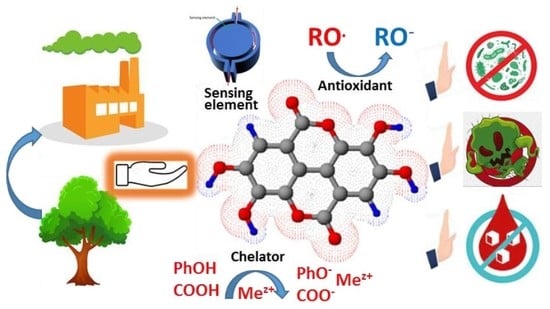
Recent Advances in the Production and Applications of Ellagic Acid and Its Derivatives. A Review
Abstract
1. Introduction
2. The Chemistry of Ellagic Acid and Ellagitannins
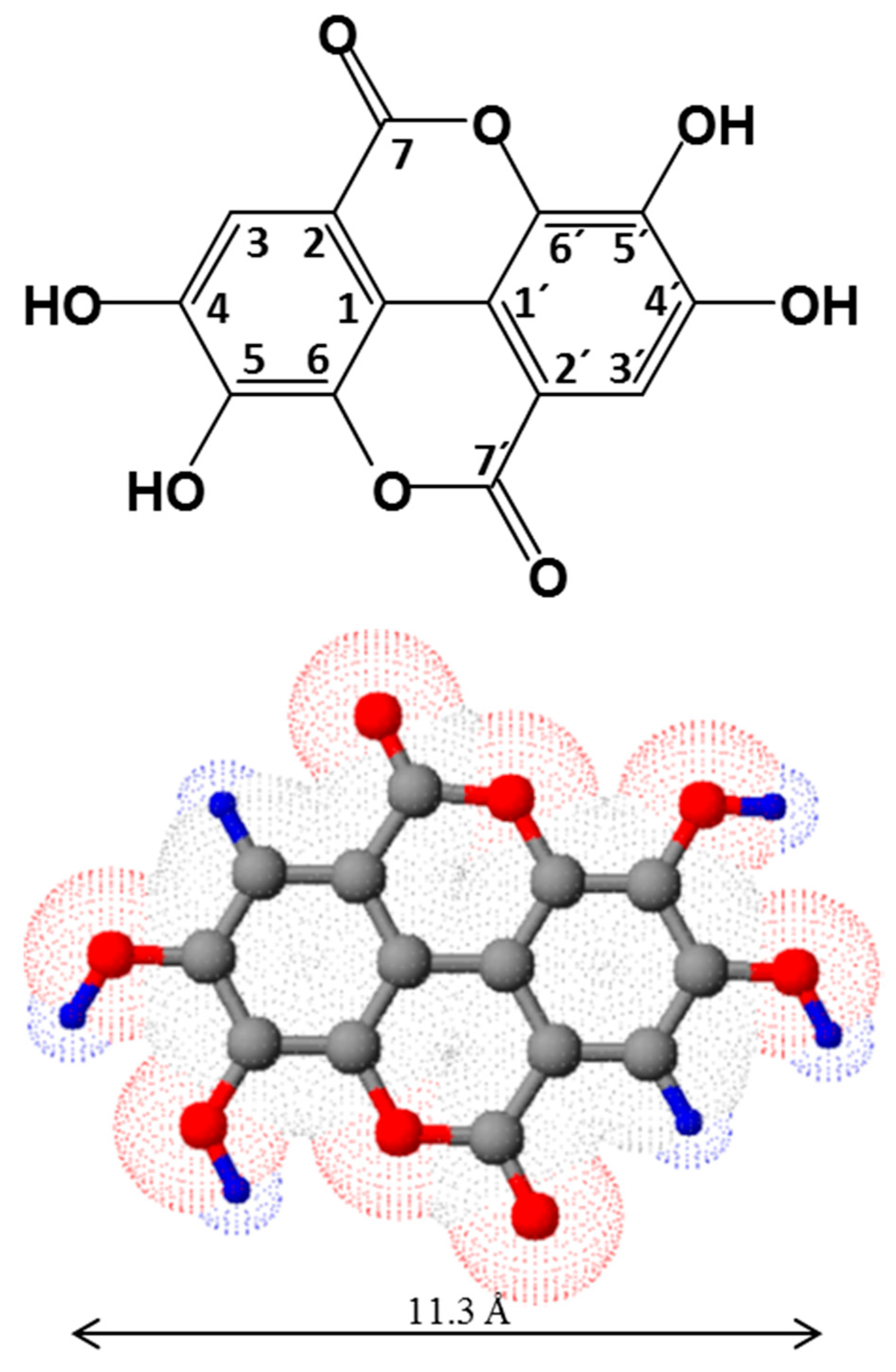
2.1. Structure and Physico-Chemical Properties of Ellagic Acid
2.2. Structure of Ellagitannins
3. Sources of Ellagic Acid and Ellagitannins
| Source | Latin Name | Total ET/EA # | Free EA | Ref. |
|---|---|---|---|---|
| Fruits | ||||
| Arctic bramble | Rubus arcticus | 3900 (fw) | - | [36] |
| Blackberry | Rubus ursinus | 1500 ± 140 (dw) | - | [54] |
| Camu-camu fruit: | Myrciaria dubia | [41] | ||
| Pulp powder | 258.5 ± 4.3 (dw) * | 56.0 ± 1.1 (dw) | ||
| Flour | 5656.6 ± 11.3 (dw) * | 764.9 ± 4.9 (dw) | ||
| Peel | 71.4 (fw) * | Nd | ||
| Pulp | 67.3 (fw) * | Nd | ||
| Seeds | 2819.8 (fw) * | 50.4 (fw) | ||
| Cloudberry | Rubus chamaemorus | 3600 (fw) | - | [36] |
| 3151 (fw) | - | [35] | ||
| Cranberries | Vaccinium | 120 ± 4 (dw) | - | [54] |
| Guava | Psidium guajava L. | 57.2–306 (dw) | - | [55] |
| Kakadu plum | Terminalia ferdinandiana | 30,510–140,250 (dw) | - | [37] |
| 8796.0 ± 156.0 (dw) | 6206.0 ± 22.0 (dw) | [38] | ||
| Muscadine grapes | Vitis rotundifolia | 360–912 (fw) | - | [43] |
| Pomegranate: | Punica granatum | [42] | ||
| Mesocarp | 40,595.4 ± 4434.2 (dw) | 234.2 ± 13.0 (dw) | ||
| Peel | 43,979.0 ± 394.8 (dw) | 637.7 ± 32.8 (dw) | ||
| Red raspberry | Rubus idaeus | 1500 ± 100 (dw) | - | [54] |
| 1900–2700 (fw) | - | [36] | ||
| 2637–3309 (fw) | - | [35] | ||
| Rose hip | Rosa rugosa | 1096 (fw) | - | [35] |
| Strawberry | Fragaria ananassa | 630 ± 90 (dw) | - | [54] |
| 650–850 (fw) | - | [36] | ||
| 683–853 (fw) | - | [35] | ||
| Processed Fruits | ||||
| Pomegranate juice | - | 87–2118.3 (mg·L−1) | 2.1–7.7 (mg·L−1) | [42] |
| Raspberry jam | - | 764 (fw) | - | [35] |
| Strawberry jam | - | 245 (fw) | - | [35] |
| Seeds and Nuts | ||||
| Pecans | Carya illinoensis | 330 ± 0.3 (dw) | - | [54] |
| Walnuts | Juglans nigra | 590 ± 0.3 (dw) | - | [54] |
| Wood | ||||
| Blue gum | Eucalyptus globulus | - | 500–1700 (dw) | [48] |
| Common Oak | Quercus robur | - | 81–228 (dw) | [56] |
| Pyrenean oak | Quercus pyrenaica | - | 66–219 (dw) | [56] |
| Rose gum | Eucalyptus grandis | - | 280–512 (dw) | [47] |
| Sessile oak | Quercus petraea | - | 109–198 (dw) | [56] |
| Sweet chestnut | Castanea sativa | - | 74–140 (dw) | [56] |
| White oak | Quercus alba | - | 132–277 (dw) | [56] |
| Wood bark | ||||
| Blue gum | Eucalyptus globulus | - | 471 (dw) | [57] |
| (Hybrid) eucalypt | Eucalyptus urograndis | - | 2243–2307 (dw) | [58] |
| Maidens Gum | Eucalyptus maidenii | - | 1130–1178 (dw) | [58] |
| Oak | Quercus robur + Quercus petraea | - | 2200–3700 (dw) | [59] |
| Sweet chestnut | Castanea sativa | - | 4300–9300 (dw) | [60] |
| Rose Gum | Eucalyptus grandis | - | 2639–2721 (dw) | [58] |
| Other sources | ||||
| Eucalypt leaves | Eucalyptus globulus | 3320.0 ± 80.0 (dw) | - | [61] |
| Filtrates from unbleached kraft wood | Eucalyptus globulus | - | 98 ± 0.7 (mg/L) | [48] |
| Sulphite spent liquor | Eucalyptus globulus | - | 1165.5 (mg/L) | [62] |
4. Production of Ellagic Acid
4.1. Extraction from Natural Sources
4.2. Organic Synthesis
5. Technical Applications of Ellagic Acid
6. Bioavailability of Ellagitannins and Ellagic Acid
7. Biological Functionality of Ellagic Acid and Its Derivatives
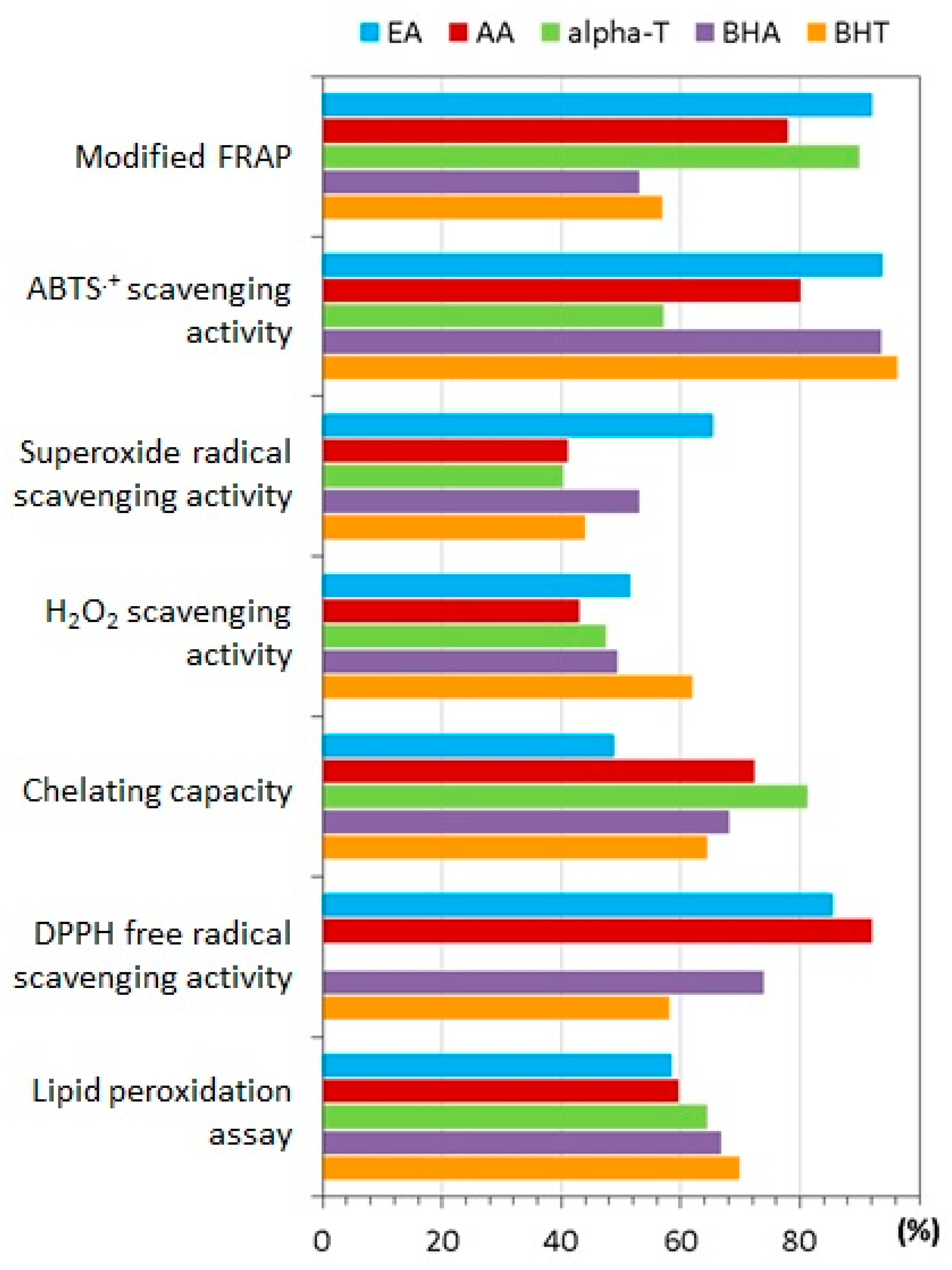
7.1. Antioxidant Properties
7.2. Biological Activity
8. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Quideau, S.; Feldman, K.S. Ellagitannin chemistry. Chem. Rev. 1996, 96, 475–504. [Google Scholar] [CrossRef] [PubMed]
- Khanbabaee, K.; van Ree, T. Tannins: Classification and definition. Nat. Prod. Rep. 2001, 18, 641–649. [Google Scholar] [CrossRef] [PubMed]
- Yamada, H.; Wakamori, S.; Hirokane, T.; Ikeuchi, K.; Matsumoto, S. Structural revisions in natural ellagitannins. Molecules 2018, 23, 1901. [Google Scholar] [CrossRef] [PubMed]
- Covington, A.D. Modern tannins chemistry. Chem. Soc. Rev. 1997, 26, 111–126. [Google Scholar] [CrossRef]
- Wu, X.; Gu, L.; Holden, J.; Haytowitz, D.B.; Gebhardt, S.E.; Beecher, G.; Prior, R.L. Development of a database for total antioxidant capacity in foods: A preliminary study. J. Food Compos. Anal. 2004, 17, 407–422. [Google Scholar] [CrossRef]
- Aslan, A.; Gok, O.; Erman, O.; Kuloglu, T. Ellagic acid impedes carbontetrachloride-induced liver damage in rats through suppression of NF-kB, Bcl-2 and regulating Nrf-2 and caspase pathway. Biomed. Pharmacother. 2018, 105, 662–669. [Google Scholar] [CrossRef]
- Ceci, C.; Tentori, L.; Atzori, M.G.; Lacal, P.M.; Bonanno, E.; Scimeca, M.; Cicconi, R.; Mattei, M.; de Martino, M.G.; Vespasiani, G.; et al. Ellagic acid inhibits bladder cancer invasiveness and in vivo tumor growth. Nutrients 2016, 8, 744. [Google Scholar] [CrossRef]
- Dhingra, D.; Chhillar, R. Antidepressant-like activity of ellagic acid in unstressed and acute immobilization-induced stressed mice. Pharmacol. Rep. 2012, 64, 796–807. [Google Scholar] [CrossRef]
- Dhingra, D.; Jangra, A. Antiepileptic activity of ellagic acid, a naturally occurring polyphenolic compound, in mice. J. Funct. Foods 2014, 10, 364–369. [Google Scholar] [CrossRef]
