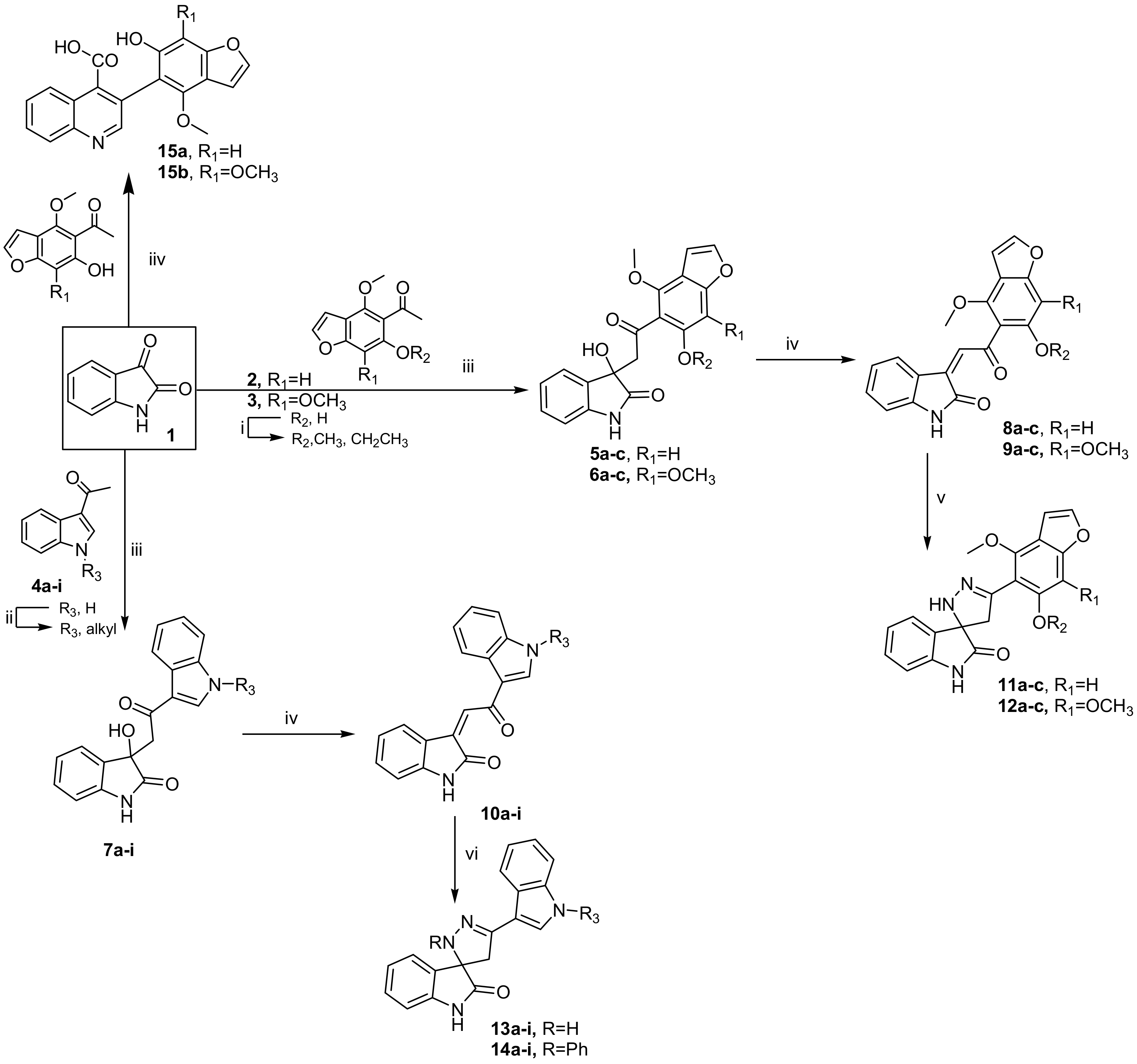
3.3. Synthesis
3.3.1. General Procedure for the Preparation of 3-hydroxy-3-(2-(aryl)-2-oxoethyl)indolin-2-ones 5a–c, 6a–c and 7a–i
Method A: To a solution of isatin (1) (10 mmol) and aryl methyl ketone 2a–c, 3a–c and 4a–i (10 mmol) in absolute ethanol (20 mL), diethylamine (10 mmol) was added. The reaction mixture was stirred at room temperature for approximately 10–15 days. The solid formed was filtered off, washed with water, air-dried and used in the next step without further purification. In the case of compounds 5a, 6a and 7a, the product was obtained only after stirring for 5h at room temperature and left overnight.
Method B: A solution of isatin (1) (10 mmol) and aryl methyl ketone 2a–c, 3a–c and 4a–i (10 mmol) in absolute ethanol (20 mL) containing diethylamine (10 mmol) was heated under reflux for ~5 h. The progress of the reaction was monitored using TLC. After the reaction accomplished, the reaction mixture was cooled, and the solid that formed was filtered off, washed with water, air-dried and used in the next step without further purification.
3.3.2. General Procedure for the Preparation of 3-(2-(aryl)-2-oxo-ethylidene)indolin-2-ones 8a–c, 9a–c and 10a–i
A solution of compound 5a–c, 6a–c and 7a–i in a mixture of glacial acetic acid (5 mL) and two drops of concentrated HCl was heated at 80 °C for 30 min. After cooling, the reaction mixture was quenched in ice-water. The solid that formed was filtered off, washed with water, air-dried and crystallized from ethyl acetate-cyclohexane.
3-(2-(6-Hydroxy-4-methoxy-benzofuran-5-yl)-2-oxo-ethylidene)indolin-2-one (8a): Yield 54%; orange powder; mp 173–5 °C; IR (KBr, cm−1): 3421 (OH), 3176 (NH), 1705, 1685 (C=O), 1545 (C=C), 1132, 1043 (C-O-C); 1H NMR (300 MHz, DMSO-d6) δ: 11.77 (s, 1H, OH), 10.27 (s, 1H, NH), 7.82 (d, 1H, H-2 furan), 7.22 (d, 1H, Ar-H), 7.20 (m, 2H, Ar-H), 6.85-6.80 (m, 2H, Ar-H), 6.72 (s, 1H, CH), 6.63 (s, 1H, CH), 4.13 (s, 3H, OCH3); 13C NMR (75 MHz, DMSO-d6) δ: 192.71, 180.53, 161.19, 156.93, 155.94, 152.03, 146.82, 143.96, 130.04, 128.72, 127.16, 112.18,110.30, 105.91, 93.85, 62.65; EI-MS: m/z (%): 335 (M+, 15); Anal Calcd for C19H13NO5 (335.31): C, 68.06; H, 3.91; N, 4.18; found: C, 67.91; H, 3.61; N, 4.01.
