1. Introduction
In recent years, proteins have been considered not only as a source of amino acids or building material for all body cells but also as a source of peptides that may have different biological properties. Peptides are delivered to the organism with protein food products. After hydrolysis in the gastrointestinal tract, they enter the bloodstream and are transported to the destinations of their action. Many them are antioxidants, inhibit the pathogenesis of metabolic syndrome, or have antimicrobial activity. Some peptides are isolated and identified from proteins, and others have been synthesized to study their properties. These compounds can be used as to reduce the development of diseases.
Metabolic syndrome (MS) is a risk factor of adverse pathophysiological conditions, including insulin resistance, disorders in sugar metabolism, visceral obesity, cardiovascular diseases, atherogenic dyslipidemia, and hypertension. The main role is played by such lifestyle factors as age, genetics, and socioeconomic status. Nowadays, there are growing numbers of data indicating that environmental risk factors, specifically air pollution, induce inflammatory processes that might play a key role in metabolic syndrome [
1,
2].
Diseases involved in MS are often associated with imbalance in the activity of enzymes that regulate the main processes in the organisms. Hypertension is the main chronic disease associated with malfunction of the most widely studied renin–angiotensin system. The angiotensin-converting enzyme (ACE) plays the most important role, as it catalyzes the formation of the potent vasoconstrictor angiotensin II (octapeptide) from inactive angiotensin I (decapeptide) and the breakdown of bradykinin, i.e., a vasodilator peptide. The excessive activity of ACE and production of angiotensin II cause a vasospasm and a direct increase in blood pressure, stimulation of cell proliferation, and excessive production of certain proteins. These changes lead to myocardial hypertrophy and damage as well as hypertrophy and stiffening of the vascular wall, which results in an increase in blood pressure and stimulation of the sympathetic nervous system. It causes blood vessels to contract, accelerates the heart rate, and increases the amount of blood ejected by the heart into vessels, which elevates pressure and, through long-term effects, causes damage to the heart and vessels. Furthermore, the secretion of the aldosterone hormone is elevated, which also increases blood pressure [
3].
In recent years, great progress has been made in inhibition of the activity of the renin–angiotensin–aldosterone (RAA) system by using drugs that affect various links in the system. Inhibition of the activity of various types of cells involved in the RAA system is widely applied in the treatment of hypertension, cardiovascular diseases, and kidney diseases. Synthetic ACE inhibitors such as captroptril, enalapril, or ramipryl are commonly used as drugs in the treatment of hypertension, although they may cause serious side effects such as dry cough, dysgeusia, and angioedema [
4]. Hence, it is necessary to search for new ACE inhibitors that may be obtained from food products and be safer. Such peptides have been identified from foods of various origins: plants [
5], animals [
6], or microorganisms. Their structure and activity differ, and the exact relationship between the structure and inhibition is still unknown. ACE inhibitory peptides represent di- or tripeptides such as the short chain peptides with sequences KVF, MKR, AKF, AMK, and GIL obtained from the enzymatic hydrolysates of lysozyme. In turn, there are several ACE inhibitory peptides with more than three amino acid residues in their structure: LIVGIIRCV from beef myofibrillar proteins [
7] or peptides with sequences GHIITVAR, IGGIGTVPVGR, HIGNILSL, FMPGVPGPIQR, PNYHPSPR, AFPAGAAHW, HIITLGR, LAGNPAGR, MPGVPGPIQR, AGALGDSVTVTR, and INTLSGR obtained from sesame protein [
8].
