Comprehensive Investigation of the Effects of Brewing Conditions in Sample Preparation of Green Tea Infusions
Abstract
:1. Introduction
2. Results and Discussion
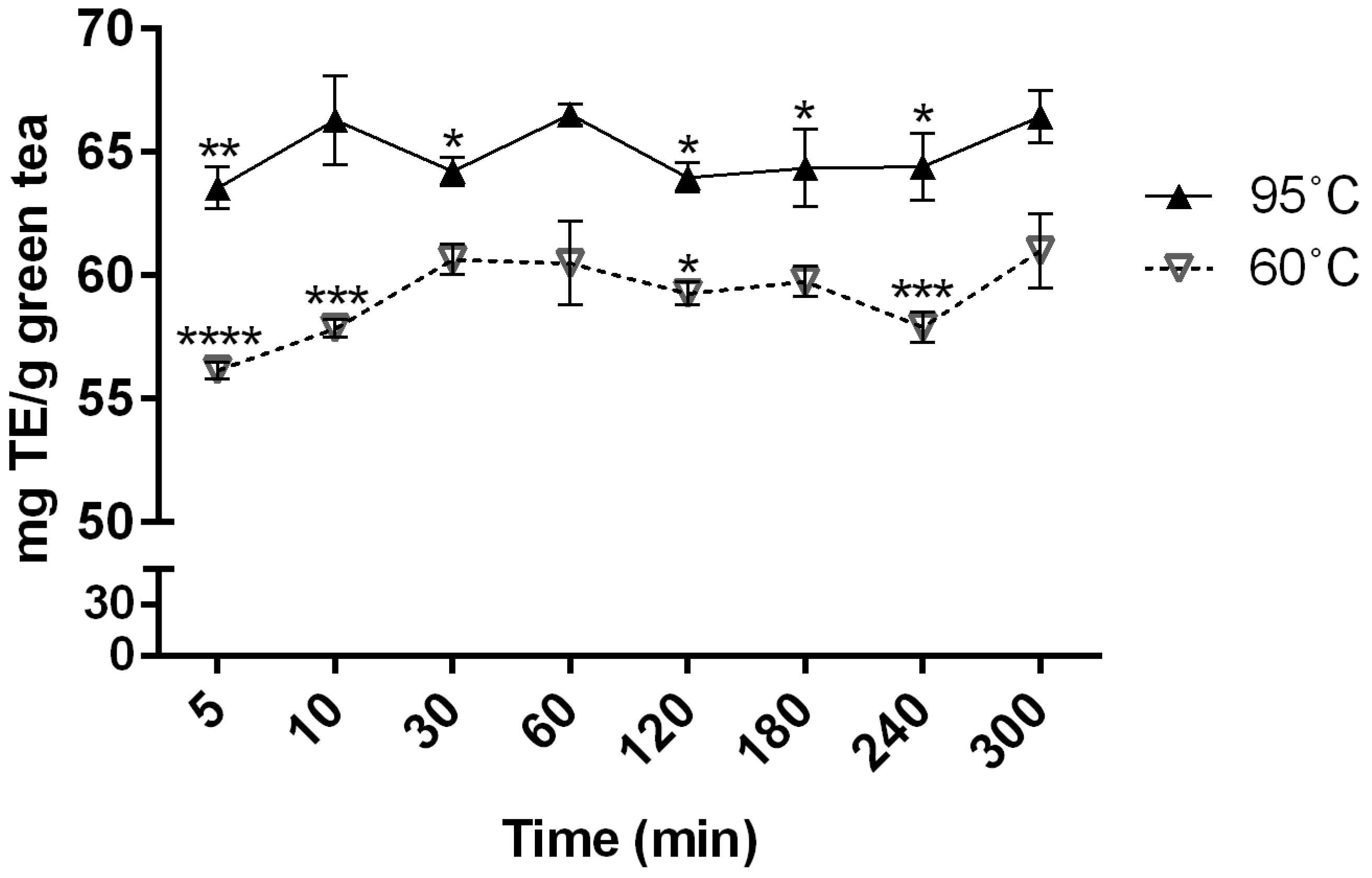
2.1. Effects of Brewing Conditions on Antioxidant Activity
2.2. Effects of Brewing Conditions on Metabolic Profiles—Investigation via PCA and OPLS-DA Studies
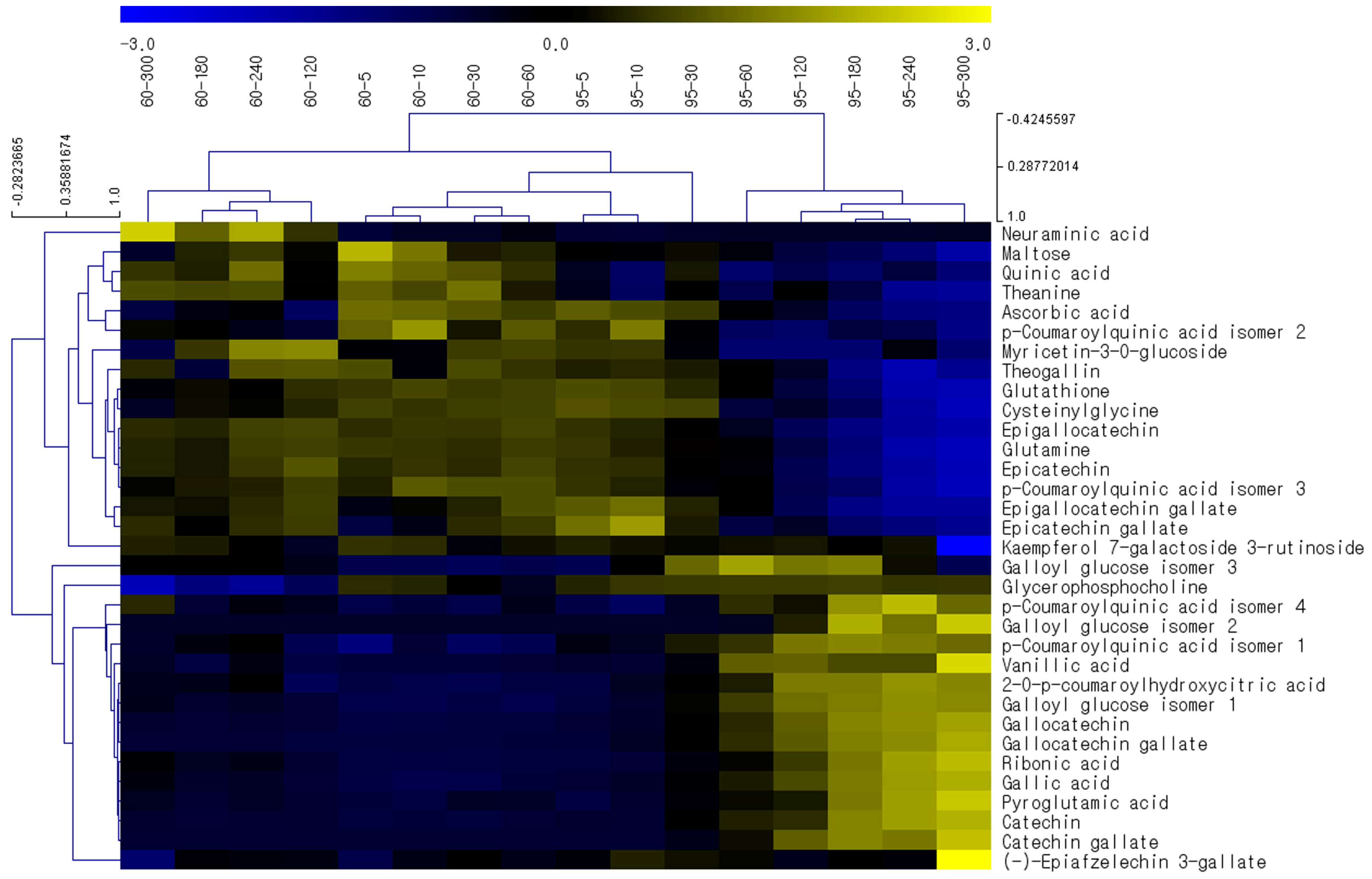
2.3. Effects of Brewing Conditions on Metabolic Profiles—Investigation via HCA Studies
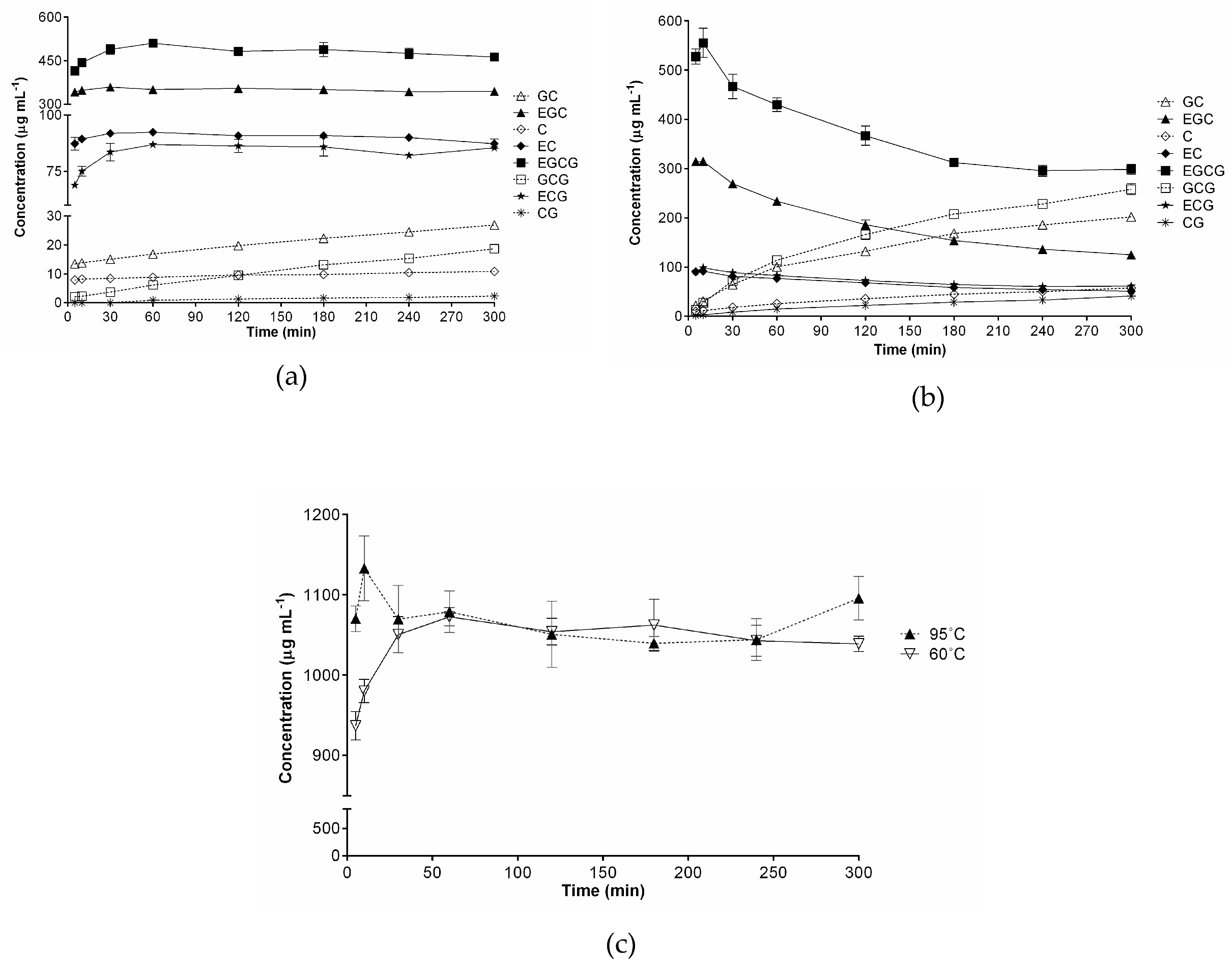
2.4. Effects of Brewing Conditions on Catechin Compositions
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Preparation of Tea Infusions
3.3. Antioxidant Assay
3.4. Instruments and Operation Conditions
3.5. Data Processing and Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Pastoriza, S.; Mesías, M.; Cabrera, C.; Rufián-Henares, J. Healthy properties of green and white teas: An update. Food Funct. 2017, 8, 2650–2662. [Google Scholar] [CrossRef] [PubMed]
- Saeed, M.; Naveed, M.; Arif, M.; Kakar, M.U.; Manzoor, R.; El-Hack, M.E.A.; Alagawany, M.; Tiwari, R.; Khandia, R.; Munjal, A. Green tea (Camellia sinensis) and l-theanine: Medicinal values and beneficial applications in humans—A comprehensive review. Biomed. Pharmacother. 2017, 95, 1260–1275. [Google Scholar] [CrossRef] [PubMed]
- Cabrera, C.; Artacho, R.; Giménez, R. Beneficial effects of green tea—A review. J. Am. Coll. Nutr. 2006, 25, 79–99. [Google Scholar] [CrossRef] [PubMed]
- Roychoudhury, S.; Agarwal, A.; Virk, G.; Cho, C.-L. Potential role of green tea catechins in the management of oxidative stress-associated infertility. Reprod. Biomed. Online 2017, 34, 487–498. [Google Scholar] [CrossRef] [PubMed]
- Fujiki, H.; Sueoka, E.; Rawangkan, A.; Suganuma, M. Human cancer stem cells are a target for cancer prevention using (−)-epigallocatechin gallate. J. Cancer Res. Clin. Oncol. 2017, 143, 2401–2412. [Google Scholar] [CrossRef]
