Creosote Bush (Larrea tridentata) Extract Assessment as a Green Antioxidant for Biodiesel
Abstract
:1. Introduction
2. Results
2.1. Extract Obtaining
2.2. Antioxidant Properties of the Extract
2.3. Biodiesel Preparation and Characterization
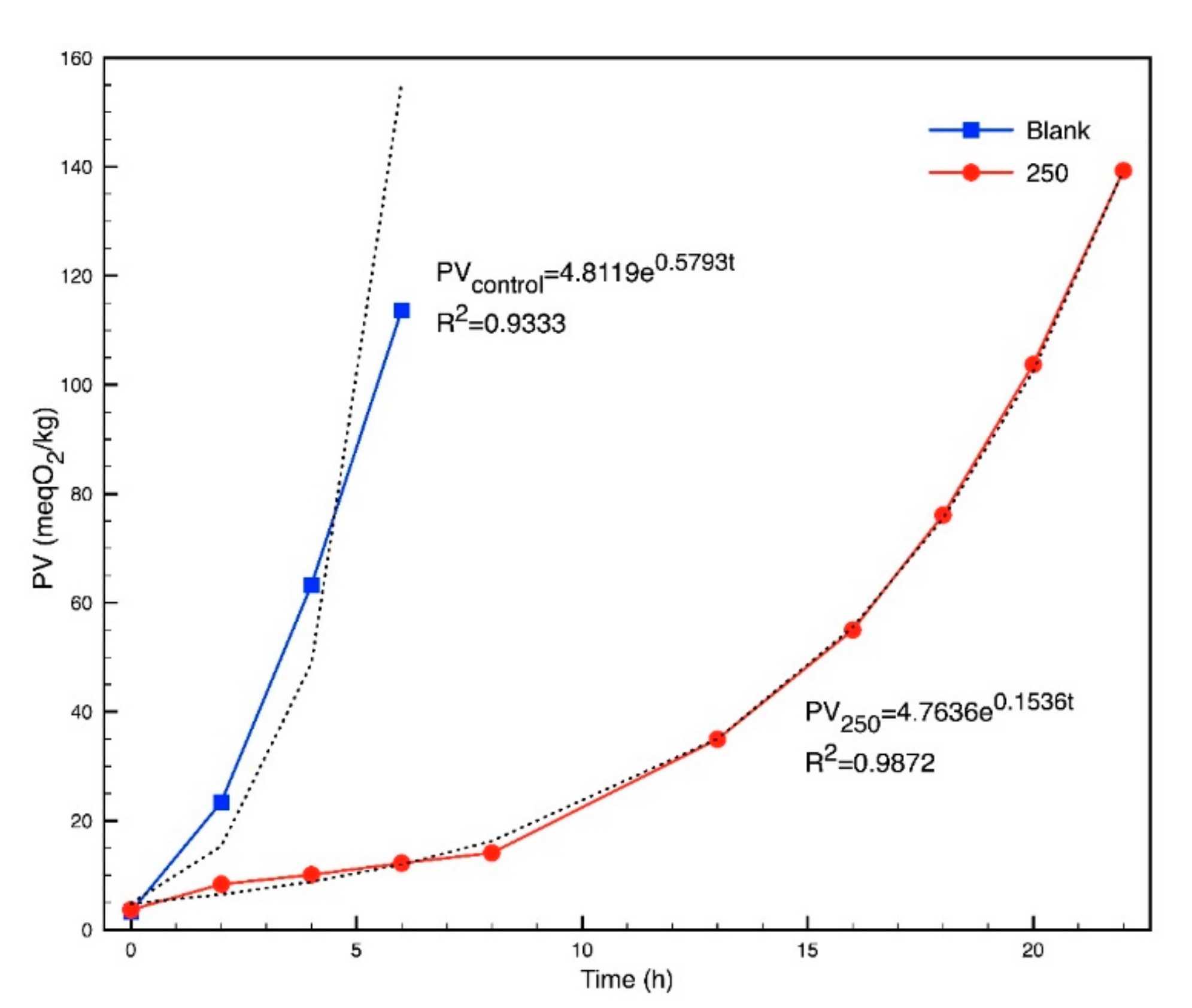
2.4. Oxidative Stability of Biodiesel
3. Materials and Methods
3.1. Extract Obtaining
3.1.1. Plant Material Obtaining
3.1.2. Maceration
3.2. Antioxidant Properties of the Extract
3.2.1. Determination of Phenolic Acids by HPLC
3.2.2. Determination of the Free Radical Scavenging Capacity Through a DPPH Assay
3.2.3. Capacity of Ferric Reduction Through FRAP Assay
3.2.4. Capacity of Absorption of Oxygen Radicals Through ORAC Assay
3.2.5. Total Phenols
3.3. Biodiesel Preparation and Characterization
3.3.1. Biodiesel Preparation
3.3.2. GC-MS
3.4. Oxidative Stability of Biodiesel
3.4.1. Peroxide Value (PV)
3.4.2. Higher Heating Value
3.4.3. Oxidative Stability Index
Effect of Temperature on OSI
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chew, K.; Haseeb, A.; Masjuki, H.; Fazal, M.; Gupta, M. Corrosion of magnesium and aluminum in palm biodiesel: A comparative evaluation. Energy 2013, 57, 478–483. [Google Scholar] [CrossRef]
- Coronado, M.; Montero, G.; García, C.; Valdez, B.; Ayala, R.; Perez, A. Quality Assessment of Biodiesel Blends Proposed by the New Mexican Policy Framework. Energies 2017, 10, 631. [Google Scholar] [CrossRef]
- Coronado, M.; Montero, G.; Valdez, B.; Stoytcheva, M.; Eliezer, A.; García, C.; Campbell, H.; Pérez, A. Degradation of nitrile rubber fuel hose by biodiesel use. Energy 2014, 68, 364–369. [Google Scholar] [CrossRef]
