Taste Processing: Insights from Animal Models
Abstract
1. Introduction
2. Taste System
3. Taste Learning
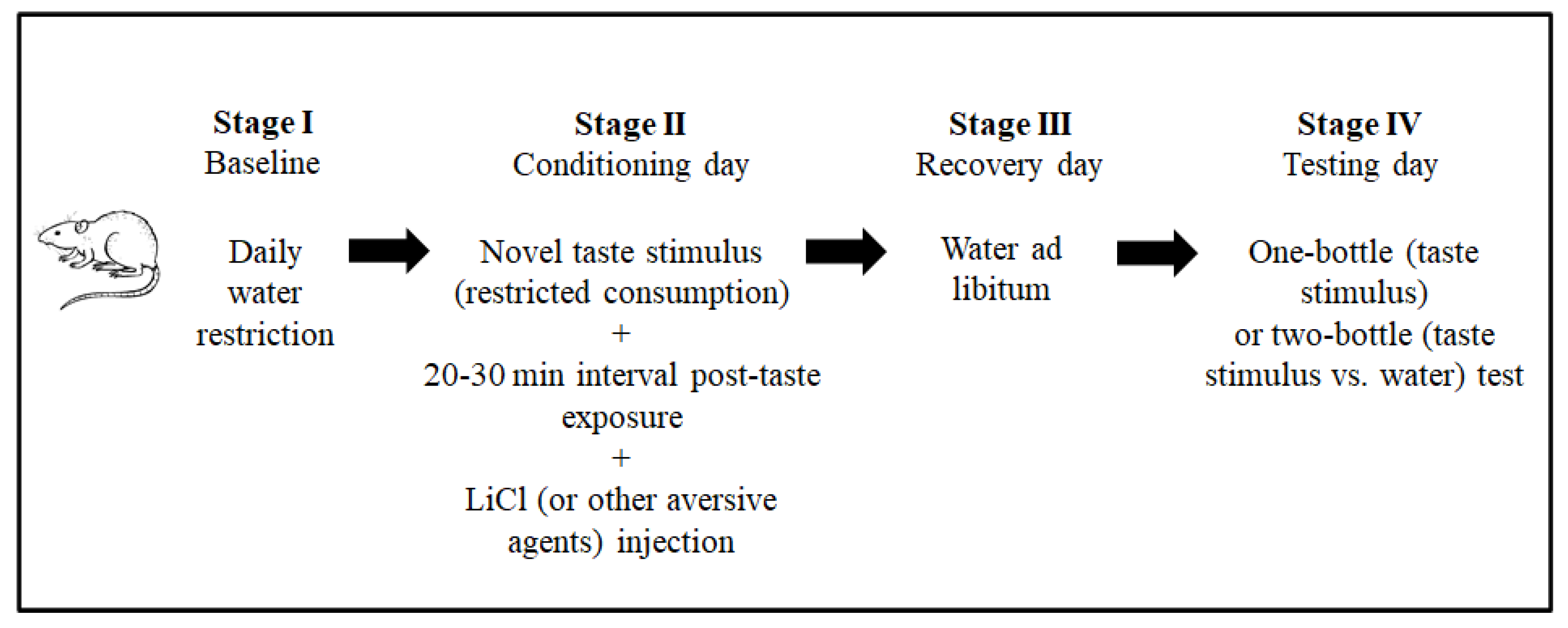
3.1. Conditioned Taste Aversion: A Peculiar Taste Learning
3.2. Neural Network of CTA
3.3. Molecular Mechanisms of CTA
4. Conclusions
Funding
Conflicts of Interest
References
- Roper, S.D.; Chaudhari, N. Taste buds: Cells, signals and synapses. Nat. Rev. Neurosci. 2017, 18, 485–497. [Google Scholar] [CrossRef] [PubMed]
- Palmer, R.K. A pharmacological perspective on the study of taste. Pharmacol. Rev. 2019, 71, 20–48. [Google Scholar] [CrossRef] [PubMed]
- Ohla, K.; Yoshida, R.; Roper, S.D.; Di Lorenzo, P.M.; Victor, J.D.; Boughter, J.D.; Fletcher, M.; Katz, D.B.; Chaudhari, N. Recognizing taste: Coding patterns along the neural axis in mammals. Chem. Senses 2019, 44, 237–247. [Google Scholar] [CrossRef] [PubMed]
- de Brito Sanchez, G.; Giurfa, M. A comparative analysis of neural taste processing in animals. Philos. Trans. R. Soc. B Biol. Sci. 2011, 366, 2171–2180. [Google Scholar] [CrossRef] [PubMed]
- De La Cruz, V.; Rodriguez-Ortiz, C.J.; Balderas, I.; Bermudez-Rattoni, F. Medial temporal lobe structures participate differentially in consolidation of safe and aversive taste memories. Eur. J. Neurosci. 2008, 28, 1377–1381. [Google Scholar] [CrossRef]
- Molero-Chamizo, A.; Nathzidy Rivera-Urbina, G. Molecular mechanisms involved in taste learning and memory. AIMS Mol. Sci. 2017, 4, 389–397. [Google Scholar] [CrossRef]
- Lee, H.; MacPherson, L.J.; Parada, C.A.; Zuker, C.S.; Ryba, N.J.P. Rewiring the taste system. Nature 2017, 548, 330–333. [Google Scholar] [CrossRef] [PubMed]
- Harrer, M.I.; Travers, S.P. Topographic organization of Fos-like immunoreactivity in the rostral nucleus of the solitary tract evoked by gustatory stimulation with sucrose and quinine. Brain Res. 1996, 711, 125–137. [Google Scholar] [CrossRef]
- Spray, K.J.; Bernstein, I.L. Afferent and efferent connections of the parvicellular subdivision of iNTS: Defining a circuit involved in taste aversion learning. Behav. Brain Res. 2004, 154, 85–97. [Google Scholar] [CrossRef]
- Scott, T.R. Learning through the taste system. Front. Syst. Neurosci. 2011, 5, 87. [Google Scholar] [CrossRef]
- Dayawansa, S.; Ruch, S.; Norgren, R. Parabrachial-hypothalamic interactions are required for normal conditioned taste aversions. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2014, 306, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Bielavska, E.; Roldan, G. Ipsilateral connections between the gustatory cortex, amygdala and parabrachial nucleus are necessary for acquisition and retrieval of conditioned taste aversion in rats. Behav. Brain Res. 1996, 81, 25–31. [Google Scholar] [CrossRef]
- Small, D.M. Taste representation in the human insula. Brain Struct. Funct. 2010, 214, 551–561. [Google Scholar] [CrossRef]
- Stehberg, J.; Moraga-Amaro, R.; Simon, F. The role of the insular cortex in taste function. Neurobiol. Learn. Mem. 2011, 96, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Wallroth, R.; Ohla, K. As soon as you taste it: Evidence for sequential and parallel processing of gustatory information. eNeuro 2018, 5. [Google Scholar] [CrossRef]
