1. Introduction
Theileriosis and babesiosis are tick-transmitted diseases with significant economic impacts in the bovine and equine industries all over the world [
1].
Babesia divergens and
B. microti mainly affect cattle and rodents, respectively, and reveal zoonotic significance [
2].
Babesia divergens is the main reason for babesiosis in Europe, mainly in immunocompromised humans, while
B. microti is the main known etiologic agent responsible for human babesiosis in the USA.
Babesia bovis, as well as
B. bigemina, are the most pathogenic agents in cattle [
1], while
Theileria equi and
Babesia caballi are considered the most devastating infections in horses. Horses and cattle that survive acute infections may become permanent sources for continuous transmission [
3].
The prevention and control of babesiosis and theileriosis rely on vector control, the use of vaccines, and drugs available on the market [
3,
4]. Unfortunately, only a few drugs are available, such as a combination of clindamycin and quinine, atovaquone-azithromycin, diminazene aceturate (DA), and imidocarb dipropionate [
5]. However, some human cases showed resistance to atovaquone and azithromycin combination, moreover, imidocarb dipropionate has shown strong toxicity to the host, and DA–resistant parasites have emerged [
2,
6]. Although several studies have documented the effectiveness of antipiroplasmic herbal extracts such as
Syzygium aromaticum methanolic extract [
7], methanolic
Cuminum cyminum seeds extract and acetonic
Myrtus communis roots [
8], and methanolic
Olea europaea and acetonic
Acacia laeta [
9] against the growth of piroplasm parasites, none of these extracts are used by veterinarians for the treatment of clinical cases [
1]. Notably, there is an urgent and continuing need to identify alternative drugs to treat piroplasmosis.
Over the past few decades, research on herbal plants has provided modern medicine with several useful chemical ingredients that have been used to manage various ailments. However, many people in developing countries, especially in Africa and Asia, still rely on crude herbal extracts to treat several human and animal ailments [
10,
11]. This is partly because these extracts are inexpensive and easily accessible [
12].
Cinnamomum genus consists of thousands of species that are distributed all over the world and is considered as one of the most important and popular spices used in cooking as well as traditional and modern medicines [
13].
Cinnamomum verum,
C.
osmophloeum,
C. cassia,
C. camphora,
C. burmannii,
C. loureiroi and
C. zeylanicum are the main economically important
Cinnamomum species. Interestingly, they are commercially used for many medicinal purposes and in the perfume industry and can be included in various types of foodstuffs. Several reports have documented antiseptic, antiviral, antibacterial, antifungal, antidiabetic, hypocholesteremic, carminative, astringent, and blood purifier activities [
13,
14]. Recently, Zhang et al. [
15] reported that
C. cassia possesses many pharmacological activities, including antibacterial, anti-inflammatory, antitumor, anti-obesity, analgesic, antidiabetic, antiviral, cardiovascular protective, neuroprotective, cytoprotective and immunoregulatory properties. The effective role of cinnamon in the inhibition of different diseases is attributed to the presence of various chemical constituents in high concentrations. For instance, camphor is one of the significant bioactive molecules extracted from
C. camphora that has been used in pharmaceuticals, mainly liniments and insecticides and shows antidiabetic affect [
16]. Barros et al. [
17] documented that essential oils extracted from
C. zeylanicum bark, significantly enhanced the osmotic fragility curve of red blood cells (RBCs), and promoting toxic effects on RBCs membranes. Another research on
C. osmophloeum (
C.
osmophloeum) indicated that the essential oil from cinnamon leaves contains a high concentration of cinnamaldehyde. (E)-cinnamaldehyde is considered one of the main bioactive molecules of essential oil extracted from
C. zeylanicum that is responsible for its activity [
14]. Cinnamaldehyde is the major constituent of
Cinnamomum spices that possesses anaesthetic, antipyretic, antiallergic, metal ion chelating, lipo-protective, antioxidant, antibacterial and antiproliferative activity against several human cancer cell lines including leukemia, ovarian, breast (T47D) and lung (NCI-H322) cancer cell lines by inhibiting cancer progression [
18].
Cinnamomum verum is a plant commonly used in traditional medicine.
C. verum belongs to the genus
Cinnamomum, which includes about 250 species of plants distributed worldwide, especially in parts of Africa and Asia [
13].
C. verum has several medicinal properties, such as anti-inflammatory, hypoglycemic, antibacterial, antioxidant, spasmolytic, antidiarrheal, antifungal, antitumor, analgesic, gastroprotective, anticancer, and anthelmintic ones [
14,
19,
20]. Interestingly, recent studies have documented the antimalarial efficacy of
C. verum extract [
21,
22]. However, to our knowledge, no data has been reported to show the efficacy of
C. verum herbal extracts against the growth of piroplasm parasites.
C. verum has shown several medicinal properties because it contains many phytochemical ingredients, such as cinnamic acid, cinnamaldehyde, cinnamate, and numerous polyphenols [
23]. More recently, Kwan et al. [
24] identified new compounds in ethanolic
C. verum extract, such as chlorogenic acid, catechin, protocatechuic acid, icariin, aesculetin, and quercetin. Therefore, it is important to use an extraction method that can harvest the effective phytochemical molecules from the
C. verum plant. Unfortunately, extraction solvents that include methanol, ethanol, acetone, and ethyl acetate have shown variation in the amount and types of bioactive molecules harvested [
25]. This study aimed to assess the effectiveness of acetonic extract of
C. verum (AECV) and ethyl acetate extract of
C. verum (EAECV) against the growth of
B. bovis,
B. bigemina,
B. divergens,
B.