- Mrabti, H.N.; Jaradat, N.; Fichtali, I.; Ouedrhiri, W.; Jodeh, S.; Ayesh, S.; Cherrah, Y.; Faouzi, M.E.A. Separation, Identification, and Antidiabetic Activity of Catechin Isolated from Arbutus unedo L. Plant Roots. Plants 2018, 7, 31. [Google Scholar] [CrossRef]
- Rashidi, B.; Malekzadeh, M.; Goodarzi, M.; Masoudifar, A.; Mirzaei, H. Green tea and its anti-angiogenesis effects. Biomed. Pharmacother. 2017, 89, 949–956. [Google Scholar] [CrossRef] [PubMed]
- Balentine, D.A.; Wiseman, S.A.; Bouwens, L.C. The chemistry of tea flavonoids. Crit. Rev. Plant Sci. 1997, 37, 693–704. [Google Scholar] [CrossRef] [PubMed]
- Harbowy, M.E.; Balentine, D.A.; Davies, A.P.; Cai, Y. Tea chemistry. Crit. Rev. Plant Sci. 1997, 16, 415–480. [Google Scholar] [CrossRef]
- Dai, W.; Qi, D.; Yang, T.; Lv, H.; Guo, L.; Zhang, Y.; Zhu, Y.; Peng, Q.; Xie, D.; Tan, J. Nontargeted analysis using ultraperformance liquid chromatography–quadrupole time-of-flight mass spectrometry uncovers the effects of harvest season on the metabolites and taste quality of tea (Camellia sinensis L.). J. Agric. Food Chem. 2015, 63, 9869–9878. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-E.; Lee, B.-J.; Chung, J.-O.; Kim, H.-N.; Kim, E.-H.; Jung, S.; Lee, H.; Lee, S.-J.; Hong, Y.-S. Metabolomic unveiling of a diverse range of green tea (Camellia sinensis) metabolites dependent on geography. Food Chem. 2015, 174, 452–459. [Google Scholar] [CrossRef]
- Han, Z.-X.; Rana, M.M.; Liu, G.-F.; Gao, M.-J.; Li, D.-X.; Wu, F.-G.; Li, X.-B.; Wan, X.-C.; Wei, S. Green tea flavour determinants and their changes over manufacturing processes. Food Chem. 2016, 212, 739–748. [Google Scholar] [CrossRef]
- Dai, Q.; He, Y.; Ho, C.-T.; Wang, J.; Wang, S.; Yang, Y.; Gao, L.; Xia, T. Effect of interaction of epigallocatechin gallate and flavonols on color alteration of simulative green tea infusion after thermal treatment. J. Food Sci. Technol. 2017, 54, 2919–2928. [Google Scholar] [CrossRef]