3-(2-(4,6-Dimethoxy-benzofuran-5-yl)-2-oxo-ethylidene)indolin-2-one (8b): Yield 33%; reddish-brown powder; mp 97–2 °C; IR (KBr, cm−1): 3220 (NH), 1702, 1695 (C=O), 1565 (C=C), 1138, 1086 (C-O-C); 1H NMR (300 MHz, DMSO-d6) δ: 10.03 (s, 1H, NH), 7.92 (d, 1H, H-2 furan), 7.56 (d, 1H, Ar-H), 7.25-6.88 (m, 6H, Ar-H), 3.83 (s, 3H, OCH3), 3.77 (s, 3H, OCH3); Anal Calcd for C20H15NO5 (349.34): C, 68.76; H, 4.33; N, 4.01; found: C, 68.55; H, 4.21; N, 3.95.
3-(2-(6-Ethoxy-4-methoxy-benzofuran-5-yl)-2-oxo-ethylidene)indolin-2-one (8c): Yield 35%; reddish-brown powder; mp 88–90 °C; IR (KBr, cm−1): 3210 (NH), 1697, 1687 (C=O), 1555 (C=C), 1135, 1051 (C-O-C); 1H NMR (300 MHz, DMSO-d6) δ: 8.19 (s, 1H, NH), 7.97 (d, 1H, H-2 furan), 7.35–7.10 (m,6H, Ar-H), 6.99 (d, 1H), 5.19 (q, 2H, CH2), 4.03 (t, 3H, CH3), 3.77 (s, 3H, OCH3); 13C NMR (75 MHz, DMSO-d6) δ: 192.07, 180.23, 160.09, 156.71, 155.11, 150.93, 144.52, 140.06, 133.21, 129.11, 127.82, 112.06, 111.13, 150.62, 105.33, 92.57, 64.32, 62.00, 25.35; EI-MS: m/z (%): 363 (M+, 29); Anal Calcd for C21H17NO5 (363.11): C, 69.41; H, 4.72; N, 3.85; found: C, 69.14; H, 4.50; N, 3.62.
3-(2-(6-Hydroxy-4,7-dimethoxy-benzofuran-5-yl)-2-oxo-ethylidene)indolin-2-one (9a): Yield 35%; purple powder; mp 138–5 °C; IR (KBr, cm−1): 3421 (OH), 3201 (NH), 1707, 1695 (C=O), 1555 (C=C), 1119, 1053 (C-O-C); 1H NMR (300 MHz, DMSO-d6) δ: 12.35 (s, 1H, OH), 10.72 (s, 1H, NH), 8.42 (d, 1H, H-2 furan), 8.07 (d, 2H, Ar-H), 7.66–7.58 (m, 3H, Ar-H), 7.16 (s, 1H, CH), 3.96 (s, 3H, OCH3), 3.79 (s, 3H, OCH3); 13C NMR (75 MHz, DMSO-d6) δ: 190.82, 179.95, 160.33, 159.01, 155.63, 142.54, 128.21, 127.42, 122.51, 121.01, 111.70, 105.91, 61.55, 61.50; EI-MS: m/z (%): 365 (M+, 45); Anal Calcd for C20H15NO6 (365.09): C, 65.75; H, 4.14; N, 3.83; found: C, 65.38; H, 3.91; N, 3.51.
3-(2-Oxo-2-(4,6,7-trimethoxy-benzofuran-5-yl)-ethylidene)indolin-2-one (9b): Yield 60%; orange powder; mp 93–5 °C; IR (KBr, cm−1): 3155 (s, 1H, NH), 1705, 1685 (C=O), 1552 (C=C), 1160, 1095 (C-O-C); 1H NMR (300 MHz, DMSO-d6) δ: 10.79 (s, 1H, NH), 8.39 (d, 1H, H-2 furan), 8.05 (d, 1H, Ar-H), 7.39 (t, 1H, Ar-H), 7.25 (d, 1H, Ar-H), 7.14 (s, 1H, CH), 7.02 (t, 1H, Ar-H), 6.90 (d, 1H, Ar-H), 4.00, 3.98, 3.81 (3s, 9H, OCH3); 13C NMR (75 MHz, DMSO-d6) δ: 190.02, 180.12, 162.04, 156.77, 155.21, 149.63, 144.60, 130.91, 129.01, 127.81, 121.71, 121.04, 110.54, 105.92, 61.00, 60.25, 60.07; EI-MS: m/z (%): 379 (M+, 42); Anal Calcd for C21H17NO6 (379.11): C, 66.49; H, 4.52; N, 3.69; found: C, 66.15; H, 4.32; N, 3.44.
3-(2-(6-Ethoxy-4,7-dimethoxy-benzofuran-5-yl)-2-oxo-ethylidene)indolin-2-one (9c): Yield 33%; orange powder; mp 118–2 °C; IR (KBr, cm−1): 3212 (s, 1H, NH), 1698, 1686 (C=O), 1566 (C=C), 1154, 1033 (C-O-C); 1H NMR (300 MHz, DMSO-d6) δ: 10.23 (s, 1H, NH), 8.12 (d, 1H, H-2 furan), 7.85 (d, 1H, Ar-H), 7.35 (t, 1H, Ar-H), 7.27–6.97 (m, 4H, Ar-H), 5.12 (q, 2H, CH2), 4.03 (t, 3H, CH3), 3.98, 3.81 (2s, 6H, OCH3); Anal Calcd for C22H19NO6 (393.39): C, 67.17; H, 4.87; N, 3.56; found: C, 67.02; H, 4.71; N, 3.44.
3-(2-(N-Ethyl-1H-indol-3-yl)-2-oxo-ethylidene)indolin-2-one (10b):Yield 92%; yellow powder; mp 197–9 °C; IR (KBr, cm−1): 3100 (NH), 1695 (C=O), 1585 (C=C); 1H NMR (300 MHz, DMSO-d6) δ: 10.90 (s, 1H, NH indolin-2-one), 8.54 (s, 1H, H-2 indole), 8.38–8.35 (m, 2H, Ar-H), 7.95 (s, 1H, CH), 7.63–7.13 (m, 6H, Ar-H), 4.37 (t, 2H, CH2), 1.54 (q, 3H, CH3); EI-MS: m/z (%): 316 (M+, 32); Anal Calcd for C20H16N2O2 (316.35): C, 75.93; H, 5.10; N, 8.86; found: C, 75.73; H, 4.92; N, 8.65.
