Otoprotective Effect of 2,3,4′,5-Tetrahydroxystilbene-2-O-β-d-Glucoside on Gentamicin-Induced Apoptosis in Mouse Cochlear UB/OC-2 Cells
Abstract
1. Introduction
2. Results
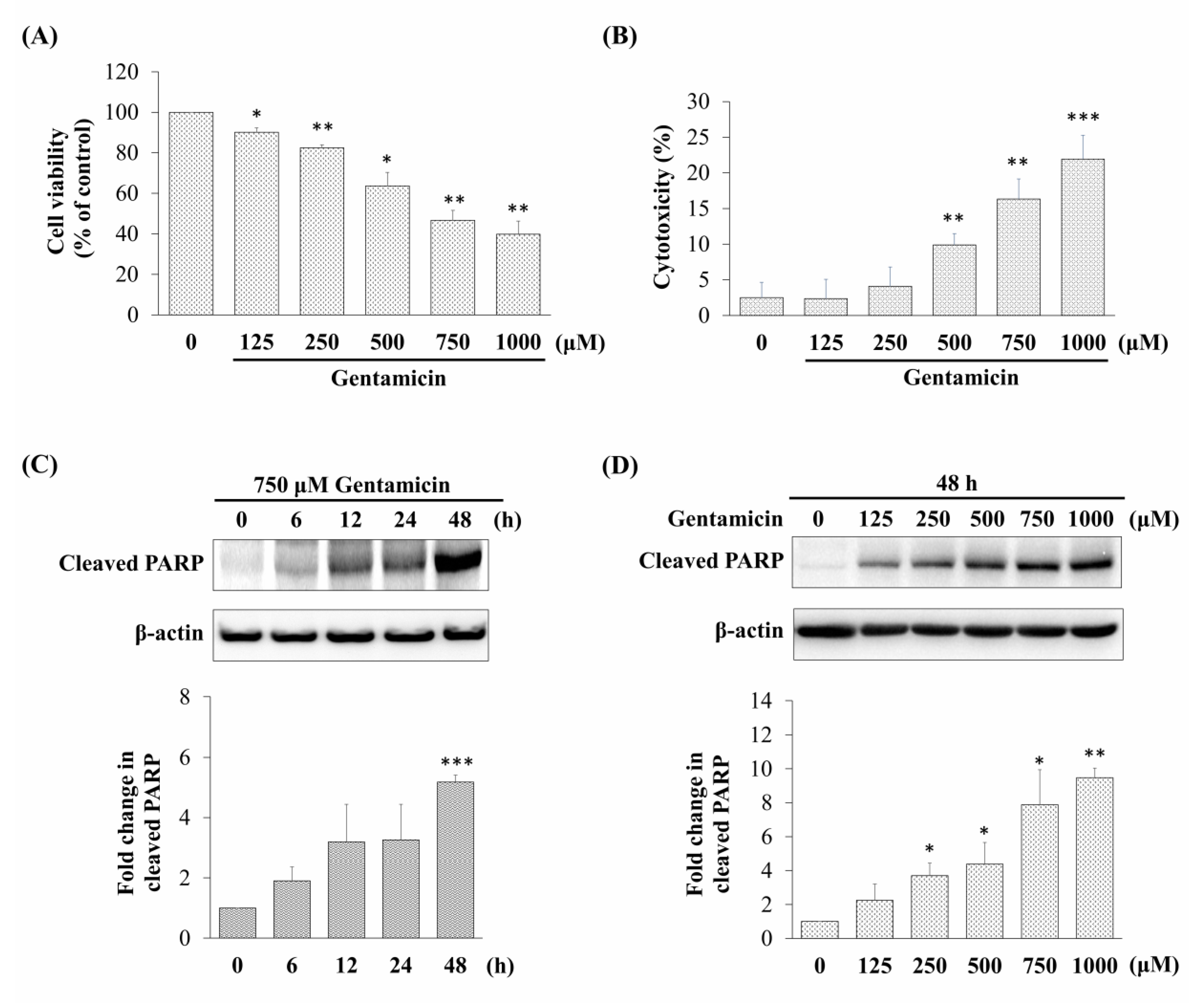
2.1. Gentamicin Suppresses Cell Viability and Induces Cell Cytotoxicity in University of Bristol/Organ of Corti-2 (UB/OC-2) Cells
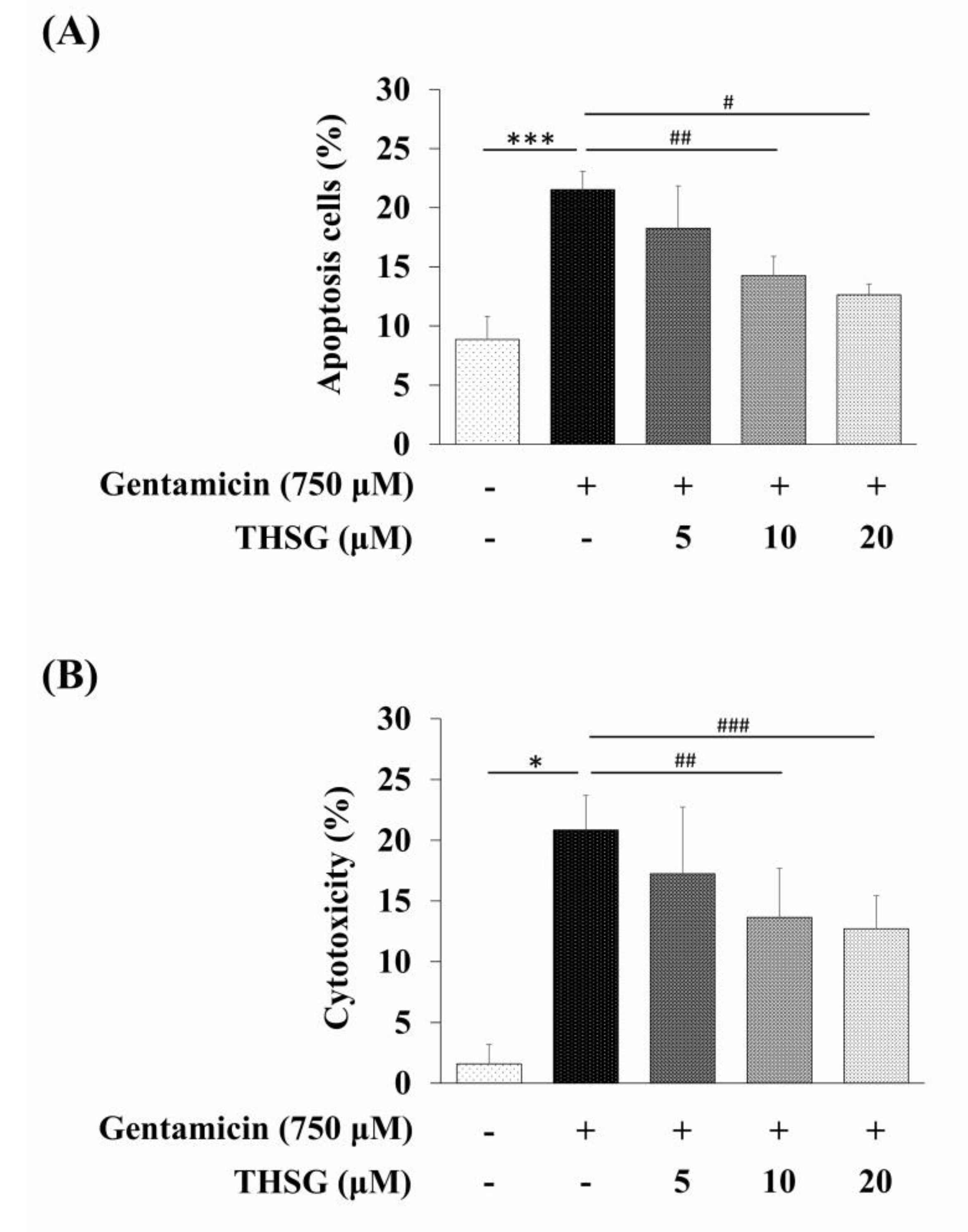