The other main risk factor of cardiovascular diseases is diabetes. There are several studies indicating a relationship between diabetes and an increased risk of hypertension [
9]. Diabetes and carbohydrate disorders may be caused by the malfunction of enzymes involved in carbohydrate metabolism. One of them is α-amylase, whose activity is directly related to the development of type 2 diabetes, i.e., the most prevalent type of diabetes [
10]. The enzyme is found in saliva and pancreas, and starch and glycogen are the substrate for its activity. Oligosaccharides released in the digestion process are further hydrolyzed into an absorbable monosaccharide: glucose. Glucose absorption into the bloodstream in patients with insulin disorders causes a sharp increase in the blood glucose level, which may lead to development of the disease [
11]. Peptides with enzyme inhibitory activity involved in polysaccharide metabolism and exhibiting a potent anti-diabetic effect were described previously: CSSV, YSPR, SAAP, PGGP, and LGGGNT [
12], or LPLLR [
13]
Fats are energy-delivering molecules. Before being absorbed in the small intestine, they should be hydrolyzed by lipase into monoglycerol and free fatty acids. The main role in this process is played by pancreatic lipase, which can hydrolyze 50–70% of food-derived fat. The inhibition of excessive activity of this enzyme can be helpful in controlling postprandial hyperglycemia and/or reducing calorie intake, thereby bringing far-reaching health benefits in type 2 diabetes and obesity treatment [
14]. One of the types of inhibitory compounds may be peptides whose activity depends on their structure and amino acid composition. There are a few peptide inhibitors, e.g., Ile-Trp-Ser and Tyr-Phe-Ser [
15], CQPHPGQTC [
16], or EITPEKNPQLR, and RKQEEDEDEEQQRE [
17].
Obesity is regarded as systemic inflammation that is strongly influenced by the cyclooxygenase (COX) and lipoxygenase (LOX) pathways. Moreover, these inflammatory markers occupy an important position as they give insights into arising risk for cardiovascular disease (CVD), diabetes, and other metabolic disorders [