- Girish, C.; Raj, V.; Arya, J.; Balakrishnan, S. Involvement of the GABAergic system in the anxiolytic-like effect of the flavonoid ellagic acid in mice. Eur. J. Pharmacol. 2013, 710, 49–58. [Google Scholar] [CrossRef]
- Gupta, P.; Mohammad, T.; Khan, P.; Alajmi, M.F.; Hussain, A.; Rehman, M.T.; Hassan, M.I. Evaluation of ellagic acid as an inhibitor of sphingosine kinase 1: A targeted approach towards anticancer therapy. Biomed. Pharmacother. 2019, 118, 109245. [Google Scholar] [CrossRef] [PubMed]
- Jha, A.B.; Panchal, S.S.; Shah, A. Ellagic acid: Insights into its neuroprotective and cognitive enhancement effects in sporadic Alzheimer’s disease. Pharmacol. Biochem. Behav. 2018, 175, 33–46. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.H.; Seo, U.M.; Zhao, B.T.; Le, D.D.; Seong, S.H.; Choi, J.S.; Min, B.S.; Woo, M.H. Ellagitannin and flavonoid constituents from Agrimonia pilosa Ledeb. with their protein tyrosine phosphatase and acetylcholinesterase inhibitory activities. Bioorg. Chem. 2017, 72, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Reitze, J.D.; Przewloka, S.R.; Shearer, B.J. The further chemistry of ellagic acid I. Synthesis of tetramethylellagic acid and associated polymer precursors. Holzforschung 2001, 55, 171–175. [Google Scholar] [CrossRef]
- Przewloka, S.R.; Shearer, B.J. The further chemistry of ellagic acid II. Ellagic acid and water-soluble ellagates as metal precipitants. Holzforschung 2002, 56, 13–19. [Google Scholar] [CrossRef]
- Zhang, N.Z.; Chen, Y.Y. Synthesis of macroporous ellagitannic acid resin and its chelating properties for metal ions. J. Macromol. Sci. Part A Chem. 1988, 25, 1455–1462. [Google Scholar] [CrossRef]
- Goriparti, S.; Harish, M.N.K.; Sampath, S. Ellagic acid—a novel organic electrode material for high capacity lithium ion batteries. Chem. Commun. 2013, 49, 7234–7236. [Google Scholar] [CrossRef]
- Gardner, J.A.F.; Hillis, W.E. The influence of extractives on the pulping of wood. In Wood Extractives and Their Significance to the Pulp and Paper Industries; Hillis, W.E., Ed.; ACADEMIC PRESS INC.: New York, NY, USA, 1962; pp. 367–403. ISBN 978-1-4832-3321-5. [Google Scholar] [CrossRef]
- Hemingway, R.W.; Hillis, W.E. Behavior of ellagitannins, gallic acid, and ellagic acid under alkaline conditions. TAPPI J. 1971, 54, 933–936. [Google Scholar]
- Sjöström, J.; Bädenlid, R.; Norborg, M.A. Short note: Analysis of ellagic acid in pulp mill deposits. Holzforschung 1993, 47, 446–448. [Google Scholar] [CrossRef]
- Pinto, P.C.R.; Sousa, G.; Crispim, F.; Silvestre, A.J.D.; Neto, C.P. Eucalyptus globulus bark as source of tannin extracts for application in leather industry. ACS Sustain. Chem. Eng. 2013, 1, 950–955. [Google Scholar] [CrossRef]
- Braconnot, H. Observations sur la préparation et la purification de l′acide gallique, et sur l′existence d′un acide nouveau dans la noix de galle. Ann. Chim. Phys. 1818, 9, 181–189. [Google Scholar]
- Berzelius, J.J. Acide ellagique (Acidum ellagicum) (I). In Traité de Chimie Minerale, Végétale et Animale, 2nd ed.; Esslinger, M., Hoefer, F., Eds.; Chez Firmin Didot frères: Paris, France, 1849; Volume 5, pp. 425–429. [Google Scholar]
- Mathieson, A.M.; Poppleton, B.J. The crystal structure of ellagic acid. Acta Crystallogr. Sect. B Struct. Crystallogr. Cryst. Chem. 1968, 24, 1456–1461. [Google Scholar] [CrossRef]
- Rossi, M.; Erlebacher, J.; Zacharias, D.E.; Carrell, H.L.; Iannucci, B. The crystal and molecular structure of ellagic acid dihydrate: A dietary anti-cancer agent. Carcinogenesis 1991, 12, 2227–2232. [Google Scholar] [CrossRef]
- Li, X.C.; Elsohly, H.N.; Hufford, C.D.; Clark, A.M. NMR assignments of ellagic acid derivatives. Magn. Reson. Chem. 1999, 37, 856–859. [Google Scholar] [CrossRef]
- Bala, I.; Bhardwaj, V.; Hariharan, S.; Kumar, M.N.V.R. Analytical methods for assay of ellagic acid and its solubility studies. J. Pharm. Biomed. Anal. 2006, 40, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Musialik, M.; Kuzmicz, R.; Pawcowski, T.S.; Litwinienko, G. Acidity of hydroxyl groups: An overlooked influence on antiradical properties of flavonoids. J. Org. Chem. 2009, 74, 2699–2709. [Google Scholar] [CrossRef]
- Simić, A.Z.; Verbić, T.Ž.; Sentić, M.N.; Vojić, M.P.; Juranić, I.O.; Manojlović, D.D. Study of ellagic acid electro-oxidation mechanism. Monatsh. Chem. 2013, 144, 121–128. [Google Scholar] [CrossRef]
- Marković, Z.; Milenković, D.; Đorović, J.; Dimitrić Marković, J.M.; Lučić, B.; Amić, D. A DFT and PM6 study of free radical scavenging activity of ellagic acid. Monatsh. Chem. Chem. Mon. 2013, 144, 803–812. [Google Scholar] [CrossRef]
- Nenadis, N.; Tsimidou, M.Z. Contribution of DFT computed molecular descriptors in the study of radical scavenging activity trend of natural hydroxybenzaldehydes and corresponding acids. Food Res. Int. 2012, 48, 538–543. [Google Scholar] [CrossRef]
- Okuda, T.; Yoshida, T.; Hatano, T.; Ito, H. Ellagitannins renewed the concept of tannins. In Chemistry and Biology of Ellagitannins: An Underestimated Class of Bioactive Plant Polyphenols; Quideau, S., Ed.; World Scientific Publishing: Singapore, 2009; pp. 1–54. ISBN 978-981-279-740-7. [Google Scholar]
- Quideau, S.; Jourdes, M.; Saucier, C.; Glories, Y.; Pardon, P.; Baudry, C. DNA topoisomerase inhibitor acutissimin a and other flavano-ellagitannins in red wine. Angew. Chem. Int. Ed. Engl. 2003, 42, 6012–6014. [Google Scholar] [CrossRef]