3-(2-(N-Benzyl-1H-indol-3-yl)-2-oxo-ethylidene)indolin-2-one (10c): Yield 95 %, orange powder; mp 205-7 °C; IR (KBr, cm-1): 3162 (NH), 1692 (C=O), 1603 (C=C); 1H NMR (300 MHz, DMSO-d6) δ: 10.72 (s, 1H, NH), 8.87 (s, 1H, H-2 indole), 8.42–8.37 (m, 2H, Ar-H), 7.67 (s, 1H, CH), 7.54–6.98 (m, 11H, Ar-H), 5.65 (s, 2H, N-CH2); 13C NMR (75 MHz, DMSO-d6) δ: 189.37, 180.62, 159.35, 140.81, 138.46, 137,04, 130.98, 126.71, 123.45, 122.61, 122.12, 120.80, 120.68, 121.60, 121.32, 119.81, 42.03; EI-MS: m/z (%): 378 (M+, 42); Anal Calcd for C25H18N2O2 (378.42): C, 79.35; H, 4.79; N, 7.40; found: C, 79.14; H, 4.57; N, 7.22.
3-(2-(N-Benzoyl-1H-indol-3-yl)-2-oxo-ethylidene)indolin-2-one (10d):Yield 62%; brown powder; mp 252–4 °C; IR (KBr, cm−1): 3201 (NH), 1705, 1985 (C=O), 1576 (C=C); 1H NMR (300 MHz, DMSO-d6) δ: 10.72 (s, 1H, NH), 8.56 (s, 1H, H-2 indole), 8.42–8.35 (m, 2H, Ar-H), 7.68 (s, 1H, CH), 7.53–6.95 (m, 11H, Ar-H); EI-MS: m/z (%): 392 (M+, 12); Anal Calcd for C25H16N2O3 (392.41): C, 76.52; H, 4.11; N, 7.14; found: C, 76.35; H, 4.02; N, 6.93.
3-(2-(N-(2-Chloro-benzoyl)-1H-indol-3-yl)-2-oxo-ethylidene)indolin-2-one (10e): Yield 40%; light brown powder; mp 228–30 °C; IR (KBr, cm−1): 3167 (NH), 1707, 1665 (C=O), 1572 (C=C), 757 (C-Cl); 1H NMR (300 MHz, DMSO-d6) δ: 10.72 (s, 1H, NH indolin-2-one), 8.56 (s, 1H, H-2 indole), 8.38–8.33 (m, 2H, Ar-H), 7.67 (s, 1H, CH), 7.35–6.85 (m, 10H, Ar-H); EI-MS: m/z (%): 426/428 (M+/M+2, 5.87/3.1); Anal Calcd for C25H15ClN2O3 (426.85): C, 70.34; H, 3.54; N, 6.56; found: C, 70.01; H, 3.34; N, 6.23.
3-(2-(N-(4-Chloro-benzoyl)-1H-indol-3-yl)-2-oxo-ethylidene)indolin-2-one (10f):Yield 65%; brown powder; mp 269–71 °C; IR (KBr, cm−1): 3155 (NH), 1705, 1668 (C=O), 1555 (C=C), 752 (C-Cl); 1H NMR (300 MHz, DMSO-d6) δ: 10.72 (s, 1H, NH indolin-2-one), 8.42 (s, 1H, H-2 indole), 8.42–8.38 (m, 2H, Ar-H), 7.67 (s, 1H, CH), 7.53–6.95 (m, 10H, Ar-H); 13C NMR (75 MHz, DMSO-d6) δ: 192.50, 185.50, 178,92, 155.74, 152.03, 146.83, 143.91, 133.25, 130.04, 128.72, 127.78, 124.33, 122.98, 121.32, 112.18, 110.50; Anal Calcd for C25H15ClN2O3 (426.85): C, 70.34; H, 3.54; N, 6.56; found: C, 70.02; H, 3.21; N, 6.23.
3-(2-(N-(4-Bromo-benzenesulfonyl)-1H-indol-3-yl)-2-oxo-ethylidene)indolin-2-one (10g): Yield 50%; brown crystal; mp 267–9 °C; IR (KBr, cm−1): 3168 (NH), 1696 (C=O), 1581 (C=C), 1375, 1117 (SO2), 782 (C-Br); 1H NMR (300 MHz, DMSO-d6) δ: 10.72 (s, 1H, NH indolin-2-one), 8.56 (s, 1H, H-2 indole), 8.42–8.35 (m, 2H, Ar-H), 7.67 (s, 1H, CH), 7.53–6.86 (m, 10H, Ar-H); 13C NMR (75 MHz, DMSO-d6) δ: 188.56, 179.90, 152.74,145.72, 140.80, 138.53, 137.00, 135.42, 130.19, 127.19, 126.33, 123.92, 122.16, 120.59, 113.17, 110.40; Anal Calcd for C24H15BrN2O4S (507.36): C, 56.82; H, 2.98; N, 5.52; found: C, 56.61; H, 3.11; N, 5.73.
3-(2-(N-(4-Chloro-benzenesulfonyl)-1H-indol-3-yl)-2-oxo-ethylidene)indolin-2-one (10h): Yield 25%; brown powder; mp 276–8 °C; IR (KBr, cm−1): 3201 (NH), 1685 (C=O), 1576 (C=C), 1375, 1119 (SO2), 775 (C-Cl); 1H NMR (300 MHz, DMSO-d6) δ: 10.72 (s, 1H, NH indolin-2-one), 8.55 (s, 1H, H-2 indole), 8.39–8.35 (m, 2H, Ar-H), 7.67 (s, 1H, CH), 7.42–6.95 (m, 10H, Ar-H); EI-MS: m/z (%): 462/464 (M+/M+2, 33/11); Anal Calcd for C24H15ClN2O4S (462.90): C, 62.27; H, 3.27; N, 6.05; found: C, 62.01; H, 3.11; N, 5.86.
3-(2-(N-(2-Nitro-benzenesulfonyl)-1H-indol-3-yl)-2-oxo-ethylidene)indolin-2-one (10i): Yield 20%; reddish-brown powder; mp 189–90 °C; IR (KBr, cm-1): 3172 (NH), 1685 (C=O), 1555 (C=C), 1376, 1117 (SO2); 1H NMR (300 MHz, DMSO-d6) δ: 10.72 (s, 1H, NH indolin-2-one), 8.56 (s, 1H, H-2 indole), 8.45–8.35 (m, 2H, Ar-H), 7.67 (s, 1H, CH), 7.41–6.96 (m, 10H, Ar-H); EI-MS: m/z (%): 473 (M+, 5); Anal Calcd for C24H15N3O6S (473.46): C, 60.88; H, 3.19; N, 8.88; found: C, 60.52; H, 2.95; N, 8.53.
