Reactivation Process of Activated Carbons: Effect on the Mechanical and Adsorptive Properties
Abstract
1. Introduction
2. Results and Discussion
2.1. Characterisation of Virgin Carbon and SAC
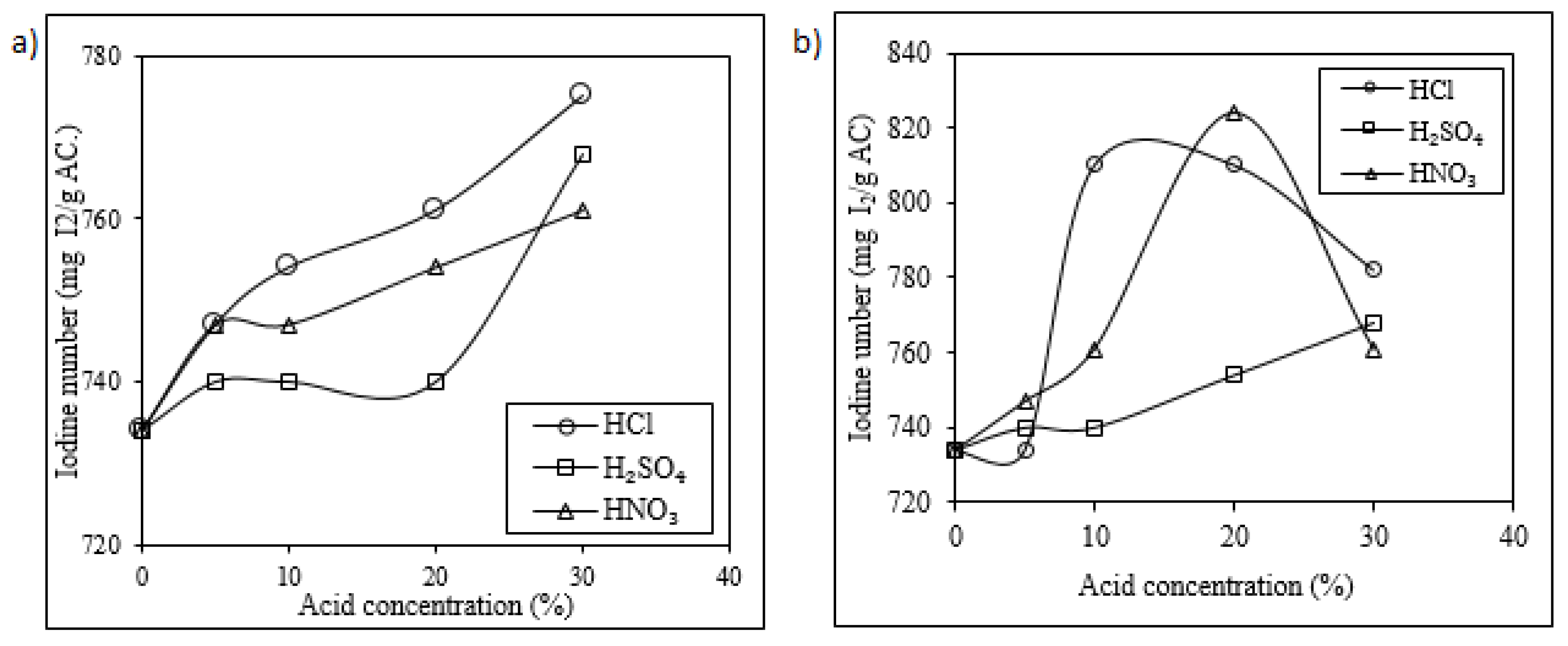
2.2. Acid Wash (Chemical Reactivation)
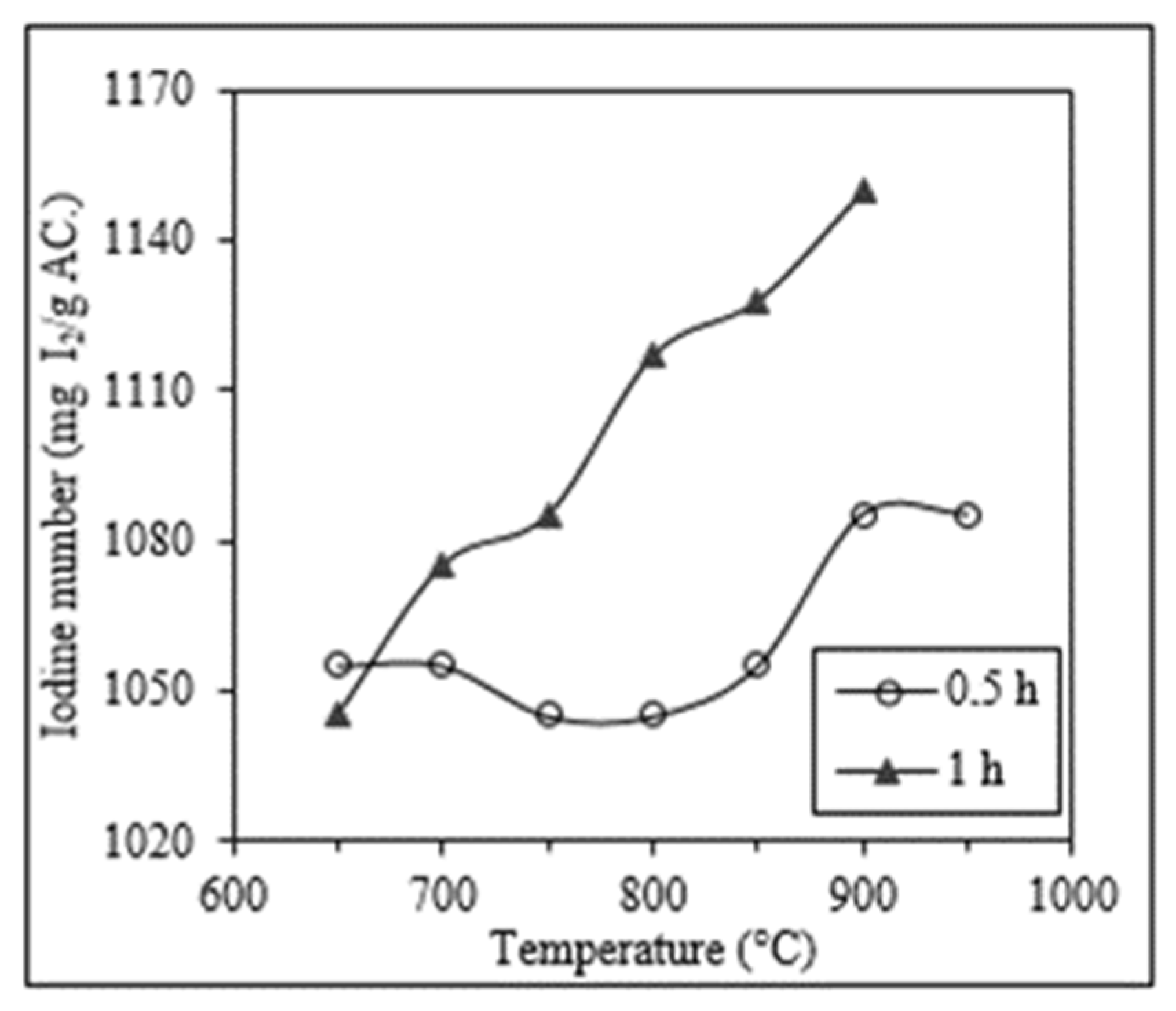
2.3. Thermal Reactivation
2.4. Specific Surface Area
2.5. Reactivation Sequence
2.6. Ball-Pan Hardness and Normal Abrasion in Pulp Resistance
2.7. Gold Adsorption Isotherms
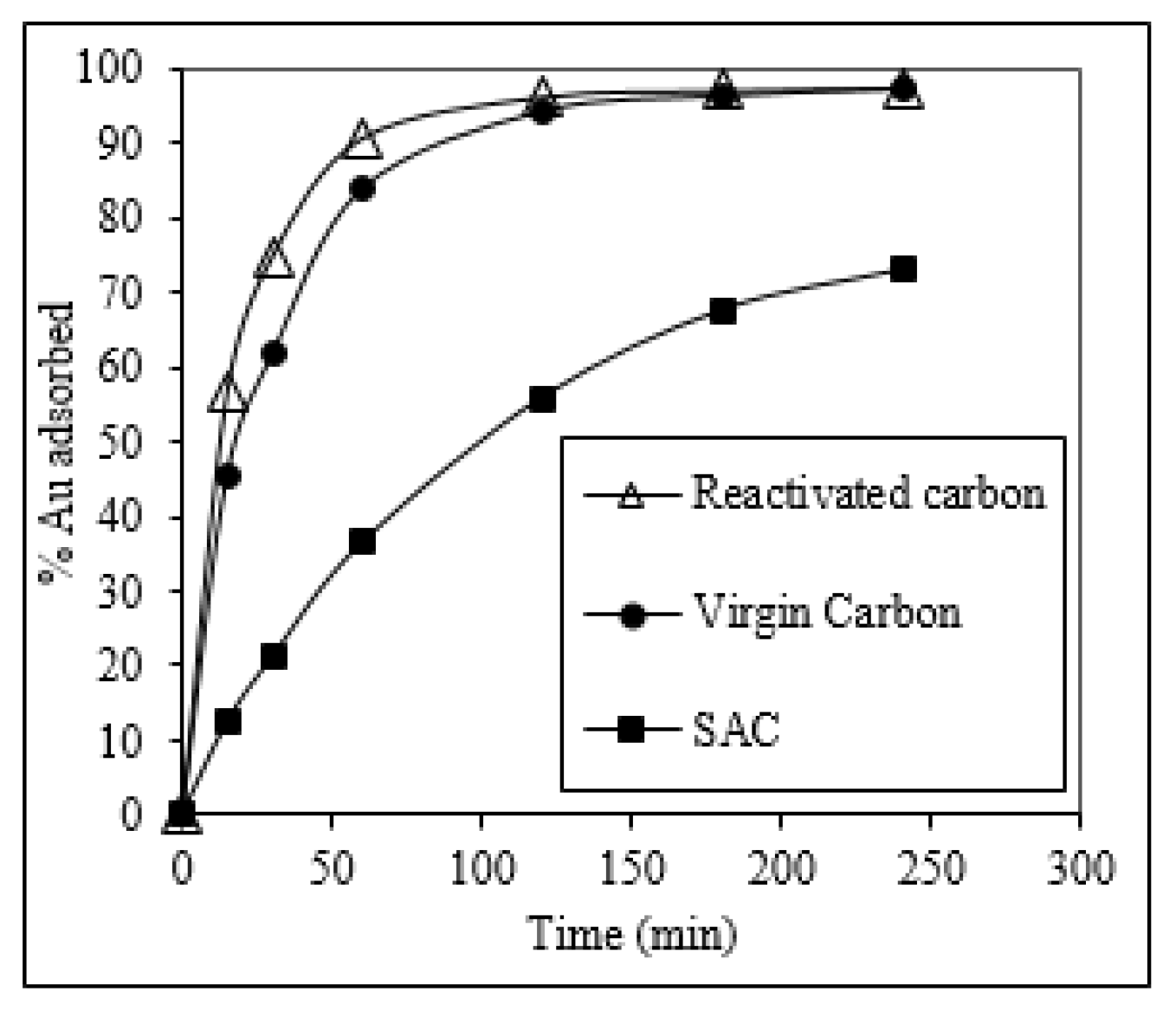
2.8. Gold Adsorption Kinetics in CIP Process
2.9. Comparison of the Properties of Virgin Carbon, SAC, and Reactivated Carbon