- Niemetz, R.; Gross, G.G. Enzymology of gallotannin and ellagitannin biosynthesis. Phytochemistry 2005, 66, 2001–2011. [Google Scholar] [CrossRef] [PubMed]
- Koponen, J.M.; Happonen, A.M.; Mattila, P.H.; Törrönen, A.R. Contents of anthocyanins and ellagitannins in selected foods consumed in Finland. J. Agric. Food Chem. 2007, 55, 1612–1619. [Google Scholar] [CrossRef] [PubMed]
- Määttä-Riihinen, K.R.; Kamal-Eldin, A.; Törrönen, A.R. Identification and quantification of phenolic compounds in berries of Fragaria and Rubus species (family Rosaceae). J. Agric. Food Chem. 2004, 52, 6178–6187. [Google Scholar] [CrossRef] [PubMed]
- Konczak, I.; Maillot, F.; Dalar, A. Phytochemical divergence in 45 accessions of Terminalia ferdinandiana (Kakadu plum). Food Chem. 2014, 151, 248–256. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.J.; Edwards, D.; Pun, S.; Chaliha, M.; Sultanbawa, Y. Profiling ellagic acid content: The importance of form and ascorbic acid levels. Food Res. Int. 2014, 66, 100–106. [Google Scholar] [CrossRef]
- Fukuda, T.; Ito, H.; Yoshida, T. Antioxidative polyphenols from walnuts (Juglans regia L.). Phytochemistry 2003, 63, 795–801. [Google Scholar] [CrossRef]
- Villarreal-Lozoya, J.E.; Lombardini, L.; Cisneros-Zevallos, L. Phytochemical constituents and antioxidant capacity of different pecan [Carya illinoinensis (Wangenh.) K. Koch] cultivars. Food Chem. 2007, 102, 1241–1249. [Google Scholar] [CrossRef]
- Fracassetti, D.; Costa, C.; Moulay, L.; Tomás-Barberán, F.A. Ellagic acid derivatives, ellagitannins, proanthocyanidins and other phenolics, vitamin C and antioxidant capacity of two powder products from camu-camu fruit (Myrciaria dubia). Food Chem. 2013, 139, 578–588. [Google Scholar] [CrossRef]
- Fischer, U.A.; Carle, R.; Kammerer, D.R. Identification and quantification of phenolic compounds from pomegranate (Punica granatum L.) peel, mesocarp, aril and differently produced juices by HPLC-DAD-ESI/MSn. Food Chem. 2011, 127, 807–821. [Google Scholar] [CrossRef]
- Lee, J.-H.; Johnson, J.V.; Talcott, S.T. Identification of ellagic acid conjugates and other polyphenolics in muscadine grapes by HPLC-ESI-MS. J. Agric. Food Chem. 2005, 53, 6003–6010. [Google Scholar] [CrossRef]
- Lu, J.; Yuan, Q. A new method for ellagic acid production from pomegranate husk. J. Food Process. Eng. 2008, 31, 443–454. [Google Scholar] [CrossRef]
- Hillis, W.E. The distribution and formation of polyphenols within the tree. In Wood Extractives and Their Significance to the Pulp and Paper Industries; Hillis, W.E., Ed.; ACADEMIC PRESS INC.: New York, NY, USA, 1962; pp. 59–131. ISBN 978-1-4832-3321-5. [Google Scholar] [CrossRef]
- Santos, S.A.O.; Villaverde, J.J.; Sousa, A.F.; Coelho, J.F.J.; Neto, C.P.; Silvestre, A.J.D. Phenolic composition and antioxidant activity of industrial cork by-products. Ind. Crops Prod. 2013, 47, 262–269. [Google Scholar] [CrossRef]
- Santos, S.A.O.; Vilela, C.; Domingues, R.M.A.; Oliveira, C.S.D.; Villaverde, J.J.; Freire, C.S.R.; Neto, C.P.; Silvestre, A.J.D. Secondary metabolites from Eucalyptus grandis wood cultivated in Portugal, Brazil and South Africa. Ind. Crops Prod. 2017, 95, 357–364. [Google Scholar] [CrossRef]
- Costa, E.V.; Lima, D.L.D.; Evtyugin, D.V.; Esteves, V.I. Development and application of a capillary electrophoresis method for the determination of ellagic acid in E. globulus wood and in filtrates from E. globulus kraft pulp. Wood Sci. Technol. 2014, 48, 99–108. [Google Scholar] [CrossRef]
- Conde, E.; Cadahia, E.; Garciavallejo, M.; Tomasbarberan, F. Low molecular weight polyphenols in wood and bark of Eucalyptus globulus. Wood Fiber Sci. 1995, 27, 379–383. [Google Scholar]
- Charrier, B.; Marques, M.; Haluk, J.P. HPLC analysis of gallic and ellagic acids in european oakwood (Quercus robur L.) and eucalyptus (Eucalyptus globulus). Holzforschung 1992, 46, 87–89. [Google Scholar] [CrossRef]
- Elgailani, I.E.H.; Ishak, C.Y. Determination of tannins of three common Acacia species of Sudan. Adv. Chem. 2014, 1–5. [Google Scholar] [CrossRef]
- Sanz, M.; Cadahía, E.; Esteruelas, E.; Muñoz, Á.M.; Fernández De Simón, B.; Hernández, T.; Estrella, I. Phenolic compounds in chestnut (Castanea sativa Mill.) heartwood. Effect of toasting at cooperage. J. Agric. Food Chem. 2010, 58, 9631–9640. [Google Scholar] [CrossRef]
- Fengel, D.; Wegener, G. Extractives. In Wood—Chemistry, Ultrastructure, Reactions; Walter de Gruyter: Berlin, Germany, 1989; pp. 182–226. ISBN 3-11-012059-3. [Google Scholar]
- Daniel, E.M.; Krupnick, A.S.; Heur, Y.H.; Blinzler, J.A.; Nims, R.W.; Stoner, G.D. Extraction, stability, and quantitation of ellagic acid in various fruits and nuts. J. Food Compos. Anal. 1989, 2, 338–349. [Google Scholar] [CrossRef]
- dos Santos, W.N.L.; da Silva Sauthier, M.C.; dos Santos, A.M.P.; de Andrade Santana, D.; Azevedo, R.S.A.; da Cruz Caldas, J. Simultaneous determination of 13 phenolic bioactive compounds in guava (Psidium guajava L.) by HPLC-PAD with evaluation using PCA and Neural Network Analysis (NNA). Microchem. J. 2017, 133, 583–592. [Google Scholar] [CrossRef]
- Alañón, M.E.; Castro-Vázquez, L.; Díaz-Maroto, M.C.; Hermosín-Gutiérrez, I.; Gordon, M.H.; Pérez-Coello, M.S. Antioxidant capacity and phenolic composition of different woods used in cooperage. Food Chem. 2011, 129, 1584–1590. [Google Scholar] [CrossRef]