2.2. Gentamicin Promotes Apoptotic Cell Death in UB/OC-2 Cells
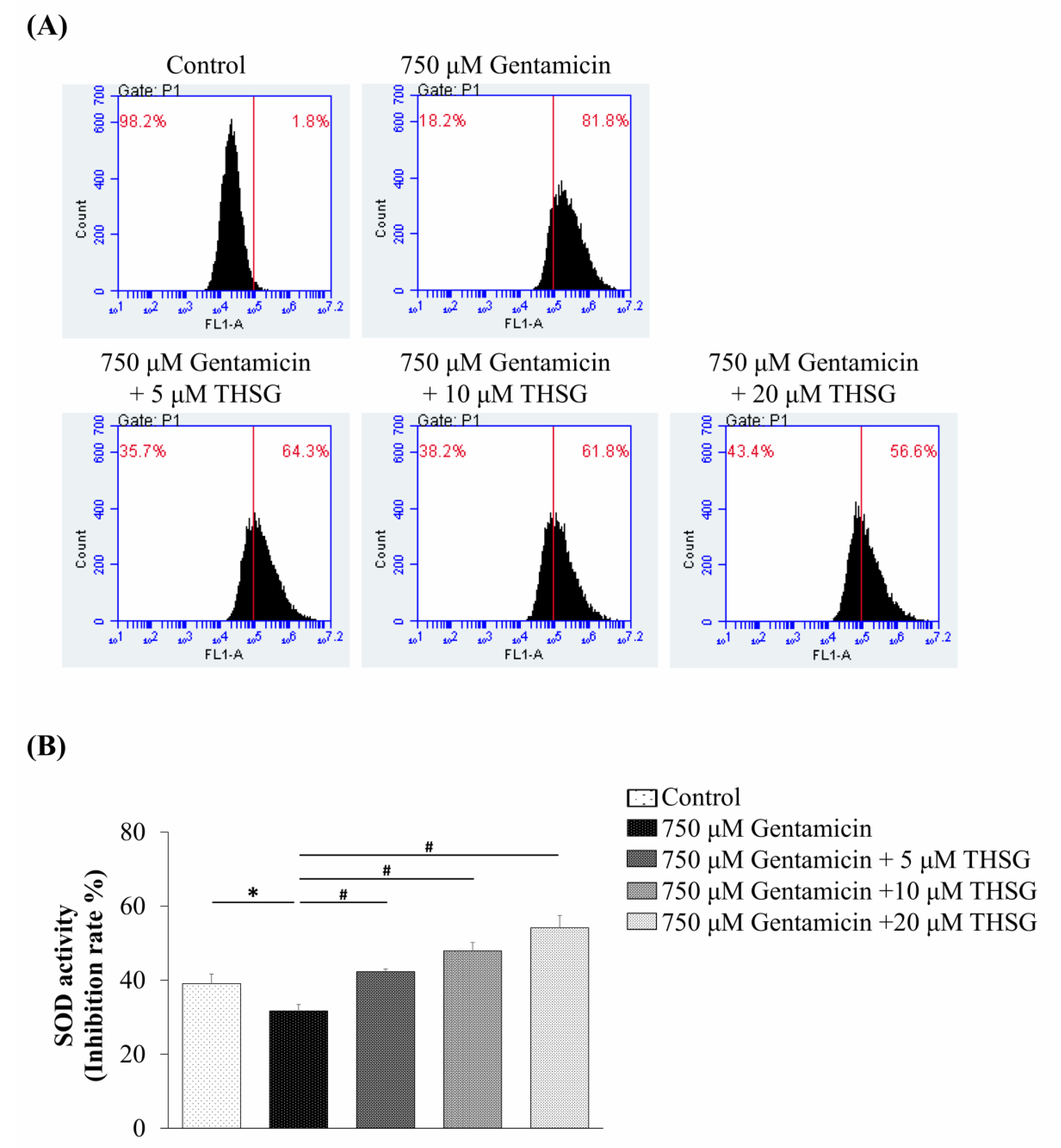
2.3. THSG Protects UB/OC-2 Cells against Gentamicin-Induced Oxidative Stress
2.4. THSG Impairs Gentamicin-Induced Mitochondrial-Dependent Apoptotic Pathway and Prevents Cell Death in UB/OC-2 Cells
3. Discussion
4. Materials and Methods
4.1. Cells and Materials
4.2. Cell Culture
4.3. Cell Viability Assay
4.4. LDH Cytotoxicity Assay
4.5. Western Blotting Analysis
4.6. ROS Detection Assay
4.7. Determination of SOD Activity
4.8. Hoechst 33258 Staining
4.9. JC-1 Staining
4.10. Cell Fractionation
4.11. Annexin V and PI Double Staining Assay
4.12. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- WHO. Deafness and Hearing Loss. Available online: https://www.who.int/health-topics/hearing-loss (accessed on 22 March 2020).