3. Materials and Methods
3.1. Acid Wash (Chemical Reactivation) and Thermal Reactivation
3.2. Characterization of Carbon Without Reactivation (CIP), Virgin Carbon, and Reactivated Carbon
3.3. Normal Abrasion in Pulp Resistance
3.4. Gold Adsorption Isotherms
3.5. Gold Adsorption Kinetics
3.6. CIP Process
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
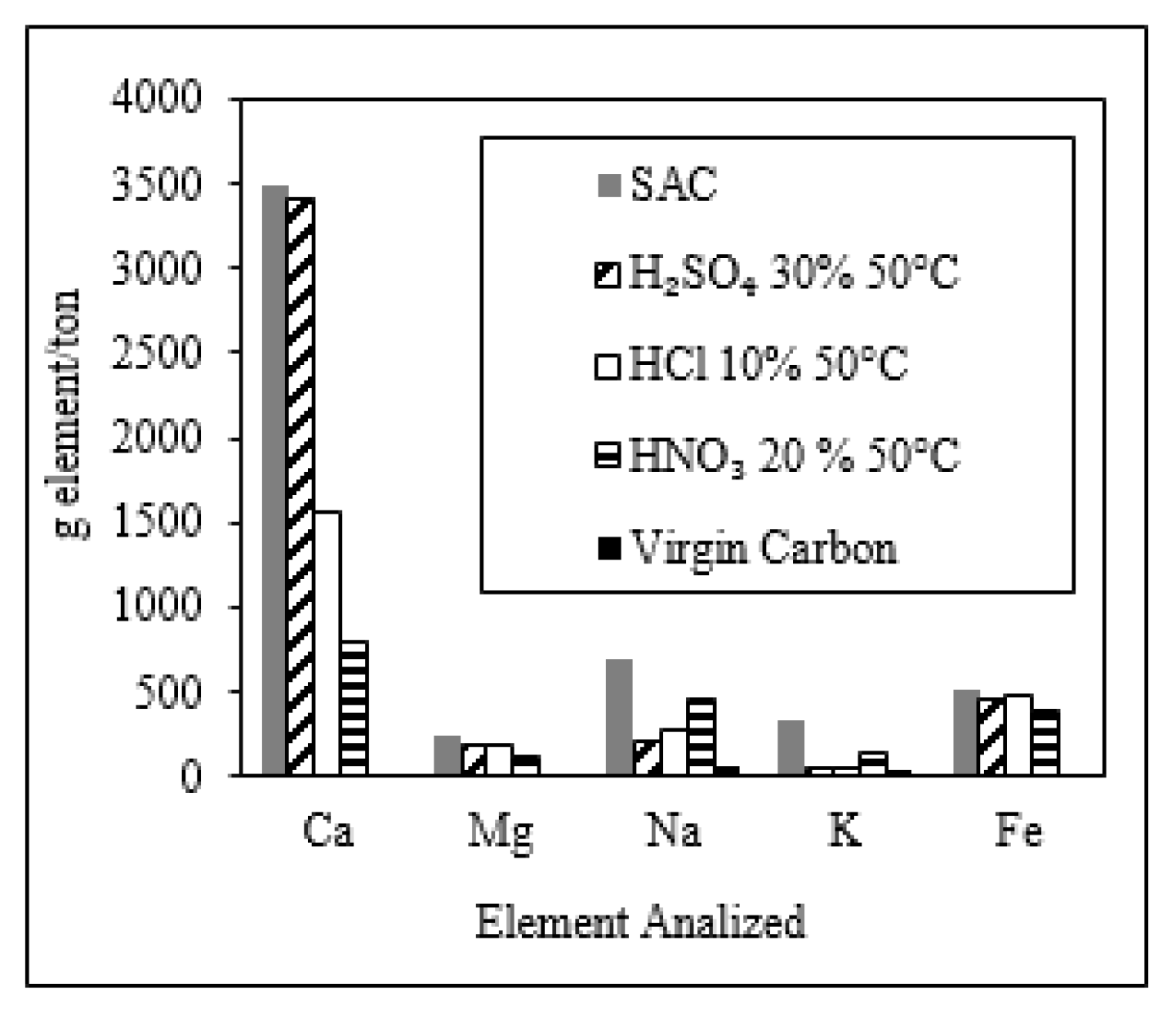
Elemental Analysis of Virgin Carbon and SAC
| Activated Carbon | C | H | N | O |
|---|---|---|---|---|
| (%) | ||||
| Virgin | 81.8 | 3.1 | 0.3 | 5.8 |
| SAC | 82.0 | 3.7 | 0.4 | 6.1 |
Appendix B
Chemical Wash of SAC Using H2SO4 at 18 °C and 50 °C
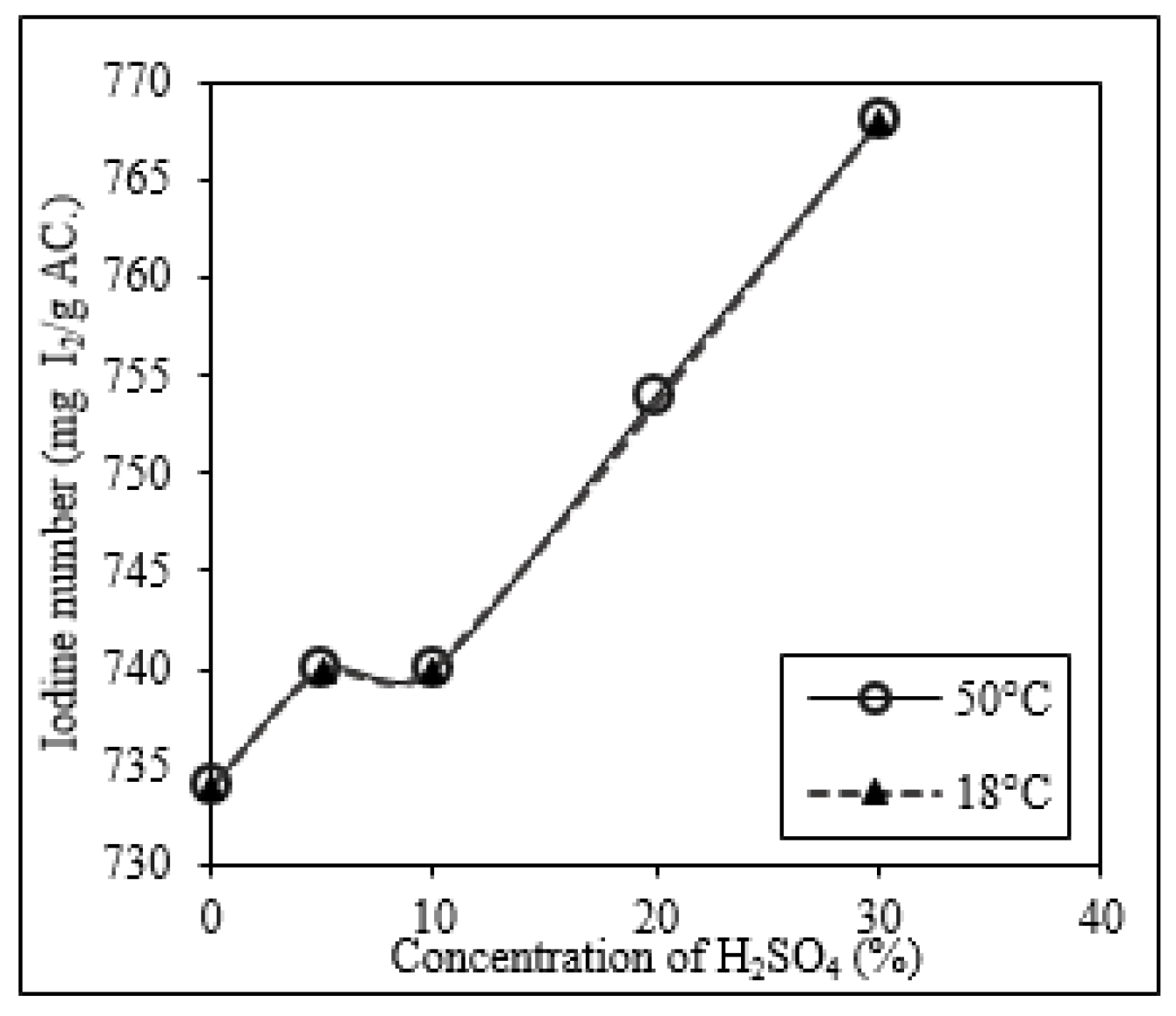
