Studying and Modeling of the Extraction Properties of the Natural Deep Eutectic Solvent and Sorbitol-Based Solvents in Regard to Biologically Active Substances from Glycyrrhizae Roots
Abstract
:1. Introduction
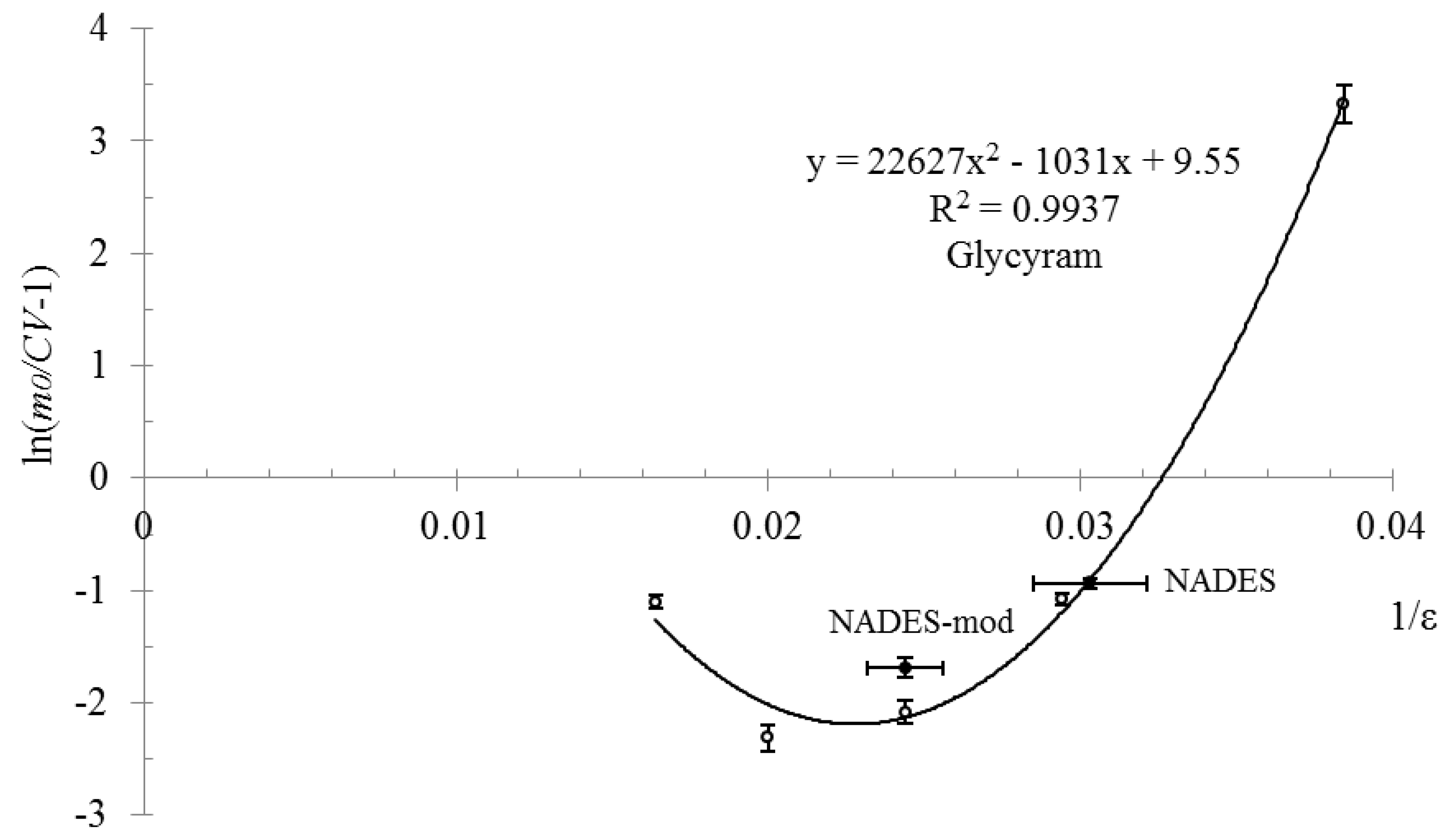
2. Results and Discussion
3. Materials and Methods
3.1. Reagents and Solvents
3.2. Methods of Analysis
3.2.1. Reverse Phase High Performance Liquid Chromatography Method (RP HPLC)
3.2.2. A Method for Obtaining NADES and Sorbitol-Based Solvents
3.2.3. A method for Extract Preparation
3.2.4. A Method for Thermal Analysis
3.2.5. A Method for Dielectric Constant Analysis
3.3. Theory
- n, n0 are quantity of BAS molecules in the liquid phase and in the extraction system in general, moles;
- C is BAS concentration in the solvent, g/mL;
- V is solvent volume, mL;
- m0 is BAS mass in the extraction system, g;
- Δμ is the change of BAS molecules’ chemical potential at the transition from plant raw material to the solvent, J/mole;
- R is gas constant, 8.314 J/(K·mol);
- T is absolute temperature, K;
- a is constant.