- Alvarado, J.C.; Fuentes-Santamaria, V.; Melgar-Rojas, P.; Valero, M.L.; Gabaldon-Ull, M.C.; Miller, J.M.; Juiz, J.M. Synergistic effects of free radical scavengers and cochlear vasodilators: A new otoprotective strategy for age-related hearing loss. Front. Aging Neurosci. 2015, 7, 86. [Google Scholar] [CrossRef] [PubMed]
- Arslan, E.; Orzan, E.; Santarelli, R. Global problem of drug-induced hearing loss. Ann. N. Y. Acad. Sci. 1999, 884, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Rybak, L.P.; Ramkumar, V. Ototoxicity. Kidney Int. 2007, 72, 931–935. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Gonzalez, S. The role of mitochondrial oxidative stress in hearing loss. Neurol. Disord. Ther. 2017, 1, 1–5. [Google Scholar] [CrossRef]
- Fujimoto, C.; Yamasoba, T. Oxidative stresses and mitochondrial dysfunction in age-related hearing loss. Oxid. Med. Cell Longev. 2014, 2014, 582849. [Google Scholar] [CrossRef]
- Ghashghaei, S.; Emtiazi, G. Increasing the antibacterial activity of gentamicin in combination with extracted polyphosphate from Bacillus megaterium. J. Appl. Microbiol. 2013, 114, 1264–1272. [Google Scholar] [CrossRef]
- Kros, C.J.; Steyger, P.S. Aminoglycoside- and Cisplatin-Induced Ototoxicity: Mechanisms and Otoprotective Strategies. Cold Spring Harb. Perspect Med. 2019, 9, a033548. [Google Scholar] [CrossRef]
- Quan, Y.; Xia, L.; Shao, J.; Yin, S.; Cheng, C.Y.; Xia, W.; Gao, W.Q. Adjudin protects rodent cochlear hair cells against gentamicin ototoxicity via the SIRT3-ROS pathway. Sci. Rep. 2015, 5, 8181. [Google Scholar] [CrossRef]
- Jiang, P.; Ray, A.; Rybak, L.P.; Brenner, M.J. Role of STAT1 and Oxidative Stress in Gentamicin-Induced Hair Cell Death in Organ of Corti. Otol. Neurotol. 2016, 37, 1449–1456. [Google Scholar] [CrossRef]
- Kalinec, G.M.; Fernandez-Zapico, M.E.; Urrutia, R.; Esteban-Cruciani, N.; Chen, S.; Kalinec, F. Pivotal role of Harakiri in the induction and prevention of gentamicin-induced hearing loss. Proc. Natl. Acad. Sci. USA 2005, 102, 16019–16024. [Google Scholar] [CrossRef]
- Ojano-Dirain, C.P.; Antonelli, P.J. Prevention of gentamicin-induced apoptosis with the mitochondria-targeted antioxidant mitoquinone. Laryngoscope 2012, 122, 2543–2548. [Google Scholar] [CrossRef] [PubMed]
- Somdas, M.A.; Korkmaz, F.; Gurgen, S.G.; Sagit, M.; Akcadag, A. N-acetylcysteine Prevents Gentamicin Ototoxicity in a Rat Model. J. Int. Adv. Otol. 2015, 11, 12–18. [Google Scholar] [CrossRef]
- Setz, C.; Benischke, A.S.; Pinho Ferreira Bento, A.C.; Brand, Y.; Levano, S.; Paech, F.; Leitmeyer, K.; Bodmer, D. Induction of mitophagy in the HEI-OC1 auditory cell line and activation of the Atg12/LC3 pathway in the organ of Corti. Hear. Res. 2018, 361, 52–65. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Tian, C.; Kim, J.; Shin, B.; Choo, O.S.; Kim, Y.S.; Choung, Y.H. Autophagic flux, a possible mechanism for delayed gentamicin-induced ototoxicity. Sci. Rep. 2017, 7, 41356. [Google Scholar] [CrossRef] [PubMed]
