Development of Coumarin-Based Hydroxamates as Histone Deacetylase Inhibitors with Antitumor Activities
Abstract
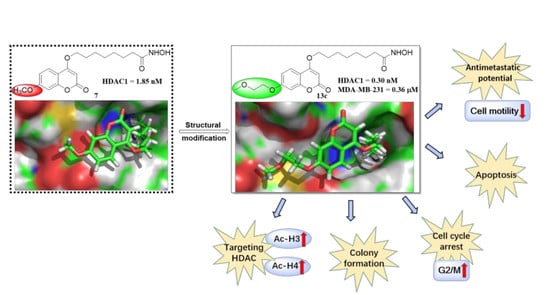
:1. Introduction
2. Chemistry
3. Results and Discussion
3.1. HDAC1 Inhibitory Activity of Synthesized Compounds
3.2. Antiproliferative Activity
3.3. Colony Formation Assay
3.4. Western Blot Analysis
3.5. Anti-Migration Assay
3.6. Cell Cycle Arrest Analysis
3.7. Apoptosis Analysis
3.8. Molecular Docking Studies
4. Experimental Section
4.1. Materials and Methods
4.2. General Procedures for Synthesis of 13a–g and 14a–s
4.3. HDAC1 Inhibitory Assay
4.4. Cell Culture and Reagents
4.5. Colony Formation Assay
4.6. Western Blot Analysis
4.7. Anti-Migration Assay
4.8. Cell Cycle Arrest Analysis and Apoptosis Analysis
4.9. Molecular Docking Studies
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Nepali, K.; Sharma, S.; Sharma, M.; Bedi, P.M.S.; Dhar, K.L. Rational approaches, design strategies, structure activity relationship and mechanistic insights for anticancer hybrids. Eur. J. Med. Chem. 2014, 77, 422–487. [Google Scholar] [CrossRef]
- Sangwan, R.; Rajan, R.; Mandal, P.K. HDAC as onco target: Reviewing the synthetic approaches with SAR study of their inhibitors. Eur. J. Med. Chem. 2018, 158, 620–706. [Google Scholar] [CrossRef]
- Marks, P.; Rifkind, R.A.; Richon, V.M.; Breslow, R.; Miller, T.; Kelly, W.K. Histone deacetylases and cancer: Causes and therapies. Nat. Rev. Cancer 2001, 1, 194–202. [Google Scholar] [CrossRef]
- Ruijter, A.J.D.; Gennip, A.H.V.; Caron, H.N.; Kemp, S.; Kuilenburg, A.B.V. Histone deacetylases (HDACs): Characterization of the classical HDAC family. Biochem. J. 2003, 370, 737–749. [Google Scholar] [CrossRef] [PubMed]
- Monneret, C. Histone deacetylase inhibitors. Eur. J. Med. Chem. 2005, 40, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Bolden, J.E.; Peart, M.J.; Johnstone, R.W. Anticancer activities of histone deacetylase inhibitors. Nat. Rev. Drug Discov. 2006, 5, 769–784. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.Y.; Tsai, A.C.; Chen, M.C.; Shen, P.J.; Cheng, Y.C.; Kuo, C.C.; Pan, S.L.; Liu, Y.M.; Liu, J.F.; Yeh, T.K.; et al. Azaindolylsulfonamides, with a more selective inhibitory effect on histone deacetylase 6 activity, exhibit antitumor activity in colorectal cancer HCT116 cells. J. Med. Chem. 2014, 57, 4009–4022. [Google Scholar] [CrossRef] [PubMed]
- Vreese, R.D.; Depetter, Y.; Verhaeghe, T.; Desmet, T.; Benoy, V.; Haeck, W.; Boschc, L.V.D.; D’hooghe, M. Synthesis and SAR assessment of novel Tubathian analogs in the pursuit of potent and selective HDAC6 inhibitors. Org. Biomol. Chem. 2016, 14, 2537–2549. [Google Scholar] [CrossRef] [PubMed]