- Santos, S.A.O.; Freire, C.S.R.; Domingues, M.R.M.; Silvestre, A.J.D.; Neto, C.P. Characterization of phenolic components in polar extracts of Eucalyptus globulus Labill. bark by high-performance liquid chromatography-mass spectrometry. J. Agric. Food Chem. 2011, 59, 9386–9393. [Google Scholar] [CrossRef]
- Santos, S.A.O.; José, J.; Freire, C.S.R.; Domingues, M.R.M.; Pascoal, C.; Silvestre, A.J.D. Phenolic composition and antioxidant activity of Eucalyptus grandis, E. urograndis (E. grandis × E. urophylla) and E. maidenii bark extracts. Ind. Crop. Prod. 2012, 39, 120–127. [Google Scholar] [CrossRef]
- Dedrie, M.; Jacquet, N.; Bombeck, P.L.; Hébert, J.; Richel, A. Oak barks as raw materials for the extraction of polyphenols for the chemical and pharmaceutical sectors: A regional case study. Ind. Crops Prod. 2015, 70, 316–321. [Google Scholar] [CrossRef]
- Comandini, P.; Lerma-García, M.J.; Simó-Alfonso, E.F.; Toschi, T.G. Tannin analysis of chestnut bark samples (Castanea sativa Mill.) by HPLC-DAD-MS. Food Chem. 2014, 157, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Chen, Z.; Han, F.; Kang, X.; Gu, H.; Yang, L. Microwave-assisted method for simultaneous hydrolysis and extraction in obtaining ellagic acid, gallic acid and essential oil from Eucalyptus globulus leaves using Brönsted acidic ionic liquid [HO3S(CH2)4mim]HSO4. Ind. Crops Prod. 2016, 81, 152–161. [Google Scholar] [CrossRef]
- Alexandri, M.; Papapostolou, H.; Vlysidis, A.; Gardeli, C.; Komaitis, M.; Papanikolaou, S.; Koutinas, A.A. Extraction of phenolic compounds and succinic acid production from spent sulphite liquor. J. Chem. Technol. Biotechnol. 2016, 91, 2751–2760. [Google Scholar] [CrossRef]
- Rana, V.; Joshi, G.; Singh, S.P.; Gupta, P.K. Eucalypts in pulp and paper industry. In Eucalypts in India; Bhojvaid, P.P., Kaushik, S., Singh, Y.P., Kumar, D., Thapliyal, M., Barthwal, S., Eds.; ENVIS Centre on Forestry, Forest Research Institute: Dehradun, India, 2014; pp. 470–506. ISBN 978-93-5174-121-3. [Google Scholar]
- Rodrigues, P.F.; Evtyugin, D.D.; Evtuguin, D.V.; Prates, A. Extractives profiles in the production of sulphite dissolving pulp from Eucalyptus globulus wood. J. Wood Chem. Technol. 2018, 38, 397–408. [Google Scholar] [CrossRef]
- Lei, Z.; Jervis, J.; Helm, R.F. Use of methanolysis for the determination of total ellagic and gallic acid contents of wood and food products. J. Agric. Food Chem. 2001, 49, 1165–1168. [Google Scholar] [CrossRef]
- Wilson, T.C.; Hagerman, A.E. Quantitative determination of ellagic acid. J. Agric. Food Chem. 1990, 38, 1678–1683. [Google Scholar] [CrossRef]
- Sádecká, J.; Tóthová, J. Spectrofluorimetric determination of ellagic acid in brandy. Food Chem. 2012, 135, 893–897. [Google Scholar] [CrossRef] [PubMed]
- Huerga-González, V.; Lage-Yusty, M.A.; Lago-Crespo, M.; López-Hernández, J. Comparison of methods for the study of ellagic acid in pomegranate juice beverages. Food Anal. Methods 2015, 8, 2286–2293. [Google Scholar] [CrossRef]
- Szmagara, A.; Krzyszczak, A.; Sadok, I.; Karczmarz, K.; Staniszewska, M.M.; Stefaniak, E.A. Determination of ellagic acid in rose matrix by spectrofluorimetry. J. Food Comp. Anal. 2019, 78, 91–100. [Google Scholar] [CrossRef]
- Matencio, A.; Navarro-Orcajada, S.; García-Carmona, F.; López-Nicolás, J.M. Ellagic acid—borax fluorescence interaction: Application for novel cyclodextrin-borax nanosensors for analyzing ellagic acid in food samples. Food Funct. 2018, 9, 3683–3687. [Google Scholar] [CrossRef] [PubMed]
- Kool, M.M.; Comeskey, D.J.; Cooney, J.M.; McGhie, T.K. Structural identification of the main ellagitannins of a boysenberry (Rubus loganbaccus × baileyanus Britt.) extract by LC-ESI-MS/MS, MALDI-TOF-MS and NMR spectroscopy. Food Chem. 2010, 119, 1535–1543. [Google Scholar] [CrossRef]
- Passos, H.; Freire, M.G.; Coutinho, J.A.P. Ionic liquid solutions as extractive solvents for value-added compounds from biomass. Green Chem. 2014, 16, 4786–4815. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, S.A.; Vijayaraghavan, R.; MacFarlane, D.R. Distillable ionic liquid extraction of tannins from plant materials. Green Chem. 2010, 12, 1023–1028. [Google Scholar] [CrossRef]
- Robledo, A.; Aguilera-Carbó, A.; Rodriguez, R.; Martinez, J.L.; Garza, Y.; Aguilar, C.N. Ellagic acid production by Aspergillus niger in solid state fermentation of pomegranate residues. J. Ind. Microbiol. Biotechnol. 2008, 35, 507–513. [Google Scholar] [CrossRef]
- Ascacio-Valdés, J.A.; Buenrostro, J.J.; De la Cruz, R.; Sepúlveda, L.; Aguilera, A.F.; Prado, A.; Contreras, J.C.; Rodríguez, R.; Aguilar, C.N. Fungal biodegradation of pomegranate ellagitannins. J. Basic Microbiol. 2014, 54, 28–34. [Google Scholar] [CrossRef]
- Seeram, N.; Lee, R.; Hardy, M.; Heber, D. Rapid large scale purification of ellagitannins from pomegranate husk, a by-product of the commercial juice industry. Sep. Purif. Technol. 2005, 41, 49–55. [Google Scholar] [CrossRef]
- Aguilera-Carbo, A.; Hernández, J.S.; Augur, C.; Prado-Barragan, L.A.; Favela-Torres, E.; Aguilar, C.N. Ellagic acid production from biodegradation of creosote bush ellagitannins by Aspergillus niger in solid state culture. Food Bioprocess. Technol. 2009, 2, 208–212. [Google Scholar] [CrossRef]