- Fazal, M.; Haseeb, A.; Masjuki, H. Comparative corrosive characteristics of petroleum diesel and palm biodiesel for automotive materials. Fuel Process. Technol. 2010, 91, 1308–1315. [Google Scholar] [CrossRef]
- Yang, J.; He, Q.; Corscadden, K.; Caldwell, C. Improvement on oxidation and storage stability of biodiesel derived from an emerging feedstock camelina. Fuel Process. Technol. 2017, 157, 90–98. [Google Scholar] [CrossRef]
- Saluja, R.; Kumar, V.; Sham, R. Stability of biodiesel—A review. Renew. Sust. Energ. Rev. 2016, 62, 866–881. [Google Scholar] [CrossRef]
- Park, J.-Y.; Kim, D.-K.; Lee, J.-P.; Park, S.-C.; Kim, Y.-J.; Lee, J.-S. Blending effects of biodiesels on oxidation stability and low temperature flow properties. Bioresource Technol. 2008, 99, 1196–1203. [Google Scholar] [CrossRef]
- Knothe, G.; Razon, L. Biodiesel fuels. Prog. Energ. Combust. 2017, 58, 36–59. [Google Scholar] [CrossRef]
- Dunn, R. Oxidative stability of biodiesel by dynamic mode pressurized−differential scanning calorimetry (p−dsc). Resour. ASABE 2006, 49, 1633–1641. [Google Scholar]
- EN 14112:2016. Fat and Oil Derivatives-Fatty Acids Methyl Esters- Determination of Oxidation Stability (Accelerated Oxidation Test). Available online: https://standards.cen.eu/dyn/www/f?p=204:110:0::::FSP_PROJECT,FSP_ORG_ID:40754,6288&cs=19EC6B976AB55D076BA6271CF645957BD (accessed on 1 March 2019).
- ASTM D2274. Standard Test Method for Oxidation Stability of Distillate Fuel Oil (Accelerated Method). Available online: https://www.astm.org/Standards/D2274.htm (accessed on 1 March 2019).