3.3.3. General Procedure for the Synthesis of 2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one Derivatives 11a–c, 12a–c and 13a–i
To a mixture of compound 8a–c, 9a–c and 10a–i (10 mmol) and hydrazine hydrate (50 mmol) in absolute ethanol (10 mL), a few drops of glacial acetic acid were added, and the reaction mixture was refluxed for 4–6 h. After cooling, the reaction mixture was poured onto ice-water, and the solid that formed was filtered off, washed with water, air-dried and crystallized from ethyl acetate-cyclohexane (1:1).
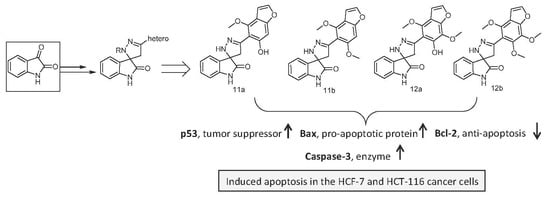
5’-(6-Hydroxy-4-methoxy-benzofuran-5-yl)-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (11a): Yield 87%; orange powder; mp 129–31 °C; IR (KBr, cm−1): 3554 (OH), 3228, 3155 (NH), 1699 (C=O), 1618 (C=N), 1550 (C=C), 1048, 1117 (C-O-C); 1H NMR (300 MHz, DMSO-d6) δ: 11.77 (s, 1H, OH), 10.99 (s, 1H, NH), 10.27 (s, 1H, NH), 7.93 (d, 1H, H-2 furan), 7.52 (d, 1H Ar-H), 7.34–7.02 (m, 3H, Ar-H), 6.97 (d, 1H, Ar-H), 6.74 (s, 1H, Ar-H), 5.95 (s, 2H, CH2), 4.14 (s, 3H, OCH3); 13C NMR (75 MHz, DMSO-d6) δ: 180.21, 165.35, 160.94, 154.62, 152.05, 145.36, 143.21, 132.32, 130.04, 128.57, 127.08, 122.06, 120.19, 105.91, 93.85, 64.50, 60.26, 45.56; EI-MS: m/z (%): 363 (M+, 13); Anal Calcd for C19H15N3O4 (349.34): C, 65.32; H, 4.33; N, 12.03; found: C, 65.17; H, 4.21; N, 11.92.
5’-(4,6-Dimethoxy-benzofuran-5-yl)-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (11b): Yield 65%; yellow powder; mp 180–2 °C; IR (KBr, cm−1): 3279, 3120 (NH), 1685 (C=O), 1617 (C=N) 1558 (C=C), 1060, 1139 (C-O-C); 1H NMR (300 MHz, DMSO-d6) δ: 11.02 (s, 1H, NH), 10.02 (s, 1H, NH), 8.01 (d, 1H, H-2 furan), 7.61 (d, 1H Ar-H), 7.51–6.66 (m, 5H, Ar-H), 5.52 (s, 2H, CH2), 4.14, 4.05 (2s, 6H, OCH3); EI-MS: m/z (%): 363 (M+, 12); Anal Calcd for C20H17N3O4 (363.37): C, 66.11; H, 4.72; N, 11.56; found: C, 66.01; H, 4.61; N, 11.44.
5’-(6-Ethoxy-4-methoxy-benzofuran-5-yl)-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (11c): Yield 51%; gray powder; mp 137–9 °C; IR (KBr, cm−1): 3222, 3124 (NH), 1693 (C=O), 1620 (C=N), 1562 (C=C), 1060, 1137 (C-O-C); 1H NMR (300 MHz, DMSO-d6) δ: 9.72 (s, 1H, NH), 8.29 (s, 1H, NH), 7.93 (d, 1H, H-2 furan), 7.62 (d, 1H Ar-H), 7.50 (t, 1H, Ar-H), 7.48 (d, 1H, Ar-H), 7.35–6.92 (m, 3H, Ar-H), 5.25 (s, 2H, CH2), 5.15 (q, 2H, OCH2), 4.03 (t, 3H, CH3), 3.77 (s, 3H, OCH3); Anal Calcd for C21H19N3O4 (377.39): C, 66.83; H, 5.07; N, 11.13; found: C, 66.65; H, 5.13; N, 11.02.
5’-(6-Hydroxy-4,7-dimethoxy-benzofuran-5-yl)-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (12a): Yield 45%; yellowish-brown powder; mp 270–2 °C; IR (KBr, cm−1): 3445 (OH), 3185, 3165 (NH), 1685 (C=O), 1618 (C=N), 1557 (C=C), 1153, 1132 (C-O-C); 1H NMR (300 MHz, DMSO-d6) δ: 10.67 (s, 1H, OH), 10.47, 10.23 (2s, 2H, 2NH), 7.95 (d, 1H, H-2 furan), 7.88 (d, 1H, Ar-H), 7.30–6.65 (m, 4H, Ar-H), 5.41 (s, 2H, CH2), 4.26, 4.19 (2s, 6H, OCH3); 13C NMR (75 MHz, DMSO-d6) δ: 184.52, 167.54, 160.19, 155.61, 145.52, 132.25, 130.05, 127.81, 127.06, 124.92, 121.25, 105.85, 63.92, 61.64, 60.25, 45.75; EI-MS: m/z (%): 379 (M+, 29); Anal Calcd for C20H17N3O5 (379.12): C, 63.32; H, 4.52; N, 11.08; found: C, 63.07; H, 4.33; N, 10.95.
5’-(4,6,7-Trimethoxy-benzofuran-5-yl)-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (12b): Yield 62%; orange powder; mp 145–7 °C; IR (KBr, cm−1): 3206, 3175 (NH), 1687 (C=O), 1618 (C=N), 1545 (C=C), 1163, 1034 (C-O-C); 1H NMR (300 MHz, DMSO-d6) δ: 12.31, 10.39 (s, 2H, 2NH), 8.40 (d, 1H, H-2 furan), 7.99 (d, 1H, Ar-H), 7.59 (t, 1H, Ar-H), 7.40–6.85 (m, 3H, Ar-H), 4.04 (s, 2H, CH2), 3.99, 3.92, 3.84 (3s, 9H, 3OCH3); Anal Calcd for C21H19N3O5 (393.39): C, 64.12; H, 4.87; N, 10.68; found: C, 64.01; H, 4.71; N, 10.53.