18]. Therefore, the inhibition of inflammation enzymes may be effective in dietary therapy of obesity. In the literature, there are a few peptides with anti-inflammatory activity. Montoya-Rodríguez and Mejía reported that pure amaranth peptides HGSEPFGPR and RPRYPWRYT reduced the expression of LOX-1 and other factors involved in the inflammatory process, such as lipopolysaccharide (LPS)-induced inflammation or transforming growth factor-α (VCAM-1)-induced inflammation [
19].
The main aim of our study was to estimate the role of synthetic peptides as inhibitors of selected enzymes associated with metabolic syndrome and determination of their antimicrobial activity and resistance to gastrointestinal enzymes. The peptides were described in our previous studies [
20,
21]. The research material was millet grains subjected to different temperature treatments (65 °C and 100 °C). Protein fractions from millet grains were isolated and hydrolyzed in gastrointestinal conditions. The hydrolyzates were a source of peptide fractions with molecular mass under 3.0 kDa with potent biopeptides.
3. Discussion
The search for natural substances in food with a potential therapeutic effect but no serious side effect has been carried out by researchers. Many studies have indicated that peptides exhibit bioactive properties, and food with high content thereof can be used to help treat metabolic syndrome diseases such as hypertension, insulin resistance, or obesity [
22,
23,
24]. The application of natural peptides in foods to prevent the development of metabolic syndrome may be difficult, since any type of peptide used for food application must be safe for ingestion. Besides, the peptides should be stable and the method for derivation thereof must be effective and cheap. Designing novel synthetic peptides based on natural native peptides may be an alternative option for the application of peptides in foods.
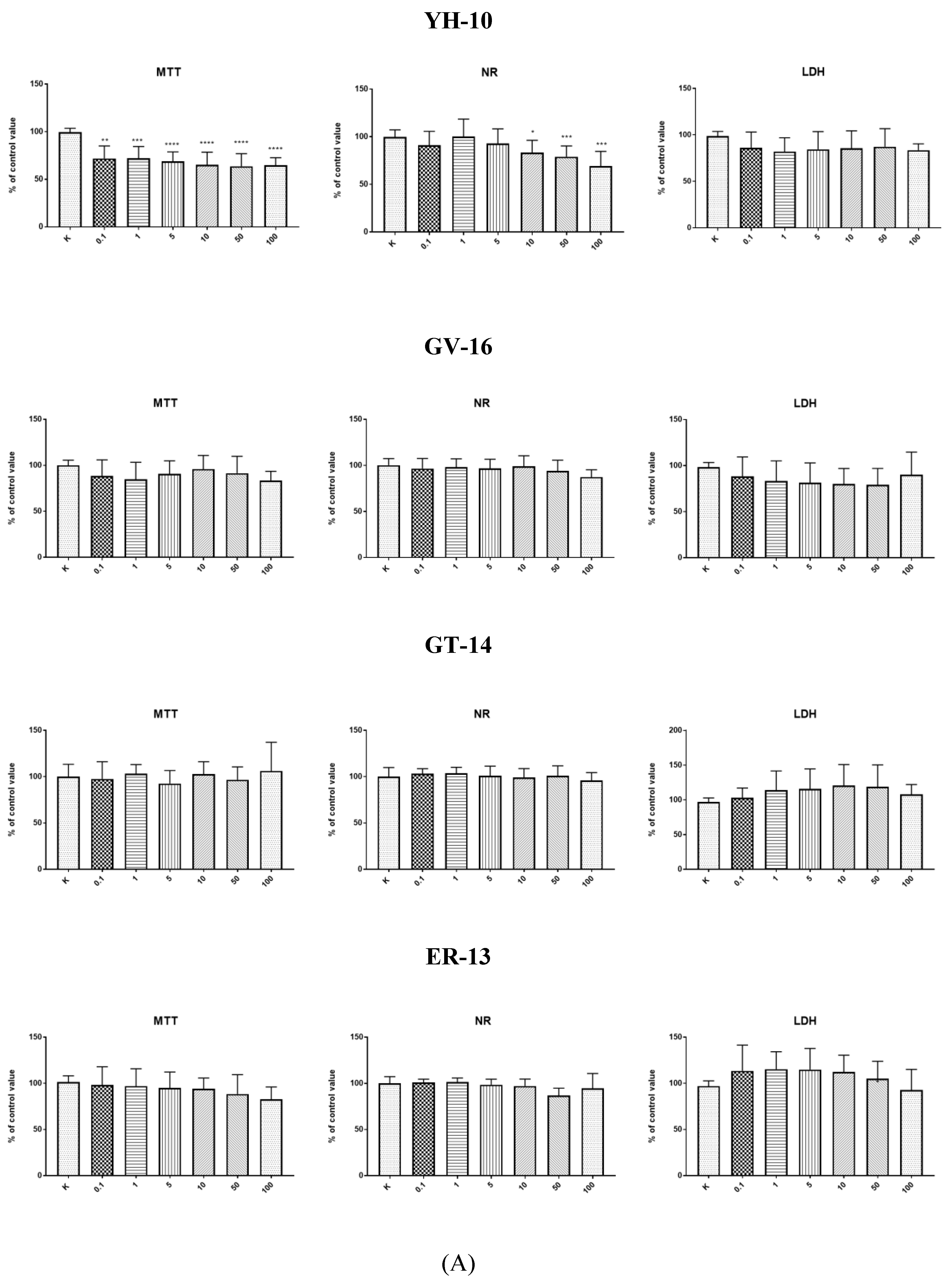
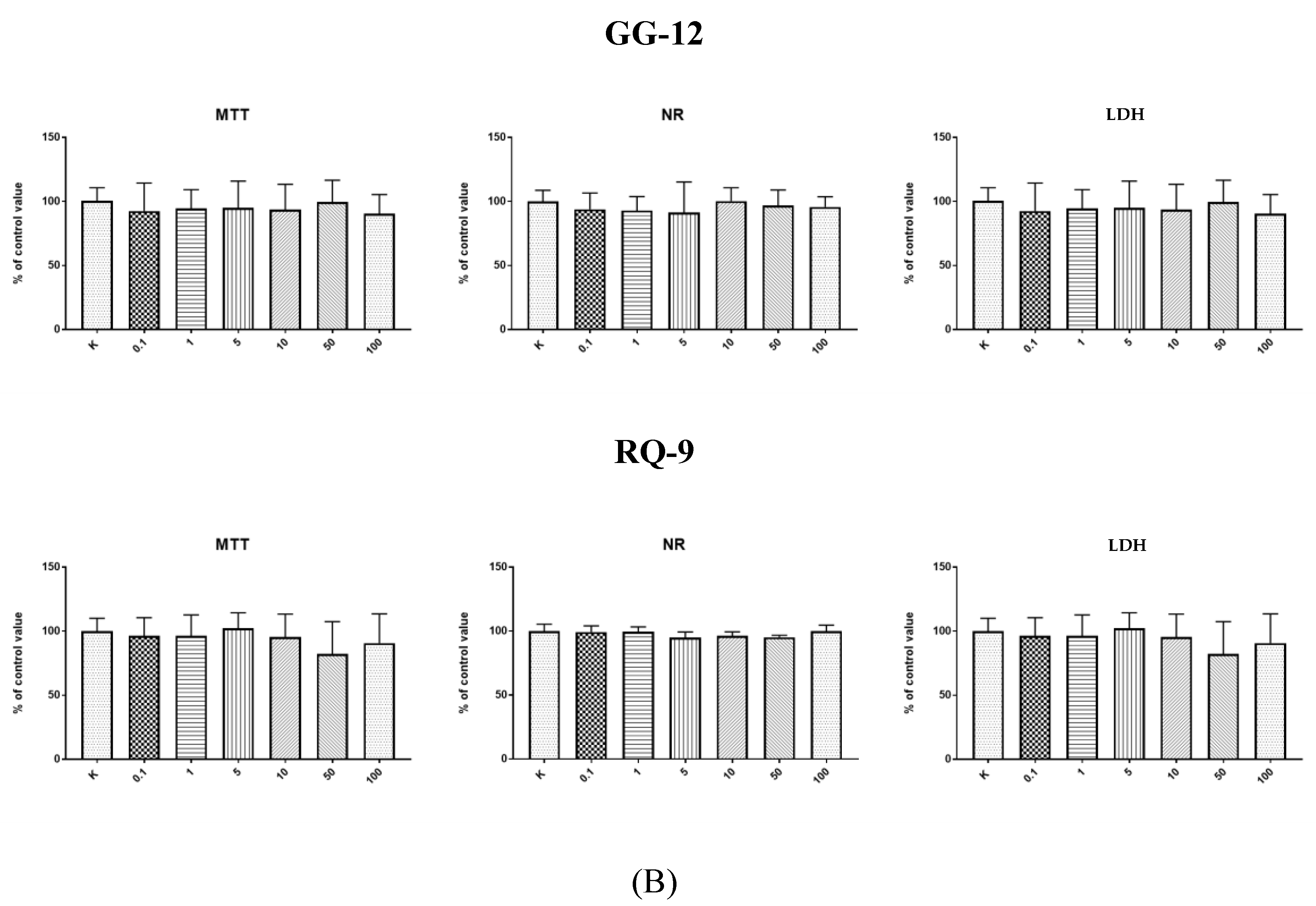
In our study, all the synthetic peptides were tested to assess their cytotoxic effect. After 24-h incubation of endothelial cells (HECa10) with peptides GV-16, GT-14, ER-13, GG-12, and RQ-9 at different concentrations (0.1–100 µg/mL), no statistically significant differences were observed in any of the tests (MTT, NR, LDH) compared to the values obtained in the control conditions (
