Bonding Analysis of Compounds with Unusual Coordination of Carbon: Proposed Symmetric Systems with Six-Coordinate Carbon
Abstract
:1. Introduction
2. Computational Methods and Software
2.1. Software
2.2. AIM and Bader Terminology
2.3. ELF
2.4. NCI
3. Density Analysis for Known Examples of Unusual Coordination of Carbon
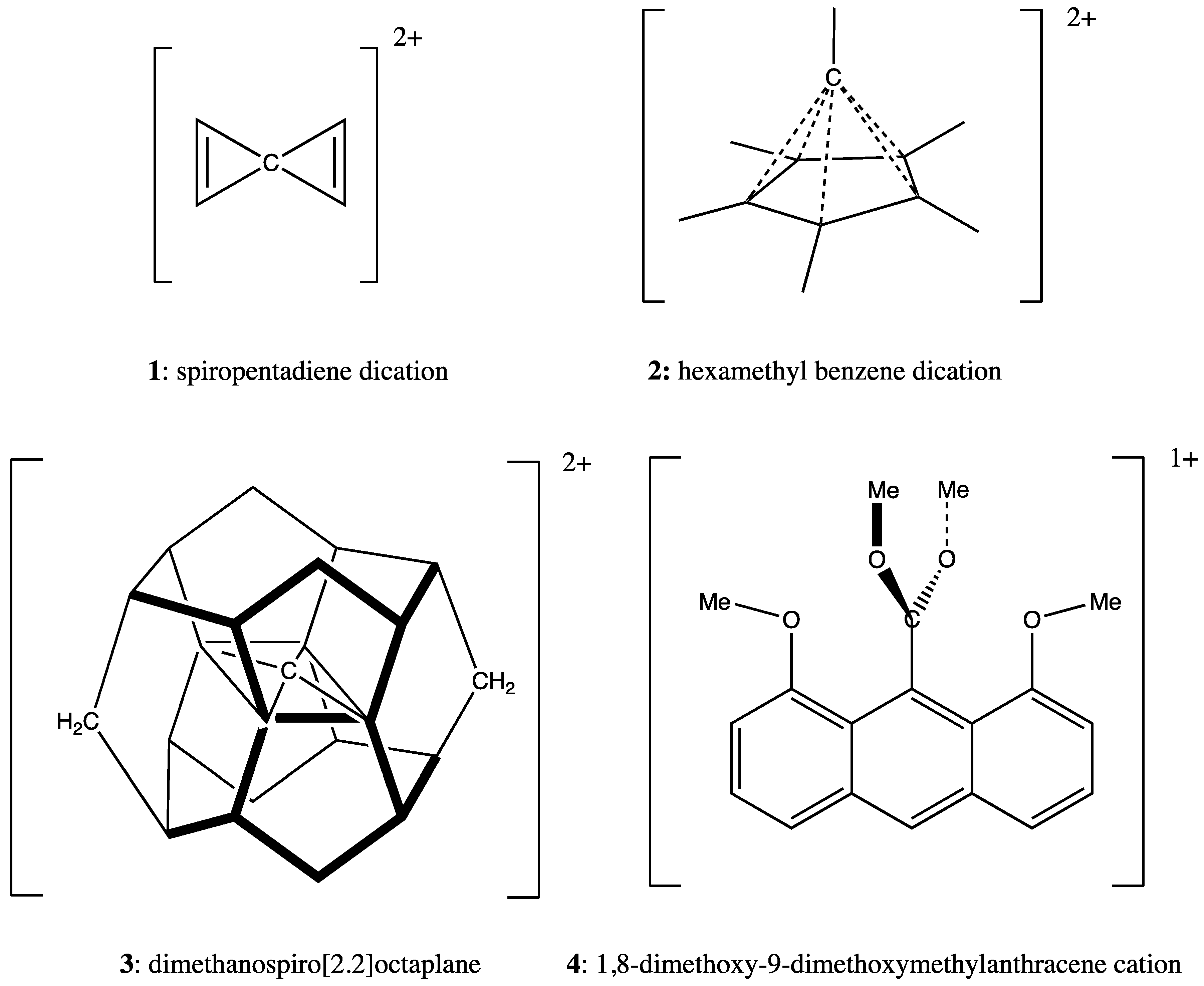
3.1. Example 1: Spiropentadiene Dication
3.1.1. AIM Analysis
3.1.2. ELF Analysis
3.1.3. NCI Analysis
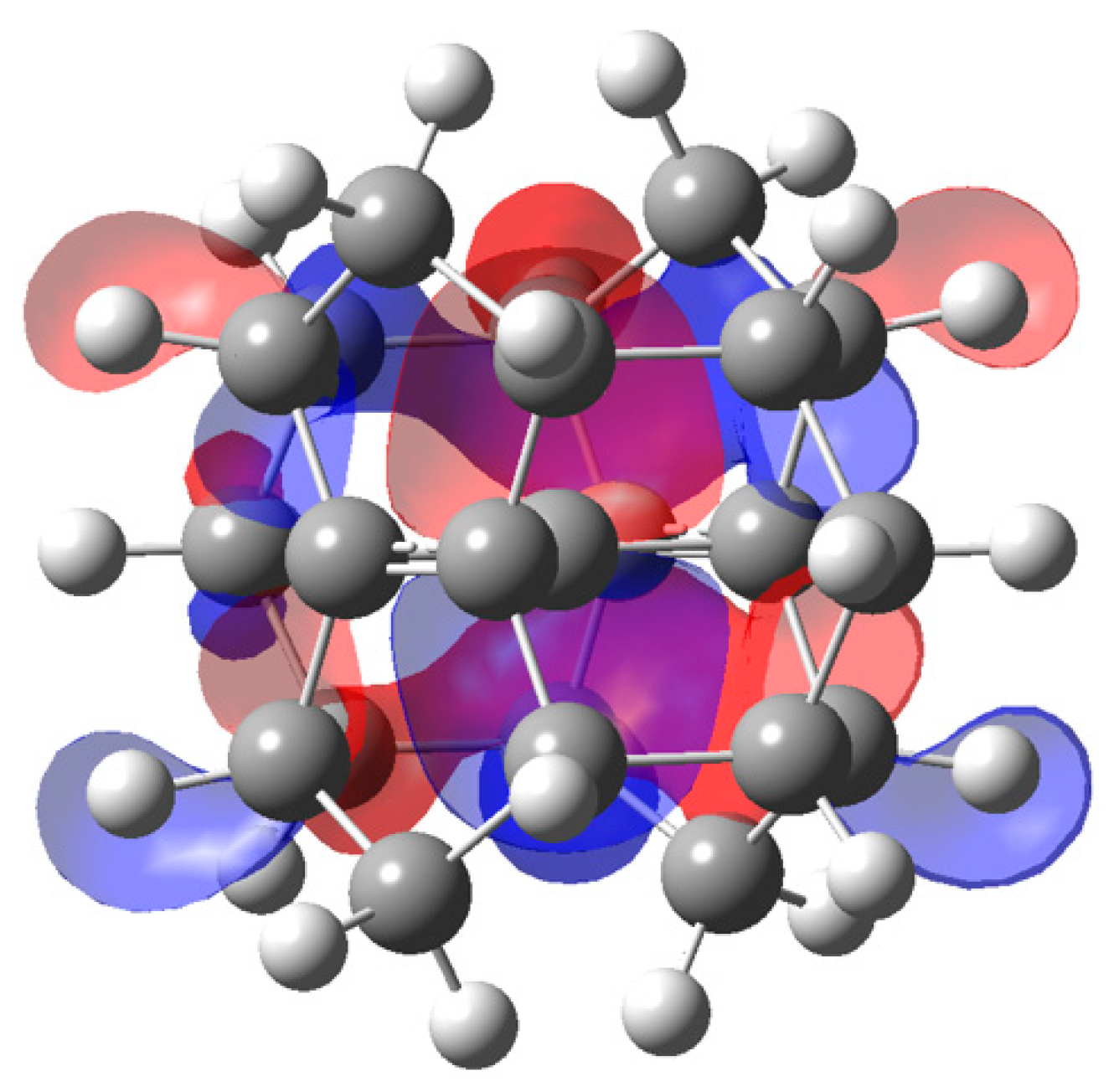
3.2. Example 2: Hexamethylbenzene Dication
3.2.1. AIM Analysis
3.2.2. ELF Analysis
3.2.3. NCI Analysis
3.3. Example 3: Dimethanospiro[2.2]octaplane
3.3.1. AIM Analysis
3.3.2. ELF Analysis
3.3.3. NCI Analysis
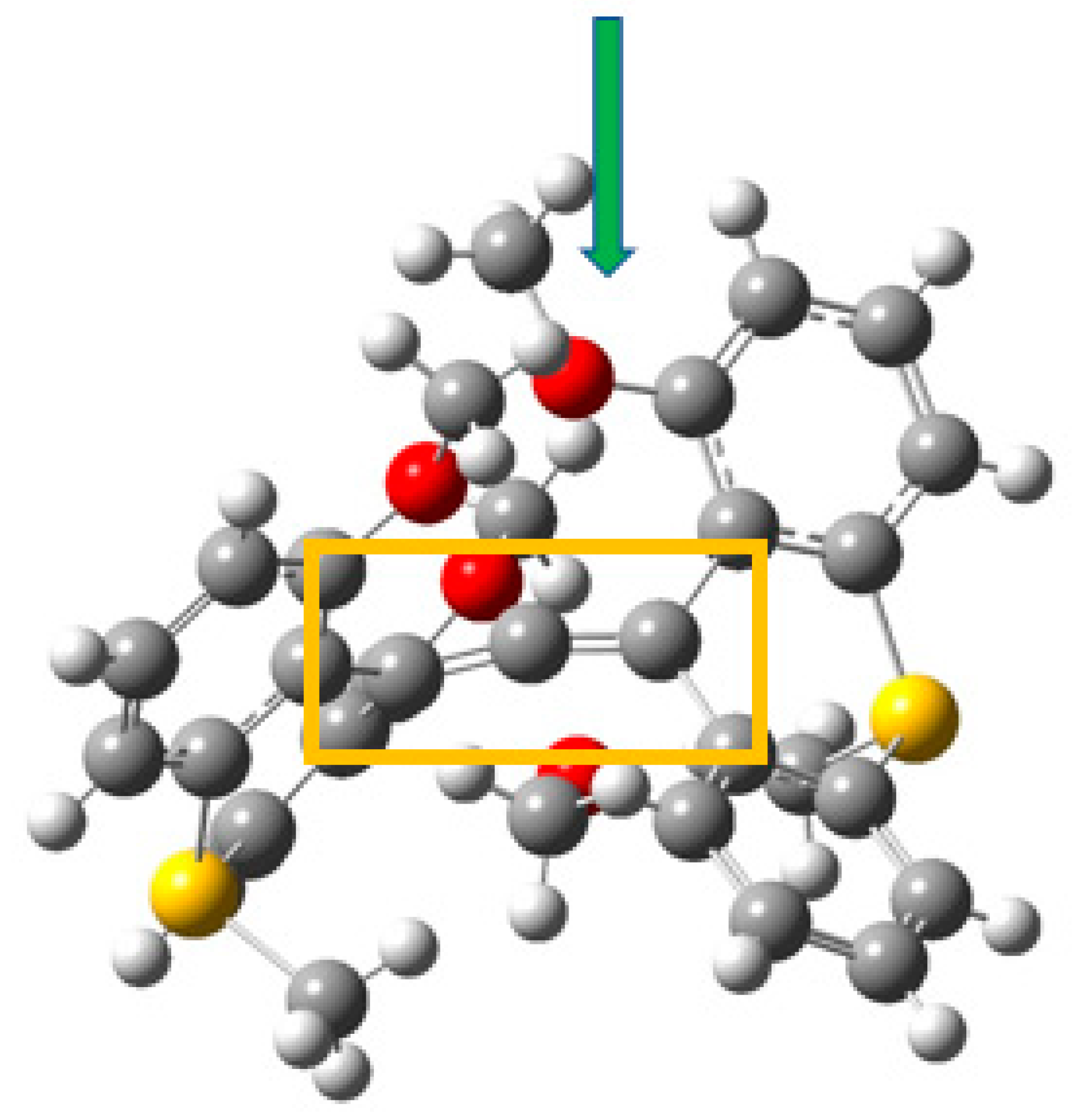
3.4. Example 4: 1,8-Dimethoxy-9-Dimethoxymethylanthracene Cation
3.4.1. AIM Analysis
3.4.2. ELF Analysis
3.4.3. NCI Analysis
4. Proposed Symmetric Variants on a Known Example of Approximately Octahedral Coordination of Carbon
4.1. A Neutral Symmetric System with Six-Coordinate Carbon
4.1.1. AIM Analysis
4.1.2. ELF Analysis
4.1.3. NCI Analysis
4.2. A Neutral System with Attractive Interactions in the Coordination Sphere
4.2.1. AIM Analysis
4.2.2. ELF Analysis
4.2.3. NCI Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hoff, V.J.H. Sur les formules de structure dans l’espace. Arch. Néerl. Sci. Exact. Nat. 1874, 9, 445–454. [Google Scholar]
- Le Bel, J.A. Sur les relations qui existent entre les formules atomiques des corps organiques et le pouvoir rotatoire de leurs dissolutions. Bull. Soc. Chim. Fr. 1874, 22, 337–347. [Google Scholar]
- Leicester, H.M. Le Bel, Joseph.Achille. In Dictionary of Scientific Biography; Gillispie, C.C., Ed.; Charles Scribner’s Sons: New York, NY, USA, 1973; Volume 8, pp. 109–110. [Google Scholar]
- Snelders, H.A.M. van’t Hoff, Jacobus Hendricus. In Dictionary of Scientific Biography; Gillispie, C.C., Ed.; Charles Scribner’s Sons: New York, NY, USA, 1976; Volume 13, pp. 575–581. [Google Scholar]