- Liu, S.; Ai, Z.; Qu, F.; Chen, Y.; Ni, D. Effect of steeping temperature on antioxidant and inhibitory activities of green tea extracts against α-amylase, α-glucosidase and intestinal glucose uptake. Food Chem. 2017, 234, 168–173. [Google Scholar] [CrossRef]
- Lantano, C.; Rinaldi, M.; Cavazza, A.; Barbanti, D.; Corradini, C. Effects of alternative steeping methods on composition, antioxidant property and colour of green, black and oolong tea infusions. J. Food Sci. Technol. 2015, 52, 8276–8283. [Google Scholar] [CrossRef]
- Lee, J.; Chambers, D. Sensory descriptive evaluation: brewing methods affect flavour of green tea. Asian J. Food Agro-Ind. 2009, 2, 427–439. [Google Scholar]
- Fernando, C.D.; Soysa, P. Extraction Kinetics of phytochemicals and antioxidant activity during black tea (Camellia sinensis L.) brewing. Nutr. J. 2015, 14, 74. [Google Scholar] [CrossRef]
- Kelebek, H. LC-DAD–ESI-MS/MS characterization of phenolic constituents in Turkish black tea: Effect of infusion time and temperature. Food Chem. 2016, 204, 227–238. [Google Scholar] [CrossRef]
- Pérez-Burillo, S.; Giménez, R.; Rufián-Henares, J.; Pastoriza, S. Effect of brewing time and temperature on antioxidant capacity and phenols of white tea: Relationship with sensory properties. Food Chem. 2018, 248, 111–118. [Google Scholar] [CrossRef]
- Saklar, S.; Ertas, E.; Ozdemir, I.S.; Karadeniz, B. Effects of different brewing conditions on catechin content and sensory acceptance in Turkish green tea infusions. J. Food Sci. Technol. 2015, 52, 6639–6646. [Google Scholar] [CrossRef] [Green Version]
- Li, N.; Taylor, L.S.; Mauer, L.J. Degradation kinetics of catechins in green tea powder: effects of temperature and relative humidity. J. Agric. Food Chem. 2011, 59, 6082–6090. [Google Scholar] [CrossRef]
- Wang, H.; Helliwell, K. Epimerisation of catechins in green tea infusions. Food Chem. 2000, 70, 337–344. [Google Scholar] [CrossRef]