- Sepúlveda, L.; Aguilera-Carbó, A.; Ascacio-Valdés, J.A.; Rodríguez-Herrera, R.; Martínez-Hernández, J.L.; Aguilar, C.N. Optimization of ellagic acid accumulation by Aspergillus niger GH1 in solid state culture using pomegranate shell powder as a support. Process. Biochem. 2012, 47, 2199–2203. [Google Scholar] [CrossRef]
- Hu, T.; Zhou, Y.; Dai, L.; Wang, Y.; Liu, D.; Zhang, J.; Liu, H. Enhanced cellulase production by solid state fermentation with polyurethane foam as inert supports. Procedia Eng. 2011, 18, 335–340. [Google Scholar] [CrossRef][Green Version]
- Silva, M.F.; Rigo, D.; Mossi, V.; Dallago, R.M.; Henrick, P.; Kuhn, G.D.O.; Rosa, C.D.; Oliveira, D.; Oliveira, J.V.; Treichel, H. Evaluation of enzymatic activity of commercial inulinase from Aspergillus niger immobilized in polyurethane foam. Food Bioprod. Process. 2013, 91, 54–59. [Google Scholar] [CrossRef]
- Buenrostro-Figueroa, J.; Huerta-Ochoa, S.; Prado-Barragán, A.; Ascacio-Valdés, J.; Sepúlveda, L.; Rodríguez, R.; Aguilera-Carbó, A.; Aguilar, C.N. Continuous production of ellagic acid in a packed-bed reactor. Process. Biochem. 2014, 49, 1595–1600. [Google Scholar] [CrossRef]
- Sepúlveda, L.; Laredo-Alcalá, E.; Buenrostro-Figueroa, J.J.; Ascacio-Valdés, J.A.; Genisheva, Z.; Aguilar, C.; Teixeira, J. Ellagic acid production using polyphenols from orange peel waste by submerged fermentation. Electron. J. Biotechn. 2020, 43, 1–7. [Google Scholar] [CrossRef]
- Mizusawa, K.; Imai, Y.; Yuasa, K.; Koyama, H.; Yamaji, N.; Kataoka, S.; Oguma, T. Process for Producing Ellagic Acid. US Patent 52,311,93A, 27 July 1993. [Google Scholar]
- Hongwei, L.; Minhua, Y.; Yanming, Y.; Fan, L.; Hai, G.; Qingqing, Z. Method for Preparing Ellagic Acid from Tara Seeds Pod. CN Patent 1,078,279,00A, 23 March 2018. [Google Scholar]
- Yefu, M. Method and Device for Preparation of Ellagic Acid from Gall Flowers. CN Patent 1,051,754,27A, 23 December 2015. [Google Scholar]
- Llano, T.; Alexandri, M.; Koutinas, A.A.; Gardeli, C.; Papapostolou, H.; Coz, A.; Quijorna, N.; Andres, A.; Komaitis, M. Liquid–liquid extraction of phenolic compounds from spent sulphite liquor. Waste Biomass Valor. 2015, 6, 1149–1159. [Google Scholar] [CrossRef]
- Pizzi, A. Tannins: Major sources, properties and applications. In Monomers, Polymers and Composites from Renewable Resources, 1st ed.; Belgacem, M.N., Gandini, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2008; pp. 179–199. ISBN 978-0-08-045316-3. [Google Scholar]
- Chowdhury, S.A.; Dean, P.M.; Vijayaraghavan, R.; MacFarlane, D.R. Efficient synthesis of ellagic acid salts using distillable ionic liquids. Aust. J. Chem. 2011, 64, 1624–1627. [Google Scholar] [CrossRef]
- Löwe, J. Ueber die bildung von ellagsäure aus gallussäure. J. prakt. Chem. 1868, 103, 464–471. [Google Scholar] [CrossRef]
- Yamada, H.; Nagao, K.; Dokei, K.; Kasai, Y.; Michihata, N. Total synthesis of (–)-Corilagin. J. Am. Chem. Soc. 2008, 130, 7566–7567. [Google Scholar] [CrossRef]
- Yamada, H.; Ohara, K.; Ogura, T. Total synthesis of Cercidinin A. Eur. J. Org. Chem. 2013, 35, 7872–7875. [Google Scholar] [CrossRef]
- Yamaguchi, S.; Ashikaga, Y.; Nishii, K.; Yamada, H. Total synthesis of the proposed structure of roxbin B; the nonidentical outcome. Org. Lett. 2012, 14, 5928–5931. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, S.; Hirokane, T.; Yoshida, T.; Tanaka, T.; Hatano, T.; Ito, H.; Nonaka, G.-I.; Yamada, H. Roxbin B is cuspinin: Structural revision and total synthesis. J. Org. Chem. 2013, 78, 5410–5417. [Google Scholar] [CrossRef] [PubMed]
- Richieu, A.; Peixoto, P.; Pouységu, L.; Denis, D.; Quideau, S. Bio-inspired total synthesis of (−)-Vescalin, a nonahydroxytriphenoylated C-glucosidic ellagitannin. Angew. Chem. Int. Ed. 2017, 56, 13833–13837. [Google Scholar] [CrossRef]
- Wang, H.; Xu, X.; Lee, C.; Johnson, C.; Sohlberg, K.; Ji, H.F. Highly selective sensing of nitroaromatics using nanomaterials of ellagic acid. J. Phys. Chem. C 2012, 116, 4442–4448. [Google Scholar] [CrossRef]
- Gonçalves, S.S.L.; Rudnitskaya, A.; Sales, A.J.M.; Costa, L.M.C.; Evtuguin, D.V. Nanocomposite Polymeric Materials Based on Eucalyptus Lignoboost® Kraft Lignin for Liquid Sensing Applications. Materials 2020, 13, 1637. [Google Scholar] [CrossRef]
- Barnaby, S.N.; Yu, S.M.; Fath, K.R.; Tsiola, A.; Khalpari, O.; Banerjee, I.A. Ellagic acid promoted biomimetic synthesis of shape-controlled silver nanochains. Nanotechnology 2011, 22, 225605. [Google Scholar] [CrossRef]
- Frayne, S.H.; Barnaby, S.N.; Nakatsuka, N.; Banerjee, I.A. Growth and properties of CdSe nanoparticles on ellagic acid biotemplates for photodegradation applications. Mater. Express 2012, 2, 335–343. [Google Scholar] [CrossRef]
- Kim, S.; Liu, Y.; Gaber, M.W.; Bumgardner, J.D.; Haggard, W.O.; Yang, Y. Development of chitosan-ellagic acid films as a local drug delivery system to induce apoptotic death of human melanoma cells. J. Biomed. Mater. Res. B Appl. Biomater. 2009, 90, 145–155. [Google Scholar] [CrossRef]
- Shaik, M.M.; Kowshik, M. Ellagic acid containing collagen-chitosan scaffolds as potential antioxidative bio-materials for tissue engineering applications. Int. J. Polym. Mater. Polym. Biomater. 2019, 68, 208–215. [Google Scholar] [CrossRef]
