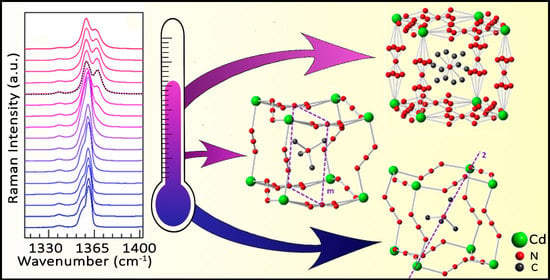
Raman Spectroscopy Studies on the Barocaloric Hybrid Perovskite [(CH3)4N][Cd(N3)3]
Abstract
:1. Introduction
2. Results and Discussion
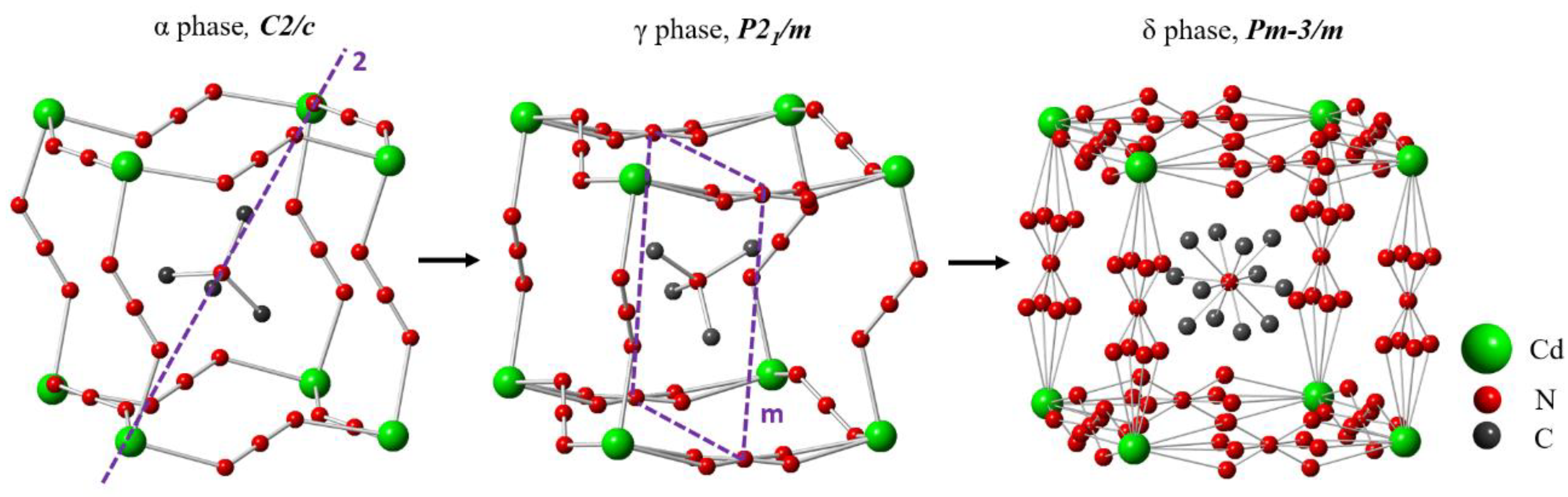
2.1. Basic Characterization and Deeper Insight into the Crystal Structure of TMACdN3
2.2. Thermal Characterization (DSC) and Barocaloric Parameters
2.3. Raman Studies
2.3.1. Room Temperature Raman Spectrum
2.3.2. Raman Spectra as a Function of Temperature
3. Materials and Methods
3.1. Synthesis
3.2. Powder X-Ray Diffraction
3.3. Hirshfeld Surface Analysis
3.4. Differential Scanning Calorimetry—DSC
3.5. Temperature-Dependent Raman Spectroscopy
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Li, W.; Wang, Z.; Deschler, F.; Gao, S.; Friend, R.H.; Cheetham, A.K. Chemically diverse and multifunctional hybrid organic–inorganic perovskites. Nat. Rev. Mater. 2017, 2. [Google Scholar] [CrossRef]
- García-fernández, A.; Juarez-perez, E.J.; Bermúdez-, J.M.; Llamas-saiz, A.L.; Artiaga, R.; López-beceiro, J.J. Hybrid Lead Halide DMAPbX3 (X = Cl- and Br-) Perovskites with Multiple Functional Properties. J. Mater. Chem. C 2019, 7, 10008–10018. [Google Scholar] [CrossRef]
- Kojima, A.; Teshima, K.; Shirai, Y.; Miyasaka, T. Organometal Halide Perovskites as Visible-Light Sensitizers for Photovoltaic Cells. J. Am. Chem. Soc. 2009, 131, 6050–6051. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Xiong, R.-G. Ferroelectric Metal–Organic Frameworks. Chem. Rev. 2011, 112, 1163–1195. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Aguirre, L.C.; Pato-Doldán, B.; Mira, J.; Castro-García, S.; Señarís-Rodríguez, M.A.; Sánchez-Andújar, M.; Singleton, J.; Zapf, V.S. Magnetic Ordering-Induced Multiferroic Behavior in [CH3NH3][Co(HCOO)3] Metal–Organic Framework. J. Am. Chem. Soc. 2016, 138, 1122–1125. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Aguirre, L.C.; Pato-Doldán, B.; Stroppa, A.; Yang, L.-M.; Frauenheim, T.; Mira, J.; Yañez-Vilar, S.; Artiaga, R.; Castro-García, S.; Sánchez-Andújar, M.; et al. Coexistence of Three Ferroic Orders in the Multiferroic Compound [(CH3 )4 N][Mn(N3 )3 ] with Perovskite-Like Structure. Chem. A Eur. J. 2016, 22, 7863–7870. [Google Scholar] [CrossRef] [PubMed]
- Bermúdez-García, J.M.; Sánchez-Andújar, M.; Señarís-Rodríguez, M.A. A New Playground for Organic–Inorganic Hybrids: Barocaloric Materials for Pressure-Induced Solid-State Cooling. J. Phys. Chem. Lett. 2017, 8, 4419–4423. [Google Scholar] [CrossRef]