Appendix C
Characterization of the Auriferous Ore Used in the Normal Abrasion in the Pulp Resistance Test
| Mineral | Content (%) |
|---|---|
| Quartz | 86.0 |
| Pyrite | - |
| Galena | - |
| Clinochlore | 7.2 |
| Butlerite | 2.1 |
| Biotite | 1.0 |
| Chalcopyrite | - |
| Albite | 3.7 |
Appendix D
Main Results of the BET Analysis of Virgin Carbon and SAC


Appendix E
FTIR Analysis of Virgin Carbon and SAC
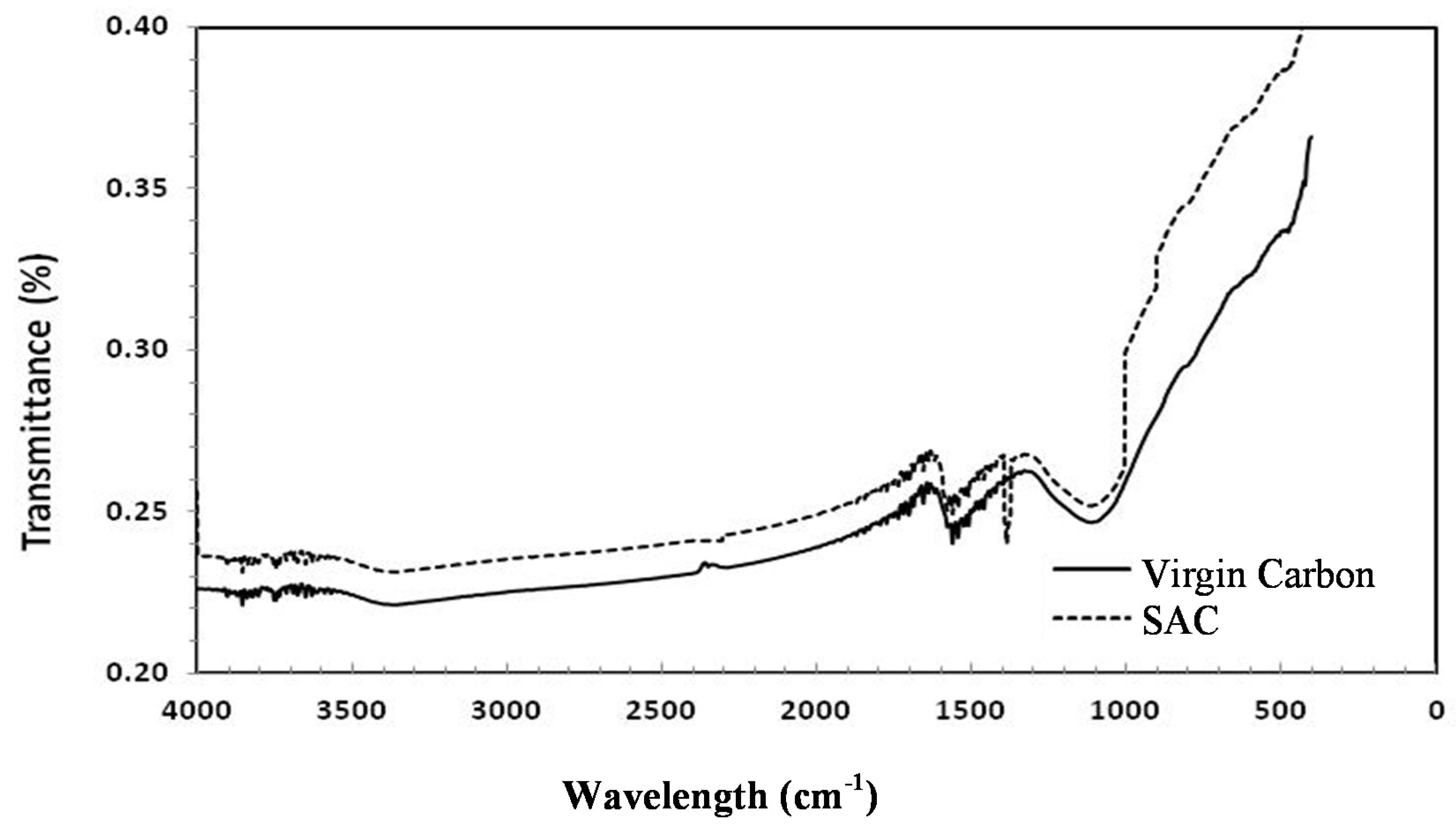
| Functional Groups | Wavelength (cm-1) | |
|---|---|---|
| Virgin Carbon | SAC | |
| OH, water phenolic-hydroxylic group | 3428 | 3400 |
| Quinone, carboxylate | 1571 | 1580 |
| Aromatic ring | 1600 | 1600 |