- Arulmozhi, V.; Pandian, K.; Mirunalini, S. Ellagic acid encapsulated chitosan nanoparticles for drug delivery system in human oral cancer cell line (KB). Colloids Surf. B Biointerfaces 2013, 110, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Mady, F.M.; Shaker, M.A. Enhanced anticancer activity and oral bioavailability of ellagic acid through encapsulation in biodegradable polymeric nanoparticles. Int. J. Nanomed. 2017, 12, 7405–7417. [Google Scholar] [CrossRef] [PubMed]
- Vilela, C.; Pinto, R.J.B.; Coelho, J.; Domingues, M.R.M.; Daina, S.; Sadocco, P.; Santos, S.A.O.; Freire, C.S.R. Bioactive chitosan/ellagic acid films with UV-light protection for active food packaging. Food Hydrocoll. 2017, 73, 120–128. [Google Scholar] [CrossRef]
- Zhang, X.-K.; He, F.; Zhang, B.; Reeves, M.J.; Liu, Y.; Zhao, X.; Duan, C.-Q. The effect of prefermentative addition of gallic acid and ellagic acid on the red wine color, copigmentation and phenolic profiles during wine aging. Food Res. Int. 2018, 106, 568–579. [Google Scholar] [CrossRef]
- González-Sarrías, A.; García-Villalba, R.; Núñez-Sánchez, M.Á.; Tomé-Carneiro, J.; Zafrilla, P.; Mulero, J.; Tomás-Barberán, F.A.; Espín, J.C. Identifying the limits for ellagic acid bioavailability: A crossover pharmacokinetic study in healthy volunteers after consumption of pomegranate extracts. J. Funct. Foods 2015, 19, 225–235. [Google Scholar] [CrossRef]
- Cerdá, B.; Tomás-Barberán, F.A.; Espín, J.C. Metabolism of antioxidant and chemopreventive ellagitannins from strawberries, raspberries, walnuts, and oak-aged wine in humans: Identification of biomarkers and individual variability. J. Agric. Food Chem. 2005, 53, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Sarrias, A.; Gimenez-Bastida, J.A.; Garcia-Conesa, M.T.; Gomez-Sanchez, M.B.; Garcia-Talavera, N.V.; Gil-Izquierdo, A.; Sanchez-Alvarez, C.; Fontana-Compiano, L.O.; Morga-Egea, J.P.; Pastor-Quirante, F.A.; et al. Occurrence of urolithins, gut microbiota ellagic acid metabolites and proliferation markers expression response in the human prostate gland upon consumption of walnuts and pomegranate juice. Mol. Nutr. Food Res. 2010, 54, 311–322. [Google Scholar] [CrossRef]
- Tomás-Barberán, F.A.; Espín, J.C.; García-Conesa, M.T. Ellagitannins bioavailability and metabolism of ellagic acid and ellagitannins. In Chemistry and Biology of Ellagitannins: An Underestimated Class of Bioactive Plant Polyphenols; Quideau, S., Ed.; World Scientific Publishing: Singapore, 2009; pp. 273–297. ISBN 978-981-279-740-7. [Google Scholar]
- Tomás-Barberán, F.A.; Gonzalez-Sarrias, A.; García-Villalba, R.; Núñez-Sánchez, M.Á.; Selma, M.V.; Garcia-Conesa, M.T.; Espín, J.C. Urolithins, the rescue of “old” metabolites to understand a “new” concept: Metabotypes as a nexus among phenolic metabolism, microbiota dysbiosis, and host health status. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef]
- Beshbishy, A.M.; Batiha, G.E.; Yokoyama, N.; Igarashi, I. Ellagic acid microspheres restrict the growth of Babesia and Theileria in vitro and Babesia microti in vivo. Parasit. Vectors 2019, 12, 269. [Google Scholar] [CrossRef]
- Bulani, V.D.; Kothavade, P.S.; Nagmoti, D.M.; Kundaikar, H.S.; Degani, M.S.; Juvekar, A.R. Characterisation and anti-inflammatory evaluation of the inclusion complex of ellagic acid with hydroxypropyl-β-cyclodextrin. J. Incl. Phenom. Macrocycl. Chem. 2015, 82, 361–372. [Google Scholar] [CrossRef]
- Ratnam, D.V.; Chandraiah, G.; Meena, A.K.; Ramarao, P.; Ravi Kumar, M.N.V. The co-encapsulated antioxidant nanoparticles of ellagic acid and coenzyme Q10 ameliorates hyperlipidemia in high fat diet fed rats. J. Nanosci. Nanotechnol. 2009, 9, 6741–6746. [Google Scholar] [CrossRef] [PubMed]
- Ruan, J.; Yang, Y.; Yang, F.; Wan, K.; Fan, D.; Wang, D. Novel oral administrated ellagic acid nanoparticles for enhancing oral bioavailability and anti-inflammatory efficacy. J. Drug. Deliv. Sci. Tec. 2018, 46, 215–222. [Google Scholar] [CrossRef]
- Savic, I.M.; Jocic, E.; Nikolic, V.D.; Popsavin, M.M.; Rakic, S.J.; Savic-Gajic, I.M. The effect of complexation with cyclodextrins on the antioxidant and antimicrobial activity of ellagic acid. Pharm. Dev. Technol. 2018, 24, 410–418. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.-T.; Chou, C.-T.; Su, N.-W. A food-grade self-nanoemulsifying delivery system for enhancing oral bioavailability of ellagic acid. J. Funct. Foods 2017, 34, 207–215. [Google Scholar] [CrossRef]
- Selma, M.V.; Beltrán, D.; García-Villalba, R.; Espín, J.C.; Tomás-Barberán, F.A. Description of urolithin production capacity from ellagic acid of two human intestinal Gordonibacter species. Food Funct. 2014, 5, 1779–1784. [Google Scholar] [CrossRef] [PubMed]
- Selma, M.V.; Beltrán, D.; Luna, M.C.; Romo-Vaquero, M.; García-Villalba, R.; Mira, A.; Espín, J.C.; Tomás-Barberán, F.A. Isolation of human intestinal bacteria capable of producing the bioactive metabolite Isourolithin A from ellagic acid. Front. Microbiol. 2017, 8, 1521. [Google Scholar] [CrossRef] [PubMed]
- Gaya, P.; Peirotén, Á.; Medina, M.; Álvarez, I.; Landete, J.M. Bifidobacterium pseudocatenulatum INIA P815: The first bacterium able to produce urolithins A and B from ellagic acid. J. Funct. Foods 2018, 45, 95–99. [Google Scholar] [CrossRef]