- Kucharava, K.; Sekulic-Jablanovic, M.; Horvath, L.; Bodmer, D.; Petkovic, V. Pasireotide protects mammalian cochlear hair cells from gentamicin ototoxicity by activating the PI3K-Akt pathway. Cell Death Dis. 2019, 10, 110. [Google Scholar] [CrossRef]
- Lai, R.; Li, W.; Hu, P.; Xie, D.; Wen, J. Role of Hsp90/Akt pathway in the pathogenesis of gentamicin-induced hearing loss. Int. J. Clin. Exp. Pathol. 2018, 11, 4431–4438. [Google Scholar]
- Heinrich, U.R.; Strieth, S.; Schmidtmann, I.; Li, H.; Helling, K. Gentamicin alters Akt-expression and its activation in the guinea pig cochlea. Neuroscience 2015, 311, 490–498. [Google Scholar] [CrossRef]
- Eshraghi, A.A.; Van de Water, T.R. Cochlear implantation trauma and noise-induced hearing loss: Apoptosis and therapeutic strategies. Anat. Rec. A Discov. Mol. Cell Evol. Biol. 2006, 288, 473–481. [Google Scholar] [CrossRef]
- Kamogashira, T.; Fujimoto, C.; Yamasoba, T. Reactive oxygen species, apoptosis, and mitochondrial dysfunction in hearing loss. Biomed. Res. Int. 2015, 2015, 617207. [Google Scholar] [CrossRef]
- Noack, V.; Pak, K.; Jalota, R.; Kurabi, A.; Ryan, A.F. An Antioxidant Screen Identifies Candidates for Protection of Cochlear Hair Cells from Gentamicin Toxicity. Front. Cell Neurosci. 2017, 11, 242. [Google Scholar] [CrossRef]
- Ling, S.; Xu, J.W. Biological Activities of 2,3,5,4′-Tetrahydroxystilbene-2-O-beta-d-Glucoside in Antiaging and Antiaging-Related Disease Treatments. Oxid. Med. Cell Longev. 2016, 2016, 4973239. [Google Scholar] [CrossRef] [PubMed]
- Lv, G.; Lou, Z.; Chen, S.; Gu, H.; Shan, L. Pharmacokinetics and tissue distribution of 2,3,5,4′-tetrahydroxystilbene-2-O-beta-d-glucoside from traditional Chinese medicine Polygonum multiflorum following oral administration to rats. J. Ethnopharmacol. 2011, 137, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Lv, L.; Shao, X.; Wang, L.; Huang, D.; Ho, C.T.; Sang, S. Stilbene glucoside from Polygonum multiflorum Thunb.: A novel natural inhibitor of advanced glycation end product formation by trapping of methylglyoxal. J. Agric. Food Chem. 2010, 58, 2239–2245. [Google Scholar] [CrossRef] [PubMed]
- Bai, H.B.; Wang, J.F.; Long, J. Study on optimizing extraction process of root of Polygonum multiflorum. Zhongguo Zhong Yao Za Zhi 2004, 29, 219–221. [Google Scholar]
- Zhang, W.; Wang, C.H.; Li, F.; Zhu, W.Z. 2,3,4′,5-Tetrahydroxystilbene-2-O-beta-d-glucoside suppresses matrix metalloproteinase expression and inflammation in atherosclerotic rats. Clin. Exp. Pharm. Physiol. 2008, 35, 310–316. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Wang, J.; Yao, W.; Chen, X.; Zhang, W. TSG (2,3,4′,5-tetrahydroxystilbene 2-O-beta-d-glucoside) suppresses induction of pro-inflammatory factors by attenuating the binding activity of nuclear factor-kappaB in microglia. J. Neuroinflam. 2013, 10, 129. [Google Scholar] [CrossRef]