5’-(6-Ethoxy-4,7-dimethoxy-benzofuran-5-yl)-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (12c): Yield 42%; orange powder; mp 135–7 °C; IR (KBr, cm−1): 3210, 3165 (NH), 1685 (C=O), 1620 (C=N), 1555 (C=C), 1163, 1035 (C-O-C); 1H NMR (300 MHz, DMSO-d6) δ: 12.23, 10.20 (s, 2H, 2NH), 8.25 (d, 1H, H-2 furan), 7.78 (d, 1H, Ar-H), 7.41 (d, 1H, Ar-H), 7.25–7.13 (m, 2H, Ar-H), 6.85 (d, 1H, Ar-H), 5.50 (s, 2H, CH2), 5.02 (q, 2H, OCH2), 4.02 (t, 3H, CH3), 3.83, 3.76 (2s, 6H, OCH3); EI-MS: m/z (%):407 (M+, 22); Anal Calcd for C22H21N3O5 (407.42): C, 64.86; H, 5.20; N, 10.31; found: C, 64.71; H, 5.05; N, 10.17.
5’-(1H-Indol-3-yl)-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (13a): Yield 65%; orange powder; mp 318–20 °C; IR (KBr, cm−1): 3261, 3157 (NH), 1696 (C=O), 1618 (C=N), 1573 (C=C); 1H NMR (300 MHz, DMSO-d6) δ: 12.54, 11.87, 10.40 (3s, 3H, 3NH), 8.91 (d, 1H, Ar-H), 8.53 (s, 1H, H-2 indole), 8.39 (d, 1H, Ar-H), 8.17 (d, 1H, Ar-H), 7.57–7.12 (m, 5H, Ar-H), 5.45 (s, 2H, CH2); 13C NMR (75 MHz, DMSO-d6) δ: 179.75, 152.62, 142.66, 138.12, 127.81, 127.65, 124.62, 122.66, 121.32, 120.65, 112.05, 111.42, 62.50, 41.73; EI-MS: m/z (%): 302 (M+, 43); Anal Calcd for C18H14N4O (302.33): C, 71.51; H, 4.67; N, 18.53; found: C, 71.33; H, 4.31; N, 18.27.
5’-(N-Ethyl-1H-indol-3-yl)-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (13b): Yield 63%; orange powder; mp 143–5 °C; IR (KBr, cm-1): 3212, 3126 (NH), 1696 (C=O), 1618 (C=N), 1573 (C=C); 1H NMR (300 MHz, DMSO-d6) δ: 12.17, 10.91 (2s, 2H, 2NH), 8.93 (d, 1H, Ar-H), 8.54 (s, 1H, Ar-H), 8.38–7.13 (m, 7H, Ar-H), 4.37–4.19 (m, 4H, 2CH2), 1.54 (t, 3H, CH3); Anal Calcd for C20H18N4O (330.38): C, 72.71; H, 5.49; N, 16.96; found: C, 72.56; H, 5.32; N, 16.80.
5’-(N-Benzyl-1H-indol-3-yl)-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (13c): Yield 80%; orange powder; mp 83–5 °C; IR (KBr, cm−1): 3284, 3127 (NH), 1676 (C=O), 1618 (C=N), 1556 (C=C); 1H NMR (300 MHz, DMSO-d6) δ: 10.72, 8.92 (2s, 2H, 2NH), 8.53 (s, 1H, H-2 indole), 8.44 (m, 2H, Ar-H), 8.20 (t, 1H, Ar-H), 7.81 (d, 1H, Ar-H), 7.67 (d, 1H, Ar-H), 7.56–6.87 (m, 8H, Ar-H), 5.56, 5.50 (2s, 4H, 2CH2); 13C NMR (75 MHz, DMSO-d6) δ: 180.05, 151.75, 142.15, 138.55, 137.72, 130.01, 128.40, 127.21, 126.04, 122.72, 121.23, 120.07, 110.71, 63.00, 44.59, 41.35; EI-MS: m/z (%): 392 (M+, 43); Anal Calcd for C25H20N4O (392.45): C, 76.51; H, 5.14; N, 14.28; found: C, 76.51; H, 5.14; N, 14.28.
5’-(N-Benzoyl-1H-indol-3-yl)-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (13d): Yield 52%; brown powder; mp 307–9 °C; IR (KBr, cm−1): 3262, 3125 (NH), 1687 (C=O), 1618 (C=N), 1576 (C=C); 1H NMR (300 MHz, DMSO-d6) δ: 11.54, 10.72 (2s, 2H, 2NH), 8.55 (d, 1H, Ar-H), 8.39–8.33 (m, 2H, Ar-H), 7.53 (m, 2H, Ar-H), 7.34–6.86 (m, 9H, Ar-H), 5.05 (s, 2H, CH2); EI-MS: m/z (%): 406 (M+, 21); Anal Calcd for C25H18N4O2 (406.44): C, 73.88; H, 4.46; N, 13.78; found: C, 73.65; H, 4.32; N, 13.63.
5’-(N-(2-Chloro-benzoyl)-1H-indol-3-yl)-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (13e): Yield 45%; brown powder; mp 219–21 °C; IR (KBr, cm-1): 3212, 3156 (NH), 1707, 1687 (C=O), 1620 (C=N), 1535 (C=C), 752 (C-Cl); 1H NMR (300 MHz, DMSO-d6) δ: 11.55, 10.72 (2s, 2H, NH), 8.23 (s, 1H, H-2 indole), 8.02 (m, 2H, Ar-H), 7.85 (d, 1H, Ar-H), 7.35–6.85 (m, 9H, Ar-H), 4.67 (s, 2H, CH2); 13C NMR (75 MHz, DMSO-d6) δ: 190.00, 179.73, 151.82, 142.62, 140.16, 139.21, 138.50, 134.21, 129.44, 127.05, 122.91, 122.51, 120.16, 112.56, 110.71, 62.65, 42.08; Anal Calcd for C25H17ClN4O2 (440.88): C, 68.11; H, 3.89; N, 12.71; found: C, 68.02; H, 3.77; N, 12.63.