- Van der Westhuizen, I.; Focke, W. Stabilizing sunflower biodiesel with synthetic antioxidants. Fuel 2018, 219, 126–131. [Google Scholar] [CrossRef]
- Coronado, M.; Montero, G.; García, C.; Schorr, M.; Valdez, B.; Eliezer, A. Equipment, Materials and Corrosion in the Biodiesel Industry. Mater. Perfor. 2019, 58, 34–38. [Google Scholar]
- Varatharajan, K.; Pushparani, D. Screening of antioxidant additives for biodiesel fuels. Renew. Sust. Energ. Rev. 2018, 82, 2017–2028. [Google Scholar] [CrossRef]
- De Souza, F.; Spinelli, A. Caffeic acid as a green corrosion inhibitor in mild steel. Corros. Sci. 2009, 51, 642–649. [Google Scholar] [CrossRef]
- Deyab, M. Corrosion inhibition of aluminum in biodiesel by ethanol extracts of rosemary leaves. J. Taiwan Inst. Chem. E. 2016, 58, 536–541. [Google Scholar] [CrossRef]
- Yang, Y.; Song, X.; Sui, X.; Qi, B.; Wang, Z.; Li, Y.; Jiang, L. Rosemary extract can be used as a synthetic antioxidant to improve vegetable oil stability. Ind. Crop Prod. 2016, 80, 141–147. [Google Scholar] [CrossRef]
- Antonijevic, M.; Petrovic, M. Copper corrosion inhibitors: A review. Int. J. Electrochem. Sci. 2008, 3, 1–28. [Google Scholar]
- Rice-Evans, C.; Miller, N.; Pananga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
- National Commission for the knowledge and use of biodiversity (Comisión nacional para el conocimiento y uso de la biodiversidad). Available online: http://www.conabio.gob.mx/conocimiento/info_especies/arboles/doctos/70-zygop2m.pdf. (accessed on 15 May 2018).
- U.S.A. Department of Agriculture, Forest Service, Rocky Mountain Research Station. Available online: https://www.fs.fed.us/database/feis/plants/shrub/lartri/all.html (accessed on 16 May 2018).
- Vargas-Aispuro, I.; Reyes, R.; Rivera, G.; Martínez, M.; Rivero, I. Antifungal lignans of creosote bush (Larrea tridentata). Ind. Crop. Prod. 2005, 22, 101–107. [Google Scholar] [CrossRef]
- Verastegui, Á.; Sánchez, C.; Heredia, N.; García, S. Antimicrobial activity of three major plants from the Chihuahua desert. J. Ethnopharmacol. 1996, 52, 175–177. [Google Scholar] [CrossRef]
- Osorio, E.; Flores, M.; Hernández, D.; Ventura, J.; Rodríguez, R.; Aguilar, C. Biological efficiency of polyphenolic extracts from pecan nuts shell (Carya Illinoensis), pomegranate husk (Punica granatum) and creosote bush leaves (Larrea tridentata Cov.) against plant pathogenic fungi. Ind. Crop. Prod. 2010, 31, 153–157. [Google Scholar] [CrossRef]
- Martins, S.; Amorim, E.; Peixoto, T.; Saraiva, A.; Pisciottano, M.; Aguilar, C.; Texeira, J.; Mussatto, S. Antibacterial activity of crude methanolic extract and fractions obtained from Larrea tridentata leaves. Ind. Crop. Prod. 2013, 41, 306–311. [Google Scholar] [CrossRef]
- Skouta, R.; Moran-Santibañez, K.; Valenzuela, C.; Vazquez, A.; Fanelon, K. Assessing the Antioxidant Properties of Larrea tridentata Extract as a Potential Molecular Therapy against Oxidative Stress. Molecules 2018, 23, 1826. [Google Scholar] [CrossRef]
- Abou, H.; Berdil, E.; Takamatsu, S.; Ferreira, D.; Khan, I. Antioxidant lignans from Larrea tridentate. Phytochemistry 2004, 65, 2499–2505. [Google Scholar] [CrossRef] [PubMed]
- Turner, S.; Davicino, R.; Alonso, R.; Ferraro, G.; Filip, R.; Anesini, C. Potential use of low-NDGA Larrea divaricata extracts as antioxidant in foods. Rev. Peru. Biol. 2011, 18, 159–164. [Google Scholar] [CrossRef]
- Prior, R.; Wu, X.; Schaich, K. Standardized Methods for the Determination of Antioxidant Capacity and Phenolics in Foods and Dietary Supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef] [PubMed]
- US National Library of Medicine, PubChem. Available online: https://pubchem.ncbi.nlm.nih.gov/ (accessed on 10 March 2019).