- Bermúdez-García, J.M.; Sánchez-Andújar, M.; Castro-García, S.; López-Beceiro, J.; Artiaga, R.; Señarís-Rodríguez, M.A. Giant barocaloric effect in the ferroic organic-inorganic hybrid [TPrA][Mn(dca)3] perovskite under easily accessible pressures. Nat. Commun. 2017, 8, 15715. [Google Scholar] [CrossRef] [Green Version]
- Saparov, B.; Mitzi, D.B. Organic–Inorganic Perovskites: Structural Versatility for Functional Materials Design. Chem. Rev. 2016, 116, 4558–4596. [Google Scholar] [CrossRef]
- Mitzi, D.B. Templating and structural engineering in organic–inorganic perovskites. J. Chem. Soc. Dalton Trans. 2001, 1–12. [Google Scholar] [CrossRef]
- Kieslich, G.; Sun, S.; Cheetham, A.K. An extended Tolerance Factor approach for organic–inorganic perovskites. Chem. Sci. 2015, 6, 3430–3433. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, X.-H.; Huang, X.-C.; Zhang, S.-L.; Shao, D.; Wei, H.-Y.; Wang, X.-Y. Cation-Dependent Magnetic Ordering and Room-Temperature Bistability in Azido-Bridged Perovskite-Type Compounds. J. Am. Chem. Soc. 2013, 135, 16006–16009. [Google Scholar] [CrossRef]
- Kieslich, G.; Sun, S.; Cheetham, A.K. Solid-state principles applied to organic–inorganic perovskites: New tricks for an old dog. Chem. Sci. 2014, 5, 4712–4715. [Google Scholar] [CrossRef]
- Du, Z.-Y.; Zhao, Y.-P.; He, C.-T.; Wang, B.-Y.; Xue, W.; Zhou, H.-L.; Bai, J.; Huang, B.; Zhang, W.-X.; Chen, X.-M. Structural Transition in the Perovskite-like Bimetallic Azido Coordination Polymers: (NMe4)2[B′·B″(N3)6] (B′ = Cr3+, Fe3+; B″ = Na+, K+). Cryst. Growth Des. 2014, 14, 3903–3909. [Google Scholar] [CrossRef]
- Du, Z.-Y.; Zhao, Y.-P.; Zhang, W.-X.; Zhou, H.-L.; He, C.-T.; Xue, W.; Wang, B.-Y.; Chen, X.-M. Above-room-temperature ferroelastic phase transition in a perovskite-like compound [N(CH3)4][Cd(N3)3]. Chem. Commun. 2014, 50, 1989–1991. [Google Scholar] [CrossRef]
- Mautner, F.A.; Cortés, R.; Lezama, L.; Lu, J. [N(CH3)4][Mn(N3)3]: A Compound with a Distorted Perovskite Structure through Azido Ligands. Angew. Chem. Int. Ed. 1996, 35, 78–80. [Google Scholar] [CrossRef]
- Ribas, J.; Monfort, M.; Díaz, C.; Bastos, C.; Solans, X. New antiferromagnetic dinuclear complexes of nickel(II) with two azides as bridging ligands. Magneto-structural correlations. Inorg. Chem. 1993, 32, 3557–3561. [Google Scholar] [CrossRef]
- Salgado-Beceiro, J.; Nonato, A.; Silva, R.X.; García-Fernández, A.; Sánchez-Andújar, M.; Castro-Garcia, S.; Stern-Taulats, E.; Señarís-Rodríguez, M.A.; Moya, X.; Bermúdez-García, J.M. Near-room-temperature reversible giant barocaloric effects in [(CH3)4N]Mn[N3]3 hybrid perovskite. Mater. Adv. 2020. [Google Scholar] [CrossRef]