- Gillespie, R.J.; Nyholm, R.S. Inorganic Stereochemistry. Quart. Rev. Chem. Soc. 1957, 11, 339–380. [Google Scholar] [CrossRef]
- Gillespie, R.J. Fifty years of the VSEPR model. Coord. Chem. Rev. 2008, 252, 1315–1327. [Google Scholar] [CrossRef]
- Hoffmann, R.; Alder, R.W.; Wilcox, C.F., Jr. Planar Tetracoordinate Carbon. J. Am. Chem. Soc. 1970, 92, 4992–4993. [Google Scholar] [CrossRef]
- Hoffmann, R. The Theoretical Design of Novel Stabilized Systems. Pure. Appl. Chem. 1970, 28, 181–194. [Google Scholar] [CrossRef]
- Wong, M.W.; Radom, L. Methane Dication: Planar but Not Square. J. Am. Chem. Soc. 1989, 111, 1155–1156. [Google Scholar] [CrossRef]
- Lammertsma, K.; Schleyer, P.v.R. Structures and energies of isomeric carbodications (C5H42+ and C6H42+). J. Phys. Chem. 1988, 92, 881–886. [Google Scholar] [CrossRef]
- Hogeveen, H.; Kwant, P.W. Direct observation of a remarkably stable dication of unusual structure: (CCH3)62+. Tetrahedron Lett. 1973, 14, 1665–1670. [Google Scholar] [CrossRef]
- Hogeveen, H.; Kwant, P.W.; Postma, J.; van Duynen, P.T. Electronic spectra of pyramidal dications, (CCH3)62+ and (CH)62+. Tetrahedron Lett. 1974, 15, 4351–4354. [Google Scholar] [CrossRef]
- Hogeveen, H.; Kwant, P.W. Chemistry and spectroscopy in strongly acidic solutions XL. (CCH3)6 2+ an unusual dication. J. Am. Chem. Soc. 1974, 96, 2208–2214. [Google Scholar] [CrossRef]
- Radom, L.; Rasmussen, D.R. The planar carbon story. Pure Appl. Chem. 1998, 70, 1977–1984. [Google Scholar] [CrossRef]
- Lyons, J.E.; Rasmussen, D.R.; McGrath, M.P.; Nobes, R.H.; Radom, L. Octaplane: A Saturated Hydrocarbon with a Remarkably Low Ionization Energy Leading to a Cation with a Planar Tetracoordinate Carbon Atom. Angew. Chem. Int. Ed. Engl. 1994, 33, 1667–1668. [Google Scholar] [CrossRef]