5’-(N-(4-Chloro-benzoyl)-1H-indol-3-yl)-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (13f): Yield 50%; off-white crystals; mp 202–4 °C; IR (KBr, cm-1): 3205, 3154 (NH), 1705, 1688 (C=O), 1618 (C=N), 1575 (C=C), 752 (C-Cl); 1H NMR (300 MHz, DMSO-d6) δ: 10.72, 9.52 (2s, 2H, 2NH), 8.32 (s, 1H, H-2 indole), 8.12 (m, 2H, Ar-H), 7.87 (m, 3H, Ar-H), 7.61–6.85 (m, 7H, Ar-H), 5.01 (s, 2H, CH2); EI-MS: m/z (%): 440/442 (M+, 21/7); Anal Calcd for C25H17ClN4O2 (440.88): C, 68.11; H, 3.89; N, 12.71; found: C, 67.94; H, 3.67; N, 12.56.
5’-(N-(4-Bromo-benzenesulfonyl)-1H-indol-3-yl)-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (13g):Yield 35%; orange powder; mp 190–2 °C; IR (KBr, cm-1): 3268 (NH), 1687 (C=O), 1620 (C=N), 1585 (C=C), 1367, 1132 (SO2), 785 (C-Br); 1H NMR (300 MHz, DMSO-d6) δ: 11.52, 10.72 (2s, 2H, 2NH), 8.31 (s, 1H, H-2 indole), 8.02–7.95 (m, 2H, Ar-H), 7.64–6.91 (m, 10H, Ar-H), 4.75 (s, 2H, CH2); 13C NMR (75 MHz, DMSO-d6) δ: 178.91, 152.05, 142.52, 140.01, 138.06, 137.27, 130.47, 129.42, 127.18, 122.06, 121.15, 120.11, 110.07, 64.00, 42.21; Anal Calcd for C24H17BrN4O3S (521.39): C, 55.29; H, 3.29; N, 10.75; found: C, 55.29; H, 3.29; N, 10.75.
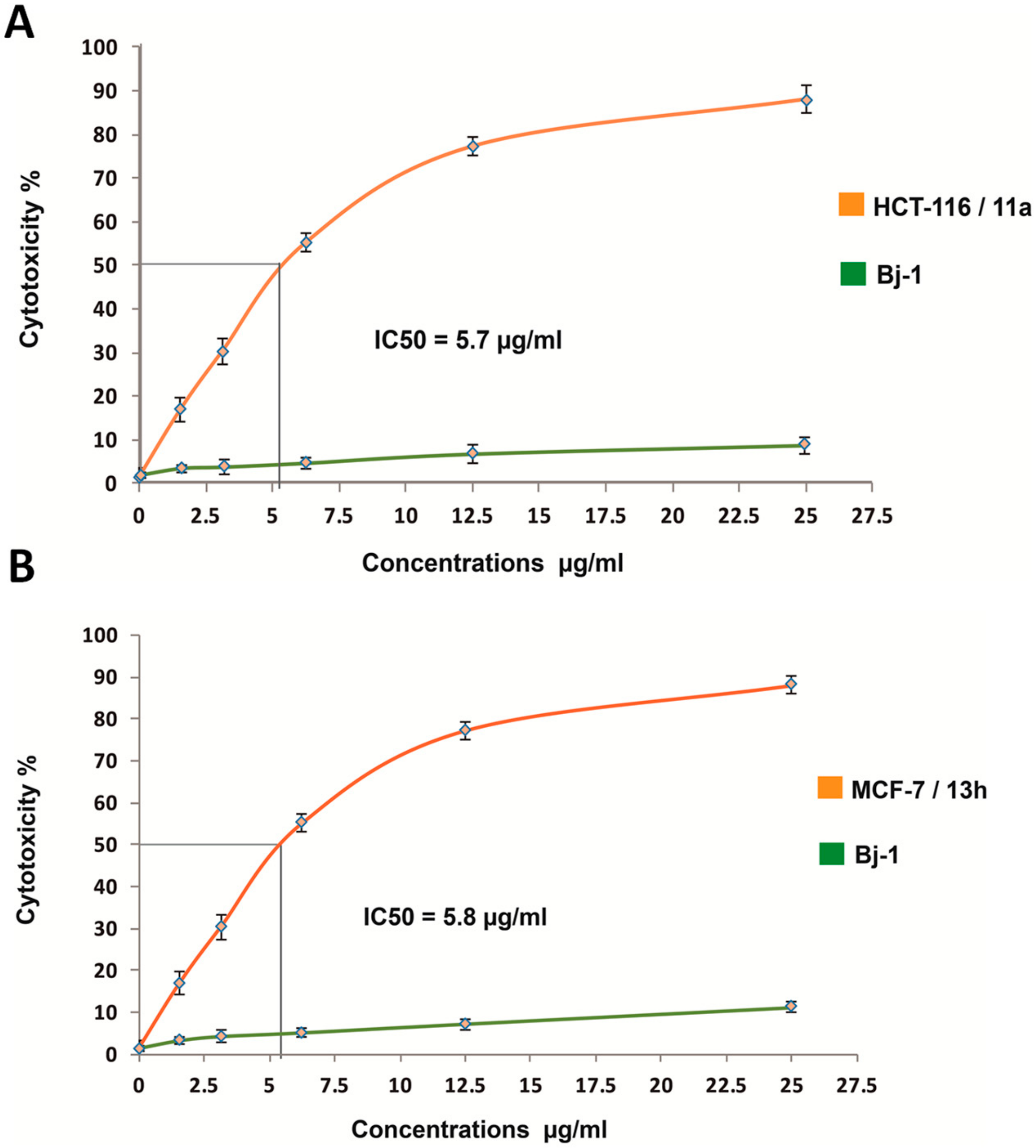
5’-(N-(4-Chloro-benzensulfonyl)-1H-indol-3-yl)-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (13h): Yield 35%; orange powder; mp 197–9 °C; IR (KBr, cm-1): 3251, 3153 (NH), 1675 (C=O), 1618 (C=N), 1565 (C=C), 1345, 1132 (SO2), 775 (C-Cl); 1H NMR (300 MHz, DMSO-d6) δ: 9.75, 10.72 (2s, 2H, 2NH), 8.45 (s, 1H, H-2 indole), 7.99–6.85 (m, 12H, Ar-H), 5.61 (s, 2H, CH2); EI-MS: m/z (%): 476/478 (M+/M+2, 27/9); Anal Calcd for. C24H17ClN4O3S (476.93): C, 60.44; H, 3.59; N, 11.75; found: C, 60.28; H, 3.41; N, 11.57.
