Pestalotiones A–D: Four New Secondary Metabolites from the Plant Endophytic Fungus Pestalotiopsis Theae
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isolation and Structure Elucidation
2.2. Bioactivities
3. Experimental Section
3.1. General Experimental Procedures
3.2. Fungal Material
3.3. Extraction and Isolation
3.4. Spectroscopic Data (uv and IR, ms)
3.5. MTS Assay
3.6. Antioxidant Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Jiang, C.S.; Zhou, Z.F.; Yang, X.H.; Lan, L.F.; Gu, Y.C.; Ye, B.P.; Guo, Y.W. Antibacterial sorbicillin and diketopiperazines from the endogenous fungus Penicillium sp. GD6 associated Chinese mangrove Bruguiera gymnorrhiza. Chin. J. Nat. Med. 2018, 16, 358–365. [Google Scholar] [CrossRef]
- Wu, Z.H.; Liu, D.; Xu, Y.; Chen, J.L.; Lin, W.H. Antioxidant xanthones and anthraquinones isolated from a marine-derived fungus Aspergillus versicolor. Chin. J. Nat. Med. 2018, 16, 219–224. [Google Scholar] [CrossRef]
- Zhang, X.; Li, Z.; Gao, J. Chemistry and biology of secondary metabolites from Aspergillus Genus. Nat. Prod. J. 2018, 8, 275–304. [Google Scholar] [CrossRef]
- Aly, A.H.; Debbab, A.; Proksch, P. Fungal endophytes-secret producers of bioactive plant metabolites. Pharmazie 2013, 68, 499–505. [Google Scholar] [PubMed]
- Kusari, S.; Spiteller, M. Are we ready for industrial production of bioactive plant secondary metabolites utilizing endophytes? Nat. Prod. Rep. 2011, 28, 1203–1207. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.N.; Rogers, L.M.; Song, Y.D.; Guo, W.J.; Kolattukudy, P.E. Homoserine and asparagine are host signals that trigger in planta expression of a pathogenesis gene in Nectria haematococca. P. Natl. Acad. Sci. USA 2005, 102, 4197–4202. [Google Scholar] [CrossRef] [Green Version]
- Young, C.A.; Felitti, S.; Shields, K.; Spangenberg, G.; Johnson, R.D.; Bryan, G.T.; Saikia, S.; Scott, B. A complex gene cluster for indole-diterpene biosynthesis in the grass endophyte Neotyphodium lolii. Fungal Genet. Biol. 2006, 43, 679–693. [Google Scholar] [CrossRef]
- Zhang, H.W.; Song, Y.C.; Tan, R.X. Biology and chemistry of endophytes. Nat. Prod. Rep. 2006, 23, 753–771. [Google Scholar] [CrossRef]
- Tan, R.X.; Zou, W.X. Endophytes: a rich source of functional metabolites. Nat. Prod. Rep. 2001, 18, 448–459. [Google Scholar] [CrossRef]
- Strobel, G.; Yang, X.S.; Sears, J.; Kramer, R.; Sidhu, R.S.; Hess, W.M. Taxol from Pestalotiopsis microspora, an endophytic fungus of Taxus wallachiana. Microbiology 1996, 142, 435–440. [Google Scholar] [CrossRef] [Green Version]
