Efficient Removal of Volatile Organic Compounds by FAU-Type Zeolite Coatings
Abstract
:1. Introduction
2. Results
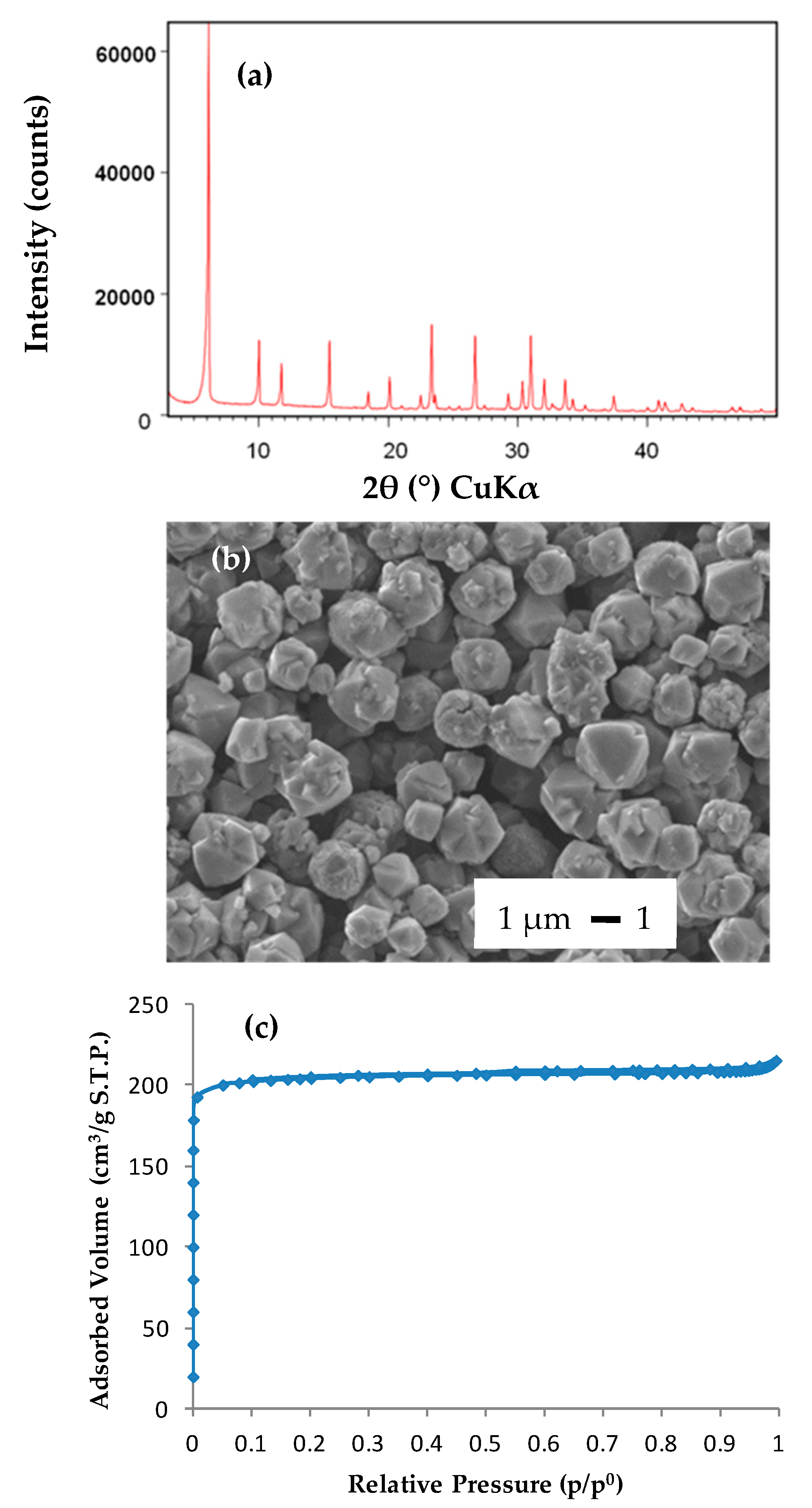
2.1. Characterization of the Reagents
2.2. Influence of the Amount of Binder on Adhesion Properties
2.3. Influence of the Nature of the Binder on the Mechanical and Adsorption Properties of the Final Zeolite Coating
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Characterization Techniques
4.3. Preparation of Zeolite Coatings
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rochow, E.G.; Gilliam, W.F. Polymeric Methyl Silicon Oxides1. J. Am. Chem. Soc. 1941, 63, 798–800. [Google Scholar] [CrossRef]
- Zhang, L.; Shi, Z.; Hu, W.; Zhang, X.; Zhu, H.; Zhao, G.; Wang, Z. Curing mechanism, heat resistance, and anticorrosion properties of a furan/methyl phenyl silicone coating. Polym. Adv. Technol. 2018, 29, 1913–1921. [Google Scholar] [CrossRef]
- Jovanovic, J.D.; Govedarica, M.N.; Dvornic, P.R.; Popovic, I. The thermogravimetric analysis of some polysiloxanes. Polym. Degrad. Stab. 1998, 61, 87–93. [Google Scholar] [CrossRef]
- Giaveri, S.; Gronchi, P.; Barzoni, A. IPN Polysiloxane-Epoxy Resin for High Temperature Coatings: Structure Effects on Layer Performance after 450 °C Treatment. Coatings 2017, 7, 213. [Google Scholar] [CrossRef] [Green Version]
- Robeyns, C.; Picard, L.; Ganachaud, F. Synthesis, characterization and modification of silicone resins: An “Augmented Review”. Prog. Org. Coat. 2018, 125, 287–315. [Google Scholar] [CrossRef]
- Xiong, G.; Kang, P.; Zhang, J.; Li, B.; Yang, J.; Chen, G.; Zhou, Z.; Li, Q. Improved adhesion, heat resistance, anticorrosion properties of epoxy resins/POSS/methyl phenyl silicone coatings. Prog. Org. Coat. 2019, 135, 454–464. [Google Scholar] [CrossRef]
- Xu, Y.; Long, J.; Zhang, R.; Du, Y.; Guan, S.; Wang, Y.; Huang, L.; Wei, H.; Liu, L.; Huang, Y. Greatly improving thermal stability of silicone resins by modification with POSS. Polym. Degrad. Stab. 2020, 174, 109082. [Google Scholar] [CrossRef]
- Zhou, W.; Yang, H.; Guo, X.; Lu, J. Thermal degradation behaviors of some branched and linear polysiloxanes. Polym. Degrad. Stab. 2006, 91, 1471–1475. [Google Scholar] [CrossRef]
- Kummer, H.; Füldner, G.; Henninger, S. Versatile siloxane based adsorbent coatings for fast water adsorption processes in thermally driven chillers and heat pumps. Appl. Therm. Eng. 2015, 85, 1–8. [Google Scholar] [CrossRef]
- Rioland, G.; Daou, T.J.; Faye, D.; Patarin, J. A new generation of MFI-type zeolite pellets with very high mechanical performance for space decontamination. Microporous Mesoporous Mater. 2016, 221, 167–174. [Google Scholar] [CrossRef]