- Sun, T.; Liu, H.; Cheng, Y.; Yan, L.; Krittanawong, C.; Li, S.; Qian, W.; Su, W.; Chen, X.; Hou, X.; et al. 2,3,5,4′-Tetrahydroxystilbene-2-O-beta-d-glucoside eliminates ischemia/reperfusion injury-induced H9c2 cardiomyocytes apoptosis involving in Bcl-2, Bax, caspase-3, and Akt activation. J. Cell Biochem. 2019, 120, 10972–10977. [Google Scholar] [CrossRef]
- Zhao, J.; Xu, S.; Song, F.; Nian, L.; Zhou, X.; Wang, S. 2,3,5,4′-tetrahydroxystilbene-2-O-beta-d-glucoside protects human umbilical vein endothelial cells against lysophosphatidylcholine-induced apoptosis by upregulating superoxide dismutase and glutathione peroxidase. Iubmb. Life 2014, 66, 711–722. [Google Scholar] [CrossRef]
- Han, X.; Ling, S.; Gan, W.; Sun, L.; Duan, J.; Xu, J.W. 2,3,5,4′-tetrahydroxystilbene-2-O-beta-d-glucoside ameliorates vascular senescence and improves blood flow involving a mechanism of p53 deacetylation. Atherosclerosis 2012, 225, 76–82. [Google Scholar] [CrossRef]
- Wu, T.Y.; Lin, J.N.; Luo, Z.Y.; Hsu, C.J.; Wang, J.S.; Wu, H.P. 2,3,4′,5-Tetrahydroxystilbene-2-O-beta-d-Glucoside (THSG) Activates the Nrf2 Antioxidant Pathway and Attenuates Oxidative Stress-Induced Cell Death in Mouse Cochlear UB/OC-2 Cells. Biomolecules 2020, 10, 465. [Google Scholar] [CrossRef]
- Rivolta, M.N.; Grix, N.; Lawlor, P.; Ashmore, J.F.; Jagger, D.J.; Holley, M.C. Auditory hair cell precursors immortalized from the mammalian inner ear. Proc. Biol. Sci. 1998, 265, 1595–1603. [Google Scholar] [CrossRef] [PubMed]
- Rivolta, M.N.; Holley, M.C. Cell lines in inner ear research. J. Neurobiol. 2002, 53, 306–318. [Google Scholar] [CrossRef] [PubMed]
- Weiss, S.; Gottfried, I.; Mayrose, I.; Khare, S.L.; Xiang, M.; Dawson, S.J.; Avraham, K.B. The DFNA15 deafness mutation affects POU4F3 protein stability, localization, and transcriptional activity. Mol. Cell Biol. 2003, 23, 7957–7964. [Google Scholar] [CrossRef] [PubMed]
- Brunetta, I.; Casalotti, S.O.; Hart, I.R.; Forge, A.; Reynolds, L.E. beta3-integrin is required for differentiation in OC-2 cells derived from mammalian embryonic inner ear. BMC Cell Biol. 2012, 13, 5. [Google Scholar] [CrossRef]
- Goncalves, A.C.; Towers, E.R.; Haq, N.; Porco, J.A., Jr.; Pelletier, J.; Dawson, S.J.; Gale, J.E. Drug-induced Stress Granule Formation Protects Sensory Hair Cells in Mouse Cochlear Explants During Ototoxicity. Sci. Rep. 2019, 9, 12501. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.L.; Zheng, M.S.; Lee, Y.K.; Moon, D.C.; Lee, C.S.; Woo, M.H.; Jeong, B.S.; Lee, E.S.; Jahng, Y.; Chang, H.W.; et al. A new stilbene glucoside from the roots of Polygonum multiflorum Thunb. Arch. Pharm. Res. 2006, 29, 946–951. [Google Scholar] [CrossRef]
- Han, M.N.; Lu, J.M.; Zhang, G.Y.; Yu, J.; Zhao, R.H. Mechanistic Studies on the Use of Polygonum multiflorum for the Treatment of Hair Graying. Biomed. Res. Int. 2015, 2015, 651048. [Google Scholar] [CrossRef]
