scholar] [CrossRef]
- Zevallos, V.F.; Raker, V.; Tenzer, S.; Jimenez-Calvente, C.; Ashfaq-Khan, M.; Rüssel, N.; Pickert, G.; Schild, H.; Steinbrink, K.; Schuppan, D. Nutritional wheat amylase-trypsin inhibitors promote intestinal inflammation via activation of myeloid cells. Gastroenterology 2017, 152, 1100–1113. [Google Scholar] [CrossRef]
- Junker, Y.; Zeissig, S.; Kim, S.-J.; Barisani, D.; Wieser, H.; Leffler, D.A.; Zevallos, V.F.; Libermann, T.A.; Freitag, T.L.; Kelly, C.P.; et al. Wheat amylase trypsin inhibitors drive intestinal inflammation via activation of toll-like receptor 4. J. Exp. Med. 2012, 209, 2395–2408. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Hettiarachchy, N.S.; Qi, M.; Burks, W.; Siebenmorgen, T. Preparation and functional properties of rice bran protein isolate. J. Agr. Food Chem. 1999, 47, 411–416. [Google Scholar] [CrossRef]
- Nivala, O.; Mäkinen, O.E.; Kruus, K.; Nordlund, E.; Dilek, E.-C. Structuring colloidal oat and faba bean protein particles via enzymatic modification. Food Chem. 2017, 231, 87–95. [Google Scholar] [CrossRef]
- Ma, C.Y.; Harwalkar, V.R. Chemical characterization and functionality assessment of oat protein fractions. J. Agric. Food Chem. 1984, 32, 144–149. [Google Scholar] [CrossRef]
- Acker, L.; Diemair, W.; Samhammer, E. The lichenin of oats. 1. Properties, preparation and composition of the muciparous polysaccharides. Z. Lebensm. Unters. For. 1955, 100, 180–188. [Google Scholar] [CrossRef]
- Zielke, C.; Kosik, O.; Ainalem, M.; Lovegrove, A.; Stradner, A.; Nilsson, L. Characterization of cereal β-glucan extracts from oat and barley and quantification of proteinaceous matter. PLoS ONE 2017, 12, e0172034. [Google Scholar] [CrossRef]
- Vasquez Mejia, S.M.; de Francisco, A.; Manique Barreto, P.L.; Mattioni, B.; Zibetti, A.W.; Molognoni, L.; Daguer, H. Physicochemical comparison of commercial vs. extracted β-glucans and structural characterization after enzymatic purification. [Comparación fisicoquímica entre β-glucanos comerciales y extraídos, y caracterización estructural después de purificación enzimática]. Vitae 2018, 25, 26–36. [Google Scholar]
- Lockhart, H.B.; Hurt, H.D. Nutrition of Oats. In Oats: Chemistry and Technology; Webster, F.H., Ed.; American Association of Cereal Chemists, Inc.: St. Paul, MN, USA, 1986; pp. 297–308. [Google Scholar]
- Youngs, V.L. Protein distribution in the oat kernel. Cereal Chem. 1972, 49, 407–411. [Google Scholar]
- Wood, P.J.; Weisz, J.; Fedec, P. Potential for b-glucan enrichment in brans derived from oat (Avena sativa L.) cultivars of different (1-3)(1-4)-b-d-glucan concentrations. Cereal Chem. 1991, 68, 48–51. [Google Scholar]
- Nnanna, I.A.; Gupta, S.V. Purification and partial characterization of oat bran globulin. J. Agr. Food Chem. 1996, 44, 3494–3499. [Google Scholar] [CrossRef]
- Walburg, G.; Larkins, B.A. Oat seed globulin: Subunit characterization and demonstration of its synthesis as a precursor. Plant Physiol. 1983, 72, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Brinegar, A.C.; Peterson, D.M. Separation and characterization of oat globulin polypeptides. Arch. Biochem. Biophys. 1982, 219, 71–79. [Google Scholar] [CrossRef]
- Nieto-Nieto, T.V.; Wang, Y.X.; Ozimek, L.; Chen, L. Effects of partial hydrolysis on structure and gelling properties of oat globular proteins. Food Res. Int. 2014, 55, 418–425. [Google Scholar] [CrossRef]
