Propofol Improved Glucose Tolerance Associated with Increased FGF-21 and GLP-1 Production in Male Sprague-Dawley Rats
Abstract
:1. Introduction
2. Results
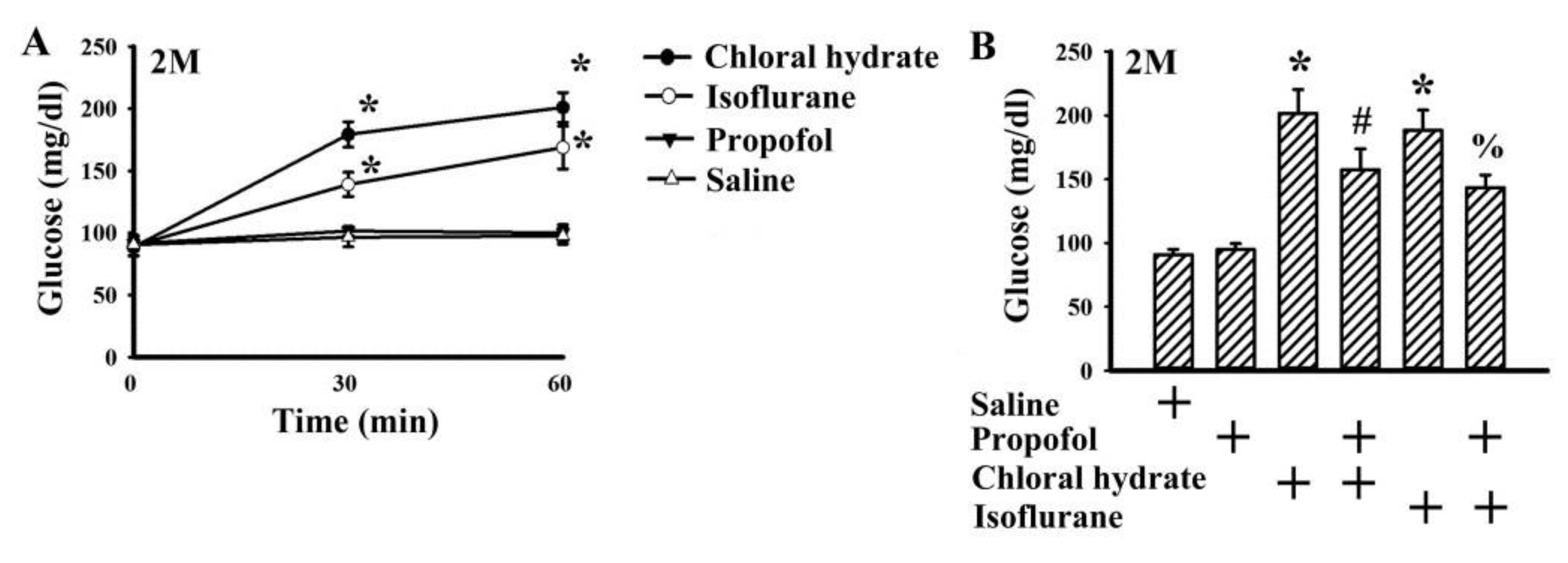
2.1. Propofol Had Minimal Effect on Basal Glucose Level
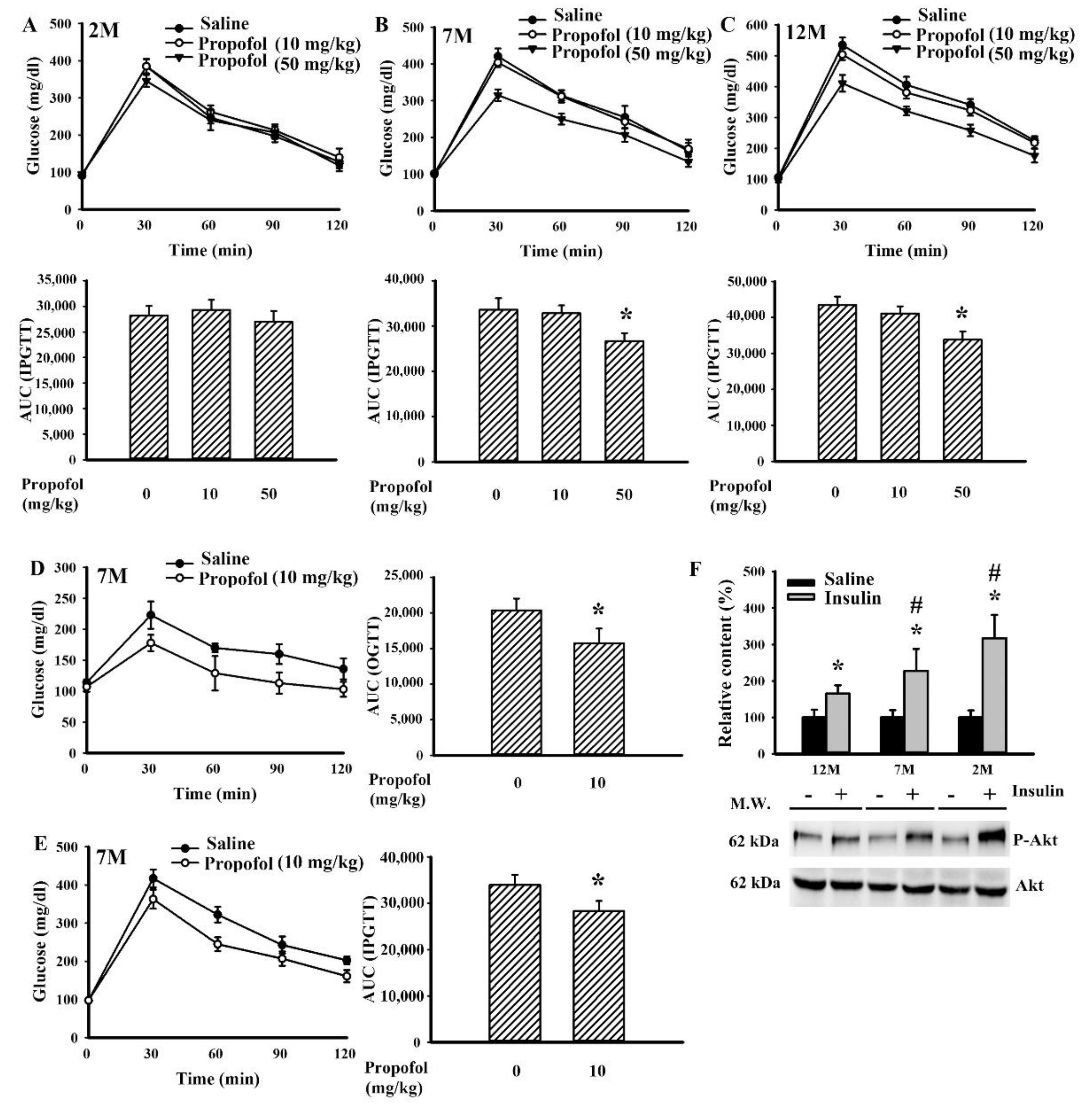
2.2. Propofol Improved Impaired Glucose Tolerance
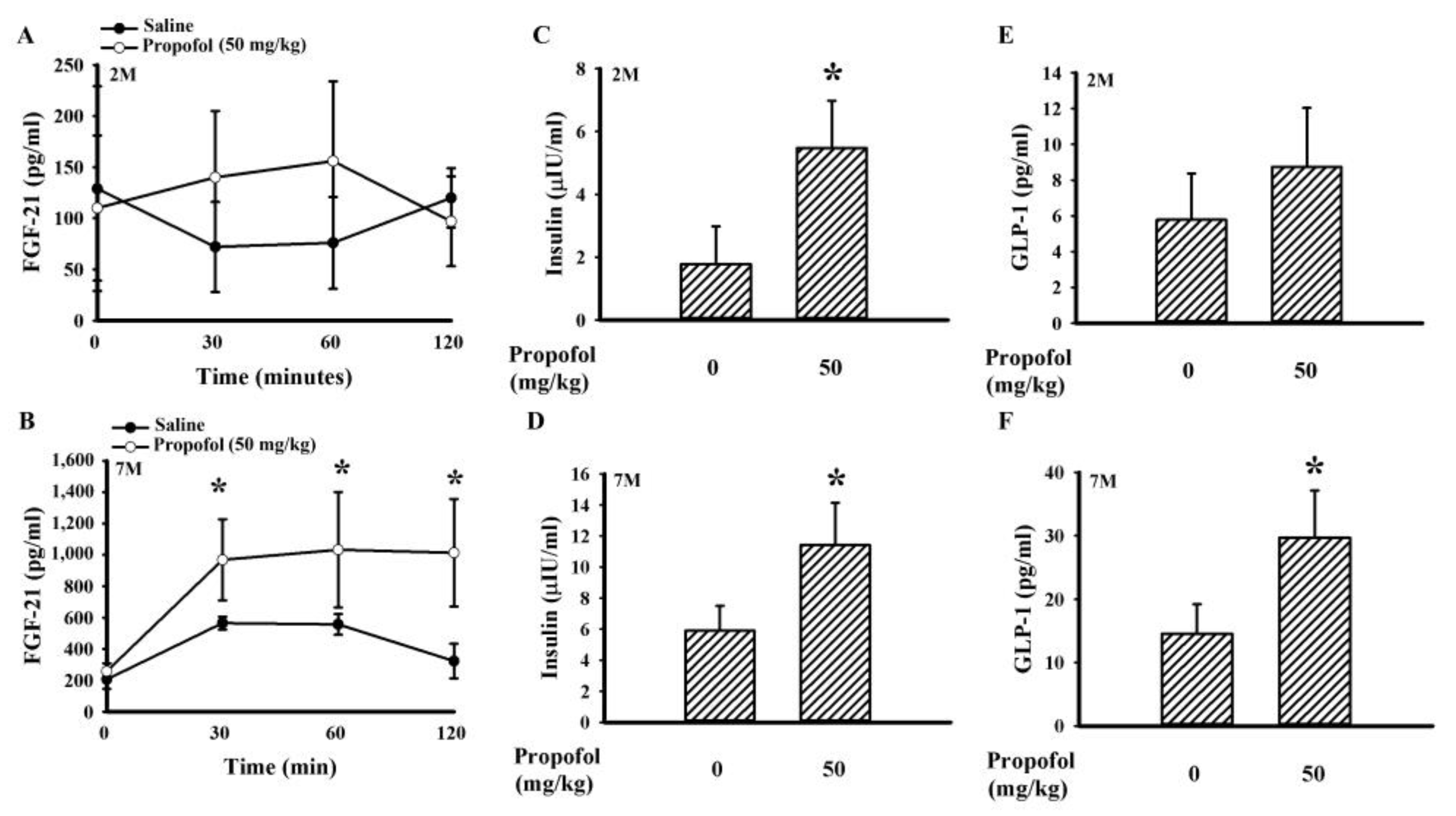
2.3. Propofol Increased the Circulating Level of Fibroblast Growth Factor-21 (FGF-21), Glucagon-like Peptide-1 (GLP-1), and Insulin
2.4. Propofol Upregulated FGF-21 Signaling in the Liver
3. Discussion
4. Materials and Methods
4.1. Animal Study
4.2. Blood Sample Reparation
4.3. Tissue Collection and Western Blot Analyses
4.4. RNA Preparation and Quantitative Real-Time Reverse Transcriptase Polymerase Chain Reaction (RT-PCR)
4.5. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Vanhorebeek, I.; Gunst, J.; Van den Berghe, G. Critical care management of stress-induced hyperglycemia. Curr. Diab. Rep. 2018, 18. [Google Scholar] [CrossRef] [PubMed]
- Swanson, C.M.; Potter, D.J.; Kongable, G.L.; Cook, C.B. Update on inpatient glycemic control in hospitals in the United States. Endocr. Pract. 2011, 17, 853–861. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Xia, T.; Xu, F.; Wu, H.; Ma, Z.; Zhao, X.; Gu, X. Isoflurane aggravates peripheral and central insulin resistance in high-fat diet/streptozocin-induced type 2 diabetic mice. Brain Res. 2020, 1727, 146511. [Google Scholar] [CrossRef] [PubMed]
- Høyer, K.F.; Nielsen, T.S.; Risis, S.; Treebak, J.T.; Jessen, N. Sevoflurane impairs insulin secretion and tissue-specific glucose uptake in vivo. Basic Clin. Pharmacol. Toxicol. 2018, 123, 732–738. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Windeløv, J.A.; Pedersen, J.; Holst, J.J. Use of anesthesia dramatically alters the oral glucose tolerance and insulin secretion in C57Bl/6 mice. Physiol. Rep. 2016, 4, e12824. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Behdad, S.; Mortazavizadeh, A.; Ayatollahi, V.; Khadiv, Z.; Khalilzadeh, S. The effects of propofol and isoflurane on blood glucose during abdominal hysterectomy in diabetic patients. Diabetes Metab. J. 2014, 38, 311–316. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.; Zhang, L.; Yin, G.; Liu, Y.; Chen, L. Stress response to propofol versus isoflurane anesthesia in patients undergoing gastric surgery. J. Coll. Physicians Surg. Pak. 2019, 29, 201–204. [Google Scholar] [CrossRef]
- Li, X.; Kitamura, T.; Kawamura, G.; Mori, Y.; Sato, K.; Araki, Y.; Sato, R.; Yamada, Y. Comparison of mechanisms underlying changes in glucose utilization in fasted rats anesthetized with propofol or sevoflurane: Hyperinsulinemia is exaggerated by propofol with concomitant insulin resistance induced by an acute lipid load. Biosci. Trends. 2014, 8, 155–162. [Google Scholar] [CrossRef] [Green Version]
- Kitamura, T.; Sato, K.; Kawamura, G.; Yamada, Y. The involvement of adenosine triphosphate-sensitive potassium channels in the different effects of sevoflurane and propofol on glucose metabolism in fed rats. Anesth. Analg. 2012, 114, 110–116. [Google Scholar] [CrossRef]
- Sato, K.; Kitamura, T.; Kawamura, G.; Mori, Y.; Sato, R.; Araki, Y.; Yamada, Y. Glucose use in fasted rats under sevoflurane anesthesia and propofol anesthesia. Anesth. Analg. 2013, 117, 627–633. [Google Scholar] [CrossRef]
- Yasuda, Y.; Fukushima, Y.; Kaneki, M.; Martyn, J.A. Anesthesia with propofol induces insulin resistance systemically in skeletal and cardiac muscles and liver of rats. Biochem. Biophys. Res. Commun. 2013, 431, 81–85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chaki, T.; Hirata, N.; Yoshikawa, Y.; Tachibana, S.; Tokinaga, Y.; Yamakage, M.J. Lipid emulsion, but not propofol, induces skeletal muscle damage and lipid peroxidation. J. Anesth. 