- Jing, J.; Shi, Y.Z.; Zhang, Q.F.; Wang, J.; Ruan, J.Y. Prediction of Chinese green tea ranking by metabolite profiling using ultra-performance liquid chromatography-quadrupole time-of-flight mass spectrometry (UPLC-Q-TOF/MS). Food Chem. 2017, 221, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Li, P.L.; Dai, W.D.; Lu, M.L.; Xie, D.C.; Tan, J.F.; Yang, C.; Zhu, Y.; Lv, H.P.; Peng, Q.H.; Zhang, Y.; et al. Metabolomic analysis reveals the composition differences in 13 Chinese tea cultivars of different manufacturing suitabilities. J. Sci. Food Agric. 2018, 98, 1153–1161. [Google Scholar] [CrossRef]
- Kellogg, J.J.; Graf, T.N.; Paine, M.F.; McCune, J.S.; Kvalheim, O.M.; Oberlies, N.H.; Cech, N.B. Comparison of Metabolomics Approaches for Evaluating the Variability of Complex Botanical Preparations: Green Tea (Camellia sinensis) as a Case Study. J. Nat. Prod. 2017, 80, 1457–1466. [Google Scholar] [CrossRef]
- Ku, K.M.; Choi, J.N.; Kim, J.; Kim, J.K.; Yoo, L.G.; Lee, S.J.; Hong, Y.S.; Lee, C.H. Metabolomics Analysis Reveals the Compositional Differences of Shade Grown Tea (Camellia sinensis L.). J. Agric. Food Chem. 2010, 58, 418–426. [Google Scholar] [CrossRef]
- Nam, M.; Choi, M.S.; Choi, J.Y.; Kim, N.; Kim, M.S.; Jung, S.; Kim, J.; Ryu, D.H.; Hwang, G.S. Effect of green tea on hepatic lipid metabolism in mice fed a high-fat diet. J. Nutr. Biochem. 2018, 51, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Li, X.L.; Yang, H.C.; Wang, F.; Kong, J.P.; Qiu, D.; Li, Z. Mass spectrometry-based metabolomics and chemometric analysis of Pu-erh teas of various origins. Food Chem. 2018, 268, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Li, N.; Ma, Z.Z.; Tu, P.F. Comparison of the Chemical Constituents of Aged Pu-erh Tea, Ripened Pu-erh Tea, and Other Teas Using HPLC-DAD-ESI-MSn. J. Agric. Food Chem. 2011, 59, 8754–8760. [Google Scholar] [CrossRef]
- Amorati, R.; Valgimigli, L. Advantages and limitations of common testing methods for antioxidants. Free Radic. Res. 2015, 49, 633–649. [Google Scholar] [CrossRef] [PubMed]