Figure 1A,B). The results of incubation of the HECa10 cells at the concentrations of 10–100 µg/mL indicated significant differences in the MTT and NR tests. It should be noted that there were no statistically significant differences in the LDH activity test. This may suggest that the changes observed in the cells are not associated with the direct cytotoxic effect of YH-10, i.e., an increase in the LDH activity after cell membrane damage. They are rather associated with the modulation of cell metabolism by synthetic peptides. An additional argument for such a thesis is the concentration-independent effect of YH-10 on cell counts demonstrated in the MTT test. It is well known that some peptides affect cell metabolism. The effect of peptides depends on the cellular target (receptors, enzymes, inhibitors, etc.). Lorenzo et al. (2012) has shown in T-cells that altered peptide ligands (APL 1 and 2) affect the proliferation, cell cycle, and secretion of growth factors and cytokines, which in turn attenuate adjuvant arthritis (AA) and collagen-induced arthritis (CIA) in rat models [
25]. In contrast, Conconi et al. reported inhibited cell adhesion, decreased cell proliferation, migration, and morphogenesis in Matrigel, and anti-angiogenic responses induced by small peptides carrying adhesion sequences (GRGDSP) (4)K [
26]. In our research, new peptides were synthesized and therefore both cell effects and cell targets are unknown.
The HECa10 cells are murine high endothelial cells (HEC) from the peripheral lymph nodes, which were established as a non-transformed cell line, displaying and keeping the characteristic phenotype of the HEC of the tissue from where they are derived [
27]. Besides their characteristic ability to recognize and select lymphocytes and specifically adhering cells, HECa10 produce characteristic blood vessel proteins, such as the angiotensin-converting enzyme, von Willebrand factor, VE-cadherin, and E-selectin [
28]. Therefore, we used these cells as a model of the blood endothelial cell line. We believe that our synthetic peptides may pass through the digestive tract without any modification, and therefore may affect the metabolism of blood endothelial cell lines. We used murine endothelial cells, because we intended to use our synthetic peptides in mice with metabolic syndromes (induced or genetically changed) to find if there is any improvement of the health of endothelial cells.