- Bouaid, A.; Martinez, M.; Aracil, J. Long storage stability of biodiesel from vegetable and used frying oils. Fuel 2007, 86, 2596–2602. [Google Scholar] [CrossRef]
- Ndana, M.; Garba, B.; Hassan, L.; Faruk, U. Effect of Storage on Stability of Biodiesel Produced from selected seed oils. Int. J. Pure Appl. Sci. Technol. 2012, 13, 10–18. [Google Scholar]
- Christensen, E.; McCormick, R. Long-term storage stability of biodiesel and biodiesel blends. Fuel Process. Technol. 2014, 128, 339–348. [Google Scholar] [CrossRef]
- Karavalakis, G.; Hilari, D.; Givalou, L.; Koranis, D.; Stournas, S. Storage stability and ageing effect of biodiesel blends treated with different antioxidants. Energy 2011, 36, 369–374. [Google Scholar] [CrossRef]
- Botella, L.; Bimbela, F.; Martin, L.; Arauzo, J.; Sánchez, J. Oxidation stability of biodiesel fuels and blends using the Rancimat and PetroOXY Methods. Effect of 4-allyl-2,6-dimethoxyphenolandcatecholasbiodiesel additives on oxidation stability. Front. Chem. 2014, 2, 1–9. [Google Scholar] [CrossRef]
- Roveda, A.; Comin, M.; Rodrigues, A.; Souza, V.; Aparecido, M. Thermal stability enhancement of biodiesel induced by a synergistic effect between conventional antioxidants and an alternative additive. Energy 2016, 109, 260–265. [Google Scholar] [CrossRef]
- Mittelbach, M.; Schober, S. The influence of antioxidants on the oxidation stability of biodiesel. J. Am. Oil Chem. Soc. 2003, 80, 817–823. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuveleir, M.; Berset, C. Use of free radical method to evaluate antioxidant activity. Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Benzie, I.; Strain, J. The Ferric Reducing Ability of Plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Ou, B.; Hampsch-Woodill, M.; Prior, R. Development and Validation of an Improved Oxygen Radical Absorbance Capacity Assay Using Fluorescein as the Fluorescent Probe. J. Agric. Food Chem. 2001, 49, 4619–4626. [Google Scholar] [CrossRef]
- Singleton, V.; Rossi, J. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am. J. Enol. Viticult. 1965, 16, 144–158. [Google Scholar]
- ASTM E 2997. Standard Test Method for Analysis of Biodiesel Products by Gas Chromatography-Mass Spectrometry. Available online: https://www.astm.org/Standards/E2997.htm (accessed on 1 March 2019).
- Shantha, N.; Decker, E. Rapid, Sensitive, Iron-Based Spectrophotometric Methods for Determination of Peroxide Values of Food Lipids. J. AOAC Int. 1994, 77, 421–424. [Google Scholar]
- Heidarpour, M.; Farhoosh, R. A preliminary Rancimat-based kinetic approach of detecting olive oil adulteration. Food Sci. Technol. 2018, 90, 77–82. [Google Scholar] [CrossRef]
- Farhoosh, R.; Niazmand, R.; Rezaei, M.; Sarabi, M. Kinetic parameter determination of vegetable oil oxidation under Rancimat test conditions. Eur. J. Lipid Sci. Tech. 2008, 110, 587–592. [Google Scholar] [CrossRef]
- Symoniuk, E.; Ratusz, K.; Krygier, K. Comparison of the oxidative stability of cold-pressed rapeseed oil using Pressure Differential Scanning Calorimetry and Rancimat methods. Eur. J. Lipid Sci. Tech. 2017, 119, 1–9. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |





| Method | Result |
|---|---|
| DPPH | 10.14 ± 0.1 TEAC/g |