2019, 33, 628–635. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.M.; Shin, J.S.; Yoon, I.H.; Min, B.H.; Jeong, W.Y.; Lee, G.E.; Kim, M.S.; Kim, J.E.; Jang, J.Y.; Park, C.G. The effect of propofol on intravenous glucose tolerance test in rhesus monkey. J. Med. Primatol. 2014, 43, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, T.; Ogawa, M.; Kawamura, G.; Sato, K.; Yamada, Y. The effects of sevoflurane and propofol on glucose metabolism under aerobic conditions in fed rats. Anesth. Analg. 2009, 109, 1479–1485. [Google Scholar] [CrossRef]
- Zhou, L.; Wang, L.; Yang, B.; Zeng, J.; Zhang, Q.; Lei, H.; Xu, S. Protective effect of pretreatment with propofol against tumor necrosis factor-α-induced hepatic insulin resistance. Exp. Ther. Med. 2015, 10, 289–294. [Google Scholar] [CrossRef] [Green Version]
- Rowlands, J.; Heng, J.; Newsholme, P.; Carlessi, R. Pleiotropic effects of GLP-1 and analogs on cell signaling, metabolism, and function. Front. Endocrinol. 2018, 9, 672. [Google Scholar] [CrossRef] [Green Version]
- Nonogaki, K.; Hazama, M.; Satoh, N. Liraglutide suppresses obesity and hyperglycemia associated with increases in hepatic fibroblast growth factor 21 production in KKAy mice. Biomed. Res. Int. 2014, 2014. [Google Scholar] [CrossRef] [Green Version]
- Yang, M.; Zhang, L.; Wang, C.; Liu, H.; Boden, G.; Yang, G.; Li, L. Liraglutide increases FGF-21 activity and insulin sensitivity in high fat diet and adiponectin knockdown induced insulin resistance. PLoS ONE 2012, 7. [Google Scholar] [CrossRef]
- Staiger, H.; Keuper, M.; Berti, L.; Hrabe de Angelis, M.; Häring, H.U. Fibroblast growth factor 21-metabolic role in mice and men. Endocr. Rev. 2017, 38, 468–488. [Google Scholar] [CrossRef]