- b, d, f are empirical constants;
- ε is a dielectric constant of the solvent.
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Marcus, Y. Deep Eutectic Solvents; Springer Nature: Cham, Switzerland, 2019. [Google Scholar]
- Nagarajan, J.; Heng, W.W.; Galanakis, C.M.; Ramanan, R.N.; Raghunandan, M.E.; Sun, J.; Ismail, A.; Beng-Ti, T.; Prasad, K.N. Extraction of phytochemicals using hydrotropic solvents. Sep. Sci. Technol. 2016, 51, 1151–1165. [Google Scholar] [CrossRef]
- Bouillot, B.; Teychené, S.; Biscans, B. An evaluation of thermodynamic models for the prediction of drug and drug-like molecule solubility in organic solvents. Fluid Phase Equilibria 2011, 309, 36–52. [Google Scholar] [CrossRef] [Green Version]
- Rahman, N.R.A.; Yunus, N.A.; Mustaffa, A.A. Selection of optimum ionic liquid solvents for flavonoid and phenolic acids extraction. In Proceedings of the IOP Conference Series: Materials Science and Engineering, Miri, Malaysia, 1–3 December 2016; pp. 1–12. [Google Scholar] [CrossRef]
- Alhadid, A.; Mokrushina, L.; Minceva, M. Modeling of solid–liquid equilibria in deep eutectic solvents: A parameter study. Molecules 2019, 24, 2334. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, B.; Row, K. Recent developments in deep eutectic solvents in chemical sciences. Mon. Chem. 2013, 144, 1427–1454. [Google Scholar] [CrossRef]
- Hiemenz, P.C.; Rajagopalan, R. Principles of Colloid and Surface Chemistry, 3rd ed.; Marcel Dekker, Inc.: New York, NY, USA, 1997. [Google Scholar]
- Boyko, N.N.; Makarevich, N.A.; Pisarev, D.I.; Zhilyakova, E.T. Simplified mathematical modeling of the distribution process of licuroside and glycyram between the extractant and Glycyrrhizae radices. Res. Result. Med. Pharm. 2018, 4, 75–80. (In Russian) [Google Scholar] [CrossRef] [Green Version]
- Boyko, N.N.; Pisarev, D.I.; Zhilyakova, E.T.; Novikov, O.O. The study of rutin and chlorogenic acid distribution process in the extraction system from the flowers of Calendula officinalis and the extractant. Vestn. Farm. 2018, 81, 52–58. (In Russian) [Google Scholar]
- Boyko, N.; Pisarev, D.; Zhilyakova, E.; Pravlotskaya, A.; Novikov, O.; Makarevich, N.; Kuznietsova, V.; Sushchuk, N. Study and modeling of the distribution process of some phenolic compounds between the solid and liquid phases. J. Adv. Pharm. Technol. Res. 2019, 10, 56–62. [Google Scholar] [CrossRef]
- Boyko, N.N.; Pisarev, D.I.; Zhilyakova, E.T.; Malyutina, A.Y.; Novikov, O.O.; Osolodchenko, T.P.; Ghazaryan, M.S. Studies relating to the development of a galenical drug with antimicrobial activity from clove buds. Probl. Biol. Med Pharm. Chem. 2019, 22, 47–52. (In Russian) [Google Scholar] [CrossRef]
- Boyko, N.N.; Zhilyakova, E.T.; Pisarev, D.I.; Malyutina, A.Y.; Novikov, O.O.; Osolodchenko, T.P. Comprehensive studies for development of galenicals with antimicrobial activity from Hypericum perforatum L. herb. Russ. J. Biopharm. 2019, 11, 89–96. (In Russian) [Google Scholar]
- Boyko, N.N.; Zhilyakova, E.T.; Malyutina, A.Y.; Naplekov, D.K.; Shestopalova, N.N.; Martceva, D.S.; Novikov, O.O.; Pisarev, D.I.; Mizina, P.G. Study of distribution of biologically active substances from flowers of Helichrysum arenarium between phases of the extraction system. Pharm. Pharmacol. 2019, 7, 271–278. (In Russian) [Google Scholar] [CrossRef]