- Wei, W.; Li, W.; Butler, K.T.; Feng, G.; Howard, C.J.; Carpenter, M.P.; Lu, P.; Walsh, A.; Cheetham, A.K. An Unusual Phase Transition Driven by Vibrational Entropy Changes in a Hybrid Organic-Inorganic Perovskite. Angew. Chem. Int. Ed. 2018, 57, 8932–8936. [Google Scholar] [CrossRef]
- Moreira, R.L.; Matinaga, F.M.; Dias, A. Raman-spectroscopic evaluation of the long-range order in Ba(B[sub 1/3][sup ʹ]B[sub 2/3][sup ʺ])O[sub 3] ceramics. Appl. Phys. Lett. 2001, 78, 428–430. [Google Scholar] [CrossRef]
- Ma̧czka, M.; Da Silva, T.A.; Paraguassu, W.; Da Silva, K.P. Raman scattering studies of pressure-induced phase transitions in perovskite formates [(CH3)2NH2][Mg(HCOO)3] and [(CH3)2NH2][Cd(HCOO)3]. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2016, 156, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, H.; Kim, H.-S.; Yang, M.; Yue, N.; Ren, G.; Zhu, K.; Liu, S.; Park, N.-G.; Zhang, Y.; et al. Multiple-Stage Structure Transformation of Organic-Inorganic Hybrid Perovskite CH3NH3PbI3. Phys. Rev. X 2016, 6, 1–20. [Google Scholar] [CrossRef]
- Ptak, M.; Mączka, M.; Gągor, A.; Sieradzki, A.; Stroppa, A.; Di Sante, D.; Perez-Mato, J.M.; Macalik, L. Experimental and theoretical studies of structural phase transition in a novel polar perovskite-like [C2H5NH3][Na0.5Fe0.5(HCOO)3] formate. Dalton Trans. 2016, 45, 2574–2583. [Google Scholar] [CrossRef] [PubMed]
- Trzebiatowska-Gusowska, M.; Ptak, M. The mechanism of phase transitions in azide perovskites probed by vibrational spectroscopy. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 214, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Leguy, A.M.A.; Goñi, A.R.; Frost, J.M.; Skelton, J.; Brivio, F.; Rodríguez-Martínez, X.; Weber, O.J.; Pallipurath, A.; Alonso, M.I.; Campoy-Quiles, M.; et al. Dynamic disorder, phonon lifetimes, and the assignment of modes to the vibrational spectra of methylammonium lead halide perovskites. Phys. Chem. Chem. Phys. 2016, 18, 27051–27066. [Google Scholar] [CrossRef] [Green Version]
- Ma̧czka, M.; Pietraszko, A.; Macalik, B.; Hermanowicz, K. Structure, Phonon Properties, and Order–Disorder Transition in the Metal Formate Framework of [NH4][Mg(HCOO)3]. Inorg. Chem. 2014, 53, 787–794. [Google Scholar] [CrossRef]
- Trzebiatowska, M.; Ma̧czka, M.; Ptak, M.; Giriunas, L.; Balciunas, S.; Šimėnas, M.; Klose, D.; Banys, J. Spectroscopic Study of Structural Phase Transition and Dynamic Effects in a [(CH3)2NH2][Cd(N3)3] Hybrid Perovskite Framework. J. Phys. Chem. C 2019, 123, 11840–11849. [Google Scholar] [CrossRef]