- Hardy, M.Y.; Tye-Din, J.A.; Stewart, J.A. Ingestion of oats and barley in patients with celiac disease mobilizes cross-reactive T cells activated by avenin peptides and immuno-dominant hordein peptides. J. Autoimmun. 2015, 56, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Arentz-Hansen, H.; Fleckenstein, B.; Molberg, Ø.; Scott, H.; Koning, F.; Günter, J.; Roepstorff, P.; Lundin, K.E.A.; Sollid, L.M. The molecular basis for oat intolerance in patients with Celiac Disease. PLoS Med. 2004, 1, e1. [Google Scholar] [CrossRef]
- Londono, D.M.; Van ’t Westende, W.P.C.; Goryunova, S.; Salentijn, E.M.J.; van den Broeck, H.C.; van der Meer, I.M.; Visser, R.G.F.; Gilissen, L.J.W.J.; Smulders, M.J.M. Avenin diversity analysis of the genus Avena (oat). Relevance for people with celiac disease. J. Cereal Sci. 2013, 58, 170–177. [Google Scholar] [CrossRef]
- Mitea, C.; Salentijn, E.M.J.; Van Veelen, P.; Goryunova, S.V.; van der Meer, I.M.; van den Broeck, H.C.; Mujico, J.R.; Montserrat, V.; Gilissen, L.J.W.J.; Wouter Drijfhout, J.; et al. A universal approach to eliminate antigenic properties of alpha-gliadin peptides in celiac disease. PLoS ONE 2010, 5, e15637. [Google Scholar] [CrossRef]
- Baakdah, M.M.; Tsopmo, A. Identification of peptides, metal binding and lipid peroxidation activities of HPLC fractions of hydrolyzed oat bran proteins. J. Food Sci. Tech. 2016, 53, 3593–3601. [Google Scholar] [CrossRef] [PubMed]
- Guan, X.; Yao, H. Optimization of viscozyme L-assisted extraction of oat bran protein using response surface methodology. Food Chem. 2008, 106, 345–351. [Google Scholar] [CrossRef]
- Tsopmo, A.; Cooper, A.; Jodayree, S. Enzymatic hydrolysis of oat flour protein isolates to enhance antioxidative properties. Adv. J. Food Sci. Tech. 2010, 4, 206–212. [Google Scholar]
- Jodayree, S.; Smith, J.C.; Tsopmo, A. Use of carbohydrase to enhance protein extraction efficiency and antioxidative properties of oat bran protein hydrolysates. Food Res. Int. 2012, 46, 69–75. [Google Scholar]
- Chen, J.; Tian, J.; Zheng, F. Effects of protein hydrolysis on pasting properties of wheat flour. Starch – Stärke 2012, 64, 524–530. [Google Scholar] [CrossRef]
- Plietz, P.; Zirwer, D.; Schlesier, B.; Gast, K.; Damaschun, G. Shape, symmetry, hydration and secondary structure of the legumin from Vicia faba in solution. Biochim. Biophys. Acta Protein Struct. Mol. Enzymol. 1984, 784, 140–146. [Google Scholar] [CrossRef]
- Immerstrand, T.; Bergenståhl, B.; Trägårdh, C.; Nyman, M.; Cui, S.; Öste, R. Extraction of β-glucan from oat bran in laboratory scale. Cereal Chem. 2009, 86, 601–608. [Google Scholar] [CrossRef]
- Autio, K.; Myllymaki, O.; Suortti, T.; Saastamoinen, M.; Poutanenm, K. Physical properties of (1-3),(1-4)- beta-d-glucan preparates isolated from finnish oat varieties. Food Hydrocoll. 1992, 5, 513–522. [Google Scholar] [CrossRef]
- Prosekov, A.; Babich, O.; Kriger, O.; Ivanova, S.; Pavsky, V.; Sukhikh, S.; Yang, Y.; Kashirskih, E. Functional properties of the enzyme-modified protein from oat bran. Food Biosci. 2018, 24, 46–49. [Google Scholar] [CrossRef]
- Brückner-Gühmann, M.; Heiden-Hecht, T.; Sözer, N.; Drusch, S. Foaming characteristics of oat protein and modification by partial hydrolysis. Eur. Food Res. Tech. 2018, 244, 2095–2106. [Google Scholar] [CrossRef]
- Harasym, J.; Brach, J.; Czarnota, J.L.; Stechman, M.; Słabisz, A.; Kowalska, A.; Chorowski, M.; Winkowski, M.; Rać, J.; Madera, A. A kit and a method of producing beta-glucan, insoluble food fibre as well as a preparation of oat proteins. European Patent EP2515672B1, 6 July 2016. [Google Scholar]
- Harasym, J.; Gromadzka-Ostrowska, J. Method for Obtaining Cereal Beta-Glucan with Low Molecular Weight, PL226915, 2017. Available online: https://grab.uprp.pl/PrzedmiotyChronione/Strony%20witryny/Wyszukiwanie%20proste.aspx (accessed on 5 April 2019).
