Hybrid Quinoline-Sulfonamide Complexes (M2+) Derivatives with Antimicrobial Activity
Abstract
:1. Introduction
2. Results and Discussion
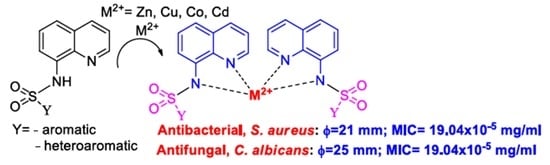
2.1. Design and Chemistry
2.2. Antimicrobial Assay
3. Experimental
3.1. Chemistry
3.1.1. General Procedure for the Synthesis of Sulfonamide Ligand 3a and its Complexes 4a–d
Synthesis of the Ligand
Synthesis of the Complexes
3.2. Antimicrobial Assay
3.2.1. Disk-Diffusion Method
3.2.2. Broth Microdilution Method
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brunton, L.; Knollmann, B.; Hilal-Dandan, R. Goodman & Gilman’s the Pharmacological Basis of Therapeutics, 13th ed.; McGraw-Hill: New York, NY, USA, 2013; ISBN 9781259584732. [Google Scholar]
- Silverman, R.B.; Holladay, M.W. The Organic Chemistry of Drug Design and Drug Action, 3rd ed.; Academic Press: Cambridge, MA, USA, 2014; ISBN 9780123820303. [Google Scholar]
- WHO Global Strategy for Containment of Antimicrobial Resistance. Available online: https://www.who.int/drugresistance/WHO_Global_Strategy_English.pdf (accessed on 14 June 2020).
- Guarner, J. Three Emerging Coronaviruses in Two Decades the Story of SARS, MERS, and Now COVID-19. Am. J. Clin. Pathol. 2020, 153, 420–421. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.; Li, X.; Liu, S.; Gao, S.J.; Gao, F. HIV-1 did not contribute to the 2019-nCoV genome. Emerg. Infect. Dis. 2020, 9, 378–381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kandeel, M.; Al-Nazawi, M. Virtual screening and repurposing of FDA approved drugs against COVID-19 main protease. Life Sci. 2020, 251, 117627. [Google Scholar] [CrossRef] [PubMed]
- Vellingiri, B.; Jayaramayya, K.; Iyer, M.; Kumar, N.S.; Subramaniam, M.D. COVID-19: A promising cure for the global panic. Sci. Total Environ. 2020, 725, 138277. [Google Scholar] [CrossRef] [PubMed]
- Yan, R.; Zhang, Y.; Li, Y.; Xia, L.; Guo, Y.; Zhou, Q. Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2. Science 2020, 367, 1444–1448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumari, L.S.; Mazumder, A.; Kumar, V.; Gupta, S. Synthesis and biological potentials of quinoline analogues: A review of literature. Mini-Rev. Org. Chem. 2019, 16, 653–688. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, S.; Ba, Y.; Xu, Z. 1,2,4-Triazole-quinoline/quinolone hybrids as potential anti-bacterial agents. Eur. J. Med. Chem. 2019, 174, 1–8. [Google Scholar] [CrossRef]
- Al Matarneh, C.; Sardaru, M.; Apostu, M.; Rosca, I.; Ciobanu, C.; Mangalagiu, I.I.; Danac, R. Synthesis and antibacterial evaluation of new pyrrolo[3’,4′:3,4]pyrrolo[1,2-a]quinoline and pyrrolo[3′,4′:3,4]pyrrolo[1,2-a]isoquinoline derivatives. Studia UBB Chem. 2019, 64, 67–80. [Google Scholar] [CrossRef]
- Kalaria, P.N.; Karad, S.C.; Raval, D.K. A review on diverse heterocyclic compounds as the privileged scaffolds in antimalarial drug discovery. Eur. J. Med. Chem. 2018, 158, 917–936. [Google Scholar] [CrossRef]
- Ajani, O.O.; Iyaye, K.T.; Audu, O.Y.; Kuye, A.O.; Olanrewaju, I.O. Microwave Assisted Synthesis and Antimicrobial Potential of Quinoline-Based 4-Hydrazide-Hydrazone Derivatives. J. Heterocycl. Chem. 2018, 55, 302–312. [Google Scholar] [CrossRef] [Green Version]
- Hu, Y.Q.; Gao, C.; Zhang, S.; Xu, L.; Xu, Z.; Feng, L.S.; Wu, X.; Zhao, F. Quinoline hybrids and their antiplasmodial and antimalarial activities. Eur. J. Med. Chem. 2017, 139, 22–47. [Google Scholar] [CrossRef]