- Lunceford, B.E.; Kubanek, J. Reception of aversive taste. Integr Comp Biol. 2015, 55, 507–517. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Reed, R.R. Signaling pathways in odorant detection. Neuron 1992, 8, 205–209. [Google Scholar] [CrossRef]
- Wackermannová, M.; Pinc, L.; Jebavý, L. Olfactory sensitivity in mammalian species. Physiol. Res. 2016, 65, 369–390. [Google Scholar] [CrossRef]
- Tham, W.W.P.; Stevenson, R.J.; Miller, L.A. The functional role of the medio dorsal thalamic nucleus in olfaction. Brain Res. Rev. 2009, 62, 109–126. [Google Scholar] [CrossRef]
- Small, D.M.; Green, B.G. A proposed model of a flavor modality. In The Neural Bases of Multisensory Processes; Murray, M.M., Wallace, M.T., Eds.; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2012; ISBN 9781439812198. [Google Scholar]
- Sakai, N.; Imada, S. Bilateral lesions of the insular cortex or of the prefrontal cortex block the association between taste and odor in the rat. Neurobiol. Learn. Mem. 2003, 80, 24–31. [Google Scholar] [CrossRef]
- Fanselow, M.S.; Birk, J. Flavor-flavor associations induce hedonic shifts in taste preference. Anim. Learn. Behav. 1982, 10, 223–228. [Google Scholar] [CrossRef]
- De Araujo, I.E.; Ferreira, J.G.; Tellez, L.A.; Ren, X.; Yeckel, C.W. The gut-brain dopamine axis: A regulatory system for caloric intake. Physiol. Behav. 2012, 106, 394–399. [Google Scholar] [CrossRef]
- Sclafani, A.; Ackroff, K. Flavor preferences conditioned by nutritive and non-nutritive sweeteners in mice. Physiol. Behav. 2017, 173, 188–199. [Google Scholar] [CrossRef] [PubMed]
- Han, W.; Tellez, L.A.; Perkins, M.H.; Perez, I.O.; Qu, T.; Ferreira, J.; Ferreira, T.L.; Quinn, D.; Liu, Z.W.; Gao, X.B.; et al. A neural circuit for gut-induced reward. Cell 2018, 175, 887–888. [Google Scholar] [CrossRef] [PubMed]
- Lenoir, M.; Serre, F.; Cantin, L.; Ahmed, S.H. Intense sweetness surpasses cocaine reward. PLoS ONE 2007, 2, e698. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, G.; Ferry, B.; Meurisse, M.; Lévy, F. Forebrain structures specifically activated by conditioned taste aversion. Behav. Neurosci. 2006, 120, 952–962. [Google Scholar] [CrossRef] [PubMed]
- Molero-Chamizo, A. Taste aversions. In Encyclopedia of Animal Cognition and Behavior; Springer: Berlin, Germany, 2018; pp. 1–7. [Google Scholar]
- Bernstein, I.L. Taste aversion learning: A contemporary perspective. Nutrition 1999, 15, 229–234. [Google Scholar] [CrossRef]
- Bures, J.; Bermudez-Rattoni, F.; Yamamoto, T. Ethology, physiological psychology, and neurobiology of CTA. In Conditioned Taste Aversion: Memory of a Special Kind; Oxford University Press: New York, NY, USA, 1998; pp. 1–10. [Google Scholar]
- Bures, J.; Bermudez-Rattoni, F.; Yamamoto, T. The CTA paradigm: Terminology, methods, and conventions. In Conditioned Taste Aversion: Memory of A Special Kind; Bures, J., Bermúdez-Rattoni, F., Yamamoto, T., Eds.; Oxford University Press: New York, NY, USA, 1998; pp. 14–25. [Google Scholar]
- Molero-Chamizo, A.; Moron, I. Latent inhibition of conditioned taste aversion in rats with excitotoxic dorsal hippocampal lesions. J. Neurosci. Res. 2015, 93, 1740–1747. [Google Scholar] [CrossRef]