- Perry, J.L. Trace Chemical Contaminant Generation Rates for Spacecraft Contamination Control System Design. NASA Tech. Memo. 108497 1995, 4–12. [Google Scholar]
- Kirsch-Rodeschini, H. Contrôle de la Contamination Moléculaire par Adsorption sur des Solides Poreux. Ph.D. Thesis, University of Haute-Alsace, Mulhouse, France, 2006. [Google Scholar]
- Faye, D.; Jakob, A.; Soulard, M.; Berlioz, P. Zeolite adsorbers for molecular contamination control in spacecraft. Opt. Eng. Appl. 2010, 7794, 77940. [Google Scholar] [CrossRef]
- Barengoltz, J.B.; Moore, S.; Soules, D.; Voecks, G. The Wide Field/Planetary Camera 2 (WFPC-2) molecular adsorber. Jet Propuls. Lab 1994, 94, 1–48. [Google Scholar]
- Kabalan, I.; Lebeau, B.; Fadlallah, M.-B.; Toufaily, J.; Hamieh, T.; Bellat, J.P.; Daou, T.J. Hierarchical Faujasite-Type Zeolite for Molecular Decontamination. J. Nanosci. Nanotechnol. 2016, 16, 9318–9322. [Google Scholar] [CrossRef]
- Dhainaut, J.; Daou, T.J.; Chappaz, A.; Bats, N.; Harbuzaru, B.; Lapisardi, G.; Chaumeil, H.; Defoin, A.; Rouleau, L.; Patarin, J. Synthesis of FAU and EMT-type zeolites using structure-directing agents specifically designed by molecular modelling. Microporous Mesoporous Mater. 2013, 174, 117–125. [Google Scholar] [CrossRef]
- Nikolakis, V.; Xomeritakis, G.; Abibi, A.; Dickson, M.; Tsapatsis, M.; Vlachos, D.G. Growth of a faujasite-type zeolite membrane and its application in the separation of saturated/unsaturated hydrocarbon mixtures. J. Membr. Sci. 2001, 184, 209–219. [Google Scholar] [CrossRef]
- Sato, K.; Sugimoto, K.; Sekine, Y.; Takada, M.; Matsukata, M.; Nakane, T. Application of FAU-type zeolite membranes to vapor/gas separation under high pressure and high temperature up to 5 MPa and 180 °C. Microporous Mesoporous Mater. 2007, 101, 312–318. [Google Scholar] [CrossRef]
- Corma, A. Application of Zeolites in Fluid Catalytic Cracking and Related Processes. Prep. Catal. V - Sci. Bases Prep. Heterog. Catal. Proc. Fifth Int. Symp. 1989, 49, 49–67. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, Y.; Xie, Z. Thoughts on the Future Development of Zeolitic Catalysts from an Industrial Point of View. Chin. J. Catal. 2012, 33, 22–38. [Google Scholar] [CrossRef]
- Giraudet, M.; Bezverkhyy, I.; Weber, G.; Dirand, C.; Macaud, M.; Bellat, J.-P. D2/H2 adsorption selectivity on FAU zeolites at 77 4K: Influence of Si/Al ratio and cationic compensation. Microporous Mesoporous Mater. 2018, 270, 211–219. [Google Scholar] [CrossRef]