The physicochemical parameters of the six synthesized peptides (YH-10, GV-16, GT-14, ER 13, GG-12, and RQ-9) were considered (
Table 1). The peptides had molecular mass between 955.12 and 1485.59 g/moL, which corresponds 0.96 to 1.46 kDa. As reported by Wang and Li [
29], this type of peptide may be transported via the paracellular route, which is the main intestinal transport pathway for high molecular mass peptides (500–1600 Da). To determine the peptide hydrophobicity, the grand average of hydropathicity (GRAVY) index defined by the sum of hydropathy values of all the amino acids divided by the peptide length was used. The peptides synthesized in this study had GRAVY indexes ranging from −1.315 to 0.011. A positive GRAVY index is obtained for a hydrophobic structure and a negative GRAVY index indicates hydrophilic compounds. The results indicate that the peptides have a globular (hydrophilic) rather than membranous (hydrophobic) structure [
30,
31]. The instability index provides an estimate of peptide stability during testing. A peptide with an instability index lower than 40 is expected to be stable. In turn, the value of >40 predicts that the peptide may be unstable [
32]. Among tested peptides, only ER-13 was characterized by a higher value than 40, indicating its instability. Solubility in water was estimated based on the number of charged residues, isoelectric point (pI), and peptide length. As demonstrated by the data obtained in our study, only two peptides ER-13 and RQ-9 have good solubility in water. However, peptides characterized by poor solubility in water can have high biological activity as well. Although the solubility of peptides GKPVAVPA (GA-8) and GKHVAVHAR (GHA-8) described by Mirzaei et al. [
32] was poor, these peptides exhibited ACE inhibitory activity.
In our study, we tested six synthesized peptides as compounds protecting against the development of metabolic syndrome. These disorders, i.e., glycemic index imbalance, hypertension leading to cardiovascular disease, dyslipidemia, and/or obesity, are an early factor of the potential future development of chronic conditions, such as type 2 diabetes [
33]. Hypertension is regarded as one of the main causes of death worldwide and is associated with increased ischemic heart failure and coronary artery disease. One of the therapeutic methods for hypertension treatment is the use of ACE inhibitors, since reduction of the concentration of the most important vasoconstrictor (angiotensin II) by suppressing ACE is a key factor to achieve blood pressure balance and water balance in the body [
34].
In our study, five tested peptides had an ACE inhibitory activity with the IC
50 value in the range from 498.79 to 728.30 (µg/mL). It corresponds to a range of 0.43 to 0.52 mM. These values are higher than those obtained for peptides described in other studies, i.e., 2.5 µg/mL for peptide Thα1 [
35] or 127 µg/mL for egg white protein (AFKDEDTEEVPFR) [
36]. These differences may be associated with the different sequences and length of the peptides. Until recently, it was thought that only di- or tri-peptides with proline at the C-terminus have ACE inhibitory activity. There are several studies describing long-chain peptides without proline at the C-terminus, exhibiting the high potential of ACE inhibition [
4,
37]. Moreover, it has been observed that the presence of charged amino acids at the C-terminal of peptides plays the main role in the interaction with the ACE enzyme. The presence of hydrophobic amino acids at the C-terminal and aliphatic amino acids at the N-terminal position contributes to the ACE inhibitory action [
38]. Several studies have also indicated that binding to ACE is influenced by hydrophobic amino acid residues (aromatic or branched chain) at three positions from the C-terminus of the peptide [
39]. The amino acids such as izoleucine and valine in the aliphatic amino acid chain are described as factors increasing the inhibitory effect [
40]. In turn, as indicated by Siow et al. [
41], the combination of both hydrophilic and hydrophobic properties allows the peptide to act as an inhibitor. The authors suggest that a peptide with a “hydrophilic head” (consisting of Arg-His) and a hydrophobic tail (consisting of PAQPNYPWTAVLVF) would act in the same way. The K
m and V
max values at the different concentrations of the peptides were determined by the Lineweaver–Burk plot. The results showed a competitive inhibition mode of four purified peptides and an uncompetitive mode of one of them (
Table 4). This result corresponds well with data reported by Villadóniga and Cantera [
4], where the inhibition kinetic analysis showed that a peptide with the TTFHTSGY sequence was a fully functional ACE competitive inhibitor. It should be noted that ACE competitive inhibitory peptides are reported in literature most frequently [
42]. A competitive inhibitor binds with free enzymes via interaction with an amino acid in the active site of the enzyme or with amino acids far from the active site. It should be noted that Captropril, i.e., the most common drug used in hypertension treatment, is also known as a competitive inhibitor of ACE [
43].