- Sreekumar, A.; Poisson, L.M.; Rajendiran, T.M.; Khan, A.P.; Cao, Q.; Yu, J.D.; Laxman, B.; Mehra, R.; Lonigro, R.J.; Li, Y.; et al. Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature 2009, 457, 910–914. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kocadağlı, T.; Özdemir, K.S.; Gökmen, V. Effects of infusion conditions and decaffeination on free amino acid profiles of green and black tea. Food Res. Int. 2013, 53, 720–725. [Google Scholar] [CrossRef]
- Zhang, H.; Li, Y.; Lv, Y.; Jiang, Y.; Pan, J.; Duan, Y.; Zhu, Y.; Zhang, S. Influence of brewing conditions on taste components in Fuding white tea infusions. J. Sci. Food Agric. 2017, 97, 2826–2833. [Google Scholar] [CrossRef] [PubMed]
- Labbé, D.; Tremblay, A.; Bazinet, L. Effect of brewing temperature and duration on green tea catechin solubilization: Basis for production of EGC and EGCG-enriched fractions. Sep. Purif. Technol. 2006, 49, 1–9. [Google Scholar] [CrossRef]
- Perva-Uzunalić, A.; Škerget, M.; Knez, Ž.; Weinreich, B.; Otto, F.; Grüner, S. Extraction of active ingredients from green tea (Camellia sinensis): Extraction efficiency of major catechins and caffeine. Food Chem. 2006, 96, 597–605. [Google Scholar] [CrossRef]
- Abourashed, E.A.; Roberson, C.L.A.; Elsharkawy, N. Content variation of catechin markers, total phenolics and caffeine in green tea dietary supplements. J. Diet. Suppl. 2016, 13, 171–184. [Google Scholar] [CrossRef]
- He, X.; Li, J.; Zhao, W.; Liu, R.; Zhang, L.; Kong, X. Chemical fingerprint analysis for quality control and identification of Ziyang green tea by HPLC. Food Chem. 2015, 171, 405–411. [Google Scholar] [CrossRef]
- Komes, D.; Horzic, D.; Belscak, A.; Ganic, K.K.; Vulic, I. Green tea preparation and its influence on the content of bioactive compounds. Food Res. Int. 2010, 43, 167–176. [Google Scholar] [CrossRef]
- Yoo, D.E.; Jeong, K.M.; Han, S.Y.; Kim, E.M.; Jin, Y.; Lee, J. Deep eutectic solvent-based valorization of spent coffee grounds. Food Chem. 2018, 255, 357–364. [Google Scholar] [CrossRef]