- Liu, J.; Yang, K.; Yang, J.; Xiao, W.; Le, Y.; Yu, F.; Gu, L.; Lang, S.; Tian, Q.; Jin, T.; et al. Liver-derived fibroblast growth factor 21 mediates effects of glucagon-like peptide-1 in attenuating hepatic glucose output. EbioMedicine. 2019, 41, 73–84. [Google Scholar] [CrossRef] [Green Version]
- Gluvic, Z.; Zaric, B.; Resanovic, I.; Obradovic, M.M.; Mitrovic, A.A.; Radak, D.; Isenovic, E.R. Link between metabolic syndrome and insulin resistance. Curr. Vasc. Pharmacol. 2017, 15, 30–39. [Google Scholar] [CrossRef]
- Tresguerres, J.A.; Cuesta, S.; Kireev, R.A.; Garcia, C.; Acuña-Castroviejo, D.; Vara, E. Beneficial effect of melatonin treatment on age-related insulin resistance and on the development of type 2 diabetes. Horm. Mol. Biol. Clin. Investig. 2013, 16, 47–54. [Google Scholar] [CrossRef]
- Pham, H.; Marathe, C.S.; Phillips, L.K.; Trahair, L.G.; Hatzinikolas, S.; Huynh, L.; Wu, T.; Nauck, M.A.; Rayner, C.K.; Horowitz, M.; et al. Longitudinal changes in fasting and glucose-stimulated GLP-1 and GIP in healthy older subjects. J. Clin. Endocrinol. Metab. 2019, 104, 6201–6206. [Google Scholar] [CrossRef] [PubMed]
- Xie, T.; Leung, P.S. Fibroblast growth factor 21: A regulator of metabolic disease and health span. Am. J Physiol. Endocrinol. Metab. 2017, 313, E292–E302. [Google Scholar] [CrossRef] [PubMed]
- Cui, D.R.; Wang, L.; Jiang, W.; Qi, A.H.; Zhou, Q.H.; Zhang, X.L. Propofol prevents cerebral ischemia-triggered autophagy activation and cell death in the rat hippocampus through the NF-κB/p53 signaling pathway. Neuroscience. 2013, 246, 117–132. [Google Scholar] [CrossRef]
- Li, J.; Yu, W.; Li, X.T.; Qi, S.H.; Li, B. The effects of propofol on mitochondrial dysfunction following focal cerebral ischemia-reperfusion in rats. Neuropharmacology 2014, 77, 358–368. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, A.; Tsuji, M.; Inagaki, M.; Tamura, Y.; Kato, M.; Niiya, A.; Usui, Y.; Oguchi, K. Neuroprotective effects of propofol on ER stress-mediated apoptosis in neuroblastoma SH-SY5Y cells. Eur. J. Pharmacol. 2014, 725, 47–54. [Google Scholar] [CrossRef]