Some of the factors that increase the risk of cardiovascular disease are overweight and obesity, which may also cause diabetes 2. The main cause of these health problems is the imbalance between the intake and conversion of calories into energy. To improve this situation, patients should incorporate physical activity it into their rhythm of the day and reduce the absorption of carbohydrates and lipids from their diet. The most important enzymes involved in carbohydrate hydrolysis and responsible for calorie absorption are α-amylase and α-glucosidase. The former hydrolyzes polysaccharides into oligosaccharides, whereas the latter catalyzes the final step to release absorbable monosaccharides [
44]. The rapid increase in blood glucose can be reduced through the inhibition of enzymes involved in the release of glucose from foods, and this approach is employed in the management of type 2 diabetes [
33]. According to the present data, all the tested peptides had α-amylase inhibitory activity but none of them inhibited α-glucosidase (
Table 2). This may indicate that these peptides may not fully inhibit glucose absorption but may help reduce oligosaccharide secretion. Peptides from food inhibiting α-amylase and α-glucosidase were demonstrated in previous studies [
13,
45,
46]. Moreover, the different amino acid compositions and sequences probably affect the bioactivity of peptides. As demonstrated by previous studies of the relationship between the structure and activity of peptides, the α-glucosidase and α-amylase inhibitory activities were strongly influenced by two hydrophobic amino acids, i.e., leucine and proline. It should be noted that the peptides analyzed in our study have a described amino acid sequence. Analysis of the relationship between the structure and activity also demonstrated that the C-terminal arginine has a positive effect on the polypeptide inhibitory activity [
12], and this amino acid also is a component of the analyzed peptides. Almost all the peptides in this study are uncompetitive inhibitors (
Table 4). These results correspond well with data described by Fu et al. [
47], where longan pericarp proanthocyanidins were characterized as uncompetitive inhibitors of α-amylase activity. It should be noted that acarbose, a sugar analog inhibitor of α-amylase activity commonly used for diabetes treatment, shows competitive inhibition. Therefore, peptides or other food compounds with different structure may exhibit different inhibition behavior.
Another enzyme involved in the hydrolysis of high-energy food compounds is pancreatic lipase. This main lipolytic hydrolase in the small intestine in the digestive system is responsible for the hydrolysis of approximately 70% of dietary fats. Triacylogilcerols are digested by pancreatic lipase into monoacylglycerol and free fatty acids, which form mixed micelles in combination with bile acids, cholesterol, and lysophosphatidic acid [
48]. An excessive amount of fatty acids flowing into cells may result in the formation of adipose tissue or the development of insulin resistance. Therefore, the inhibition of pancreatic lipase activity may prevent the occurrence of obesity as well and glucose metabolism disorders. Orlistat is one of the drugs used in obesity treatment whose action is based on the inhibition of pancreatic lipase activity. Although it shows satisfying effects on weight control, the serious side effects such as pancreatic damage, kidney disease, gastrointestinal disorders, and high cancer risk have limited the clinical application of this drug, especially for long-term treatment [
49]. Therefore, new pancreatic lipase inhibitors in food compounds are being investigated. In our study, all the tested peptides exhibited pancreatic lipase inhibitory activity (
Table 2) and were mostly characterized as uncompetitive inhibitors (
Table 4). The activity of inhibitors depends on their structure and amino acid compositions. Peter and Bywater [
50] suggest that the hydrophilic character is the main determinant of high inhibitory activity of peptides. This has explained the inhibitory activity of the tested peptides, which may be due to the varied amounts of hydrophilic residues (e.g., arginine, glutamic acid, glutamine, or threonine) in the peptide sequences.
Moreover, one of the factors of obesity and overweight is the inflammatory process in tissue. Lipid-mediated inflammation is strongly influenced by the cyclooxygenase (COX) and lipoxygenase (LOX) pathways, and these inflammatory markers play an important role as risk factors of diabetes and cardiovascular diseases [
18].