- Ding, G.; Zheng, Z.H.; Liu, S.C.; Zhang, H.; Guo, L.D.; Che, Y.S. Photinides A-F, cytotoxic benzofuranone-derived α-lactones from the plant endophytic fungus Pestalotiopsis photiniae. J. Nat. Prod. 2009, 72, 942–945. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, L.; Si, Y.K.; Jiang, X.J.; Guo, L.D.; Che, Y.S. Virgatolides A-C, benzannulated spiroketals from the plant endophytic fungus Pestalotiopsis virgatula. Org. Lett. 2011, 13, 2670–2673. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wu, X.F.; Ding, G.; Feng, Y.; Jiang, X.J.; Guo, L.D.; Che, Y.S. α-Pyrones and pyranes from the plant pathogenic fungus Pestalotiopsis scirpina. Eur. J. Org. Chem. 2012, 2445–2452. [Google Scholar] [CrossRef]
- Yang, X.L.; Zhang, J.Z.; Luo, D.Q. The taxonomy, biology and chemistry of the fungal Pestalotiopsis genus. Nat. Prod. Rep. 2012, 29, 622–641. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Yang, X.B.; Lin, Q. Chemistry and biology of Pestalotiopsis-derived natural products. Fungal Divers. 2014, 66, 37–68. [Google Scholar] [CrossRef]
- Strobel, G.A.; Hess, W.M.; Li, J.Y.; Ford, E.; Sears, J.; Sidhu, R.S.; Summerell, B. Pestalotiopsis guepinii, a taxol-producing endophyte of the Wollemi pine, Wollemia nobilis. Aust. J. Bot. 1997, 45, 1073–1082. [Google Scholar] [CrossRef]
- Metz, A.M.; Haddad, A.; Worapong, J.; Long, D.M.; Ford, E.J.; Hess, W.M.; Strobel, G.A. Induction of the sexual stage of Pestalotiopsis microspora, a taxol-producing fungus. Microbiology 2000, 146, 2079–2089. [Google Scholar] [CrossRef] [Green Version]
- Akone, S.H.; El Amrani, M.; Lin, W.H.; Lai, D.W.; Proksch, P. Cytosporins F-K, new epoxyquinols from the endophytic fungus Pestalotiopsis theae. Tetrahedron Lett. 2013, 54, 6751–6754. [Google Scholar] [CrossRef]
- Nagata, T.; Ando, Y.; Hirota, A. Phytotoxins from tea gray blight fungi, Pestalotiopsis longiseta and Pestalotiopsis theae. Biosci. Biotech. Bioch. 1992, 56, 810–811. [Google Scholar] [CrossRef] [Green Version]
- Li, E.W.; Tian, R.R.; Liu, S.C.; Chen, X.L.; Guo, L.D.; Che, Y.S. Pestalotheols A-D, bioactive metabolites from the plant endophytic fungus Pestalotiopsis theae. J. Nat. Prod. 2008, 71, 664–668. [Google Scholar] [CrossRef]