- Rioland, G.; Bullot, L.; Daou, T.J.; Simon-Masseron, A.; Chaplais, G.; Faye, D.; Fiani, E.; Patarin, J. Elaboration of FAU-type zeolite beads with good mechanical performances for molecular decontamination. RSC Adv. 2016, 6, 2470–2478. [Google Scholar] [CrossRef]
- Rioland, G.; Nouali, H.; Daou, T.J.; Faye, D.; Patarin, J. Adsorption of volatile organic compounds in composite zeolites pellets for space decontamination. Adsorption 2017, 23, 395–403. [Google Scholar] [CrossRef]
- Lauridant, N.; Daou, T.J.; Arnold, G.; Soulard, M.; Nouali, H.; Patarin, J.; Faye, D. Key steps influencing the formation of ZSM-5 films on aluminum substrates. Microporous Mesoporous Mater. 2012, 152, 1–8. [Google Scholar] [CrossRef]
- Lauridant, N.; Daou, T.J.; Arnold, G.; Nouali, H.; Patarin, J.; Faye, D. Zeolite hybrid films for space decontamination. Microporous Mesoporous Mater. 2013, 172, 36–43. [Google Scholar] [CrossRef]
- Lauridant, N.; Daou, T.J.; Arnold, G.; Patarin, J.; Faye, D. MFI/∗BEA hybrid coating on aluminum alloys. Microporous Mesoporous Mater. 2013, 166, 79–85. [Google Scholar] [CrossRef]
- Li, X.; Wang, C.; Liu, S.; Xin, W.; Wang, Y.; Xie, S.; Xu, L. Influences of alkaline treatment on the structure and catalytic performances of ZSM-5/ZSM-11 zeolites with alumina as binder. J. Mol. Catal. A: Chem. 2011, 336, 34–41. [Google Scholar] [CrossRef]
- Dubey, A.; Mailapalli, D.R. Zeolite coated urea fertilizer using different binders: Fabrication, material properties and nitrogen release studies. Environ. Technol. Innov. 2019, 16, 100452. [Google Scholar] [CrossRef]
- Cañizares, P.; Durán, A.; Dorado, F.; Carmona, M. The role of sodium montmorillonite on bounded zeolite-type catalysts. Appl. Clay Sci. 2000, 16, 273–287. [Google Scholar] [CrossRef]
- Workneh, S.; Shukla, A. Synthesis of sodalite octahydrate zeolite-clay composite membrane and its use in separation of SDS. J. Membr. Sci. 2008, 309, 189–195. [Google Scholar] [CrossRef]
- Minelli, M.; Papa, E.; Medri, V.; Miccio, F.; Benito, P.; Doghieri, F.; Landi, E. Characterization of novel geopolymer – Zeolite composites as solid adsorbents for CO2 capture. Chem. Eng. J. 2018, 341, 505–515. [Google Scholar] [CrossRef]
- Sudagar, A.J.; Andrejkovičová, S.; Patinha, C.; Velosa, A.; McAdam, A.; Da Silva, E.F.; Rocha, F. A novel study on the influence of cork waste residue on metakaolin-zeolite based geopolymers. Appl. Clay Sci. 2018, 152, 196–210. [Google Scholar] [CrossRef]
- Straka, S.A.; Peters, W.; Hasegawa, M.; Novo-Gradac, K.; Wong, A. Development of molecular adsorber coatings. Opt. Eng. Appl. 2010, 7794, 77940. [Google Scholar] [CrossRef]