- Hanna, S.; Mautner, F.; Koppelhuber-Bitschnau, B.; Abu-Youssef, M. Preparation and Study of Phase Transformation of the Azido Complex [N(CH3)4][CD(N3)3] by X-Ray Powder Diffraction and DSC. Mater. Sci. Forum 2000, 321, 1098–1101. [Google Scholar] [CrossRef]
- Mañosa, L.; Planes, A. Materials with Giant Mechanocaloric Effects: Cooling by Strength. Adv. Mater. 2016, 29, 11. [Google Scholar] [CrossRef]
- Mhiri, A.; Krichen, F.; Oueslati, A.; Lhoste, J.; Goutenoire, F.; Gargouri, M.; Bulou, A. Synthesis, structural characterization and spectroscopic studies of bis tetramethylammonium hexabromostannate [N(CH3)4]2SnBr6. J. Alloys Compd. 2019, 772, 546–556. [Google Scholar] [CrossRef]
- Ma̧czka, M.; Ptak, M.; Macalik, L. Infrared and Raman studies of phase transitions in metal–organic frameworks of [(CH3)2NH2][M(HCOO)3] with M=Zn, Fe. Vib. Spectrosc. 2014, 71, 98–104. [Google Scholar] [CrossRef]
- Simonis, G.J.; Hathaway, C.E. Raman spectrum and phase transition in sodium azide. Phys. Rev. B 1974, 10, 4419–4433. [Google Scholar] [CrossRef]
- Chitnis, A.; Bhatt, H.; Ma̧czka, M.; Deo, M.N.; Garg, N.; Deo, M.N. Remarkable resilience of the formate cage in a multiferroic metal organic framework material: Dimethyl ammonium manganese formate (DMAMnF). Dalton Trans. 2018, 47, 12993–13005. [Google Scholar] [CrossRef]
- Trzebiatowska-Gusowska, M.; Gągor, A. The order-disorder state of diaminoalkanes in Cu-based metal-organic materials. J. Coord. Chem. 2017, 70, 1536–1547. [Google Scholar] [CrossRef]
- Szymborska-Małek, K.; Trzebiatowska-Gusowska, M.; Mączka, M.; Gągor, A. Temperature-dependent IR and Raman studies of metal–organic frameworks [(CH3)2NH2][M(HCOO)3], M=Mg and Cd. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2016, 159, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Aghdaee, S.R.; Rae, A.I.M. The phase transition in sodium azide. J. Chem. Phys. 1983, 79, 4558. [Google Scholar] [CrossRef]
- Spackman, M.A.; Jayatilaka, D. Hirshfeld surface analysis. Cryst. Eng. Commun. 2009, 11, 19–32. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compound [(CH3)4N][Cd(N3)3] is available from the authors. |







| Parameters | Heating | Cooling | ||
|---|---|---|---|---|
| Tα→β/Tβ→γ | Tγ→δ | Tα←β/Tβ←γ | Tγ←δ | |
| Tt (K) | 270.4/277.6 | 322.0 | 263.9/269.6 | 319.5 |
| |∆Hib| (J·g−1) | 8.40 | 8.46 | 9.22 | 9.14 |
| |∆Sib| (J·Kg−1K−1) | 29.83 | 25.97 | 32.26 | 28.51 |
| N | 2.9 | 2.5 | 3.2 | 2.8 |
| δTt/δP (K kbar−1) | 12.39 | −6.52 | - | - |
| C2/c (80 K) | P21/m (298 K) | Pm-3m (373 K) | Assignment |