In our study, all the examined peptides exerted a potential anti-inflammatory effect (
Table 3), but ER-13 was characterized by the highest activity. It should be noted that the type of inhibition and potential anti-inflammatory activity are influenced by the structure and amino acid sequence of peptides. There a few reports about peptides with LOX and COX inhibitory activity, but the direct mechanism of this process has still not been elucidated. The results obtained in our previous study indicated that glycine-rich peptide fractions obtained from millet grains had potent anti-inflammatory activities [
21]. As reported by Montoya-Rodríguez et al. [
19], pure peptides described as HGSEPFGPR and RPRYPWRYT inhibited the expression of LOX-1 and other factors involved in the inflammatory process.
Although the peptides in this study are characterized by high inhibitory properties, there are a few strategies to improve their inhibitory effects. These include (1) the use of a mixture of peptides—there are literature reports on the synergistic effect of peptides [
41,
51,
52], (2) effective transport of peptides, and (3) an effective method of encapsulation in the material from which the peptides will be released at the target site (for it may be used e.g., as edible films [
53]).
Food-borne pathogens cause a great number of diseases with significant effects on human health.
E. coli, S. aureus, S. enteritidis, and
L. monocytogenes are responsible for food-borne infections [
54].
C. albicans yeast is a commensal microorganism commonly colonizing the skin, gastrointestinal tract, genitourinary system, oropharynx, and upper respiratory tract without causing harm to healthy individuals [
55,
56]. However, in some cases, the yeast is associated with opportunistic infections in both animals and humans, especially in immunologically weak and immunocompromised patients such as those with HIV/AIDS [
56,
57]. In susceptible patients,
C. albicans can enter the bloodstream by translocation across the mucosa of the gastrointestinal tract [
55]. In recent years, more information about synthetic peptides is available, especially in relation to pathogenic microorganisms that cause e.g., harmful food-borne diseases in humans [
56]. Antimicrobial peptides bind to bacterial cell membranes or mitochondrial membranes, which causes their disintegration and, consequently, cell death [
58]. The antimicrobial activity of peptides is related not only to their physicochemical properties but the number and type of amino acids to [