| Carbonate | - | 1383 |
| Phenol | 1100 | 1100 |
References
- Bansal, R.C.; Goyal, M. Activated Carbon Adsorption; CRC Press: New York, NY, USA, 2005. [Google Scholar]
- Snyder, S.A.; Adham, S.; Redding, A.M.; Cannon, F.S.; DeCarolis, J.; Oppenheimer, J.; Yoon, Y. Role of membranes and activated carbon in the removal of endocrine disruptors and pharmaceuticals. Desalination 2007, 202, 156–181. [Google Scholar] [CrossRef]
- Jurado-Sánchez, B.; Sattayasamitsathit, S.; Gao, W.; Santos, L.; Fedorak, Y.; Singh, V.; Orozco, J.; Galarnyk, M.; Wang, J. Self-Propelled Activated Carbon Janus Micromotors for Efficient Water Purification. Small 2015, 11, 385–506. [Google Scholar] [CrossRef] [PubMed]
- Roosta, M.; Ghaedi, M.; Daneshfar, A.; Sahraei, R.; Asghari, A. Optimization of the ultrasonic assisted removal of methylene blue by gold nanoparticles loaded on activated carbon using experimental design methodology. Ultrason. Sonochem. 2014, 21, 242–252. [Google Scholar] [CrossRef] [PubMed]
- Hameed, B.H.; Din, A.M.; Ahmad, A.L. Adsorption of methylene blue onto bamboo-based activated carbon: Kinetics and equilibrium studies. J. Hazard. Mater. 2007, 141, 819–825. [Google Scholar] [CrossRef] [PubMed]
- Karnib, M.; Kabbani, A.; Holail, H.; Olama, Z. Heavy metals removal using activated carbon, silica and silica activated carbon composite. Energy Procedia 2014, 50, 113–120. [Google Scholar] [CrossRef]
- Sounthararajah, D.P.; Loganathan, P.; Kandasamy, J.; Vigneswaran, S. Adsorptive removal of heavy metals from water using sodium titanate nanofibres loaded onto GAC in fixed-bed columns. J. Hazard. Mater. 2015, 287, 306–316. [Google Scholar] [CrossRef]
- Ministerio de Minería (Ministry of Mining Ecuador), ARCOM Agencia de Regulación y Control Minero, Instituto Nacional de Investigación Geológico Minero Metalúrgico (2016) Plan Nacional de Desarollo del Sector Minero. Available online: http://extwprlegs1.fao.org/docs/pdf/ecu166602anx.pdf (accessed on 1 August 2016).