- Mantu, D.; Antoci, V.; Moldoveanu, C.; Zbancioc, G.; Mangalagiu, I.I. Hybrid imidazole (benzimidazole)/pyridine (quinoline) derivatives and evaluation of their anticancer and antimycobacterial activity. J. Enz. Inhib. Med. Chem. 2016, 31, 96–103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhong, F.; Geng, G.; Chen, B.; Pan, T.; Li, Q.; Zhang, H.; Bai, C. Identification of benzenesulfonamide quinoline derivatives as potent HIV-1 replication inhibitors targeting Rev protein. Org. Biomol. Chem. 2015, 13, 1792–1799. [Google Scholar] [CrossRef] [PubMed]
- Sen, C.; Sahoo, T.; Singh, H.; Suresh, E.; Ghosh, S.C. Visible light-promoted photocatalytic C-5 carboxylation of 8-aminoquinoline amides and sulfonamides via a single electron transfer pathway. J. Org. Chem. 2019, 84, 9869–9896. [Google Scholar] [CrossRef] [PubMed]
- Pascual-Álvarez, A.; Topala, T.; Estevan, F.; Sanz, F.; Alzuet-Piña, G. Photoinduced and self-activated nuclease activity of copper(II) complexes with N-(quinolin-8-yl)quinolin-8-sulfonamide-DNA and bovine serum albumin binding. Eur. J. Inorg. Chem. 2016, 2016, 982–994. [Google Scholar] [CrossRef]
- Meeusen, J.W.; Tomasiewicz, H.; Nowakowski, A.; Petering, D.H. TSQ (6-methoxy-8-p-toluenesulfonamido-quinoline), a common fluorescent sensor for cellular zinc, images zinc proteins. Inorg. Chem. 2011, 50, 7563–7573. [Google Scholar] [CrossRef] [Green Version]
- Rouffet, M.; De Oliveira, C.A.F.; Udi, Y.; Agrawal, A.; Sagi, I.; McCammon, J.A.; Cohen, S.M. From sensors to silencers: Quinoline- and benzimidazole-sulfonamides as inhibitors for zinc proteases. J. Am. Chem. Soc. 2010, 132, 8232–8233. [Google Scholar] [CrossRef]
- Macías, B.; García, I.; Villa, M.V.; Borrás, J.; Castineiras, A.; Sanz, F. Synthesis and structural characterization of zinc complexes with sulfonamide containing 8-aminoquinoline. Z. Anorg. Allg. Chem. 2003, 629, 255–260. [Google Scholar] [CrossRef]
- Fahrni, C.J.; O’Halloran, T.V. Aqueous coordination chemistry of quinoline-based fluorescence probes for the biological chemistry of zinc. J. Am. Chem. Soc. 1999, 121, 11448–11458. [Google Scholar] [CrossRef]
- Da Silva, L.E.; de Sousa, P.T., Jr.; Joussef, A.C.; Piovezan, C.; Neves, A. Synthesis, structure and physicochemical properties of zinc and copper complexes based on sulfonamides containing 8-aminoquinoline ligands. Quim. Nova 2008, 31, 1161–1164. [Google Scholar] [CrossRef]
- Da Silva, L.E.; Joussef, A.C.; Pacheco, L.K.; Da Silva, D.G.; Steindel, M.; Rebelo, R.A.; Schmidt, B. Synthesis and in vitro evaluation of leishmanicidal and trypanocidal activities of N-quinolin-8-yl arylsulfonamides. Bioorg. Med. Chem. 2007, 15, 7553–7560. [Google Scholar] [CrossRef]
- Da Silva, L.E.; de Sousa, P.T., Jr.; Maciel, E.N.; Numes, R.K.; Eger, I.; Steindel, M.; Rebelo, R.A. In vitro antiprotozoal evaluation of zinc and copper complexes based on sulfonamides containing 8-aminoquinoline ligands. Lett. Drug Des. Discov. 2010, 7, 679–685. [Google Scholar] [CrossRef]
- Diaz, J.R.A.; Baldo, M.F.; Echeverría, G.; Baldoni, H.; Vullo, D.; Soria, D.B.; Supuran, C.T.; Cami, G.E. A substituted sulfonamide and its Co (II), Cu (II), and Zn (II) complexes as potential antifungal agents. J. Enz. Inhib. Med. Chem. 2016, 31, 51–62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Antoci, V.; Cucu, D.; Zbancioc, G.; Moldoveanu, C.; Mangalagiu, V.; Amariucai-Mantu, D.; Aricu, D.; Mangalagiu, I.I. Bis-(imidazole/benzimidazole)-pyridine derivatives: Synthesis, structure and antimycobacterial activity. Future Med. Chem. 2020, 12, 207–222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olaru, A.M.; Vasilache, V.; Danac, R.; Mangalagiu, I.I. Antimycobacterial activity of nitrogen heterocycles derivatives: 7-(pyridine-4-yl)-indolizine derivatives. Part VII. J. Enz. Inhib. Med. Chem. 2017, 32, 1291–1298. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mantu, D.; Antoci, V.; Nicolescu, A.; Delenu, C.; Vasilache, V.; Mangalagiu, I.I. Synthesis, stereochemical studies and antimycobacterial activity of new acetyl-hydrazines pyridazinone. Curr. Org. Synth. 2017, 14, 112–119. [Google Scholar] [CrossRef] [Green Version]