59]. Previously data indicated that a lack of Arg residue in the sequence of peptides is connected with less antibacterial ability [
60]. It is in agreement with our data. On the other hand, Sousa et al. [
61] reported that glycine/leucine-rich peptide inhibited the growth of Gram-negative bacteria such as
E. coli. Our study indicated that the RQ peptide rich in Arg and Lys was characterized by higher antimicrobial activity compared with other tested peptides. Lum et al. [
56] tested the anticandidal activity of synthetic peptides. Their results showed that hybrid peptides, KU2 and KU3, containing a mixed backbone of KABT-AMP and Uperin 3.6 had the most potent anticandidal activity against
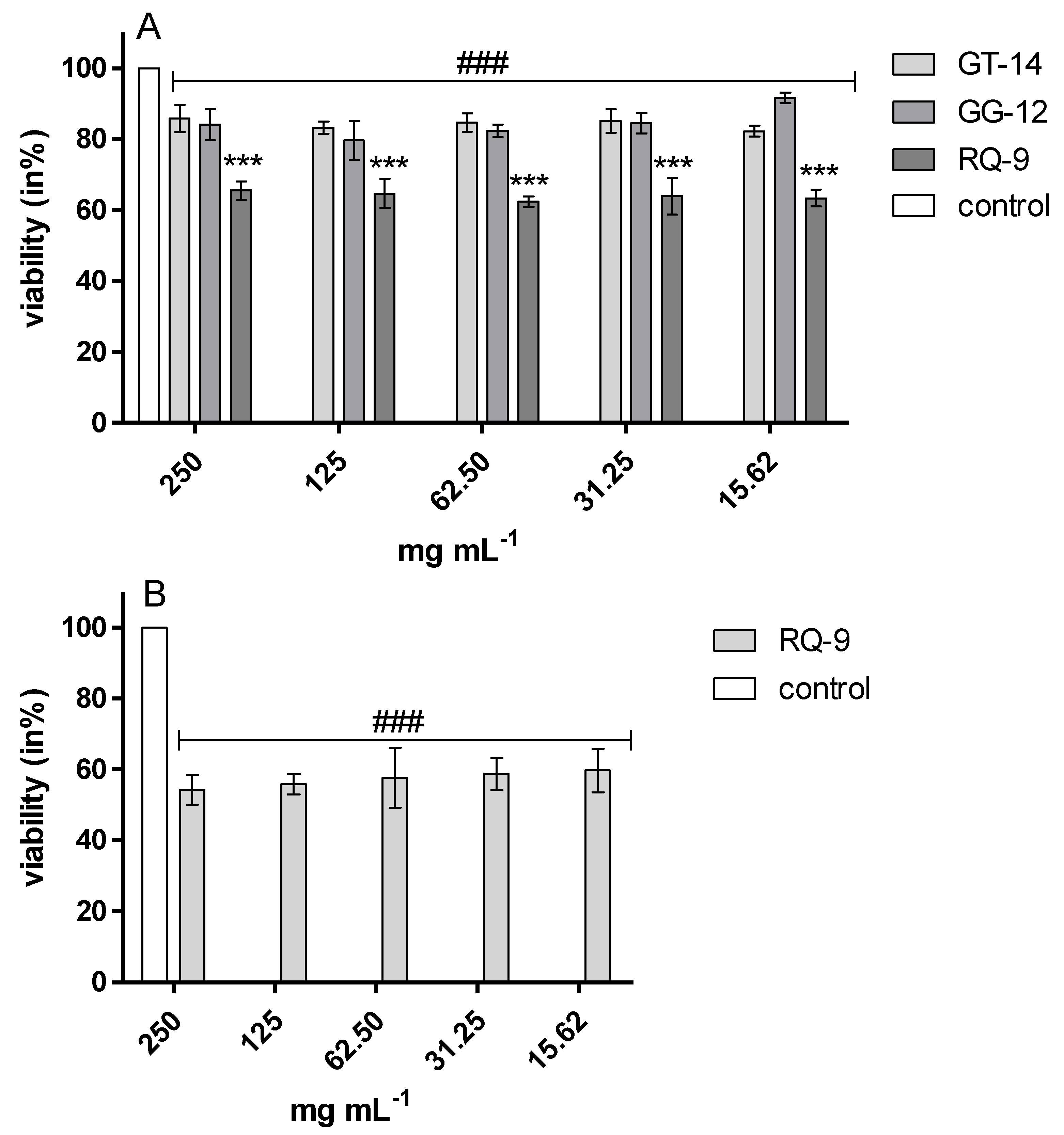
C. albicans 90028 with MIC values ranging from 16 to 128 mg/L. (= 0.016–0.128 mg/mL) [
56]. Our results are in agreement with those reported in the previous study. We used resasurin assay based on the detection of metabolic activity of the cells. The redox dye resazurin (7-hydroxy-3H-phenoxazin-3-one 10-oxide) enters the cell in the oxidized form (blue) and is converted to the reduced form, resorufin (pink), mainly by enzymes in the electron transport system [
62]. We estimated the anticandidal activity of synthetic peptide RQ-9, but with higher MIC values (15.62 mg/mL). Our study indicated antibacterial activity of RQ-9, GT-14, and GG-12 against
E. coli ATCC 25922 (MIC = 250 mg/mL), YH-10, GV-16, GT-14, ER-13, GG-12 against Y,
S. aureus ATCC 29737 (MIC = 250 mg/mL), and RG-9 against
L. monocytogenes ATCC BAA-2660 (MIC = 15.62–250 mg/mL). The previous study indicated the antibacterial activity of peptide 35409 against
E. coli ML 35 (43827) and
S. aureus ATCC 29213 with MIC 22 and 350 µM, respectively. Cusimano et al. have demonstrated the antibacterial activity of synthetic peptides H2 and Tag against different strains of
L. monocytogenes (MIC > 5 mg/mL) [
63]. The growth of
S. enterica was inhibited only by peptide RQ-9 (MIC = 15.62 mg/mL).
It should be emphasized that foods supplemented with the peptides described in this study are not only functional foods with health benefits but also sources of substances that inhibit the growth of pathogenic bacteria responsible for food poisoning. There are several studies of hydrolyzates or peptide fractions. In this study, the analysis was carried out only with peptides, as we do not have other compounds that could additionally affect enzyme activity.