- Ding, G.; Jiang, L.H.; Guo, L.D.; Chen, X.L.; Zhang, H.; Che, Y.S. Pestalazines and pestalamides, bioactive metabolites from the plant pathogenic fungus Pestalotiopsis theae. J. Nat. Prod. 2008, 71, 1861–1865. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Han, Y.; Xiao, J.H.; Li, L.; Guo, L.D.; Jiang, X.J.; Kong, L.Y.; Che, Y.S. Chlorotheolides A and B, spiroketals generated via Diels-Alder Reactions in the endophytic fungus Pestalotiopsis theae. J. Nat. Prod. 2016, 79, 2616–2623. [Google Scholar] [CrossRef] [PubMed]
- Shimada, A.; Takahashi, I.; Kawano, T.; Kimura, Y. Chloroisosulochrin, chloroisosulochrin dehydrate, and pestheic acid, plant growth regulators, produced by Pestalotiopsis theae. Z. Naturforsch B. 2001, 56, 797–803. [Google Scholar] [CrossRef]
- Hamasaki, T.; Kimura, Y. Isolation and structures of four new metabolites from Aspergillus wentii. Agric. Bioi. Chem. 1983, 47, 163–165. [Google Scholar] [CrossRef] [Green Version]
- Klaiklay, S.; Rukachaisirikul, V.; Tadpetch, K.; Sukpondma, Y.; Phongpaichit, S.; Buatong, J.; Sakayaroj, J. Chlorinated chromone and diphenyl ether derivatives from the mangrove-derived fungus Pestalotiopsis sp. PSU-MA69. Tetrahedron 2012, 68, 2299–2305. [Google Scholar] [CrossRef]
- Du, L.; Zhu, T.; Liu, H.; Fang, Y.; Zhu, W.; Gu, Q. Cytotoxic polyketides from a marine-derived fungus Aspergillus glaucus. J. Nat. Prod. 2008, 71, 1837–1842. [Google Scholar] [CrossRef]
- Li, Y.; Li, X.; Lee, U.; Kang, J.S.; Choi, H.D.; Sona, B.W. A new radical scavenging anthracene glycoside, asperflavin ribofuranoside, and polyketides from a marine isolate of the fungus microsporum. Chem. Pharm. Bull. 2006, 54, 882–883. [Google Scholar] [CrossRef] [Green Version]
- Serianni, A.S.; Barker, R. [13C]-Enriched tetroses and tetrofuranosides: An evaluation of the relationship between NMR parameters and furanosyl ring conformation. J. Org. Chem. 1984, 49, 3292–3300. [Google Scholar] [CrossRef]
- Ness, R.K.; Diehl, H.W.; Fletcher, H.G., Jr. New benzoyl derivatives of d-ribofuranose and aldehydo-d-ribose. The preparation of crystalline 2, 3, 5-Tri-O-benzoyl-β-d-ribose from d-Ribose1. J. Am.Chem. Soc. 1954, 76, 763–767. [Google Scholar] [CrossRef]
- Xu, X.X.; Liu, L.; Zhang, F.; Wang, W.Z.; Li, J.Y.; Guo, L.D.; Che, Y.S.; Liu, G. Identification of the first diphenyl ether gene cluster for pestheic acid biosynthesis in plant endophyte Pestalotiopsis fici. Chembiochem 2014, 15, 284–292. [Google Scholar] [CrossRef]
- Gobbi, S.; Zimmer, C.; Belluti, F.; Rampa, A.; Hartmann, R.W.; Recanatini, M.; Bisi, A. Novel highly potent and selective nonsteroidal aromatase inhibitors: Synthesis, biological evaluation and structure-activity relationships investigation. J. Med. Chem. 2010, 53, 5347–5351. [Google Scholar] [CrossRef] [PubMed]