- Wang, Z.X.; Von Ragué Schleyer, P. The Theoretical Design of Neutral Planar Tetracoordinate Carbon Molecules with C(C)4 Substructures. J. Am. Chem. Soc. 2002, 124, 11979–11982. [Google Scholar] [CrossRef] [PubMed]
- Von Ragué Schleyer, P.; Boldyrev, A.I. A new, general strategy for achieving planar tetracoordinate geometries for carbon and other second row periodic elements. J. Chem. Soc. Chem. Comm. 1991, 1536–1538. [Google Scholar] [CrossRef]
- Li, X.; Wang, L.S.; Boldyrev, A.I.; Simons, J. Tetracoordinated planar carbon in the Al4C-anion. A combined photoelectron spectroscopy and ab initio study. J. Am. Chem. Soc. 1999, 121, 6033–6038. [Google Scholar] [CrossRef]
- Boldyrev, A.I.; Simons, J. Tetracoordinated Planar Carbon in Pentaatomic Molecules. J. Am. Chem. Soc. 1998, 120, 7967–7972. [Google Scholar] [CrossRef]
- Keese, R. Carbon Flatland: Planar Tetracoordinate Carbon and Fenestranes. Chem. Rev. 2006, 106, 4787–4808. [Google Scholar] [CrossRef]
- Wu, X.-F.; Cheng, Y.-X.; Guo, J.-C. CLi2AlE (E = P, As, Sb, Bi): Planar Tetracoordinate Carbon Clusters with 16 and 14 Valence Electrons. ACS Omega 2019, 4, 21311–21318. [Google Scholar] [CrossRef] [Green Version]
- Exner, K.; Schleyer, P.v.R. Planar Hexacoordinate Carbon: A Viable Possibility. Science 2000, 290, 1937–1940. [Google Scholar] [CrossRef]
- Bader, R.F.W. Atoms in Molecules: A Quantum Theory, 1st ed.; Oxford University Press: Oxford, UK, 1994. [Google Scholar]
- Becke, A.D.; Edgecombe, K.E. A simple measure of electron localization in atomic and molecular systems. J. Chem. Phys. 1990, 92, 5397–5403. [Google Scholar] [CrossRef]
- Johnson, E.R.; Keinan, S.; Mori-Sánchez, P.; Contreras-García, J.; Cohen, A.J.; Yang, W. Revealing noncovalent interactions. J. Am. Chem. Soc. 2010, 132, 6498–6506. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 09, Revision A.02; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 16, Revision C.01; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Chai, J.