- Abraham, N.S.; Hasegawa, M.M. Development and testing of molecular adsorber coatings. Opt. Eng. Appl. 2012, 8492, 849203. [Google Scholar] [CrossRef]
- Bendix, P.B.; Henninger, S.; Henning, H.-M. Temperature and Mechanical Stabilities and Changes in Porosity of Silicone Binder Based Zeolite Coatings. Ind. Eng. Chem. Res. 2016, 55, 4942–4947. [Google Scholar] [CrossRef]
- Bendix, P.; Füldner, G.; Möllers, M.; Kummer, H.; Schnabel, L.; Henninger, S.; Henning, H.-M. Optimization of power density and metal-to-adsorbent weight ratio in coated adsorbers for adsorptive heat transformation applications. Appl. Therm. Eng. 2017, 124, 83–90. [Google Scholar] [CrossRef]
- Dorado, F.; Romero, R.; Cañizares, P. Hydroisomerization of n-butane over Pd/HZSM-5 and Pd/Hβ with and without binder. Appl. Catal. A: Gen. 2002, 236, 235–243. [Google Scholar] [CrossRef]
- Sánchez, P.; Dorado, F.; Fúnez, A.; Jimenez, V.; Ramos, M.J.; Valverde, J.L.; Marcos, M.J.R. Effect of the binder content on the catalytic performance of beta-based catalysts. J. Mol. Catal. A: Chem. 2007, 273, 109–113. [Google Scholar] [CrossRef]
- Joshi, U.; Joshi, P.; Tamhankar, S.; Joshi, V.; Shiralkar, V. Adsorption Behavior of N2, Water, C6 Hydrocarbons, and Bulkier Benzene Derivative (TMB) on Na–X Zeolite and Its K+-, Rb+-, and Cs+-Exchanged Analogues. J. Colloid Interface Sci. 2001, 235, 135–143. [Google Scholar] [CrossRef]
- Cosseron, A.F.; Daou, T.J.; Tzanis, L.; Nouali, H.; Deroche, I.; Coasne, B.; Tchamber, V. Adsorption of organic volatile compounds in pure silica CHA, *BEA, MFI and STT-type zeolites. Microporous Mesoporous Mater. 2013, 173, 147–154. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef] [Green Version]
- Chen, D.; Hu, X.; Shi, L.; Cui, Q.; Wang, H.; Yao, H. Synthesis and characterization of zeolite X from lithium slag. Appl. Clay Sci. 2012, 59, 148–151. [Google Scholar] [CrossRef]
Sample Availability: Not available. |




| Binder | Temperature of the First Weight Loss (°C) |
|---|---|
| SILRES® HK 46 | 250 |
| SILRES® MP 50 E | 300 |
| DOWSIL® 2405 | 100 |
| DOWSIL® RSN-0805 | 300 |
| DOWSIL® RSN-0806 | 350 |
| VINNAPAS® EP 8010 | 250 |
| Zeolite Coating | 13X Zeolite Content(wt.%) 1 | Binder SILRES® HK 46 Content(wt.%) 1 | Adhesion Note 2 |
|---|---|---|---|
| 1 | 50 | 50 | 0 |
| 2 | 60 | 40 | 0 |
| 3 | 70 | 30 | 0 |
| 4 | 80 | 20 | 0 |
| 5 | 90 | 10 | 3 |
| Zeolite Coating Code | 13X Zeolite Content(wt.%) 1 | Binder SILRES® HK 46 Content (wt.%) 1 | Adhesion Note 2 | Coating Thickness (µm) 3 | Curing Conditions |
|---|---|---|---|---|---|
| FAU-HK46 | 80 | 20 | 0 | 46 ± 2 | 250 °C/30 min |
| FAU-MP50E | 80 | 20 | 0 | 28 ± 11 | 250 °C/30 min |