caballi and
T. equi using the in vitro fluorescence assay. Furthermore, the chemotherapeutic potential of AECV and EAECV on
B. microti in mice was investigated.
3. Discussion
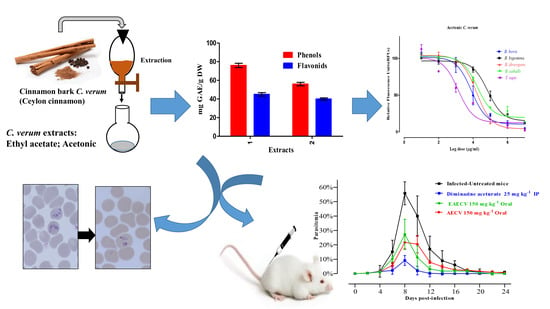
The current study documents the efficacy of various
C. verum extracts, namely, acetonic and ethyl acetate against the in vitro growth of
B. bovis,
B. bigemina,
B. divergens,
B. caballi, and
T. equi in vitro and on
B. microti in vivo. Initially, AECV and EAECV were examined for the existence of various biologically active compounds and the preliminary and qualitative screening emphasized the presence of significant amounts of polyphenols, terpenoids, alkaloid, flavonoids, and tannin. It has been shown that all these secondary metabolites have many therapeutic and antiprotozoal properties against several parasites namely
Plasmodium,
Leishmania,
Trypanosoma,
Schistosoma and
Trichomonas Vaginalis and they are known to be pharmacologically active components [
26].
The GC-MS analysis was used to identify and detect phytochemical constituents present in AECV and EAECV. The analysis revealed that AECV consisted of 17 compounds and the main chemical components detected were (
E)-cinnamaldehyde (52.87%), chromen-2-one (10.63%),
o-methoxycinnamaldehyde, (5.04%), γ-muurolene (4.92%), cadina-1(10),4-diene (4.64%) and acetic acid cinnamyl ester (4.35%), while EAECV was found to possess 26 compounds and the main chemical components identified were (
E)-cinnamaldehyde (53.81%), coumarin (9.92%), γ-muurolene (5.37%),
p-methoxycinnamaldehyde, (4.91%), acetic acid cinnamyl ester (4.83%), cadina-1(10),4-diene (4.78%) and cinnamyl alcohol (4.27%). Our results indicating that EAECV consist of slightly higher percent of (
E)-cinnamaldehyde, γ-muurolene, and acetic acid cinnamyl ester, suggesting that ethyl acetate is the better solvent of extraction. This finding is consistent with Dvorackova et al. [
25] who revealed that different extraction solvents have shown variation in the amount and types of bioactive molecules harvested.
AECV and EAECV showed varying levels of effectiveness against all tested species. For instance, AECV was most effective against
B. bovis,
B. divergens,
B. caballi and
T. equi, while EAECV was most effective against
B. bigemina. Different
C. verum extracts exhibited varying IC
50 values against
Babesia and
Theileria species, which implies that the mode of extraction is a key determinant of the effectiveness or concentration of active molecules in the final extract. Moreover, the preliminary and qualitative screening results showed that AECV contains higher total phenolic and flavonoid content than EAECV and these results explaining the highest antipiroplasmic and inhibitory effect of AECV. This finding was consistent with a previous study performed by Nkanwen et al. [
21] who tested ethyl acetate/
n-hexane
C. verum extract for anti-plasmodial efficacy and showed an inhibitory effect on the
Plasmodium falciparum enoyl-ACP reductase enzyme. More recently, Parvazi et al. [
22] showed that aqueous
C. verum extract possesses inhibitory activity against
P. falciparum in vitro, by alteration of the following metabolites:
l-aspartic acid, succinic acid, β-alanine, glutathione, and 2-methylbutyryl glycine.
Recently, several studies documented the antiprotozoal activities of different phytochemical molecules detected by our GC-MS analysis. For example, von Son-de Fernex et al. [
27] and Williams et al. [
28] documented the in vitro anthelmintic activity of 2
H-chromen-2-one and cinnamaldehyde, the main chemical component in AECV and EAECV against
Cooperia Punctata and the swine nematode
Ascaris suum, whereas Kpadonou et al. [
29] disclosed the antitrypanosomal efficacy of caryophyllene oxide against
Trypanosoma brucei brucei with IC
50 value of 17.67 µg/mL. Moreover, Bosquiroli et al. [
30] proved antileishmanial activity of caryophyllene and caryophyllene oxide against promastigotes of
L. infantum. In addition to that, Le et al. [
31] reported the anti-protozoal activity of
C. verum essential oil and its constituents (cinnamaldehyde and caryophyllene) against
Leishmania,
Plasmodium and
Trypanosoma. Therefore, we hypothesized that cinnamaldehyde, caryophyllene, 2
H-chromen-2-one, and caryophyllene oxide are the main active compounds responsible for the antipiroplasmic activity of