-D.; Head-Gordon, M. Long-range corrected hybrid density functionals with damped dispersion corrections. Phys. Chem. Chem. Phys. 2008, 10, 6615–6620. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dunning, T.H. Gaussian basis sets for use in correlated molecular calculations. I. The atoms boron through neon and hydrogen. J. Chem. Phys. 1989, 90, 1007–1023. [Google Scholar] [CrossRef]
- Todd, A.; Keith, T.K. AIMAll, Version 17.11.14; Gristmill Software: Overland Park, KS, USA, 2017. [Google Scholar]
- Lu, T.; Chen, F. Multiwfn: A Multifunctional Wavefunction Analyzer. J. Comp. Chem. 2012, 33, 580–592. Available online: http://sobereva.com/multiwfn/ (accessed on 21 August 2020). [CrossRef]
- Contreras-García, J.; Johnson, E.R.; Keinan, S.; Chaudret, R.; Piquemal, J.P.; Beretan, D.; Yang, W. NCIPLOT: A program for plotting noncovalent interaction regions. J. Chem. Theory Comp. 2011, 7, 625–632. [Google Scholar]
- Kumar, P.S.V.; Raghavendra, V.; Subramanian, V.J. Bader’s theory of atoms in molecules (AIM) and its applications to chemical bonding. J. Chem. Sci. 2016, 128, 1527–1536. [Google Scholar] [CrossRef]
- Grabowski, S.J. What is the Covalency of Hydrogen Bonding? Chem. Rev. 2011, 111, 2597–2625. [Google Scholar] [CrossRef]
- Grimme, S.; Mueck-Lichtenfeld, C.; Erker, G.; Kehr, G.; Wang, H.; Beckers, H.; Willner, H. When do interacting atoms form a chemical bond? Spectroscopic measurements and theoretical analyses of dideuteriophenanthrene. Angew. Chem. Int. Ed. 2009, 48, 2592–2595. [Google Scholar] [CrossRef]
- Bader, R.F.W. Bond Paths are Not Chemical Bonds. J. Phys. Chem. A 2009, 113, 10391–10396. [Google Scholar] [CrossRef] [Green Version]
- Cerpa, E.; Andreas Krapp, A.; Flores-Moreno, R. Influence of Endohedral Confinement on the Electronic Interaction between He atoms: A He2@C20H20 Case Study. Chem. Eur. J. 2009, 15, 1985–1990. [Google Scholar] [CrossRef]