| FAU-2405 | 70 | 25 | 1 | 46 ± 7 | RT/1 h (with catalyst) 4 |
| FAU-RSN0805 | 80 | 20 | 0 | 33 ± 5 | 250 °C/30 min |
| FAU-RSN0806 | 80 | 20 | 0 | 46 ± 7 | 250 °C/30 min |
| FAU-EP8010 | 75 | 25 | 1 | 42 ± 6 | RT/1 h |
| Sample 1 | 13X Zeolite Content (wt.%) 2 | Micropore Volume of Zeolite Coating in Powder Form (cm3·g−1) | Accessible Micropore Volume of Zeolite (cm3·g−1) 3 | Porosity Accessibility (%) 3 |
|---|---|---|---|---|
| FAU powder | 100 | 0.30 | 0.30 | 100 |
| FAU-HK46 | 80 | 0.23 | 0.29 | 95 |
| FAU-MP50E | 80 | 0.24 | 0.30 | 100 |
| FAU-2405 | 70 | 0.21 | 0.30 | 100 |
| FAU-RSN0805 | 80 | 0.17 | 0.21 | 70 |
| FAU-RSN0806 | 80 | 0.19 | 0.24 | 80 |
| FAU-EP8010 | 75 | 0.23 | 0.30 | 100 |
| Sample 1 | Adsorbed Amount of n-Hexane at Saturation Stage (mg/ganhydrous zeolite) |
|---|---|
| FAU powder | 180 ± 2 |
| FAU-HK46 | 175 ± 2 |
| FAU-MP50E | 164 ± 3 |
| FAU-2405 | 160 ± 2 |
| FAU-RSN0805 | 131 ± 3 |
| FAU-RSN0806 | 141 ± 4 |
| FAU-EP8010 | 152 ± 5 |
| Commercial Binder 1 | Supplier | Solid Content (wt.%) 2 | Solvent 3 | Curing Conditions | Zeolite Coating Code 4 |
|---|---|---|---|---|---|
| SILRES® HK 46 | IMCD | 50 | m-xylene | 250 °C/30 min | FAU-HK46 |
| SILRES® MP 50 E | IMCD | 50 | water | 250 °C/30 min | FAU-MP50E |
| DOWSIL® 2405 | Univar | 72 | m-xylene | RT/1 h (with catalyst) 5 | FAU-2405 |
| DOWSIL® RSN-0805 | Univar | 50 | m-xylene | 250 °C/30 min | FAU-RSN0805 |
| DOWSIL® RSN-0806 | Univar | 50 | m-xylene | 250 °C/30 min | FAU-RSN0806 |
| VINNAPAS® EP 8010 | IMCD | 60 | water | RT/1 h | FAU-EP8010 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diboune, M.; Nouali, H.; Soulard, M.; Patarin, J.; Rioland, G.; Faye, D.; Daou, T.J. Efficient Removal of Volatile Organic Compounds by FAU-Type Zeolite Coatings. Molecules 2020, 25, 3336. https://doi.org/10.3390/molecules25153336
Diboune M, Nouali H, Soulard M, Patarin J, Rioland G, Faye D, Daou TJ. Efficient Removal of Volatile Organic Compounds by FAU-Type Zeolite Coatings. Molecules. 2020; 25(15):3336. https://doi.org/10.3390/molecules25153336
Chicago/Turabian StyleDiboune, Mathieu, Habiba Nouali, Michel Soulard, Joël Patarin, Guillaume Rioland, Delphine Faye, and T. Jean Daou. 2020. "Efficient Removal of Volatile Organic Compounds by FAU-Type Zeolite Coatings" Molecules 25, no. 15: 3336. https://doi.org/10.3390/molecules25153336
APA StyleDiboune, M., Nouali, H., Soulard, M., Patarin, J., Rioland, G., Faye, D., & Daou, T. J. (2020). Efficient Removal of Volatile Organic Compounds by FAU-Type Zeolite Coatings. Molecules, 25(15), 3336. https://doi.org/10.3390/molecules25153336