- Hata, K.; Kozawa, M.; Baba, K. A new stilbene glucoside from Chinese crude drug "Heshouwu," the roots of Polygonum multiflorum Thunb. Yakugaku Zasshi 1975, 95, 211–213. [Google Scholar] [CrossRef][Green Version]
- Frombaum, M.; Le Clanche, S.; Bonnefont-Rousselot, D.; Borderie, D. Antioxidant effects of resveratrol and other stilbene derivatives on oxidative stress and *NO bioavailability: Potential benefits to cardiovascular diseases. Biochimie 2012, 94, 269–276. [Google Scholar] [CrossRef]
- Lin, E.Y.; Bayarsengee, U.; Wang, C.C.; Chiang, Y.H.; Cheng, C.W. The natural compound 2,3,5,4′-tetrahydroxystilbene-2-O-beta-d-glucoside protects against adriamycin-induced nephropathy through activating the Nrf2-Keap1 antioxidant pathway. Environ. Toxicol. 2018, 33, 72–82. [Google Scholar] [CrossRef]
- Cheng, J.; Wang, H.; Zhang, Z.; Liang, K. Stilbene glycoside protects osteoblasts against oxidative damage via Nrf2/HO-1 and NF-kappaB signaling pathways. Arch. Med. Sci. 2019, 15, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Ahn, S.M.; Wang, Z.; Choi, Y.W.; Shin, H.K.; Choi, B.T. Neuroprotective effects of 2,3,5,4′-tetrahydoxystilbene-2-O-beta-d-glucoside from Polygonum multiflorum against glutamate-induced oxidative toxicity in HT22 cells. J. Ethnopharmacol. 2017, 195, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Clerici, W.J.; Hensley, K.; DiMartino, D.L.; Butterfield, D.A. Direct detection of ototoxicant-induced reactive oxygen species generation in cochlear explants. Hear. Res. 1996, 98, 116–124. [Google Scholar] [CrossRef]
- Abi-Hachem, R.N.; Zine, A.; Van De Water, T.R. The injured cochlea as a target for inflammatory processes, initiation of cell death pathways and application of related otoprotectives strategies. Recent Pat. CNS Drug Discov. 2010, 5, 147–163. [Google Scholar] [CrossRef]
- El Mouedden, M.; Laurent, G.; Mingeot-Leclercq, M.P.; Taper, H.S.; Cumps, J.; Tulkens, P.M. Apoptosis in renal proximal tubules of rats treated with low doses of aminoglycosides. Antimicrob. Agents Chemother. 2000, 44, 665–675. [Google Scholar] [CrossRef]
- Jia, Z.; He, Q.; Shan, C.; Li, F. Tauroursodeoxycholic acid attenuates gentamicin-induced cochlear hair cell death in vitro. Toxicol. Lett. 2018, 294, 20–26. [Google Scholar] [CrossRef]
- Zhou, M.; Sun, G.; Zhang, L.; Zhang, G.; Yang, Q.; Yin, H.; Li, H.; Liu, W.; Bai, X.; Li, J.; et al. STK33 alleviates gentamicin-induced ototoxicity in cochlear hair cells and House Ear Institute-Organ of Corti 1 cells. J. Cell Mol. Med. 2018, 22, 5286–5299. [Google Scholar] [CrossRef]
- Wang, J.; Ladrech, S.; Pujol, R.; Brabet, P.; Van De Water, T.R.; Puel, J.L. Caspase inhibitors, but not c-Jun NH2-terminal kinase inhibitor treatment, prevent cisplatin-induced hearing loss. Cancer Res. 2004, 64, 9217–9224. [Google Scholar] [CrossRef]