- Clark, T.; Murray, J.S.; Politzer, P. A perspective on quantum mechanics and chemical concepts in describing noncovalent interactions. Phys. Chem. Chem. Phys. 2018, 20, 30076–30082. [Google Scholar] [CrossRef]
- Narth, C.; Maroun, Z.; Boto, C.R.; Bonnet, M.-L.; Piquemal, J.-P.; Contreras-Garcia, J. A Complete NCI Perspective from new bonds to reactivity. In Applications of Topological Methods in Molecular Chemistry; Alikhani, E., Chauvin, R., Lepetit, C., Silvi, B., Eds.; Springer: Berlin, Germany, 2016; Volume 22, pp. 491–527. [Google Scholar]
- Esteves, P.M.; Ferreira, N.B.P.; Corrêa, R.J. Neutral Structures with a Planar Tetracoordinated Carbon Based on Spiropentadiene Analogues. J. Am. Chem. Soc. 2005, 127, 8680–8685. [Google Scholar] [CrossRef] [PubMed]
- Firme, C.L.; Barreiro, N.B.P.; Esteves, P.M.; Corrêa, R.J. Understanding the Planar Tetracoordinate Carbon Atom: Spiropentadiene Dication. J. Phys. Chem. A 2008, 112, 686–692. [Google Scholar] [CrossRef] [PubMed]
- Firme, C.L.; Antunes, O.A.C.; Esteves, P.M.; Corrêa, R.J. Derivatives of Spiropentadiene Dication: New Species with Planar Tetracoordinate Carbon (ptC) atom. J. Phys. Chem. A 2009, 113, 3171–3176. [Google Scholar] [CrossRef] [PubMed]
- Malischewski, M.; Seppelt, K. Crystal Structure Determination of the Pentagonal-Pyramidal Hexamethylbenzene Dication (CH3)6C62+. Angew. Chem. Int. Ed. 2017, 56, 368–370. [Google Scholar] [CrossRef] [PubMed]
- Schleyer, P.v.R.; Maerker, C.; Dransfeld, A.; Jiao, H.; van Eikema Hommes, N.J. Nucleus-Independent Chemical Shifts: A Simple and Efficient Aromaticity Probe. J. Am. Chem. Soc. 1996, 118, 6317–6318. [Google Scholar] [CrossRef]
- Klein, J.E.M.N.; Havenith, R.W.A.; Knizia, G. The Pentagonal-Pyramidal Hexamethylbenzene Dication: Many Shades of Coordination Chemistry at Carbon. Chem. Eur. J. 2018, 24, 12340–12345. [Google Scholar] [CrossRef] [Green Version]