- Fujita, Y.; Komagoe, K.; Sasaki, Y.; Uehara, I.; Okuda, T.; Yoshida, T. Studies on inhibition mechanism of autoxidation by tannins and flavonoids. I. Inhibition mechanism of tannins on Cu (II)-catalyzed autoxidation of ascorbic acid. Yakugaku Zasshi 1987, 107, 17–22. [Google Scholar] [CrossRef]
- Kilic, I.; Yeşiloǧlu, Y.; Bayrak, Y. Spectroscopic studies on the antioxidant activity of ellagic acid. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 130, 447–452. [Google Scholar] [CrossRef]
- Dröge, W. Free radicals in the physiological control of cell function. Physiol. Rev. 2002, 82, 47–95. [Google Scholar] [CrossRef]
- Jakus, V. The role of free radicals, oxidative stress and antioxidant systems in diabetic vascular disease. Bratisl. Lek. Listy 2000, 101, 541–551. [Google Scholar] [PubMed]
- Levonen, A.-L.; Vahakangas, E.; Koponen, J.K.; Yla-Herttuala, S. Antioxidant gene therapy for cardiovascular disease: Current status and future perspectives. Circulation 2008, 117, 2142–2150. [Google Scholar] [CrossRef] [PubMed]
- Morel, Y.; Barouki, R. Repression of gene expression by oxidative stress. Biochem. J. 1999, 342, 481–496. [Google Scholar] [CrossRef]
- Rice-Evans, C.; Miller, N.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant. Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
- Vattem, D.A.; Shetty, K. Biological functionality of ellagic acid: A review. J. Food Biochem. 2005, 29, 234–266. [Google Scholar] [CrossRef]
- Boehning, A.L.; Essien, S.A.; Underwood, E.L.; Dash, P.K.; Boehning, D. Cell type-dependent effects of ellagic acid on cellular metabolism. Biomed. Pharmacother. 2018, 106, 411–418. [Google Scholar] [CrossRef]
- Zeb, A. Ellagic acid in suppressing in vivo and in vitro oxidative stresses. Mol. Cell. Biochem. 2018, 448, 27–41. [Google Scholar] [CrossRef] [PubMed]
- Ríos, J.L.; Giner, R.M.; Marín, M.; Recio, M.C. A pharmacological update of ellagic acid. Planta Med. 2018, 84, 1068–1093. [Google Scholar] [CrossRef]
- Bialonska, D.; Kasimsetty, S.G.; Schrader, K.K.; Ferreira, D. The effect of pomegranate (Punica granatum L.) byproducts and ellagitannins on the growth of human gut bacteria. J. Agric. Food Chem. 2009, 57, 8344–8349. [Google Scholar] [CrossRef]
- Funatogawa, K.; Hayashi, S.; Shimomura, H.; Yoshida, T.; Hatano, T.; Ito, H.; Hirai, Y. Antibacterial activity of hydrolyzable tannins derived from medicinal plants against Helicobacter pylori. Microbiol. Immunol. 2004, 48, 251–261. [Google Scholar] [CrossRef]
- Asres, K.; Bucar, F.; Edelsbrunner, S.; Kartnig, T.; Höger, G.; Thiel, W. Investigations on antimycobacterial activity of some Ethiopian medicinal plants. Phyther. Res. 2001, 15, 323–326. [Google Scholar] [CrossRef] [PubMed]
- Kolodziej, H.; Kayser, O.; Kiderlen, A.; Ito, H.; Hatano, T.; Yoshida, T.; Foo, L. Antileishmanial activity of hydrolyzable tannins and their modulatory effects on nitric oxide and tumour necrosis factor-alpha release in macrophages in vitro. Planta Med. 2001, 67, 825–832. [Google Scholar] [CrossRef]
- Soh, P.N.; Witkowski, B.; Olagnier, D.; Nicolau, M.L.; Garcia-Alvarez, M.C.; Berry, A.; Benoit-Vical, F. In vitro and in vivo properties of ellagic acid in malaria treatment. Antimicrob. Agents Chemother. 2009, 53, 1100–1106. [Google Scholar] [CrossRef]
- Ascacio-Valdés, J.; Burboa, E.; Aguilera-Carbo, A.F.; Aparicio, M.; Pérez-Schmidt, R.; Rodríguez, R.; Aguilar, C.N. Antifungal ellagitannin isolated from Euphorbia antisyphilitica Zucc. Asian Pac. J. Trop. Biomed. 2013, 3, 41–46. [Google Scholar] [CrossRef]
- Vilhelmova-Ilieva, N.; Jacquet, R.; Quideau, S.; Galabov, A.S. Ellagitannins as synergists of ACV on the replication of ACV-resistant strains of HSV 1 and 2. Antiviral Res. 2014, 110, 104–114. [Google Scholar] [CrossRef]
- BenSaad, L.A.; Kim, K.H.; Quah, C.C.; Kim, W.R.; Shahimi, M. Anti-inflammatory potential of ellagic acid, gallic acid and punicalagin A&B isolated from Punica granatum. BMC Complement. Altern. Med. 2017, 17, 47. [Google Scholar] [CrossRef]
- Akileshwari, C.; Raghu, G.; Muthenna, P.; Mueller, N.H.; Suryanaryana, P.; Petrash, J.M.; Reddy, G.B. Bioflavonoid ellagic acid inhibits aldose reductase: Implications for prevention of diabetic complications. J. Funct. Foods 2014, 6, 374–383. [Google Scholar] [CrossRef]
- Al-Sayed, E.; El-Naga, R.N. Protective role of ellagitannins from Eucalyptus citriodora against ethanol-induced gastric ulcer in rats: Impact on oxidative stress, inflammation and calcitonin-gene related peptide. Phytomedicine 2015, 22, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Kannan, M.M.; Quine, S.D. Ellagic acid inhibits cardiac arrhythmias, hypertrophy and hyperlipidaemia during myocardial infarction in rats. Metabolism 2013, 62, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Rogerio, A.P.; Fontanari, C.; Borducchi, É.; Keller, A.C.; Russo, M.; Soares, E.G.; Albuquerque, D.A.; Faccioli, L.H. Anti-inflammatory effects of Lafoensia pacari and ellagic acid in a murine model of asthma. Eur. J. Pharmacol. 2008, 580, 262–270. [Google Scholar] [CrossRef]
- Ortiz-Ruiz, C.V.; Berna, J.; Tudela, J.; Varon, R.; Garcia-Canovas, F. Action of ellagic acid on the melanin biosynthesis pathway. J. Dermatol. Sci. 2016, 82, 115–122. [Google Scholar] [CrossRef] [PubMed]


| Activity | Active Compound | Main Features | Ref. |
|---|---|---|---|