- Roman, C.; Lin, J.Y.; Reilly, S. Conditioned taste aversion and latent inhibition following extensive taste preexposure in rats with insular cortex lesions. Brain Res. 2009, 1259, 68–73. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Molero-Chamizo, A. Excitotoxic lesion of the hippocampus of Wistar rats disrupts the circadian control of the latent inhibition of taste aversion learning. Brain Res. 2013, 1533, 105–113. [Google Scholar] [CrossRef]
- Molero-Chamizo, A. Excitotoxic lesion of the posterior part of the dorsal striatum does not affect the typically dopaminergic phenomenon of latent inhibition in conditioned taste aversion. Neurosci. Res. 2015, 91, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Molero-Chamizo, A.; Rivera-Urbina, G.N. Effects of temporal contexts and contextual habituation on latent inhibition. Psicothema 2017, 29, 346–351. [Google Scholar] [CrossRef]
- Molero-Chamizo, A. Circadian-temporal context and latent inhibition of conditioned taste aversion: Effect of restriction in the intake of the conditioned taste stimulus. Learn. Behav. 2016, 45, 157–163. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Molero-Chamizo, A. Changes in the time of day of conditioning with respect to the pre-exposure interfere with the latent inhibition of conditioned taste aversion in rats. Behav. Processes 2018, 146, 22–26. [Google Scholar] [CrossRef] [PubMed]
- Traverso, L.M.; Ruiz, G.; De La Casa, L.G. MK-801 induces a low intensity conditioned taste aversion. Pharmacol. Biochem. Behav. 2012, 100, 645–651. [Google Scholar] [CrossRef] [PubMed]
- St. John, S.J.; Pour, L.; Boughter, J.D. Phenylthiocarbamide produces conditioned taste aversions in mice. Chem. Senses 2005, 30, 377–382. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yamamoto, T.; Ueji, K. Brain mechanisms of flavor learning. Front. Syst. Neurosci. 2011, 5, 76. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T. Neural mechanisms of taste aversion learning. Neurosci. Res. 1993, 16, 181–185. [Google Scholar] [CrossRef]
- Clark, E.W.; Bernstein, I.L. Establishing aversive, but not safe, taste memories requires lateralized pontine-cortical connections. Behav. Brain Res. 2009, 197, 356–363. [Google Scholar] [CrossRef][Green Version]
- Marotta, R.; Fenu, S.; Scheggi, S.; Vinci, S.; Rosas, M.; Falqui, A.; Gambarana, C.; De Graziella, M.G.; Acquas, E. Acquisition and expression of conditioned taste aversion differentially affects extracellular signal regulated Kinase and glutamate receptor phosphorylation in rat prefrontal cortex and nucleus accumbens. Front. Behav. Neurosci. 2014, 8, 153. [Google Scholar] [CrossRef][Green Version]
- Grau-Perales, A.; Gómez-Chacón, B.; Morillas, E.; Gallo, M. Flavor recognition memory related activity of the posterior piriform cortex in adult and aged rats. Behav. Brain Res. 2019, 360, 196–201. [Google Scholar] [CrossRef]
- Morillas, E.; Gómez-Chacón, B.; Gallo, M. Flavor and object recognition memory impairment induced by excitotoxic lesions of the perirhinal cortex. Neurobiol. Learn. Mem. 2017, 144, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Reilly, S.; Bornovalova, M.A. Conditioned taste aversion and amygdala lesions in the rat: A critical review. Neurosci. Biobehav. Rev. 2005, 29, 1067–1088. [Google Scholar] [CrossRef]
- Schafe, G.E.; Bernstein, I.L. Forebrain contribution to the induction of a brainstem correlate of conditioned taste aversion: I. The amygdala. Brain Res. 1996, 741, 109–116. [Google Scholar] [CrossRef]