- Akiba, K.; Yamashita, M.; Yamamoto, Y.; Nagase, S. Synthesis and Isolation of Stable Hypervalent Carbon Compound (10-C-5) Bearing a 1,8-Dimethoxyanthracene Ligand. J. Am. Chem. Soc. 1999, 121, 10644–10645. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Akiba, K. Synthesis of Hypervalent Pentavalent Carbon and Boron Compounds. J. Synth. Org. Chem. Jpn. 2004, 62, 1128–1137. [Google Scholar] [CrossRef]
- Yamashita, M.; Yamamoto, Y.; Akiba, K.; Hashizume, D.; Iwasaki, F.; Tagaki, N.; Nagase, S. Synthesis and Structures of Hypervalent Carbon and Boron Compounds Bearing an Anthracene Skeleton–Elucidation of Hypervalent Interaction Based on X-ray Analysis and DFT Calculation. J. Am. Chem. Soc. 2005, 127, 4354–4371. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, T.; Yamamoto, Y.; Kinoshita, D.; Akiba, K.; Zhang, Y.; Reed, C.A.; Hashizune, D.; Iwasaki, F. Synthesis and Structure of a Hexacoordinate Carbon Compound. J. Amer. Chem. Soc. 2008, 130, 6894–6895. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamaguchi, T.; Yamamoto, Y. Substituent effects on the structure of hexacoordinate carbon bearing two thioxanthene ligands. Pure Appl. Chem. 2013, 85, 671–682. [Google Scholar] [CrossRef]
- Nakai, H.; Okoshi, M.; Atsumi, T.; Kikuchi, T.; Akiba, K. Theoretical Design of Hexacoordinate Hypervalent Carbon Compounds by Analyzing Substituent Effects. Bull. Chem. Soc. Jpn. 2011, 84, 505–510. [Google Scholar] [CrossRef]
- Kikuchi, Y.; Ishii, M.; Akiba, K.; Nakai, H. Discovery of hexacoordinate hypervalent carbon compounds: Density functional study. Chem. Phys. Lett. 2008, 460, 37–41. [Google Scholar] [CrossRef]




















| Critical Point | Density ρ | Laplacian | G | V | |2G/V| |
|---|---|---|---|---|---|
| tpC-Cx: (3, −1) | 0.2323 0.230 | −0.1184 −0.113 | +0.1685 | −0.3506 | 0.961 |
| Cx-Cx (3, −1) | 0.3708 0.369 | −1.1167 −1.109 | +0.1872 | −0.6535 | 0.573 |
| Cx-H (3, −1) | 0.2716 0.275 | −1.0302 −1.050 | +0.0134 | −0.2844 | 0.094 |