- Zhao, J.; Jin, Y.; Shin, Y.; Jeong, K.M.; Lee, J. Indirect enantioseparation of fluoxetine in mouse serum by derivatization with 1R-(–)-menthyl chloroformate followed by ultra high performance liquid chromatography and quadrupole time-of-flight mass spectrometry. JSS 2016, 39, 1041–1049. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds may be available from the authors. |
| No. | tR (min) | Compounds | Formula | Monoisotopic Molecular Mass | Detected Mass | Mass Error (mDa) |
|---|---|---|---|---|---|---|
| 1 | 0.79 | Glutamine 1 | C5H10N2O3 | 146.0691 | 147.0761 a | 0.8 |
| 2 | 0.79 | Glycerophosphocholine 1 | C8H20NO6P | 257.1028 | 258.1079 a | 2.7 |
| 3 | 0.8 | Ribonic acid 2 | C5H10O6 | 166.0477 | 165.0399 b | 0.0 |
| 4 | 0.8 | Maltose 1 | C12H22O11 | 342.1162 | 341.1081 b | 0.3 |
| 5 | 0.81 | Quinic acid 1 | C7H12O6 | 192.0634 | 191.0555 b | 0.1 |
| 6 | 0.83 | Ascorbic acid 2 | C6H8O6 | 176.0321 | 175.0241 b | 0.2 |
| 7 | 0.83 | Glutathione 2 | C10H17N3O6S | 307.0838 | 308.0853 a | 6.3 |
| 8 | 0.84 | Galloyl glucose isomer 1 2, d | C13H16O10 | 332.0743 | 331.0660 b | 0.5 |
| 9 | 0.84 | 2-O-p-Coumaroylhydroxycitric acid 2 | C15H14O10 | 354.0587 | 355.0668 a | −0.3 |
| 10 | 1.15 | Theanine 1 | C7H14N2O3 | 174.1004 | 173.0922 b | 0.4 |
| 11 | 1.15 | Cysteinylglycine 2 | C5H10N2O3S | 178.0412 | 179.0480 a | 1.0 |
| 12 | 1.15 | Galloyl glucose isomer 2 2, d | C13H16O10 | 332.0743 | 331.0663 b | 0.2 |
| 13 | 1.19 | Pyroglutamic acid 2 | C5H7NO3 | 129.0426 | 128.0350 b | −0.2 |
| 14 | 1.19 | Neuraminic acid 2 | C9H17NO8 | 267.0954 | 268.1043 a | −1.1 |
| 15 | 1.4 | Galloyl glucose isomer 3 2, d | C13H16O10 | 332.0743 | 331.0661 b | 0.4 |
| 16 | 1.64 | Theogallin 2 | C14H16O10 | 344.0743 | 345.0809 a | 1.2 |
| 17 | 1.69 | Gallic acid 1 | C7H6O5 | 170.0215 | 171.0285 a; 169.0135 b | 0.8; 0.2 |
| 18 | 2.19 | Vanillic acid 2 | C8H8O4 | 168.0423 | 169.0497 a | 0.4 |
| 19 | 2.25 | Gallocatechin 1 | C15H14O7 | 306.0740 | 307.0813 a; 305.0660 b | 0.5; 0.2 |
| 20 | 3.16 | Epigallocatechin 1 | C15H14O7 | 306.0740 | 307.0815 a; 305.0663 b | 0.3; −0.1 |
| 21 | 3.44 | p-Coumaroylquinic acid isomer 1 2, d | C16H18O8 | 338.1002 | 361.0886 c; 337.0925 b | 1.4; −0.1 |