- Boyko, N.N.; Pisarev, D.I.; Zhilyakova, E.T.; Novikov, O.O. Study and modeling of solvent influence on isosalipurposide extraction from Helichrysi arenarii flowers. Pharm. Pharmacol. 2018, 6, 340–350. (In Russian) [Google Scholar] [CrossRef]
- Boyko, N.N.; Pisarev, D.I.; Zhilyakova, E.T.; Novikov, O.O.; Tsvetkova, Z.E.; Kuznietsova, V.Y.; Sushchuk, N.A. The role of solvent dielectric constant in modeling of the extraction process of phenolic compounds from Silybum marianum L. fruits. J. Pharm. Res. 2018, 12, 440–445. [Google Scholar]
- Boyko, N.; Pisarev, D.; Zhilyakova, E.; Novikov, O.; Kuznietsova, V.; Sushchuk, N. Modeling of solvent effects on phytocompounds’ extraction from Glycyrrhizae radix. J. Adv. Pharm. Technol. Res. 2018, 9, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Wohlfarth, C. Static Dielectric Constants of Pure Liquids and Binary Liquid Mixtures; Springer: Berlin/Heidelberg, Germany, 2015. [Google Scholar]
- Sorby, D.L.; Liu, G.; Horowitz, K.N. Dielectric constants of complex pharmaceutical solvent systems II. Water-ethanol-sucrose and water-ethanol-sorbitol. J. Pharm. Sci. 1965, 54, 1811–1813. [Google Scholar] [CrossRef]
- Ali, M.C.; Chen, J.; Zhang, H.; Li, Z.; Zhao, L.; Qiu, H. Effective extraction of flavonoids from Lycium barbarum L. fruits by deep eutectic solvents-based ultrasound-assisted extraction. Talanta 2019, 203, 16–22. [Google Scholar] [CrossRef]
- Xiong, Z.; Wang, M.; Guo, H.; Xu, J.; Ye, J.; Zhao, J.; Zhao, L. Ultrasound-assisted deep eutectic solvent as green and efficient media for the extraction of flavonoids from Radix scutellariae. New J. Chem 2019, 43, 644–650. [Google Scholar] [CrossRef]
- Jiang, Z.-M.; Wang, L.-J.; Gao, Z.; Zhuang, B.; Yin, Q.; Liu, E.-H. Green and efficient extraction of different types of bioactive alkaloids using deep eutectic solvents. Microchem. J. 2019, 145, 345–353. [Google Scholar] [CrossRef]
- Dai, Y.; Rozema, E.; Verpoorte, R.; Choi, Y.H. Application of natural deep eutectic solvents to the extraction of anthocyanins from Catharanthus roseus with high extractability and stability replacing conventional organic solvents. J. Chromatogr. A. 2016, 1434, 50–56. [Google Scholar] [CrossRef]
- González, C.G.; Mustafa, N.R.; Wilson, E.G.; Verpoorte, R.; Choi, Y.H. Application of natural deep eutectic solvents for the “green” extraction of vanillin from vanilla pods. Flavour Fragr. J. 2017, 33, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Ribeiro, B.D.; Coelho, M.A.Z.; Marrucho, I.M. Extraction of saponins from sisal (Agave sisalana) and juá (Ziziphusjoazeiro) with cholinium-based ionic liquids and deep eutectic solvents. Eur. Food Res. Technol. 2013, 237, 965–975. [Google Scholar] [CrossRef]
- Espino, M.; Fernández, M.; Gomez, F.; Silva, M. Natural designer solvents for greening analytical chemistry. Trac Trends Anal. Chem. 2015, 76, 126–136. [Google Scholar] [CrossRef]
- Reregister of Medicines of Russia. Available online: https://www.rlsnet.ru/tn_index_id_5789.htm (accessed on 23 December 2019). (In Russian).
- Electronic Medicines Compendium UK. Natures aid Herbal Mucus Cough Syrup. Available online: https://www.medicines.org.uk/emc/product/7984/smpc#companyDetails (accessed on 23 December 2019).