| Basin Charges | |||||
| Charge (|e|) | H = +0.3794 | Cx = +0.1691 | tpC = −0.1932 | ||
| Critical Point | Density | Laplacian | G | V | |2G/V| |
|---|---|---|---|---|---|
| C ring-C apex: (3, −1) | 0.1551 | +0.0205 | +0.0913 | −0.1765 | 1.0346 |
| C ring-C ring (3, −1) | 0.2823 | −0.7869 | +0.0894 | −0.3766 | 0.4748 |
| C apex-Me (3, −1) | 0.2705 | −0.5607 | +0.0962 | −0.3254 | 0.5913 |
| C ring-Me (3, −1) | 0.2722 | −0.6231 | +0.0819 | −0.3194 | 0.5128 |
| Basin Charges | |||||
| Charge (|e|) | hcC −0.088 | Ring C −0.019 | Apical methyl C −0.016 | Ring methyl C −0.037 | |
| Critical Point | Density | Laplacian | G | V | |−2G/V| |
|---|---|---|---|---|---|
| rC–tcC: (3, −1) | 0.2286 | −0.2823 | +0.1435 | −0.3576 | 0.8026 |
| rC–rC (3, −1) | 0.2981 | −0.8509 | +0.1084 | −0.4295 | 0.5048 |
| sC–sC (3, −1) | 0.2493 | −0.6234 | +0.0729 | −0.3016 | 0.4834 |
| Critical Point | Density | Laplacian | G | V | |2G/V| |
|---|---|---|---|---|---|
| tbpC-OMe: (3, −1) | 0.3537 | −0.3326 | +0.4764 | −1.0359 | 0.9198 |
| tbpC…OMe (3, −1) | 0.0235 0.022 | +0.0898 +0.078 | +0.0209 | −0.0193 | 2.1658 |
| C-O (3, −1) | 0.2400 | −0.6625 | +0.2650 | −0.5445 | 0.9734 |
| C=C (3, −1) | 0.2618 | −1.0337 | +0.0708 | −0.3073 | 0.4608 |
| Critical Point | Density ρ | Laplacian ∇2ρ | Kinetic Energy Density G | Potential Energy Density V | |2G/V| | ||
|---|---|---|---|---|---|---|---|
| Allenic hcC=Ca | +0.3444 | −1.0187 | +0.1551 | −0.5648 | 0.5492 | ||
| Allenic hcC=Cb | +0.3385 | −0.9761 | +0.1515 | −0.5470 | 0.5539 | ||
| CH3Oa…hcC | +0.0182 | +0.0584 | +0.0128 | −0.0105 | 2.4381 | ||
| CH3Ob…hcC | +0.0192 | +0.0739 | +0.0165 | −0.0146 | 2.2603 | ||
| CH3Oc…hcC | +0.0140 | +0.0593 | +0.0155 | −0.0136 | 2.2794 | ||
| CH3Od…hcC | +0.0133 | +0.0602 | +0.0123 | −0.0099 | 2.4848 | ||
| Basin Charges | |||||||
| Atom | hcC | Allene Ca | Allene Cb | CH3Oa | CH3Ob | CH3Oc | CH3Od |
| AIM Q | −0.1943 | +0.0926 | +0.0898 | −1.0736 | −1.0884 | −1.0753 | −1.0720 |