| 22 | 3.78 | Catechin 1 | C15H14O6 | 290.0790 | 291.0863 a; 289.0711 b | 0.5; 0.1 |
| 23 | 4.9 | p-Coumaroylquinic acid isomer 2 2, d | C16H18O8 | 338.1002 | 361.0897 c; 337.0923 b | 0.3; 0.1 |
| 24 | 5.36 | Epicatechin 1 | C15H14O6 | 290.0790 | 291.0867 a; 289.0712 b | 0.1; 0.0 |
| 25 | 5.57 | p-Coumaroylquinic acid isomer 3 2, d | C16H18O8 | 338.1002 | 361.0889 c; 337.0922 b | 1.1; 0.2 |
| 26 | 5.66 | Epigallocatechin gallate 1 | C22H18O11 | 458.0849 | 459.0921 a; 457.0766 b | 0.6; 0.5 |
| 27 | 6.43 | Gallocatechin gallate 1 | C22H18O11 | 458.0849 | 459.0920 a; 457.0767 b | 0.7; 0.4 |
| 28 | 7.03 | p-Coumaroylquinic acid isomer 4 2, d | C16H18O8 | 338.1002 | 361.0905 c; 337.0941 b | −0.5; −1.7 |
| 29 | 7.33 | Myricetin-3-O-glucoside 2 | C21H20O13 | 480.0904 | 479.0814 b | 1.2 |
| 30 | 9.42 | Epicatechin gallate 1 | C22H18O10 | 442.0900 | 443.0972 a; 441.0814 b | 0.6; 0.8 |
| 31 | 9.8 | Catechin gallate 1 | C22H18O10 | 442.0900 | 443.0977 a; 441.0824 b | 0.1; −0.2 |
| 32 | 10.39 | Kaempferol 7-galactoside 3-rutinoside 2 | C33H40O20 | 756.2113 | 755.2025 b | 1.0 |
| 33 | 11.61 | (−)-Epiafzelechin 3-gallate 2 | C22H18O9 | 426.0951 | 425.0863 b | 1.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, Y.; Zhao, J.; Kim, E.M.; Kim, K.H.; Kang, S.; Lee, H.; Lee, J. Comprehensive Investigation of the Effects of Brewing Conditions in Sample Preparation of Green Tea Infusions. Molecules 2019, 24, 1735. https://doi.org/10.3390/molecules24091735
Jin Y, Zhao J, Kim EM, Kim KH, Kang S, Lee H, Lee J. Comprehensive Investigation of the Effects of Brewing Conditions in Sample Preparation of Green Tea Infusions. Molecules. 2019; 24(9):1735. https://doi.org/10.3390/molecules24091735
Chicago/Turabian StyleJin, Yan, Jing Zhao, Eun Mi Kim, Ki Hyun Kim, Seulgi Kang, Heesoo Lee, and Jeongmi Lee. 2019. "Comprehensive Investigation of the Effects of Brewing Conditions in Sample Preparation of Green Tea Infusions" Molecules 24, no. 9: 1735. https://doi.org/10.3390/molecules24091735