- Palmeira, A.; Paiva, A.; Sousa, E.; Seca, H.; Almeida, G.M.; Lima, R.T.; Fernandes, M.X.; Pinto, M.; Vasconcelos, M.H. Insights into the in vitro antitumor mechanism of action of a new pyranoxanthone. Chem. Biol. Drug Des. 2010, 76, 43–58. [Google Scholar] [CrossRef] [PubMed]
- Honda, N.; Pavan, F.R.; Coelho, R.; de Andrade Leite, S.; Micheletti, A.; Lopes, T.; Misutsu, M.; Beatriz, A.; Brum, R.; Leite, C.Q.F. Antimycobacterial activity of lichen substances. Phytomedicine 2010, 17, 328–332. [Google Scholar] [CrossRef] [PubMed]
- Pinto, M.; Sousa, M.; Nascimento, M. Xanthone derivatives: New insights in biological activities. Curr. Med. Chem. 2005, 12, 2517–2538. [Google Scholar] [CrossRef] [PubMed]
- Shi, T.; Qi, J.; Shao, C.L.; Zhao, D.L.; Hou, X.M.; Wang, C.Y. Bioactive Diphenyl Ethers and Isocoumarin Derivatives from a Gorgonian-Derived Fungus Phoma sp. (TA07-1). Mar. Drugs 2017, 15, 146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, L.; Chen, X.Y.; Li, D.; Zhang, Y.; Li, L.; Guo, L.D.; Cao, Y.; Che, Y.S. Bisabolane sesquiterpenoids from the plant endophytic fungus Paraconiothyrium brasiliense. J. Nat. Prod. 2015, 78, 746–753. [Google Scholar] [CrossRef]
- Tan, C.B.; Liu, Z.M.; Chen, S.H.; Huang, X.S.; Cui, H.; Long, Y.H.; Lu, Y.J.; She, Z.G. Antioxidative polyketones from the mangrove-derived fungus Ascomycota sp. SK2YWS-L. Sci. Rep. 2016, 6, 36609. [Google Scholar] [CrossRef] [Green Version]
Sample Availability: Not available. |
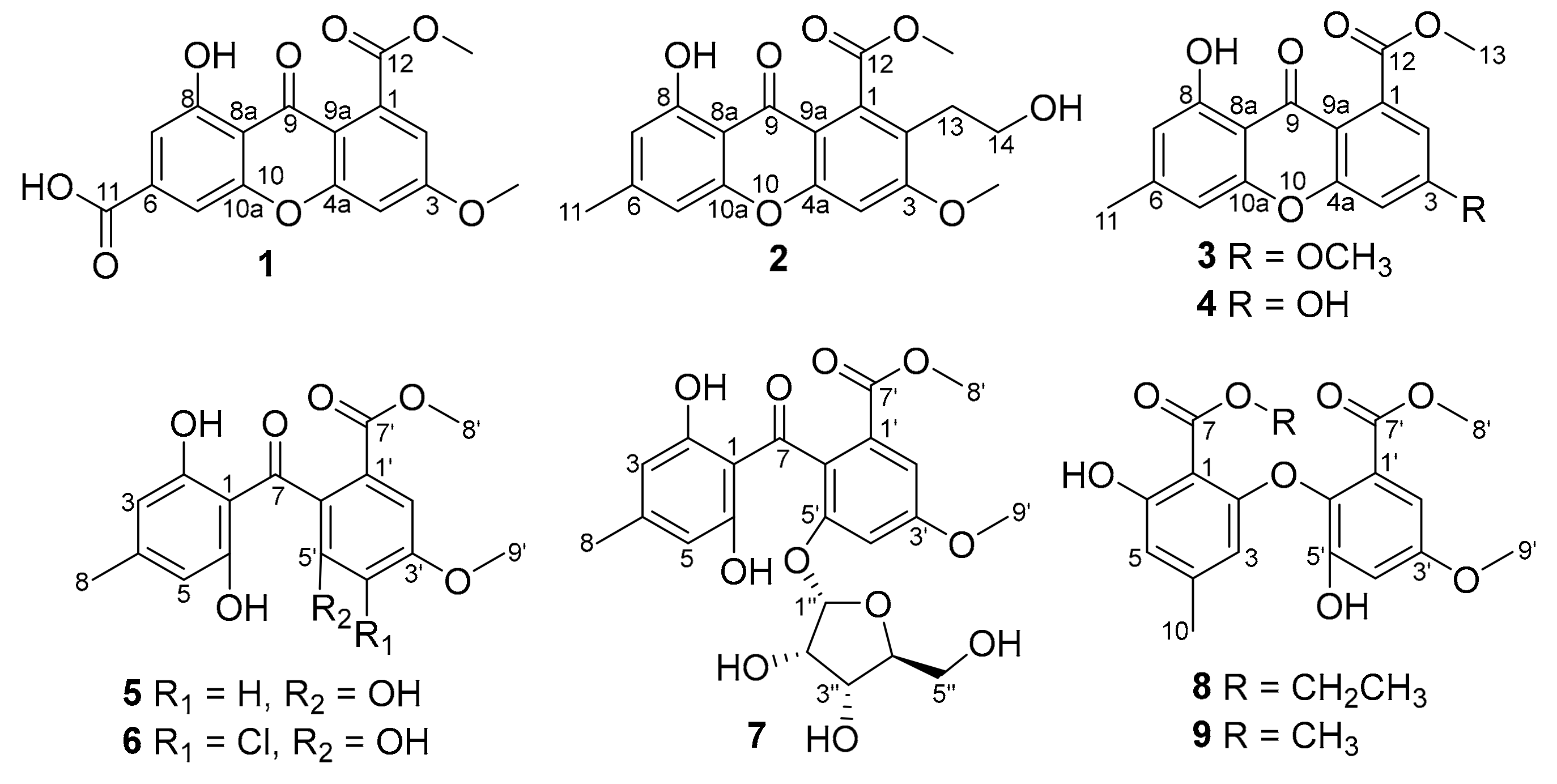


| Pos. | 1 | 2 | ||||
|---|---|---|---|---|---|---|
| δHa (J in Hz) | δCb | HMBCc | δHa (J in Hz) | δCb | HMBCc | |
| 1 | 134.7, qC | 141.2, qC | ||||
| 2 | 7.13, d (2.2) | 112.9, CH | 3, 4, 9a, 12 | 118.5, qC | ||
| 3 | 165.3, qC | 166.7, qC | ||||
| 4 | 7.30, d (2.2) | 101.9, CH | 2, 3, 4a, 9, 9a | 7.22, s | 101.3, CH | 2, 3, 4a, 9, 9a |
| 4a | 158.1, qC | 159.5, qC | ||||
| 5 | 7.45, s | 107.5, CH | 6, 7, 9, 10a, 11 | 6.83, s | 108.2, CH | 7, 8a, 9, 10a, 11 |
| 6 | 139.4, qC | 150.3, qC | ||||
| 7 | 7.23, s | 110.6, CH | 5, 6, 8, 8a, 9, 11 | 6.63, s | 112.4, CH | 5, 8, 8a, 11, |
| 8 | 160.7, qC | 162.3, qC | ||||
| 8a | 110.1, qC | 107.1, qC | ||||
| 9 | 179.5, qC | 180.3, qC | ||||
| 9a | 110.3, qC | 111.7, qC | ||||
| 10a | 155.3, qC | 156.8, qC | ||||
| 11 | 165.8, qC | 2.43, s | 22.4, CH3 | 5, 6, 7 | ||
| 12 | 167.9, qC | 168.4, qC | ||||
| 13a | 3.00, m | 38.0, CH2 | 2, 14 | |||
| 13b | 2.87, m | 2, 14 | ||||
| 14a | 3.56, m | 61.5, CH2 | 13 | |||