| Critical Point | ρ | Laplacian | G | V | |2G/V| |
|---|---|---|---|---|---|
| C=O bond (3, −1) | 0.3976 | +0.1874 | +0.7174 | −1.3880 | 1.0337 |
| C=C bond (3, −1) | 0.3455 | −1.0337 | +0.1576 | −0.5736 | 0.5495 |
| H3C-O bond (3, −1) | 0.2523 | −0.2481 | +0.3985 | −0.6692 | 1.1910 |
| MeO … C (3, −1) | 0.0203 | +0.0755 | 0.0171 | −0.0154 | 2.2207 |
| Critical Point | ρ | Laplacian | G | V | |2G/V| |
|---|---|---|---|---|---|
| C=O bond (3, −1) | 0.4045 | −0.0281 | +0.6700 | −1.3470 | 0.9948 |
| C=C bond (3, −1) | 0.3448 | −1.0315 | +0.1624 | −0.5826 | 0.5575 |
| H3C-S bond (3, −1) | 0.1845 | −0.2965 | +0.0497 | −0.1736 | 0.5726 |
| MeS … C (3, −1) | 0.0201 | +0.0619 | +0.0141 | −0.0128 | 2.3125 |
| Basis cc-pVTZ | |||
|---|---|---|---|
| Charges (O) | C(allyl) | hcC | O atom |
| Species 6 | +0.2193 | −0.3279 | −1.1729 |
| Charges (S) | C(allyl) | hcC | S atom |
| Species 7 | +0.1552 | −0.3484 | +0.1130 |
| Basis aug-cc-pVTZ | |||
| Charges (O) | C(allyl) | hcC | O atom |
| Species 6 | +0.2213 | −0.3256 | −1.1815 |
| Charges (S) | C(allyl) | hcC | S atom |
| Species 7 | +0.2077 | −0.4591 | +0.1087 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trindle, C.; Altun, Z.; Bleda, E.A. Bonding Analysis of Compounds with Unusual Coordination of Carbon: Proposed Symmetric Systems with Six-Coordinate Carbon. Molecules 2020, 25, 3937. https://doi.org/10.3390/molecules25173937
Trindle C, Altun Z, Bleda EA. Bonding Analysis of Compounds with Unusual Coordination of Carbon: Proposed Symmetric Systems with Six-Coordinate Carbon. Molecules. 2020; 25(17):3937. https://doi.org/10.3390/molecules25173937
Chicago/Turabian StyleTrindle, Carl, Zikri Altun, and Erdi Ata Bleda. 2020. "Bonding Analysis of Compounds with Unusual Coordination of Carbon: Proposed Symmetric Systems with Six-Coordinate Carbon" Molecules 25, no. 17: 3937. https://doi.org/10.3390/molecules25173937