- St. Andre, J.; Reilly, S. Effects of central and basolateral amygdala lesions on conditioned taste aversion and latent inhibition. Behav. Neurosci. 2007, 121, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Sako, N.; Sakai, N.; Iwafune, A. Gustatory and visceral inputs to the amygdala of the rat: Conditioned taste aversion and induction of c-fos-like immunoreactivity. Neurosci. Lett. 1997, 226, 127–130. [Google Scholar] [CrossRef]
- Molero-Chamizo, A.; Rivera-Urbina, G.N. Effects of lesions in different nuclei of the amygdala on conditioned taste aversion. Exp. Brain Res. 2017, 235, 3517–3526. [Google Scholar] [CrossRef]
- Molero-Chamizo, A. Modulation of the magnitude of conditioned taste aversion in rats with excitotoxic lesions of the basolateral amygdala. Neurobiol. Learn. Mem. 2017, 137, 56–64. [Google Scholar] [CrossRef]
- Reilly, S. Central gustatory system lesions and conditioned taste aversion. In Conditioned Taste Aversion: Behavioral and Neural Processes; Reilly, S., Schachtman, T.R., Eds.; Oxford University Press: New York, NY, USA, 2009; pp. 309–327. [Google Scholar]
- Lavi, K.; Jacobson, G.A.; Rosenblum, K.; Lüthi, A. Encoding of conditioned taste aversion in cortico-amygdala circuits. Cell Rep. 2018, 24, 278–283. [Google Scholar] [CrossRef]
- Kayyal, H.; Yiannakas, A.; Kolatt Chandran, S.; Khamaisy, M.; Sharma, V.; Rosenblum, K. Activity of insula to basolateral amygdala projecting neurons is necessary and sufficient for taste valence representation. J. Neurosci. 2019, 39, 9369–9382. [Google Scholar] [CrossRef]
- Bermúdez-Rattoni, F.; Ramírez-Lugo, L.; Gutiérrez, R.; Miranda, M.I. Molecular signals into the insular cortex and amygdala during aversive gustatory memory formation. Cell. Mol. Neurobiol. 2004, 24, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Miranda, M.I.; McGaugh, J.L. Enhancement of inhibitory avoidance and conditioned taste aversion memory with insular cortex infusions of 8-Br-cAMP: Involvement of the basolateral amygdala. Learn. Mem. 2004, 11, 312–317. [Google Scholar] [CrossRef]
- Ming, T.K.; Bernstein, I.L. Mapping conditioned taste aversion associations using c-Fos reveals a dynamic role for insular cortex. Behav. Neurosci. 2005, 119, 388–398. [Google Scholar] [CrossRef]
- Vincis, R.; Fontanini, A. Central taste anatomy and physiology. Handb Clin Neurol. 2019, 164, 187–204. [Google Scholar] [CrossRef]
- Inberg, S.; Jacob, E.; Elkobi, A.; Edry, E.; Rappaport, A.; Simpson, T.I.; Armstrong, J.D.; Shomron, N.; Pasmanik-Chor, M.; Rosenblum, K. Fluid consumption and taste novelty determines transcription temporal dynamics in the gustatory cortex. Mol. Brain 2016, 9, 13. [Google Scholar] [CrossRef] [PubMed]
- Miranda, M.I.; Ferreira, G.; Ramírez-Lugo, L.; Bermúdez-Rattoni, F. Role of cholinergic system on the construction of memories: Taste memory encoding. Neurobiol Learn Mem. 2003, 80, 211–222. [Google Scholar] [CrossRef]
- Stern, E.; Chinnakkaruppan, A.; David, O.; Sonenberg, N.; Rosenblum, K. Blocking the eIF2α kinase (PKR) enhances positive and negative forms of cortex-dependent taste memory. J. Neurosci. 2013, 33, 2517–2525. [Google Scholar] [CrossRef] [PubMed]