- Shahriar, S.; Topal, E. An overview of global gold market and gold price forecasting. Resour. Policy 2010, 35, 178–189. [Google Scholar]
- Marsden, J.; House, C. Solution purification and concentration. In The Chemistry of Gold Extraction; SME: Dearborn, MI, USA, 2006. [Google Scholar]
- Stange, W. The process design of gold leaching and carbon-in-pulp circuits. J. S. Afr. Inst. Min. Metall. 1999, 99, 13–25. [Google Scholar]
- Wan, R.; Miller, J. Carbon adsorption technology. Miner. Process. Extr. Metall. Rev. 2007, 6, 153–164. [Google Scholar]
- Bahawan, D.; Poodari, S.; Ravi, K.; Golla, S.; Anand, C.; Banda, K.; Himabindu, V.; Vidyavathi, S. Reactivation and recycling of spent carbon using solvent desorption followed by thermal treatment (TR). Mater. Cycles Waste Manag. 2014, 17, 185–193. [Google Scholar]
- Klymenko, N.; Samsoni-Todrova, E.; Savchyna, L.; Patiuk, L. Restoration of activated carbon adsorption capacity after a long-term use of filters for add-on treatment of tap water. J. Water Chem. Technol. 2012, 35, 159–164. [Google Scholar] [CrossRef]
- Zuo, Y.; Zhabg, L.; Peng, J.; Srinivasakannan, C.; Liu, B.; Ma, A. Regeneration of waste activated carbon after extracting gold with steam under microwave heating: Optimization using response surface methodology. J. Cent. South Univ. 2014, 21, 3233–3240. [Google Scholar] [CrossRef]
- Cazetta, A.; Junior, O.; Vargas, A.; Da silva, A.; Zou, X.; Asefa, T.; Almeida, V. Thermal regeneration study of high surface area activated carbon obtained from coconut Shell: Characterization and application of response surface methodology. J. Anal. Appl. Pyrolysis. 2013, 101, 53–60. [Google Scholar] [CrossRef]
- Caliskan, E.; Bermúdez, J.; Parra, J.; Menéndez, J.; Mahramanhoglu, M.; Ania, C. Low temperature regeneration of activated carbons using microwaves: Revising conventional wisdom. J. Environ. Manag. 2012, 102, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Xia, H.; Wu, J.; Xia, Y.; Zhang, L.; Peng, J.; Wang, S.; Zheng, Z.; Zhang, S. Microwave assisted regeneration of spent activated carbon from petrochemical plant using response surface methodology. J. Porous Mater. 2014, 22, 137–146. [Google Scholar] [CrossRef]
- Cho, J.; Kim, Y.; Jeon, S.; Jung, J.; Oh, K. Improvement of thermal regeneration of spent granular activated carbon using air agent: Application of sintering and deoxygenation. Korean J. Chem. 2014, 31, 1641–1650. [Google Scholar] [CrossRef]
- Foo, K.; Hameed, B. A cost effective method for regeneration of durian shell and jackfruit peel activated carbons by microwave irradiation. Chem. Eng. J. 2012, 193, 404–409. [Google Scholar] [CrossRef]
- Foo, K.; Hameed, B. Microwave-assisted regeneration of activated carbon. Water Res. 2013, 119, 234–240. [Google Scholar] [CrossRef]
- Zhang, L.; Jiang, H.; Ma, C.; Dong, Y. Microwave regeneration characteristics of activated carbon for flue gas desulfurization. J. Fuel Chem. Technol. 2012, 40, 1366–1371. [Google Scholar] [CrossRef]
- Waranabe, N.; Takata, M.; Takemine, S.; Yamamoto, K. Thermal mineralization behavior of PFOA, PFHxA and PFOS during reactivation of granular activated carbon (GAC) in nitrogen atmosphere. Environ. Sci. Pollut. Res. 2018, 25, 7200–7205. [Google Scholar] [CrossRef]
- Román, S.; Ledesma, B.; Álvarez, A.; González, J. Comparative study on the thermal reactivation of spent adsorbents. Fuel Process. Technol. 2013, 116, 358–365. [Google Scholar] [CrossRef]
- Duan, X.; Srinivasakannan, C.; Qu, W.W.; Wang, X.; Peng, J.; Zhang, L. Regeneration of microwave assisted spent activated carbon: Process optimization, adsorption isotherms and kinetics. Chem. Eng. Process. Process Intensif. 2012, 53, 53–62. [Google Scholar]
- Seung, N.; Wang, S.; Young-Kwon, P.; Sang, K. Thermal and chemical regeneration of spent activated carbon and its adsorption property for toluene. Chem. Eng. J. 2012, 210, 500–509. [Google Scholar]
- Samonin, V.; Podvyaznikov, M.; Solov’ev, V.; Kiseleva, C.; Khrylova, E.; Spiridonova, E. Study of the possibility of regeneration of activated carbon spent in water treatment processes using the chemical regeneration and thermal reactivation. Russ. J. Appl. Chem. 2013, 86, 1220–1224. [Google Scholar] [CrossRef]
- Kononova, M.; Vorob’ev-Desyatovskii, N.; Ibragimova, R.; Kubyshkin, S. Effect of Inorganic Compounds in the activated carbon phase and in solution on the adsorption of Gold (I) cyanide complex. Russ. J. Appl. Chem. 2009, 82, 173–182. [Google Scholar] [CrossRef]