| 14b | 3.50, m | 13 | ||||
| CH3O-3 | 3.98, s | 56.8, CH3 | 3 | 4.14, s | 57.9, CH3 | 3 |
| CH3O-12 | 3.89, s | 52.8, CH3 | 12 | 3.97, s | 53.1, CH3 | 12 |
| HO-8 | 12.3, s | 7, 8, 8a | 12.3, s | 7, 8, 8a | ||
| Pos. | δHa (J in Hz) | δCb | HMBCc |
|---|---|---|---|
| 1 | 110.9, qC | ||
| 2 | 163.2, qC | ||
| 3 | 6.13, s | 109.0, CH | 1, 2, 5, 7, 8 |
| 4 | 149.6, qC | ||
| 5 | 6.13, s | 109.0, CH | 1, 3, 6, 7, 8 |
| 6 | 163.2, qC | ||
| 7 | 201.1, qC | ||
| 8 | 2.20, s | 22.1, CH3 | 3, 4, 5 |
| 1′ | 130.6, qC | ||
| 2′ | 7.17, d (2.4) | 109.0, CH | 1′, 3′, 4′, 6′, 7′ |
| 3′ | 161.6, qC | ||
| 4′ | 7.03, d (2.4) | 108.2, CH | 2′, 3′, 5′, 6′ |
| 5′ | 155.7, qC | ||
| 6′ | 130.4, qC | ||
| 7′ | 167.7, qC | ||
| 8′ | 3.70, s | 52.6, CH3 | 7′ |
| 9′ | 3.86, s | 56.2, CH3 | 3′ |
| 1′′ | 5.56, d (4.4) | 103.3, CH | 5′, 2′′, 3′′, 4′′ |
| 2′′ | 4.00, dd (6.4, 4.4) | 73.3, CH | 1′′ |
| 3′′ | 3.90, dd (6.4, 3.2) | 70.7, CH | 1′′ |
| 4′′ | 3.96, dd (6.8, 3.2) | 88.1, CH | |
| 5′′ | 3.55, m | 63.0, CH2 | 3′′, 4′′ |
| Pos. | δHa (J in Hz) | δCb | HMBCc |
|---|---|---|---|
| 1 | 102.1, qC | ||
| 2 | 158.2, qC | ||
| 3 | 5.94, d (1.2) | 107.1, CH | 1, 2, 4, 5, 7, 10 |
| 4 | 146.6, qC | ||
| 5 | 6.49, d (1.2) | 112.5, CH | 1, 3, 6, 7, 10 |
| 6 | 162.1, qC | ||
| 7 | 169.7, qC | ||
| 8 | 4.47, q (7.2) | 62.4, CH2 | 7, 9 |
| 9 | 1.40, t (7.2) | 14.3, CH3 | 8 |
| 10 | 2.16, s | 22.2, CH3 | 3, 4, 5 |
| 1′ | 125.5, qC | ||
| 2′ | 7.05, d (3.2) | 107.3, CH | 1′, 3′, 4′, 6′, 7′ |
| 3′ | 157.5, qC | ||
| 4′ | 6.81, d (3.2) | 107.3, CH | 2′, 3′, 5′, 6′ |
| 5′ | 150.7, qC | ||
| 6′ | 135.4, qC | ||
| 7′ | 165.4, qC | ||
| 8′ | 3.74, s | 52.5, CH3 | 7′ |
| 9′ | 3.84, s | 55.9, CH3 | 3′ |
| OH-6 | 10.54, s | 1, 5, 6 | |
| OH-5′ | 6.84, br s | 4′, 6′ |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, L.; Lin, J.; Niu, S.; Liu, S.; Liu, L. Pestalotiones A–D: Four New Secondary Metabolites from the Plant Endophytic Fungus Pestalotiopsis Theae. Molecules 2020, 25, 470. https://doi.org/10.3390/molecules25030470
Guo L, Lin J, Niu S, Liu S, Liu L. Pestalotiones A–D: Four New Secondary Metabolites from the Plant Endophytic Fungus Pestalotiopsis Theae. Molecules. 2020; 25(3):470. https://doi.org/10.3390/molecules25030470
Chicago/Turabian StyleGuo, Longfang, Jie Lin, Shubin Niu, Shuchun Liu, and Ling Liu. 2020. "Pestalotiones A–D: Four New Secondary Metabolites from the Plant Endophytic Fungus Pestalotiopsis Theae" Molecules 25, no. 3: 470. https://doi.org/10.3390/molecules25030470