|---|---|---|---|
| 3044 m | νsCH3 | ||
| 3037 s | 3032 m | 3031m | νasCH3 νsCH3 |
| 2982 s | 2978 m | 2978 m | νsCH3 |
| 2972 m | 2969 sh | 2970 sh | νasCH3 νsCH3 |
| 2961 m/2949 s | 2953 m | 2954 | νsCH3 |
| 2917 vw | 2921 m | 2923 m | νasCH3 νsCH3 |
| 2821 vw | 2820 w | 2824 w | 2xδasCH3 |
| 2812 vw | 2813 w | 2814 vw | 2xνs(ν1)N3− |
| 1479 vw | 1481 vw | 1471 vw | δasCH3 δsCH3 |
| 1462 vw | 1453 w | 1451 w | δasCH3 δsCH3 |
| 1454 s/1438 s/1418 vw | 1440 vw | 1440 vw/ | combination |
| 1399 vw | combination | ||
| 1366 vs | 1367 s | νs(ν1)N3− | |
| 1360 vs/135 5m | 1359 vs | 1359 vs/ | νs(ν1)N3 |
| 1338 w | 1337 vw | 1337 w | |
| 1290 m | 1289 w | 1285 vw | ρCH3 |
| 1273 w | 1267 vw | ρCH3 | |
| 1217 w | 1218 vw | ρCH3 | |
| 1179 w | 1171 vw | 1174 | ρCH3 |
| 1170 w | ρCH3 | ||
| 1045 w | 1047 vw | 1043 w/1047 vw | ρCH3 |
| 958 m/952 m | 952 w | 950 w | νasNC4 |
| 762 m | 758 m | 759 m | νsNC4 |
| 755 m | 752 sh | 753 w-sh | |
| 730 vw | 741 vw | 741 vw | |
| 609 w | 609 w | ||
| 463 w/453 w | 458 vw | 458 vw | δNC4 |
| 378 w/369 w | 370 vw | 373 vw | δNC4 τCH3 |
| 302 vw-sh | 291 vw-sh | ||
| 275 s | 274 m | 261 w | τCH3 T′(Cd) |
| 223 vs | 220 m | 217 w | LN3− |
| 208 s | 186 w | 189 w | LN3− |
| 168 s | 161 w | 159 w | T’N3− T′(Cd) |
| 154 m | 149 m | 147 m | |
| 134 w | 137 w | 134 w | |
| 121 m | 121 w | 117 m | T’(TMA) |
| 116 s/115 m | T’(TMA) | ||
| 98 w/90 m | 101 w/92 vw | 101 w/91 w |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
da Silva, R.X.; de Araujo Paschoal, C.W.; Costa dos Santos, C.; García-Fernández, A.; Salgado-Beceiro, J.; Señarís-Rodríguez, M.A.; Sanchez-Andujar, M.; Nonato Almeida de Abreu Silva, A. Raman Spectroscopy Studies on the Barocaloric Hybrid Perovskite [(CH3)4N][Cd(N3)3]. Molecules 2020, 25, 4754. https://doi.org/10.3390/molecules25204754
da Silva RX, de Araujo Paschoal CW, Costa dos Santos C, García-Fernández A, Salgado-Beceiro J, Señarís-Rodríguez MA, Sanchez-Andujar M, Nonato Almeida de Abreu Silva A. Raman Spectroscopy Studies on the Barocaloric Hybrid Perovskite [(CH3)4N][Cd(N3)3]. Molecules. 2020; 25(20):4754. https://doi.org/10.3390/molecules25204754
Chicago/Turabian Styleda Silva, Rosivaldo Xavier, Carlos William de Araujo Paschoal, Clenilton Costa dos Santos, Alberto García-Fernández, Jorge Salgado-Beceiro, María Antonia Señarís-Rodríguez, Manuel Sanchez-Andujar, and Ariel Nonato Almeida de Abreu Silva. 2020. "Raman Spectroscopy Studies on the Barocaloric Hybrid Perovskite [(CH3)4N][Cd(N3)3]" Molecules 25, no. 20: 4754. https://doi.org/10.3390/molecules25204754