C. verum extracts.
The viability assay showed that AECV and EAECV were effective against
Babesia and
Theileria parasites. This result was comparable with that of Parvazi et al. [
22] who showed that aqueous
C. verum extract inhibited the growth of
P. falciparum by preventing amino acid biosynthesis through inhibiting carbon dioxide fixation and disabling the synthesis of alanine, aspartame, and glutamate. Based on this finding, we suggest that
C. verum extracts have multiple mechanisms against piroplasm parasites. Compared with previous studies on the viability of treated piroplasm parasites, EAECV suppressed the regrowth of
Babesia and
Theileria parasites at lower concentrations than the methanolic extract of
O. europaea and chalcone hydrate [
6,
9]. Furthermore, AECV suppressed the regrowth of
B. bovis and
B. divergens at lower concentrations than the methanolic extract of
S. aromaticum [
7]. Nevertheless, not all
C. verum extracts completely inhibited the regrowth of piroplasm parasites at low concentrations. For instance, EAECV is able to clear all the parasites which indicate that the ethyl acetate extraction method harvested more bioactive molecules that can eliminate
Babesia and
Theileria parasites.
Morphological observations in micrographs of
C. verum-treated parasites showed that the
C. verum extract-treated
Babesia and
Theileria parasites were not able to eject and died inside the RBCs. Additionally, the malformations observed in the piroplasm parasites might be due to the ability of
C. verum extracts to damage the parasite metabolome. The suggested mode of action for
P. falciparum was reported previously by Parvazi et al. [
22] who revealed that aqueous
C. verum extract disrupts the thioredoxin and glutathione system, resulting in a change in the parasite metabolome, inhibiting its growth.
The experiments to determine cytotoxicity showed that
C. verum extracts affected the viability of the MDBK, NIH/3T3, and HFF cell lines at a slightly high selectivity index. This means that the bioactive compounds that exist in
C. verum extracts were more likely to affect piroplasm parasites than the host cells.
C. verum extracts contain a mixture of polyphenols with cinnamaldehyde that plays a significant therapeutic role in cancer cell lines by depolarization of the mitochondrial membrane potential, leading to cellular apoptosis without cytotoxic activities on host cells [
32]. One possible explanation of the modest selectivity index of AECV and EAECV is due to the presence of alkaloids and terpenoids [
30,
33]. The aforementioned results indicating the safety of
C. verum extracts for the treatment of animals and humans following further clinical studies.
Drugs with a combination of
C. verum extracts with DA, MESA, and MEOE were assessed in vitro against
B. bovis,
B. bigemina,
B. divergens,
B. caballi, and
T. equi. The combined application of
C. verum extracts with DA, MESA, and MEOE showed synergistic and additive effects. One possible explanation is that
C. verum extracts contain many bioactive ingredients that may interact differently in combination treatments [
23].
Acetonic and ethyl acetate
C. verum extracts produced promising in vivo antibabesial activity. Oral administration of 150 mg/kg of AECV and EAECV resulted in 61.1% and 51.7% inhibition, respectively compared with 83.7% inhibition showed by 25 mg/kg DA on day 8 p.i. The growth inhibitory effects of AECV and EAECV on
B. microti were lower than the 80 % inhibition revealed by 150 mg/kg MEOE, 64% inhibition shown by acetonic
Acacia laeta extracts [
9] and 69.2% shown by MESA [
7]. However, the inhibitory efficacy of AECV on the multiplication of
B. microti was higher than the 60% revealed by 150 mg/kg methanolic
Peganum harmala seed extract, 55.1% inhibition noticed by 150 mg/kg ethanolic
Artemisia absinthium leaf extract [
34] and 42.4% inhibition shown by methanolic
Camellia sinensis extract [
7]. Furthermore, the inhibitory efficacy of EAECV on the multiplication of
B. microti was higher than 42.4% shown by methanolic
Camellia sinensis extract [
7]. The chemosuppression effect produced by AECV and EAECV on
B. microti may indicate the presence of potential compounds with higher antibabesial activity.
C. verum extracts have strong anti-inflammatory [
15] and antioxidant effects and improve reduced glutathione synthesis in the liver [
35,
36]. Such medicinal effects are useful because piroplasmosis infection is related to reactive oxygen and nitrogen species overproduction, which leads to oxidative stress [
37]. Thus, identifying the active compound is necessary for contriving a higher chemosuppression effect from these extracts for the future discovery of a novel potential drug against piroplasmosis. The limitation of this study is the use of the whole extract to confirm its antipiroplasmic efficacy, and it is recommended to assess the antipiroplasmic efficacy of the GC-MS identified compounds for the future discovery of a novel lead potential drug against piroplasmosis. As well, evaluate the actual mode of action employed against the recovery of piroplasm parasites.