- Zhou, R.; Yang, Z.; Tang, X.; Tan, Y.; Wu, X.; Liu, F. Propofol protects against focal cerebral ischemia via inhibition of microglia-mediated proinflammatory cytokines in a rat model of experimental stroke. PLoS ONE 2013, 12. [Google Scholar] [CrossRef] [Green Version]
- Tanaka, K.; Kawano, T.; Tsutsumi, Y.M.; Kinoshita, M.; Kakuta, N.; Hirose, K.; Kimura, M.; Oshita, S. Differential effects of propofol and isoflurane on glucose utilization and insulin secretion. Life. Sci. 2011, 88, 96–103. [Google Scholar] [CrossRef]
- Maruyama, R.; Shimizu, M.; Hashidume, T.; Inoue, J.; Itoh, N.; Sato, R. FGF21 alleviates hepatic endoplasmic reticulum stress under physiological conditions. J. Nutr. Sci. Vitaminol. 2018, 64, 200–208. [Google Scholar] [CrossRef] [Green Version]
- Liu, F.; Zhu, S.; Ni, L.; Huang, L.; Wang, K.; Zhou, Y. Dexmedetomidine alleviates insulin resistance in hepatocytes by reducing endoplasmic reticulum stress. Endocrine 2020, 67, 87–94. [Google Scholar] [CrossRef] [Green Version]
- Nakajima, S.; Hira, T.; Hara, H. Postprandial glucagon-like peptide-1 secretion is increased during the progression of glucose intolerance and obesity in high-fat/high-sucrose diet-fed rats. Br. J. Nutr. 2015, 113, 1477–1488. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krasner, N.M.; Ido, Y.; Ruderman, N.B.; Cacicedo, J.M. Glucagon-like peptide-1 (GLP-1) analog liraglutide inhibits endothelial cell inflammation through a calcium and AMPK dependent mechanism. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.H.; Wen, X.; Cho, H.; Koo, S.H. CREB/CRTC2 controls GLP-1-dependent regulation of glucose homeostasis. FASEB J. 2018, 32, 1566–1578. [Google Scholar] [CrossRef] [Green Version]