- Gal-Ben-Ari., S.; Rosenblum, K. Molecular mechanisms underlying memory consolidation of taste information in the cortex. Front. Behav. Neurosci. 2012, 5, 87. [Google Scholar] [CrossRef]
- Elkobi, A.; Ehrlich, I.; Belelovsky, K.; Barki-Harrington, L.; Rosenblum, K. ERK-dependent PSD-95 induction in the gustatory cortex is necessary for taste learning, but not retrieval. Nat. Neurosci. 2008, 11, 1149–1151. [Google Scholar] [CrossRef] [PubMed]
- Rosenblum, K.; Meiri, N.; Dudai, Y. Taste memory: The role of protein synthesis in gustatory cortex. Behav. Neural Biol. 1993, 59, 49–56. [Google Scholar] [CrossRef]
- Merhav, M.; Rosenblum, K. Facilitation of taste memory acquisition by experiencing previous novel taste is protein-synthesis dependent. Learn. Mem. 2008, 15, 501–507. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Levitan, D.; Gal-Ben-Ari, S.; Heise, C.; Rosenberg, T.; Elkobi, A.; Inberg, S.; Sala, C.; Rosenblum, K. The differential role of cortical protein synthesis in taste memory formation and persistence. NPJ Sci. Learn. 2016, 1, 16001. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Okuno, H. Regulation and function of immediate-early genes in the brain: Beyond neuronal activity markers. Neurosci. Res. 2011, 69, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Plath, N.; Ohana, O.; Dammermann, B.; Errington, M.L.; Schmitz, D.; Gross, C.; Mao, X.; Engelsberg, A.; Mahlke, C.; Welzl, H.; et al. Arc/Arg3.1 is essential for the consolidation of synaptic plasticity and memories. Neuron 2006, 52, 437–444. [Google Scholar] [CrossRef]
- Inberg, S.; Elkobi, A.; Edri, E.; Rosenblum, K. Taste familiarity is inversely correlated with Arc/Arg3.1 hemispheric lateralization. J. Neurosci. 2013, 33, 11734–11743. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Olvera, A.; Rodríguez-Durán, L.F.; Escobar, M.L. Conditioned taste aversion prevents the long-lasting BDNF-induced enhancement of synaptic transmission in the insular cortex: A metaplastic effect. Neurobiol. Learn. Mem. 2016, 130, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Doron, G.; Rosenblum, K. C-Fos expression is elevated in GABAergic interneurons of the gustatory cortex following novel taste learning. Neurobiol. Learn. Mem. 2010, 94, 21–29. [Google Scholar] [CrossRef]
- Saddoris, M.P.; Holland, P.C.; Gallagher, M. Associatively learned representations of taste outcomes activate taste-encoding neural ensembles in gustatory cortex. J. Neurosci. 2009, 29, 15386–15396. [Google Scholar] [CrossRef]
- Berman, D.E. Modulation of taste-induced Elk-1 activation by identified neurotransmitter systems in the insular cortex of the behaving rat. Neurobiol. Learn. Mem. 2003, 79, 122–126. [Google Scholar] [CrossRef]
- Bermúdez-Rattoni, F. Molecular mechanisms of taste-recognition memory. Nat. Rev. Neurosci. 2004, 5, 209–217. [Google Scholar] [CrossRef]
- Bermudez-Rattoni, F. The forgotten insular cortex: Its role on recognition memory formation. Neurobiol. Learn. Mem. 2014, 109, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Barki-Harrington, L.; Elkobi, A.; Tzabary, T.; Rosenblum, K. Tyrosine phosphorylation of the 2B subunit of the NMDA receptor is necessary for taste memory formation. J. Neurosci. 2009, 29, 9219–9226. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Serrano, L.M.; Ramírez-León, B.; Rodríguez-Durán, L.F.; Escobar, M.L. Acute infusion of brain-derived neurotrophic factor in the insular cortex promotes conditioned taste aversion extinction. Neurobiol. Learn. Mem. 2014, 116, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, T.; Elkobi, A.; Rosenblum, K. mAChR-dependent decrease in proteasome activity in the gustatory cortex is necessary for novel taste learning. Neurobiol. Learn. Mem. 2016, 135, 115–124. [Google Scholar] [CrossRef]