5’-(N-(2-Nitro-benzensulfonyl)-1H-indol-3-yl)-2’,4’-dihydrospiro (indoline-3,3’-pyrazol)-2-one (13i):Yield 35%; brown powder; mp 210–2 °C; IR (KBr, cm-1): 3227, 3156 (NH), 1685 (C=O), 1618 (C=N), 1575 (C=C), 1376, 1117 (SO2); 1H NMR (300 MHz, DMSO-d6) δ: 11.51, 10.72 (2s, 2H, 2NH), 8.50 (s, 1H, H-2 indole), 8.37–7.14 (m, 12H, Ar-H), 5.32 (2s, 2H, CH2); EI-MS: m/z (%): 487(M+, 21); Anal Calcd for. C24H17N5O5S (487.49): C, 59.13; H, 3.51; N, 14.37; found: C, 59.01; H, 3.43; N, 14.26.
3.3.4. General Procedure for the Synthesis of 2’-phenyl-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-ones 14a–i
Compounds 14a–i prepared according to previously described methods for compounds 13a–i using an equimolar mixture of compounds 10a–i (10 mmol) and phenyl hydrazine (50 mmol).
5’-(1H-Indol-3-yl)-2’-phenyl-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (14a): Yield 52%; orange powder; mp 124–6 °C; IR (KBr, cm-1): 3186 (NH), 1701 (C=O), 1620 (C=N), 1565 (C=C); 1H NMR (300 MHz, DMSO-d6) δ: 11.62, 10.72 (2s, 2H, 2NH), 8.22 (d, 1H, Ar-H), 8.05 (s, 1H, H-2), 7.92 (m, 1H, Ar-H), 7.89 (d, 1H, Ar-H), 7.82 (d, 1H, Ar-H), 7.64 (m, 1H, Ar-H),7.53 (d, 1H, Ar-H), 7.46–7.12 (m, 7H, Ar-H), 5.28 (s, 2H, CH2); 13C NMR (75 MHz, DMSO-d6) δ: 197.72, 152.31, 144.05, 142.15, 139.61, 137.72, 128.46, 128.04, 127.26, 122.71, 121.36, 121.01, 110.07, 68.71, 40.95; EI-MS: m/z (%):378 (M+, 22); Anal Calcd for. C24H18N4O (378.43): C, 76.17; H, 4.79; N, 14.81; found: C, 76.02; H, 4.61; N, 14.65.
5’-(N-Ethyl-1H-indol-3-yl)-2’-phenyl-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (14b): Yield 51%; reddish-brown powder; mp 108–10 °C; IR (KBr, cm-1): 3196 (NH), 1678 (C=O), 1618 (C=N), 1556 (C=C); 1H NMR (300 MHz, DMSO-d6) δ: 10.52 (s, H, NH), 8.2 (d, 1H, Ar-H), 8.22 (s, 1H, Ar-H), 8.04–7.12 (m, 7H, Ar-H), 5.21 (s, 2H, CH2), 4.21 (q, 2H, CH2), 1.54 (t, 3H, CH3); EI-MS: m/z (%): 406 (M+, 22); Anal Calcd for C26H22N4O (406.48): C, 76.83; H, 5.46; N, 13.78; found: C, 76.71; H, 5.26; N, 13.50.
5’-(N-Benzyl-1H-indol-3-yl)-2’-phenyl-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (14c): Yield 60%; brown powder; mp 96–8 °C; IR (KBr, cm-1): 3165 (NH), 1676 (C=O), 1618 (C=N), 1558 (C=C); 1H NMR (300 MHz, DMSO-d6) δ: 10.72 (s, H, NH), 8.24 (s, 1H, H-2 indole), 8.12 (m, 2H, Ar-H), 7.98–7.02 (m, 16H, Ar-H), 5.55, 5.52 (2s, 4H, 2CH2); 13C NMR (75 MHz, DMSO-d6) δ: 179.91, 153.065–110.09, 69.11, 43.35, 41.04; EI-MS: m/z (%): 468 (M+, 12); Anal Calcd for C31H24N4O (468.55): C, 79.46; H, 5.16; N, 11.96; found: C, 79.46; H, 5.16; N, 11.96.
5’-(N-Benzoyl-1H-indol-3-yl)-2’-phenyl-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (14d): Yield 56%; brown powder; mp 100–2 °C; IR (KBr, cm-1): 3175 (NH), 1798, 1975 (C=O), 1534 (C=C); 1H NMR (300 MHz, DMSO-d6) δ: 10.45 (s, 1H, NH), 8.45 (s, 1H, H-2 indole), 8.06 (d, 1H, Ar-H), 7.98 (m, 2H, Ar-H), 7.85–7.02 (m, 15H, Ar-H), 5.50 (s, 2H, CH2); 13C NMR (75 MHz, DMSO-d6) δ: 187.60, 176.51, 151.91, 143.51, 142.24, 137.71, 136.15, 132.61, 130.01, 128.42, 127.91, 126.35, 122.36, 122.21, 121.31, 116.09, 112.14, 111.81, 68.95, 42.01; EI-MS: m/z (%): 482 (M+, 6); Anal Calcd for C31H22N4O2 (482.53): C, 77.16; H, 4.60; N, 11.61; found: C, 77.01; H, 4.52; N, 11.45.
5’-(N-(2-Chlorobenzoyl)-1H-indol-3-yl)-2’-phenyl-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (14e): Yield 40%; reddish-brown powder; mp 78–80 °C; IR (KBr, cm-1): 3176 (NH), 1710, 1695 (C=O), 1620 (C=N), 1555 (C=C), 755 (C-Cl); 1H NMR (300 MHz, DMSO-d6) δ: 10.72 (s, 1H, NH), 8.06 (m, 2H, Ar-H), 7.92 (d, 1H, Ar-H), 7.81–6.92 (m, 15H, Ar-H), 5.21 (s, 2H, CH2); EI-MS: m/z (%): 516/518 (M+/M++2, 13/6); Anal Calcd for C31H21ClN4O2 (516.98): C, 72.02; H, 4.09; N, 10.84; found: C, 72.11; H, 4.19; N, 10.73.
5’-(N-(4-Chlorobenzoyl)-1H-indol-3-yl)-2’-phenyl-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (14f): Yield 52%; brown powder; mp 162–4 °C; IR (KBr, cm-1): 3210 (NH), 1797, 1685 (C=O), 1618 (C=N), 1575 (C=C), 757 (C-Cl); 1H NMR (300 MHz, DMSO-d6) δ: 10.72 (s, 1H, NH), 8.55 (1H, d, Ar-H), 8.21 (s, 1H, H-2 indole), 8.01 (m, 2H, Ar-H), 7.92–6.75 (m, 14H, Ar-H), 5.56 (s, 2H, CH2); EI-MS: m/z (%): 516/518 (M+/M++2, 11/4); Anal Calcd for C31H21ClN4O2 (516.98): C, 72.02; H, 4.09; N, 10.84; found: C, 71.93; H, 3.98; N, 10.71.