- Gómez, A.V.; Biswas, A.; Tadini, C.C.; Furtado, R.F.; Alves, C.R.; Cheng, H.N. Use of Natural Deep Eutectic Solvents for Polymerization and Polymer Reactions. J. Braz. Chem. Soc. 2019, 30, 717–726. [Google Scholar] [CrossRef]
- Dai, Y.; van Spronsen, J.; Witkamp, G.-J.; Verpoorte, R.; Choi, Y.H. Natural deep eutectic solvents as new potential media for green technology. Anal. Chim. Acta 2013, 766, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Baraz, V.R.; Pegashkin, V.F. Using the MS Excel for the Statistical Analysis of Data; NTI (filial) UrFU: Nizhny Tagil, Russia, 2014. (In Russian) [Google Scholar]
- Zhilyakova, E.T.; Novikov, O.O.; Pisarev, D.I.; Malyutina, A.Y.; Boyko, N.N. Studying the polyphenolic structure of Laurus Nobilis L. leaves. Indoam J. Pharmsci. 2017, 4, 3066–3074. [Google Scholar]
- State Pharmacopoeia of the Russian Federation, XIVth ed.; Ministry of Healthcare of Russian Federation: Moscow, Russia, 2018; Volume I. (In Russian)
Sample Availability: Samples of the compounds are not available from the authors. |

| Compound | Mole Ratio | Tm, °C | Tg, °C |
|---|---|---|---|
| 1. Sorbitol | - | 97.3 ± 0.5 | - |
| 2. Malic acid | - | 130.1 ± 0.5 | - |
| 3. Sorbitol-based NADES (sorbitol:malic acid:water) | 1:1:3 | - | −55.9 ± 1.5 |
| Parameter | Pharmacopoeia Limit [32] | Licuroside * | Glycyram * |
|---|---|---|---|
| 1. Retention time, min | - | 24.2 ± 0.2 | 37.3 ± 0.3 and 37.9 ± 0.3 |
| 2. Asymmetry parameter | 0.8–1.5 | 0.82 | 0.84 |
| 3. Resolution between the peaks | ≥1.5 | 2.5 | 1.7 and 1.5 |
| 4. Relative standard deviation, RSD, % | ≤2.0 | 1.6 | 1.9 and 1.9 |
| 5. LOD, g/mL | - | 2.0·10−5 | 8.9·10−5 |
| 6. LOQ, g/mL | - | 6.1·10−5 | 2.7·10−4 |
| 7. Determination coefficient, r2 | ≥0.98 | 0.9999 | 0.9997 |
| 8. Linear regression equation, C(g/mL) = f(S(mAU·s)) | - | C = (3.36 ± 0.04)·10−7·S | C = (1.77 ± 0.06)·10−6·S |
| No. | Content, % wt. * | Density, g/mL * | Dielectric Constant (ε) | ||||
|---|---|---|---|---|---|---|---|
| Sorbitol | Ethanol | Water | Malic Acid | Glycerin | |||
| 1 | 2.00 ± 0.01 | 91.0 ± 0.5 | 7.00 ± 0.04 | 0 | 0 | 0.815 ± 0.006 | 26 |
| 2 | 14.0 ± 0.1 | 66.0 ± 0.3 | 20.0 ± 0.1 | 0 | 0 | 0.900 ± 0.006 | 34 |
| 3 | 34.0 ± 0.2 | 42.0 ± 0.2 | 24.0 ± 0.1 | 0 | 0 | 1.018 ± 0.006 | 41 |
| 4 | 51.0 ± 0.3 | 22.0 ± 0.1 | 27.0 ± 0.1 | 0 | 0 | 1.130 ± 0.006 | 50 |
| 5 | 72.0 ± 0.4 | 0 | 28.0 ± 0.1 | 0 | 0 | 1.298 ± 0.006 | 61 |
| 6 ** | 49.0 ± 0.3 | 0 | 15.0 ± 0.1 | 36.0 ± 0.2 | 0 | 1.404 ± 0.006 | 33 ± 2 *** |
| 7 ** | 42.7 ± 0.3 | 0 | 4.20 ± 0.03 | 31.5 ± 0.2 | 21.6 ± 0.1 | 1.381 ± 0.006 | 41 ± 2 *** |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boyko, N.; Zhilyakova, E.; Malyutina, A.; Novikov, O.; Pisarev, D.; Abramovich, R.; Potanina, O.; Lazar, S.; Mizina, P.; Sahaidak-Nikitiuk, R. Studying and Modeling of the Extraction Properties of the Natural Deep Eutectic Solvent and Sorbitol-Based Solvents in Regard to Biologically Active Substances from Glycyrrhizae Roots. Molecules 2020, 25, 1482. https://doi.org/10.3390/molecules25071482
Boyko N, Zhilyakova E, Malyutina A, Novikov O, Pisarev D, Abramovich R, Potanina O, Lazar S, Mizina P, Sahaidak-Nikitiuk R. Studying and Modeling of the Extraction Properties of the Natural Deep Eutectic Solvent and Sorbitol-Based Solvents in Regard to Biologically Active Substances from Glycyrrhizae Roots. Molecules. 2020; 25(7):1482. https://doi.org/10.3390/molecules25071482
Chicago/Turabian StyleBoyko, Nikolay, Elena Zhilyakova, Anastasiya Malyutina, Oleg Novikov, Dmitriy Pisarev, Rimma Abramovich, Olga Potanina, Simon Lazar, Praskovia Mizina, and Rita Sahaidak-Nikitiuk. 2020. "Studying and Modeling of the Extraction Properties of the Natural Deep Eutectic Solvent and Sorbitol-Based Solvents in Regard to Biologically Active Substances from Glycyrrhizae Roots" Molecules 25, no. 7: 1482. https://doi.org/10.3390/molecules25071482