- Ojha, R.; Huang, H.L.; HuangFu, W.C.; Wu, Y.W.; Nepali, K.; Lai, M.J.; Su, C.J.; Sung, T.E.; Chen, Y.L.; Pan, S.L.; et al. 1-Aroylindoline-hydroxamic acids as anticancer agents, inhibitors of HSP90 and HDAC. Eur. J. Med. Chem. 2018, 150, 667–677. [Google Scholar] [CrossRef]
- Abdizadeh, T.; Kalani, M.R.; Abnous, K.; Tayarani-Najaran, Z.; Khashyarmanesh, B.Z.; Abdizadeh, R.; Ghodsi, R.; Hadizadeh, F. Design, synthesis and biological evaluation of novel coumarin-based benzamides as potent histone deacetylase inhibitors and anticancer agents. Eur. J. Med. Chem. 2017, 132, 42–62. [Google Scholar] [CrossRef]
- Bertrand, P. Inside HDAC with HDAC inhibitors. Eur. J. Med. Chem. 2010, 45, 2095–2116. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhao, S.; Li, H.M.; Wang, X.; Geng, A.; Cui, H.; Lu, T.; Chen, Y.D.; Zhu, Y. Design, synthesis and biological evaluation of novel isoindolinone derivatives as potent histone deacetylase inhibitors. Eur. J. Med. Chem. 2019, 168, 110–122. [Google Scholar] [CrossRef] [PubMed]
- Dietz, K.C.; Casaccia, P. HDAC inhibitors and neurodegeneration: At the edge between protection and damage. Pharmacol. Res. 2010, 62, 11–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, C.H.; Yun, F.; He, J.; Ullah, S.; Yuan, Q.P. Design, synthesis and biological evaluation of novel thioquinazolinone-based 2-aminobenzamide derivatives as potent histone deacetylase (HDAC) inhibitors. Eur. J. Med. Chem. 2019, 173, 185–202. [Google Scholar] [CrossRef]
- Gao, L.; Cueto, M.A.; Asselbergs, F.; Atadja, P. Cloning and functional characterization of HDAC11, a novel member of the human histone deacetylase family. J. Biol. Chem. 2002, 277, 25748–25755. [Google Scholar] [CrossRef] [Green Version]
- Bottomley, M.J.; Lo Surdo, P.; Di Giovine, P.; Cirillo, A.; Scarpelli, R.; Ferrigno, F.; Jones, P.; Neddermann, P.; De Francesco, R.; Steinkuhler, C.; et al. Structural and functional analysis of the human HDAC4 catalytic domain reveals a regulatory structural zinc-binding domain. J. Biol. Chem. 2008, 283, 26694–26704. [Google Scholar] [CrossRef] [Green Version]
- Haigis, M.C.; Guarente, L.P. Mammalian sirtuins-emerging roles in physiology, aging, and calorie restriction. Genes Dev. 2006, 20, 2913–2921. [Google Scholar] [CrossRef] [Green Version]
- Liu, T.; Liu, P.Y.; Marshall, G.M. The critical role of the class III histone deacetylase SIRT1 in cancer. Cancer Res. 2009, 69, 1702–1705. [Google Scholar] [CrossRef] [Green Version]
- Choo, Q.Y.; Paul, C.H.; Tanaka, Y.; Lin, H.S. Histone deacetylase inhibitors MS-275 and SAHA induced growth arrest and suppressed lipopolysaccharide-stimulated NF-kappaB P65 nuclear accumulation in human rheumatoid arthritis synovial fibroblastic E11 cells. Rheumatology 2010, 49, 1447–1460. [Google Scholar] [CrossRef] [Green Version]