Sample Availability: Samples of the compounds are not available from the authors. |
| Sample | Starch (%/db) | Glucose (%/db) | ||
|---|---|---|---|---|
| Treatment | HM | LM | HM | LM |
| C | 15.55 ± 0.21 D, b | 9.26 ± 0.35 D, a | 3.70 ± 0.12 D, b | 1.60 ± 0.14 C, a |
| PC | 9.19 ± 0.89 C, b | 5.69 ± 0.33 C, a | 3.45 ± 0.03 D, b | 1.05 ± 0.06 B, a |
| T | 1.65 ± 0.07 B, b | 0.65 ± 0.06 B, a | 1.25 ± 0.01 A, b | 0.35 ± 0.07 A, a |
| A | 0.45 ± 0.04 A, b | 0.25 ± 0.03 A, a | 1.20 ± 0.14 A, b | 0.30 ± 0.14 A, a |
| S | nd | nd | 2.50 ± 0.14 BC, b | 1.50 ± 0.28 C, a |
| AA | nd | nd | 2.75 ± 0.02 C, b | 1.15 ± 0.09 B, a |
| P | nd | nd | 2.55 ± 0.04 BC, b | 0.95 ± 0.03 B, a |
| AAA | nd | nd | 2.40 ± 0.28 B, b | 0.50 ± 0.14 A, a |
| Second order interactions p values | ||||
| Sample | *** | *** | ||
| Treatment | *** | *** | ||
| Sample x treatment | *** | *** | ||
| C | 15.55 ± 0.21 D | 9.26 ± 0.35 D | 3.70 ± 0.14 C | 1.60 ± 0.14 C |
| PC | 9.19 ± 0.89 C | 5.69 ± 0.33 C | 3.45 ± 0.07 C | 1.05 ± 0.07 B |
| T | 1.65 ± 0.07 B | 0.65 ± 0.07 B | 1.25 ± 0.07 A | 0.35 ± 0.07 A |
| A | 0.45 ± 0.07 A | 0.25 ± 0.07 AB | 1.20 ± 0.14 A | 0.30 ± 0.14 A |
| S | nd | nd | 2.50 ± 0.14 B | 1.50 ± 0.28 C |
| Second order interactions p values | ||||
| Sample | *** | *** | ||
| Treatment | *** | *** | ||
| Sample x Treatment | *** | *** | ||
| Sample | Soluble Proteins (%/db) | Nitrogen x 5,83 (%/db) | ||
|---|---|---|---|---|
| Treatment | HM | LM | HM | LM |
| C | 5.85 ± 0.07 E, b | 3.30 ± 0.14 D, a | 6.16 ± 0.13 G, b | 3.94 ± 0.04 F, a |
| PC | 2.25 ± 0.21 D, b | 1.00 ± 0.14 C, a | 3.94 ± 0.04 F, b | 3.21 ± 0.08 E, a |
| T | 1.70 ± 0.14 C, b | 0.45 ± 0.07 B, a | 2.71 ± 0.04 E, b | 1.93 ± 0.08 D, a |
| A | 0.70 ± 0.14 B, b | 0.09 ± 0.01 A, a | 1.17 ± 0.08 D, a | 1.14 ± 0.12 C, a |
| S | 0.04 ± 0.01 A, a | 0.02 ± 0.01 A, a | 0.61 ± 0.04 C, b | 0.47 ± 0.08 B, a |
| AA | 0.01 ± 0.00 A | nd | 0.41 ± 0.00 B, b | 0.02 ± 0.01 A, a |
| P | 0.01 ± 0.00 A, a | 0.01 ± 0.00 A, a | 0.03 ± 0.01 A, b | 0.01 ± 0.00 A, a |
| AAA | 0.01 ± 0.00 A | nd | 0.01 ± 0.00 A | nd |
| Second order interactions p value | ||||
| Sample | *** | *** | *** | *** |
| Treatment | *** | *** | *** | *** |
| Sample x treatment | *** | *** | *** | *** |
| C | 5.85 ± 0.07 E | 3.30 ± 0.14 D | 5.16 ± 0.13 E | 3.94 ± 0.04 E |
| PC | 2.25 ± 0.21 D | 1.00 ± 0.14 C | 3.94 ± 0.04 D | 3.21 ± 0.08 D |
| T | 1.70 ± 0.14 C | 0.45 ± 0.07 B | 2.71 ± 0.04 C | 1.93 ± 0.08 C |
| A | 0.70 ± 0.14 B | 0.09 ± 0.01 A | 1.17 ± 0.08 B | 1.14 ± 0.12 B |
| S | 0.04 ± 0.01 A | 0.02 ± 0.00 A | 0.61 ± 0.04 A | 0.47 ± 0.08 A |
| Second order interactions p value | ||||
| Sample | *** | *** | *** | *** |
| Treatment | *** | *** | *** | *** |
| Sample x treatment | *** | *** | *** | *** |
| Sample | β-Glucan (%/db) | Molar Mass (g/mol) | ||
|---|---|---|---|---|
| Treatment | HM | LM | HM | LM |