- Al Matarneh, C.M.; Shova, S.; Mangalagiu, I.I.; Danac, R. Synthesis, structure, antimycobacterial and anticancer evaluation of new pyrrolo-(phenanthroline) derivatives. J. Enz. Inhib. Med. Chem. 2016, 31, 470–480. [Google Scholar] [CrossRef] [Green Version]
- Aricu, A.; Ciocarlan, A.; Lungu, L.; Barba, A.; Shova, S.; Zbancioc, G.; Mangalagiu, I.I.; D’Ambrosio, M.; Vornicu, N. Synthesis of new antibacterial and antifungal drimane sesquiterpenoids with azaheterocyclic units. Med. Chem. Res. 2016, 25, 2316–2323. [Google Scholar] [CrossRef]
- Al Matarneh, C.; Ciobanu, C.I.; Mangalagiu, I.I.; Danac, R. Design, synthesis and antimycobacterial activity of some new azaheterocycles: 4,7-phenanthroline with p-halogeno-benzoyl skeleton. Part VI. J. Serb. Chem. Soc. 2016, 81, 133–140. [Google Scholar]
- Danac, R.; Al Matarneh, C.M.; Shova, S.; Daniloaia, T.; Balan, M.; Mangalagiu, I.I. New indolizines with phenanthroline skeleton: Synthesis, structure, antimycobacterial and anticancer evaluation. Bioorg. Med. Chem. 2015, 23, 2318–2327. [Google Scholar] [CrossRef]
- Danac, R.; Daniloaia, T.; Antoci, V.; Vasilache, V.; Mangalagiu, I.I. Design, synthesis and antimycobacterial activity of some new azaheterocycles: Phenanthroline with p-halo-benzoyl skeleton. Part, V. Lett. Drug Des. Discov. 2015, 12, 14–19. [Google Scholar] [CrossRef]
- Balan, A.M.; Miron, A.; Tuchilus, C.; Rotinberg, P.; Mihai, C.T.; Mangalagiu, I.I.; Zbancioc, G. Synthesis and in vitro analysis of novel dihydroxyacetophenone derivatives with antimicrobial and antitumor activities. Med. Chem. 2014, 10, 476–483. [Google Scholar]
- Kuchkova, K.; Aricu, A.; Barba, A.; Vlad, P.; Shova, S.; Secara, E.; Ungur, N.; Tuchilus, C.; Zbancioc, G.; Mangalagiu, I.I. Design, syntheses and antimicrobial activity of some novel homodrimane sesquiterpenoids with diazine skeleton. Med. Chem. Res. 2014, 23, 1559–1568. [Google Scholar] [CrossRef]
- Tucaliuc, R.; Cotea, V.; Niculaua, M.; Tuchilus, C.; Mantu, D.; Mangalagiu, I.I. New pyridazine–fluorine derivatives: Synthesis, chemistry and biological activity. Part II. Eur. J. Med. Chem. 2013, 67, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Mantu, D.; Antoci, V.; Mangalagiu, I.I. Design, synthesis and antituberculosis activity of some new pyridazine derivatives: Bis-pyridazine. Part IV. Infect. Disord. Drug Targets 2013, 13, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Mantu, D.; Luca, M.C.; Moldoveanu, C.; Zbancioc, G.; Mangalagiu, I.I. Synthesis and antituberculosis activity of some new pyridazine derivatives. Part II. Eur. J. Med. Chem. 2010, 45, 5164–5168. [Google Scholar] [CrossRef]
- Balan, A.M.; Florea, O.; Moldoveanu, C.; Zbancioc, G.; Iurea, D.; Mangalagiu, I.I. Diazinium salts with dihydroxyacetophenone skeleton: Syntheses and antimicrobial activity. Eur. J. Med. Chem. 2009, 44, 2275–2279. [Google Scholar] [CrossRef]
- CLSI Document M07-A11. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically; Approved Standard, 11th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2018; Available online: https://clsi.org/media/1928/m07ed11_sample.pdf (accessed on 14 June 2020).