| FRAP | 172.1 ± 0.13 TEAC/g |
| ORAC | 50,770 ± 4.2 TEAC/g |
| Total Phenols | 211.18 ± 0.39 GAE/g |
| Compound | Concentration (mg/g) |
|---|---|
| Chlorogenic Acid | 0.920 |
| Caffeic Acid | 2.288 |
| p-Coumaric Acid | 0.916 |
| Ferulic Acid | 0.849 |
| Catechin | 1.967 |
| Epicatechin | 1.866 |
| Total | 8.806 |
| RT (min) | Methyl Ester Name | Relative Abundance (%) |
|---|---|---|
| 32.24 | 9-Octadecenoic acid (z) methyl ester | 46.35 |
| 32.14 | 9,12-Octadecadienoic acid, methyl ester | 23.54 |
| 30.24 | Hexadecanoic acid, methyl ester | 9.04 |
| 32.49 | Octadecanoic acid, methyl ester | 5.46 |
| 34.08 | 11-Eicosenoic acid, methyl ester | 3.12 |
| Others | 4.20 | |
| B100 | B100ox | Biodiesel 250 mg/L | Biodiesel 500 mg/L | Biodiesel 1000 mg/L | |
|---|---|---|---|---|---|
| Average (MJ/kg) | 36.11 | 31.17 | 35.94 | 35.59 | 36.48 |
| SD | 0.08 | 1.35 | 0.14 | 0.19 | 0.25 |
| Sample Concentration (mg/L) | T (°C) | OSI (h) | Linear Equation | Ea (kJ/mol) | Q10 | ||
|---|---|---|---|---|---|---|---|
| m | ln A | R2 | |||||
| Blank | 100 | 2.11 | 8457 | −21.90 | 0.98 | 70.32 | 1.76 |
| 110 | 1.25 | ||||||
| 120 | 0.58 | ||||||
| 130 | 0.42 | ||||||
| 250 | 100 | 27.90 | 12,296 | −29.81 | 0.93 | 102.23 | 2.38 |
| 110 | 8.80 | ||||||
| 120 | 3.04 | ||||||
| 130 | 2.65 | ||||||
| 500 | 100 | 27.88 | 14,249 | −34.64 | 0.96 | 118.47 | 2.65 |
| 110 | 16.89 | ||||||
| 120 | 5.61 | ||||||
| 130 | 1.69 | ||||||
| 1000 | 100 | 68.41 * | 10,989 | −25.23 | 0.99 | 91.37 | 2.08 |
| 110 | 32.27 | ||||||
| 120 | 14.22 | ||||||
| 130 | 7.89 | ||||||
| Material | Antioxidant | Concentration (mg/L) | Blank OSI (h) | OSI with Antioxidant (h) | Reference |
|---|---|---|---|---|---|
| Canola biodiesel | 4-allyl-2,6-dimethoxyphenol | 1000 | 5.2 | 7.45 | Botella et al. [35] |
| Catechol | 1000 | 5.2 | 9.51 | ||
| Soybean biodiesel | BHT | 500 | 3.7 | 7 | Roveda et al. [36] |
| Propyl gallate | 500 | 3.7 | 10 | ||
| Soybean oil | Rosemary extract | 400 | 2.2 | 3.40 | Yang et al. [17] |
| BHA+BHT | 400 | 2.2 | 2.90 | ||
| Canola biodiesel | THBQ | 1000 | 9.15 | 38.53 | Mittelbach et al. [37] |
| Propyl gallate | 1000 | 9.15 | 27.36 | ||
| BHA | 1000 | 9.15 | 24.30 | ||
| Canola biodiesel | L. tridentata methanolic extract | 1000 | 1.25 | 32.27 | Current authors |
| 500 | 1.25 | 16.89 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sagaste, C.A.; Montero, G.; Coronado, M.A.; Ayala, J.R.; León, J.Á.; García, C.; Rojano, B.A.; Rosales, S.; Montes, D.G. Creosote Bush (Larrea tridentata) Extract Assessment as a Green Antioxidant for Biodiesel. Molecules 2019, 24, 1786. https://doi.org/10.3390/molecules24091786
Sagaste CA, Montero G, Coronado MA, Ayala JR, León JÁ, García C, Rojano BA, Rosales S, Montes DG. Creosote Bush (Larrea tridentata) Extract Assessment as a Green Antioxidant for Biodiesel. Molecules. 2019; 24(9):1786. https://doi.org/10.3390/molecules24091786
Chicago/Turabian StyleSagaste, Carlos A., Gisela Montero, Marcos A. Coronado, José R. Ayala, José Á. León, Conrado García, Benjamín A. Rojano, Stephania Rosales, and Daniela G. Montes. 2019. "Creosote Bush (Larrea tridentata) Extract Assessment as a Green Antioxidant for Biodiesel" Molecules 24, no. 9: 1786. https://doi.org/10.3390/molecules24091786