- Lee, H.Z.; Kwitkowski, V.E.; Del Valle, P.L.; Ricci, M.S.; Saber, H.; Habtemariam, B.A.; Bullock, J.; Bloomquist, E.; Shen, Y.L.; Chen, X.H. FDA approval: Belinostat for the treatment of patients with relapsed or refractory peripheral T-cell lymphoma. Clin. Cancer Res. 2015, 21, 2666–2670. [Google Scholar] [CrossRef] [Green Version]
- Garnock-Jones, K.P. Panobinostat: First global approval. Drugs 2015, 25, 695–704. [Google Scholar] [CrossRef]
- Furumai, R.; Matsuyama, A.; Kobashi, N.; Lee, K.H.; Nishiyama, M.; Nakajima, H.; Tanaka, A.; Komatsu, Y.; Nishino, N.; Yoshida, M. FK228 (depsipeptide) as a natural prodrug that inhibits classI histone deacetylases. Cancer Res. 2002, 62, 4916–4921. [Google Scholar]
- Moradei, O.; Vaisburg, A.; Martell, R.E. Histone deacetylase inhibitors in cancer therapy: New compounds and clinical update of benzamide-type agents. Curr. Top. Med. Chem. 2008, 8, 841–858. [Google Scholar] [CrossRef]
- Taddei, M.; Cini, E.; Giannotti, L.; Giannini, G.; Battistuzzi, G.; Vignola, D.; Vesci, L.; Cabri, W. Lactam based 7-amino suberoylamide hydroxamic acids as potent HDAC inhibitors. Bioorganic Med. Chem. 2014, 24, 61–64. [Google Scholar] [CrossRef]
- Huang, M.L.; Zhang, J.; Yan, C.J.; Li, X.H.; Zhang, J.L.; Ling, R. Small molecule HDAC inhibitors: Promising agents for breast cancer treatment. Bioorganic Chem. 2019, 91, 103184. [Google Scholar] [CrossRef] [PubMed]
- Jin, P.; Chen, X. Current status of epigenetics and anticancer drug discovery. Anti-Cancer Agents Med. Chem. 2016, 16, 699–712. [Google Scholar] [CrossRef]
- Manal, M.; Chandrasekar, M.J.N.; Priya, J.G.; Nanjan, M.J. Inhibitors of histone deacetylase as antitumor agents: A critical review. Bioorganic Chem. 2016, 67, 18–42. [Google Scholar] [CrossRef] [PubMed]
- Zhan, P.; Wang, X.; Liu, X.; Suzuki, T. Medicinal chemistry insights into novel HDAC inhibitors: An updated patent review (2012–2016). Recent pat. Anti-Cancer Drug Discov. 2017, 12, 16–34. [Google Scholar] [CrossRef] [PubMed]
- Bansal, Y.; Sethi, P.; Bansal, G. Coumarin: A potential nucleus for anti-inflammatory molecules. Med. Chem. Res. 2013, 22, 3049–3060. [Google Scholar] [CrossRef]
- Amin, K.M.; Awadalla, F.M.; Eissa, A.A.M.; Abou-Seri, S.M.; Hassan, G.S. Design, synthesis and vasorelaxant evaluation of novel coumarin-pyrimidine hybrids. Bioorganic Med. Chem. 2011, 19, 6087–6097. [Google Scholar] [CrossRef] [PubMed]