- Kavanagh, A.; Ramu, S.; Gong, Y.; Copper, M.A.; Blaskovich, M.A.T. Effects of microplate type and broth additives on microdilution MIC susceptibility assays. Antimicrob. Agents Ch. 2019, 63, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Supuran, C.T. Carbonic anhydrases: Novel therapeutic applications for inhibitors and activators. Nat. Rev. Drug Discov. 2008, 7, 168–181. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |







| ν (cm−1) | Sulfonamide Ligand (3a) | Zinc Complex (4a) | Copper Complex (4b) | Cobalt Complex (4c) | Cadmium Complex (4d) |
|---|---|---|---|---|---|
| νN-H | 3267 (m) | - | - | - | - |
| νasSO2 | 1372 (s-m) | 1389 (s-m) | 1385 (s-m) | 1386 (s-m) | 1392 (s-m) |
| νsymSO2 | 1177 (s) | 1138 (s) | 1155 (s) | 1151 (s) | 1136 (s) |
| νC-N | 1584 (m) | 1583 (m) | 1583 (m) | 1584 (m) | 1578 (m) |
| νS-N | 927 (m) | 954 (m) | 954 (m) | 961 (m) | 957 (m) |
| νC-S | 624 (s) | 624 (s) | 629 (s) | 624 (s) | 621 (s) |
| Strain | Diameter of | Inhibition Zone | |||
|---|---|---|---|---|---|
| a 3a | a 4a (Zn) | a 4b (Cu) | a 4c (Co) | a 4d (Cd) | |
| S. aureus ATCC 25923 | 18 ± 1.73 | 11 ± 1.73 | 16 ± 1 | 17 ± 2 | 21± 2 |
| E. coli ATCC 25922 | 0 | 12 ± 2 | 14.5 ± 2 | 10.7 ± 2 | 19 ± 1.73 |
| C. albicans ATCC 10231 | 26.5± 1.80 | 12 ± 2 | 0 | 11 ± 1 | 25± 1.15 |
| Strain | MIC (mg/mL) | ||||
|---|---|---|---|---|---|
| 3a | 4a (Zn) | 4b (Cu) | 4c (Co) | 4d (Cd) | |
| S. aureus ATCC 25923 | 12.5 | 25 | 0.04875 | 12.5 | 0.00019 |
| E. coli ATCC 25922 | - | 25 | 0.04875 | 12.5 | 0.00609 |
| C. albicans ATCC 10231 | 12.5 | 12.5 | - | 12.5 | 0.00019 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diaconu, D.; Mangalagiu, V.; Amariucai-Mantu, D.; Antoci, V.; Giuroiu, C.L.; Mangalagiu, I.I. Hybrid Quinoline-Sulfonamide Complexes (M2+) Derivatives with Antimicrobial Activity. Molecules 2020, 25, 2946. https://doi.org/10.3390/molecules25122946
Diaconu D, Mangalagiu V, Amariucai-Mantu D, Antoci V, Giuroiu CL, Mangalagiu II. Hybrid Quinoline-Sulfonamide Complexes (M2+) Derivatives with Antimicrobial Activity. Molecules. 2020; 25(12):2946. https://doi.org/10.3390/molecules25122946
Chicago/Turabian StyleDiaconu, Dumitrela, Violeta Mangalagiu, Dorina Amariucai-Mantu, Vasilichia Antoci, Cristian Levente Giuroiu, and Ionel I. Mangalagiu. 2020. "Hybrid Quinoline-Sulfonamide Complexes (M2+) Derivatives with Antimicrobial Activity" Molecules 25, no. 12: 2946. https://doi.org/10.3390/molecules25122946