- Mauna, J.C.; Miyamae, T.; Pulli, B.; Thiels, E. Protein phosphatases 1 and 2A are both required for long-term depression and associated dephosphorylation of cAMP response element binding protein in hippocampal area CA1 in vivo. Hippocampus 2011, 21, 1093–1104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, Q.; Zhao, Y.; Duan, W.; Liu, Y.; Chen, X.; Zhu, M. Propofol inhibits high glucose-induced PP2A expression. in human umbilical vein endothelial cells. Vascul. Pharmacol. 2017, 91, 18–25. [Google Scholar] [CrossRef]
- Nair, A.B.; Jacob, S. A simple practice guide for dose conversion between animals and human. J. Basic Clin. Pharm. 2016, 7, 27–31. [Google Scholar] [CrossRef] [Green Version]
Sample Availability: Samples of the compounds are not available from the authors. |

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, C.-C.; Hung, C.-J.; Wang, Y.-Y.; Lin, S.-Y.; Chen, W.-Y.; Kuan, Y.-H.; Liao, S.-L.; Yang, C.-P.; Chen, C.-J. Propofol Improved Glucose Tolerance Associated with Increased FGF-21 and GLP-1 Production in Male Sprague-Dawley Rats. Molecules 2020, 25, 3229. https://doi.org/10.3390/molecules25143229
Wu C-C, Hung C-J, Wang Y-Y, Lin S-Y, Chen W-Y, Kuan Y-H, Liao S-L, Yang C-P, Chen C-J. Propofol Improved Glucose Tolerance Associated with Increased FGF-21 and GLP-1 Production in Male Sprague-Dawley Rats. Molecules. 2020; 25(14):3229. https://doi.org/10.3390/molecules25143229
Chicago/Turabian StyleWu, Chih-Cheng, Chih-Jen Hung, Ya-Yu Wang, Shih-Yi Lin, Wen-Ying Chen, Yu-Hsiang Kuan, Su-Lan Liao, Ching-Ping Yang, and Chun-Jung Chen. 2020. "Propofol Improved Glucose Tolerance Associated with Increased FGF-21 and GLP-1 Production in Male Sprague-Dawley Rats" Molecules 25, no. 14: 3229. https://doi.org/10.3390/molecules25143229