- Parkes, S.L.; De la Cruz, V.; Bermúdez-Rattoni, F.; Coutureau, E.; Ferreira, G. Differential role of insular cortex muscarinic and NMDA receptors in one-trial appetitive taste learning. Neurobiol. Learn. Mem. 2014, 116, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-García, G.; Miranda, M.I. Opposing roles of cholinergic and GABAergic activity in the insular cortex and nucleus basalis magnocellularis during novel recognition and familiar taste memory retrieval. J. Neurosci. 2016, 36, 1879–1889. [Google Scholar] [CrossRef] [PubMed]
- Berman, D.E.; Hazvi, S.; Neduva, V.; Dudai, Y. The role of identified neurotransmitter systems in the response of insular cortex to unfamiliar taste: Activation of ERK1-2 and formation of a memory trace. J. Neurosci. 2000, 20, 7017–7023. [Google Scholar] [CrossRef]
- Núñez-Jaramillo, L.; Jimenez, B.; Ramirez-Munguía, N.; Delint-Ramírez, I.; Luna-Illades, C.; Tapia, R.; Bermúdez-Rattoni, F. Taste novelty induces intracellular redistribution of NR2A and NR2B subunits of NMDA receptor in the insular cortex. Brain Res. 2008, 1215, 116–122. [Google Scholar] [CrossRef]
- David, O.; Barrera, I.; Chinnakkaruppan, A.; Kaphzan, H.; Nakazawa, T.; Yamamoto, T.; Rosenblum, K. Dopamine-induced tyrosine phosphorylation of NR2B (Tyr1472) is essential for ERK1/2 activation and processing of novel taste information. Front. Mol. Neurosci. 2014, 7, 66. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Moreno, A.; Rodríguez-Durán, L.F.; Escobar, M.L. Late protein synthesis-dependent phases in CTA long-term memory: BDNF requirement. Front. Behav. Neurosci. 2011, 5, 61. [Google Scholar] [CrossRef] [PubMed]
- Castillo, D.V.; Figueroa-Guzmán, Y.; Escobar, M.L. Brain-derived neurotrophic factor enhances conditioned taste aversion retention. Brain Res. 2006, 1067, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Bi, A.L.; Wang, Y.; Zhang, S.; Li, B.Q.; Sun, Z.P.; Bi, H.S.; Chen, Z.Y. Myosin II regulates actin rearrangement-related structural synaptic plasticity during conditioned taste aversion memory extinction. Brain Struct. Funct. 2015, 220, 813–825. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Durán, L.F.; Escobar, M.L. NMDA receptor activation and PKC but not PKA lead to the modification of the long-term potentiation in the insular cortex induced by conditioned taste aversion: Differential role of kinases in metaplasticity. Behav. Brain Res. 2014, 266, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Adaikkan, C.; Rosenblum, K. A molecular mechanism underlying gustatory memory trace for an association in the insular cortex. eLife 2015, 4, e07582. [Google Scholar] [CrossRef]
- Gómez-Chacón, B.; Gámiz, F.; Foster, T.C.; Gallo, M. Increased N-ethylmaleimide-sensitive factor expression in amygdala and perirhinal cortex during habituation of taste neophobia. Neural Plast. 2016, 2016, 2726745. [Google Scholar] [CrossRef][Green Version]