| Antibacterial (Gram-Positive) | Commercial extract of pomegranate byproduct (POMx) and punicalagin | Inhibited the growth of pathogenic Clostridium and Staphyloccocus aureus | [130] |
| Antibacterial (Gram-Positive) | Ellagic acid | Action against Bacillus luteus and Listeria monocytogenes | [114] |
| Antibacterial (Gram-Negative) | Tellimagrandin I | Time- and dose-dependent bactericidal activity against Helicobacter pylori | [131] |
| Antibacterial (Gram-Negative) | Ellagic acid | EA—cyclodextrin complex expressed activity against Escherichia coli and Pseudomonas aeruginosa | [114] |
| Antimycobacterial | Punicalagin | Inhibited the growth of Mycobacterium tuberculosis typus humanus ATCC 27294 and patient strain of Mycobacterium tuberculosis sensitive to the standard antituberculosis drugs | [132] |
| Antileishmanial | Geraniin, phyllanthusiin B and elaeocarpusin | Exhibited effect against protozoa Leishmania donovani, comparable to that of the amphotericin B | [133] |
| Antimalarial | Ellagic acid | In vitro against all Plasmodium falciparum strains. In vivo against Plasmodium vinckei petteri; potentiates the activity of chloroquine, mefloquine, artesunate and atovaquone | [134] |
| Antibabesial | Ellagic acid | In vivo against Babesia microti; EA nanoparticles as an alternative antiparasitic agent | [110] |
| Antifungal | Candelitannin (ellagitannin) isolated from E. antisyphilitica Zucc. | Effective against Alternaria alternata, Fusarium oxyzporum, Colletotrichum gloeosporoides and Rhizoctnia solani | [135] |
| Antifungal | Ellagic acid | Action against Candida albicans | [114] |
| Antiviral | Castalagin, vescalagin and grandinin. | Action against acyclovir (ACV)—resistant strains of Herpes simplex virus HSV−1 and HSV-2; synergistic effects when used in combination with ACV | [136] |
| Prebiotic effect | Commercial extract of pomegranate byproduct (POMx) and punicalagin | Enhanced growth of Bifidobacterium breve and Bifidobacterium infantis | [130] |
| Anti-inflammatory | Ellagic acid, gallic acid and punicalagin A&B | Potential inhibition of LPS-induced NO, PGE-2 and IL-6 production | [137] |
| Anti-inflammatory | Ellagic acid | Enhancement of EA’s anti-inflammatory properties in vivo by inclusion complex of EA with hydroxypropyl-β-cyclodextrin | [111] |
| Treatment of Type 2 diabetes mellitus | Ellagic acid and ETs from Agrimonia pilosa Ledeb. | Inhibition of protein tyrosine phosphatases (PTP1B) | [13] |
| Prevention of diabetic complications | Ellagic acid | ALR2 (aldose reductase) inhibition and antiglycating effect of EA could possibly delay progression of cataract | [138] |
| Anticancerous agent | Ellagic acid | Inhibition of SphK1 (sphingosine kinase 1) | [11] |
| Antiangiogenic and antiproliferative effect | Ellagic acid | Reduction in metastatic potential of bladder cancer and enhancement of the efficacy of anti-VEGF-A therapies | [7] |
| Gastroprotective | Ellagitannin-rich fraction obtained from E. citriodora | Possibly due to their antioxidant, anti-inflammatory and anti-apoptotic properties. Partially mediated by attenuating induced oxidative stress and by the reduction of pro-inflammatory markers. | [139] |
| Hepatoprotective | Ellagic acid | Suppression of caspase-3, bcl-2, NF-kB and Nrf-2 | [6] |
| Antiarrhythmic | Ellagic acid | Antilipid peroxidation property and antihyperlipidemic activity through 3-hydroxy-3 methyl glutaryl CoA reductase inhibition; cardioprotective effect | [140] |
| Antiasthmatic | L. pacari extract and ellagic acid | Effective eosinophilic inflammation suppressors | [141] |
| Antihyperlipidemic | Ellagic acid | EA-CoQ10 nanoparticles effectively attenuated induced hyperlipidemia in rats | [112] |
| Antiepileptic | Ellagic acid | Possibly achieved through increase of brain GABA levels | [9] |
| Antianxiety | Ellagic acid | Possible involvement of GABAergic system in the anxiolytic action | [10] |
| Antidepressant | Ellagic acid | Possible interaction through adrenergic and serotonergic systems or through inhibition of inducible NOS | [8] |
| Neuroprotective in SAD | Ellagic acid | Diminished oxidative stress profile, pro-inflammatory markers, acetylcholinesterase activity, and amyloid-β plaque level in induced SAD (Sporadic Alzheimer’s Disease) rats | [12] |
| Skin-whitening agent | Ellagic acid | EA acts as an alternative substrate of tyrosinase, inhibiting the melanogenesis process | [142] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Evtyugin, D.D.; Magina, S.; Evtuguin, D.V. Recent Advances in the Production and Applications of Ellagic Acid and Its Derivatives. A Review. Molecules 2020, 25, 2745. https://doi.org/10.3390/molecules25122745
Evtyugin DD, Magina S, Evtuguin DV. Recent Advances in the Production and Applications of Ellagic Acid and Its Derivatives. A Review. Molecules. 2020; 25(12):2745. https://doi.org/10.3390/molecules25122745
Chicago/Turabian StyleEvtyugin, Dmitry D., Sandra Magina, and Dmitry V. Evtuguin. 2020. "Recent Advances in the Production and Applications of Ellagic Acid and Its Derivatives. A Review" Molecules 25, no. 12: 2745. https://doi.org/10.3390/molecules25122745
APA StyleEvtyugin, D. D., Magina, S., & Evtuguin, D. V. (2020). Recent Advances in the Production and Applications of Ellagic Acid and Its Derivatives. A Review. Molecules, 25(12), 2745. https://doi.org/10.3390/molecules25122745