- Basanagouda, M.; Jambagi, V.B.; Barigidad, N.N.; Laxmeshwar, S.S.; Devaru, V.; Narayana, C. Synthesis, structure-activity relationship of iodinated-4-aryloxymethyl-coumarins as potential anti-cancer and anti-mycobacterial agents. Eur. J. Med. Chem. 2014, 74, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Riveiro, M.E.; Moglioni, A.; Vazquez, R.; Gomez, N.; Facorro, G.; Piehl, L.; De Celis, E.R.; Shayo, C.; Davio, C. Structural insights into hydroxycoumarin-induced apoptosis in U-937 cells. Bioorganic Med. Chem. 2008, 16, 2665–2675. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.F.; Zhao, N.; Song, J.L.; Zhu, K.K.; Jiang, C.S.; Shan, P.P.; Zhang, H. Design, synthesis and biological evaluation of novel coumarin-based hydroxamate derivatives as histone deacetylase (hdac) inhibitors with antitumor activities. Molecules 2019, 24, 2569. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ge, D.; Han, L.N.; Yang, F.F.; Zhao, N.; Yang, Y.; Zhang, H.; Chen, Y.H. Development of hydroxamate-based histone deacetylase inhibitors of bis-substituted aromatic amides with antitumor activities. MedChemComm 2019, 10, 1828–1837. [Google Scholar] [CrossRef]
Sample Availability: Samples of compounds 13a–g and 14a–s are available from the authors. |










| Compounds | R | IC50 (nM) |
|---|---|---|
| 13a |  | 0.48 |
| 13b |  | 0.31 |
| 13c |  | 0.30 |
| 13d |  | 0.33 |
| 13e |  | 0.77 |
| 13f |  | 0.60 |
| 13g |  | 5.01 |
| 14a |  | 0.96 |
| 14b |  | 1.59 |
| 14c |  | 1.97 |
| 14d |  | 1.72 |
| 14e |  | 0.75 |
| 14f |  | 0.99 |
| 14g |  | 0.88 |
| 14h |  | 2.40 |
| 14i |  | 4.25 |
| 14j |  | 2.52 |
| 14k |  | 3.02 |
| 14l |  | 5.50 |
| 14m |  | 0.50 |
| 14n |  | 0.78 |
| 14o |  | 3.22 |
| 14p |  | 1.56 |
| 14q |  | 2.95 |
| 14r |  | 2.50 |
| 14s |  | 5.19 |
| 7 | 1.85 | |
| 1 | 12.36 |
| Compounds | MDA-MB-231 | H157 | A549 | MCF-7 |
|---|---|---|---|---|
| 13a | 0.73 ± 0.23 | 1.04 ± 0.08 | 5.62 ± 1.21 | 2.91 ± 0.43 |
| 13b | 1.97 ± 0.56 | 1.20 ± 0.17 | 4.09 ± 1.54 | 2.93 ± 0.49 |
| 13c | 0.36 ± 0.08 | 0.59 ± 0.03 | 2.79 ± 0.87 | 1.84 ± 0.17 |
| 13d | 1.70 ± 0.39 | 2.36 ± 0.50 | 4.11 ± 0.65 | 5.23 ± 1.63 |
| 14m | 1.95 ± 0.29 | 4.81 ± 0.8 | 7.58 ± 2.05 | 6.05 ± 1.33 |
| 1 | 1.33 ± 0.61 | 1.66 ± 0.14 | 5.34 ± 1.09 | 4.15 ± 0.74 |
| Compounds | Beas-2B | L-02 |
|---|---|---|
| 13a | >50 | >50 |
| 13b | >50 | >20 |
| 13c | >100 | >100 |
| 13d | >20 | >20 |
| 1 | >50 | >50 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, N.; Yang, F.; Han, L.; Qu, Y.; Ge, D.; Zhang, H. Development of Coumarin-Based Hydroxamates as Histone Deacetylase Inhibitors with Antitumor Activities. Molecules 2020, 25, 717. https://doi.org/10.3390/molecules25030717
Zhao N, Yang F, Han L, Qu Y, Ge D, Zhang H. Development of Coumarin-Based Hydroxamates as Histone Deacetylase Inhibitors with Antitumor Activities. Molecules. 2020; 25(3):717. https://doi.org/10.3390/molecules25030717
Chicago/Turabian StyleZhao, Na, Feifei Yang, Lina Han, Yuhua Qu, Di Ge, and Hua Zhang. 2020. "Development of Coumarin-Based Hydroxamates as Histone Deacetylase Inhibitors with Antitumor Activities" Molecules 25, no. 3: 717. https://doi.org/10.3390/molecules25030717