| C | 76.75 ± 0.21 A, a | 87.10 ± 0.28 A, b | 2054520 ± 77342.6 E | 72260.0 ± 3258.4 E |
| PC | 79.75 ± 0.92 B, a | 91.35 ± 0.64 B, b | 2042120 ± 73876.4 E | 67166.0 ± 410.1 D |
| T | 91.20 ± 1.27 C, a | 95.30 ± 1.41 C, b | 1776840 ± 31325.5 D | 65877.5 ± 190.2 CD |
| A | 95.40 ± 0.28 D, a | 98.15 ± 1.06 D, b | 1759600 ± 7030.8 D | 61731.5 ± 2395.0 C |
| S | 97.50 ± 0.42 E, a | 99.25 ± 0.35 D, b | 1705400 ± 142.8 D | 61486.0 ± 2118.5 C |
| AA | 98.20 ± 0.14 E, a | 99.25 ± 0.35 D, b | 987284 ± 1003.4 C | 21100.5 ± 1101.0 B |
| P | 98.05 ± 0.21 E, a | 99.40 ± 0.14 D, b | 752639 ± 5236.8 B | 14888.0 ± 2531.4 A |
| AAA | 97.95 ± 0.07 E, a | 99.00 ± 0.14 D, b | 333128 ± 17920.9 A | 11924.0 ± 1303.9 A |
| Second order interactions p value | ||||
| Sample | *** | *** | ||
| Treatment | *** | *** | ||
| Sample x Treatment | *** | *** | ||
| C | 76.75 ± 0.21 A | 87.10 ± 0.28 A | 2054520 ± 77342.6 B | 72260.0 ± 3258.4 C |
| PC | 79.75 ± 0.92 B | 90.35 ± 0.64 B | 2042120 ± 73876.4 B | 67166.0 ± 410.1B C |
| T | 91.20 ± 1.27 C | 95.30 ± 1.41 C | 1776840 ± 31325.5 A | 65877.5 ± 190.2A B |
| A | 95.40 ± 0.28 D | 98.15 ± 1.06 D | 1759600 ± 7030.8 A | 61731.5 ± 2395.0 A |
| S | 97.50 ± 0.42 E | 99.25 ± 0.35 D | 1705400 ± 142.8 A | 61486.0 ± 2118.5 A |
| Second order interactions p value | ||||
| Sample | *** | *** | ||
| Treatment | *** | *** | ||
| Sample x Treatment | *** | *** | ||
| Starch | Glucose | Proteins | Amine N | β-Glucan | Molar Mass | ||
|---|---|---|---|---|---|---|---|
| Starch | HM | - | 0.96 *** | 0.93 *** | −0.97 *** | - | |
| LM | - | 0.95 *** | 0.92 ** | −0.97 *** | - | ||
| Glucose | HM | - | - | - | - | - | |
| LM | - | - | - | - | - | ||
| Proteins | HM | 0.99 ** | - | 0.94 *** | −0.92 ** | - | |
| LM | - | - | 0.86 ** | −0.92 ** | - | ||
| Amine N | HM | - | - | 0.95 * | −0.98 *** | 0.79 * | |
| LM | - | - | - | −0.98 *** | 0.79 * | ||
| β-glucan | HM | −0.97 * | - | 0.98 * | −0.98 * | −0.71 * | |
| LM | - | - | - | −0.96 * | - | ||
| Molar Mass | HM | - | - | - | - | - | |
| LM | - | - | - | 0.98 * | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harasym, J.; Żyła, E.; Dziendzikowska, K.; Gromadzka-Ostrowska, J. Proteinaceous Residue Removal from Oat β-Glucan Extracts Obtained by Alkaline Water Extraction. Molecules 2019, 24, 1729. https://doi.org/10.3390/molecules24091729
Harasym J, Żyła E, Dziendzikowska K, Gromadzka-Ostrowska J. Proteinaceous Residue Removal from Oat β-Glucan Extracts Obtained by Alkaline Water Extraction. Molecules. 2019; 24(9):1729. https://doi.org/10.3390/molecules24091729
Chicago/Turabian StyleHarasym, Joanna, Ewa Żyła, Katarzyna Dziendzikowska, and Joanna Gromadzka-Ostrowska. 2019. "Proteinaceous Residue Removal from Oat β-Glucan Extracts Obtained by Alkaline Water Extraction" Molecules 24, no. 9: 1729. https://doi.org/10.3390/molecules24091729