5’-(N-(4-Bromo-benzenesulfonyl)-1H-indol-3-yl)-2’-phenyl-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (14g): Yield 61%; reddish-brown powder; mp 132–4 °C; IR (KBr, cm-1): 3186 (NH), 1692 (C=O), 1618 (C=N), 1555 (C=C), 1353, 1123 (SO2), 787 (C-Br); 1H NMR (300 MHz, DMSO-d6) δ: 10.54 (s, 1H, NH), 8.78 (d, 1H, Ar-H), 8.22 (s, 1H, H-2 indole), 8.14 (m, 2H, Ar-H), 7.88 (d, 1H, Ar-H), 7.77–6.87 (m, 13H, Ar-H), 5.25 (s, 2H, CH2); 13C NMR (75 MHz, DMSO-d6) δ: 180.05, 152.72, 144.35, 142.20, 139.51, 138.27, 136.06, 130.25, 128.26, 126.05, 122.31, 121.16, 112.53, 110.04, 112.55, 68.62, 41.16; Anal Calcd for C30H21BrN4O3S (597.48): C, 60.31; H, 3.54; N, 9.38; found: C, 60.24; H, 3.41; N, 9.22.
5’-(N-(4-Chloro-benzenesulfonyl)-1H-indol-3-yl)-2’-phenyl-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (14h): Yield 54%; orange powder; mp 110–2 °C; IR (KBr, cm-1): 3175 (NH), 1687 (C=O), 1618 (C=N), 1575 (C=C), 1355, 1132 (SO2), 775 (C-Cl); 1H NMR (300 MHz, DMSO-d6) δ: 10.72 (s, 1H, NH), 8.75 (d, 1H, Ar-H), 8.35 (s, 1H, H-2 indole), 8.11 (m, 2H, Ar-H), 7.98 (d, 1H, Ar-H), 7.87–6.75 (m, 13H, Ar-H), 5.54 (s, 2H, CH2); EI-MS: m/z (%): 553/555 (M+/M+2, 13/5); Anal Calcd for C30H21ClN4O3S (553.03): C, 65.15; H, 3.83; N, 10.13; found: C, 65.01; H, 3.72; N, 10.02.
5’-(N-(2-Nitro-benzenesulfonyl)-1H-indol-3-yl)-2’-phenyl-2’,4’-dihydrospiro(indoline-3,3’-pyrazol)-2-one (14i): Yield 41%; reddish-brown powder; mp 152–4 °C; IR (KBr, cm-1): 3177 (NH), 1695 (C=O), 1620 (C=N), 1585 (C=C), 1353, 1127 (SO2); 1H NMR (300 MHz, DMSO-d6) δ: 10.72 (s, 1H, NH), 8.57 (d, 1H, Ar-H), 8.15 (s, 1H, H-2 indole), 7.92 (m, 2H, Ar-H), 7.87 (d, 1H, Ar-H), 7.82 (m, 2H, Ar-H), 7.53–7.05 (m, 11H, Ar-H), 5.65 (2s, 2H, CH2); EI-MS: m/z (%): 563 (M+,24); Anal Calcd for C30H21N5O5S (563.58): C, 63.93; H, 3.76; N, 12.43; found: C, 63.75; H, 3.61; N, 12.35.
3.3.5. General Procedure for Synthesis of Quinoline-4-carboxylic acids 15a, b
To a solution of isatin (1) (10 mmol) in ethanol (10 mL), a solution of potassium hydroxide (10 mL, 33%) was added, and the mixture was heated under reflux for 10 min. After cooling, the reaction mixture was acidified to pH 2–3 using 0.38 mL of concentrated hydrochloric acid. To the reaction mixture, compound 2a and 3a (10 mmol) were added and then heated under reflux for 6–12 h. After cooling, the reaction mixture was poured into ice-water, and the solid formed was filtered off, washed with water, air-dried and crystallized from ethanol.
3-(6-Hydroxy-4-methoxybenzofuran-5-yl)quinoline-4-carboxylic acid (15a): Yield 70%; pale yellow powder; mp 217–9 °C; IR (KBr, cm-1): 3450 (OH), 1710 (C=O), 1595 (C=C), 1135, 1029 (C-O-C); 1H NMR (300 MHz, DMSO-d6) δ: 11.85 (s, 2H, 2OH), 8.47 (d, 1H, Ar-H), 8.30 (s, 1H, Ar-H), 8.13 (d, 1H, Ar-H), 7.82 (m, 3H, Ar-H), 7.17 (t, 1H, Ar-H), 6.91 (s, 1H, Ar-H), 4.51 (s, 3H, OCH3); 13C NMR (75 MHz, DMSO-d6) δ: 203.01, 167.53, 156.93, 155.62, 146.83, 143.52, 136.14, 128.61, 127.78, 125.38, 122.98, 122.09, 112.18, 110.33, 105.35, 93.35, 62.00; EI-MS: m/z (%):335 (M+, 33); Anal Calcd for C19H13NO5 (335.31): C, 68.06; H, 3.91; N, 4.18; found: C, 67.91; H, 3.84; N, 4.07.
3-(6-Hydroxy-4,6-dimethoxybenzofuran-5-yl)quinoline-4-carboxylic acid (15b): Yield 53%; yellow powder; mp 275–7 °C; IR (KBr, cm-1): 3510 (OH), 1709 (C=O), 1587 (C=C), 1155, 1031 (C-O-C); 1H NMR (300 MHz, DMSO-d6) δ: 12.31 (s, 2H, 2OH), 8.42 (d, 1H, Ar-H), 8.07 (d, 1H, Ar-H), 7.91 (s, 1H, Ar-H), 7.66–7.58 (m, 2H, Ar-H), 7.16 (s, 2H, Ar-H), 3.96, 3.79 (2s, 6H, 2OCH3); 13C NMR (75 MHz, DMSO-d6) δ: 200.93, 161.33, 152.70, 147.23, 143.61, 138.51, 132.71, 128.35, 126.90, 124.44, 123.66, 105.91, 61.50, 59.25; EI-MS: m/z (%):365 (M+, 33); Anal Calcd for C20H15NO6 (365.34): C, 65.75; H, 4.14; N, 3.83; found: C, 65.61; H, 4.03; N, 3.65.