- Slouzkey, I.; Rosenblum, K.; Maroun, M. Memory of conditioned taste aversion is erased by inhibition of PI3K in the insular cortex. Neuropsychopharmacology 2013, 38, 1143–1153. [Google Scholar] [CrossRef]
- Miranda, M.I.; Rangel-Hernández, J.A.; Vera-Rivera, G.; García-Medina, N.E.; Soto-Alonso, G.; Rodríguez-García, G.; Núñez-Jaramillo, L. The role of dopamine D2 receptors in the nucleus accumbens during taste-aversive learning and memory extinction after long-term sugar consumption. Neuroscience 2017, 359, 142–150. [Google Scholar] [CrossRef]
- Rosenblum, K.; Berman, D.E.; Hazvi, S.; Lamprecht, R.; Dudai, Y. NMDA receptor and the tyrosine phosphorylation of its 2B subunit in taste learning in the rat insular cortex. J. Neurosci. 1997, 17, 5129–5135. [Google Scholar] [CrossRef]
- Rosenberg, T.; Elkobi, A.; Dieterich, D.C.; Rosenblum, K. NMDAR-dependent proteasome activity in the gustatory cortex is necessary for conditioned taste aversion. Neurobiol. Learn. Mem. 2016, 130, 7–16. [Google Scholar] [CrossRef]

| Receptors | Protein Expression | Molecular Signals |
|---|---|---|
| NMDAr (GIC; CTA acquisition and taste learning) [56,75,76,77] AMPAr (GIC; CTA acquisition) [56,75,76] GluR2 (amygdala and PEC; SP and taste memory) [78] mACh (GIC; neophobia, SP and CTA acquisition) [56,75,76,79,80,81] DA (GIC; novel taste memory) [76,82] GABA-A (GIC; taste recognition) [56,75,76,80,81] NR1 (PFC; SP in CTA acquisition) [44] NR2A-2B (GIC; CTA acquisition and taste processing) [83] TrkB (GIC; taste memory) [78] β-adrenergic (GIC; novel taste memory) [82] D1 (GIC; novel taste processing and memory) [84] | c-fos (GIC; SP and LT taste memory) [72] BDNF (GIC and amygdala; SP in taste memory and CTA) [71,78,85,86] Homer 1a (GIC; SP and LT taste memory) [73] Arc/Arg3.1 (GIC; CTA memory) [60,68,69,70] Transcription factor Elk-1 (GIC; SP and LT taste memory) [74] | cAMP (GIC; SP and taste memory) [76] Adenylyl cyclase (GIC; SP and taste memory) [76] Actin (IFC; structural plasticity and CTA memory) [87] PKA (GIC and amygdala CTA memory) [76,88] CaMKIIα (GIC; SP, novel taste memory, CTA acquisition) [89] PKC (GIC and amygdala; LT plasticity and CTA memory) [76,88] NSF (amygdala and PEC; SP and taste memory) [90] ERK1/2 (GIC; novel taste processing, SP and taste memory) [82,84] Myosin II (IFC; structural plasticity and CTA memory) [87] PSD-95 (GIC; SP and taste learning) [64,77] PI3K (GIC; SP and CTA memory) [91] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molero-Chamizo, A.; Rivera-Urbina, G.N. Taste Processing: Insights from Animal Models. Molecules 2020, 25, 3112. https://doi.org/10.3390/molecules25143112
Molero-Chamizo A, Rivera-Urbina GN. Taste Processing: Insights from Animal Models. Molecules. 2020; 25(14):3112. https://doi.org/10.3390/molecules25143112
Chicago/Turabian StyleMolero-Chamizo, Andrés, and Guadalupe Nathzidy Rivera-Urbina. 2020. "Taste Processing: Insights from Animal Models" Molecules 25, no. 14: 3112. https://doi.org/10.3390/molecules25143112
APA StyleMolero-Chamizo, A., & Rivera-Urbina, G. N. (2020). Taste Processing: Insights from Animal Models. Molecules, 25(14), 3112. https://doi.org/10.3390/molecules25143112