- Ibragimova, R.; Mil’chenko, A.; Vorob’ev-Desyatovskii, N. Criteria for choice of a brand of activated carbon for hydrometallurgical recovery of gold from ore pulps in carbon-in-leaching and carbon-in-pulp processes. Russ. J. Appl. Chem. 2007, 6, 919–931. [Google Scholar] [CrossRef]
- Seon-Ha, C.; Seong-Su, K.; Woochang, J.; No-suk, P. Evaluation of physical properties and adsorption capacity of regenerated granular activated carbons (GACs). Korean J. Chem. 2012, 30, 891–897. [Google Scholar]
- Prauchner, M.J.; Rodriguez-Reinoso, F. Chemical versus physical activation of coconut shell: A comparative study. Microporous Mesoporous Mater. 2012, 152, 163–171. [Google Scholar] [CrossRef]
- Marsh, H.; Reinoso, F.R. Activated Carbon; Elsevier: Great Britain, UK, 2006. [Google Scholar]
- Perry, R.; Greeen, D.; Maloney, J. Handbook of Chemical Engineering, 6th ed.; Mc Graw Hill: New York, NY, USA, 1999. [Google Scholar]
- Guo, Y.; Du, E. The effects of thermal regeneration conditions and inorganic compounds on the characteristics of activated carbon used in a power plant. Chem. Eng. J. 2012, 17, 444–449. [Google Scholar] [CrossRef]
- Romero, A.; Sampaio, C.H. Characterization of activated carbons for the extractive metallurgy of gold. Turk. XML Min. Congr. 1993, 427–438. [Google Scholar]
- Bansal, R.; Donnet, J.; Stoeckli, F. Active Carbon; Marcel Dekker: New York, NY, USA, 1998. [Google Scholar]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of gases in multimolecular layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Horváth, G.; Kawazoe, K. Method for the calculation of effective pore size distribution in molecular sieve carbon. J. Chem. Eng. Jpn. 1983, 16, 470–475. [Google Scholar] [CrossRef]
- Groen, J.; Peffer, L.; Pérez-Ramírez, J. Pore size determination in modified micro and mesoporous materials. Pitfalls and limitations in gas adsorption data analysis. Microporous Mesoporous Mater. 2003, 60, 1–17. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |



| Virgin Carbon | SAC | |
|---|---|---|
| d80 (mm) | 3.1 | 2.5 |
| Iodine number (mg I2/g AC) | 1036 | 734 |
| Specific surface area (m2/g) | 1012 | 869 |
| Ball-pan hardness (%) | 98.3 | 98.8 |
| Carbon | Specific Surface Area (m2/g) |
|---|---|
| Virgin carbon | 1012 |
| SAC | 869 |
| Reactivated carbon 1 | 1079 |
| Carbon | Iodine Number (mg I2/g AC) | |
|---|---|---|
| Chemical-Thermal | Thermal-Chemical | |
| Acid wash: HCl 10% v/v 50 °C | 1085 | 1035 |
| HNO3 20% v/v 50 °C | 1199 | 1045 |
| H2SO4 30% v/v 50 °C | 1128 | 1055 |
| Carbon | Ball-Pan Hardness (%) | Normal Abrasion in Pulp Resistance (%) |
|---|---|---|
| Virgin carbon | 98.3 | 92.5 |
| SAC | 98.8 | 97.1 |
| Chemical reactivation 1 | 98.4 | 96.4 |
| Thermal reactivation 2 | 97.3 | 90.1 |
| Reactivated carbon 3 | 98.1 | 96.3 |
| Carbon | Virgin Carbon | SAC | Reactivated Carbon 1 |
|---|---|---|---|
| Moisture content (%) | 0.20 | 0.81 | 0.54 |
| Volatile content (%) | 5.27 | 8.35 | 6.10 |
| Ash content (%) | 3.09 | 12.08 | 3.35 |
| Fixed carbon (%) | 91.94 | 80.33 | 89.30 |
| Iodine number (mg I2/g AC) | 1036 | 734 | 1199 |
| Methylene blue index (mg/100g AC) | 27.91 | 27.84 | 27.90 |
| Sugar discoloration index (UBR) | 34.94 | 34.05 | 33.18 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
B. Cevallos Toledo, R.; F. Aragón-Tobar, C.; Gámez, S.; de la Torre, E. Reactivation Process of Activated Carbons: Effect on the Mechanical and Adsorptive Properties. Molecules 2020, 25, 1681. https://doi.org/10.3390/molecules25071681
B. Cevallos Toledo R, F. Aragón-Tobar C, Gámez S, de la Torre E. Reactivation Process of Activated Carbons: Effect on the Mechanical and Adsorptive Properties. Molecules. 2020; 25(7):1681. https://doi.org/10.3390/molecules25071681
Chicago/Turabian StyleB. Cevallos Toledo, Rita, Carlos F. Aragón-Tobar, Sebastián Gámez, and Ernesto de la Torre. 2020. "Reactivation Process of Activated Carbons: Effect on the Mechanical and Adsorptive Properties" Molecules 25, no. 7: 1681. https://doi.org/10.3390/molecules25071681
APA StyleB. Cevallos Toledo, R., F. Aragón-Tobar, C., Gámez, S., & de la Torre, E. (2020). Reactivation Process of Activated Carbons: Effect on the Mechanical and Adsorptive Properties. Molecules, 25(7), 1681. https://doi.org/10.3390/molecules25071681