- Li, P.; Nijhawan, D.; Budihardjo, I.; Srinivasula, S.M.; Ahmad, M.; Alnemri, E.S.; Wang, X. Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 1997, 91, 479–489. [Google Scholar] [CrossRef]
- Guo, C.; Sun, L.; Chen, X.; Zhang, D. Oxidative stress, mitochondrial damage and neurodegenerative diseases. Neural Regen. Res. 2013, 8, 2003–2014. [Google Scholar]
- Denamur, S.; Tyteca, D.; Marchand-Brynaert, J.; Van Bambeke, F.; Tulkens, P.M.; Courtoy, P.J.; Mingeot-Leclercq, M.P. Role of oxidative stress in lysosomal membrane permeabilization and apoptosis induced by gentamicin, an aminoglycoside antibiotic. Free Radic. Biol. Med. 2011, 51, 1656–1665. [Google Scholar] [CrossRef] [PubMed]
- Bodmer, D.; Brors, D.; Pak, K.; Bodmer, M.; Ryan, A.F. Gentamicin-induced hair cell death is not dependent on the apoptosis receptor Fas. Laryngoscope 2003, 113, 452–455. [Google Scholar] [CrossRef]
- Guan, M.X. Molecular pathogenetic mechanism of maternally inherited deafness. Mitochondrial Pathogenesis 2004, 1011, 259–271. [Google Scholar]
- Lin, C.D.; Kao, M.C.; Tsai, M.H.; Lai, C.H.; Wei, I.H.; Tsai, M.H.; Tang, C.H.; Lin, C.W.; Hsu, C.J.; Lin, C.Y. Transient ischemia/hypoxia enhances gentamicin ototoxicity via caspase-dependent cell death pathway. Lab. Investig. 2011, 91, 1092–1106. [Google Scholar] [CrossRef] [PubMed]
- Joza, N.; Susin, S.A.; Daugas, E.; Stanford, W.L.; Cho, S.K.; Li, C.Y.; Sasaki, T.; Elia, A.J.; Cheng, H.Y.; Ravagnan, L.; et al. Essential role of the mitochondrial apoptosis-inducing factor in programmed cell death. Nature 2001, 410, 549–554. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |



© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wen, Y.-H.; Lin, J.-N.; Wu, R.-S.; Yu, S.-H.; Hsu, C.-J.; Tseng, G.-F.; Wu, H.-P. Otoprotective Effect of 2,3,4′,5-Tetrahydroxystilbene-2-O-β-d-Glucoside on Gentamicin-Induced Apoptosis in Mouse Cochlear UB/OC-2 Cells. Molecules 2020, 25, 3070. https://doi.org/10.3390/molecules25133070
Wen Y-H, Lin J-N, Wu R-S, Yu S-H, Hsu C-J, Tseng G-F, Wu H-P. Otoprotective Effect of 2,3,4′,5-Tetrahydroxystilbene-2-O-β-d-Glucoside on Gentamicin-Induced Apoptosis in Mouse Cochlear UB/OC-2 Cells. Molecules. 2020; 25(13):3070. https://doi.org/10.3390/molecules25133070
Chicago/Turabian StyleWen, Yu-Hsuan, Jia-Ni Lin, Rong-Shuan Wu, Szu-Hui Yu, Chuan-Jen Hsu, Guo-Fang Tseng, and Hung-Pin Wu. 2020. "Otoprotective Effect of 2,3,4′,5-Tetrahydroxystilbene-2-O-β-d-Glucoside on Gentamicin-Induced Apoptosis in Mouse Cochlear UB/OC-2 Cells" Molecules 25, no. 13: 3070. https://doi.org/10.3390/molecules25133070
APA StyleWen, Y.-H., Lin, J.-N., Wu, R.-S., Yu, S.-H., Hsu, C.-J., Tseng, G.-F., & Wu, H.-P. (2020). Otoprotective Effect of 2,3,4′,5-Tetrahydroxystilbene-2-O-β-d-Glucoside on Gentamicin-Induced Apoptosis in Mouse Cochlear UB/OC-2 Cells. Molecules, 25(13), 3070. https://doi.org/10.3390/molecules25133070





