Hyaluronic Acid Derivative Effect on Niosomal Coating and Interaction with Cellular Mimetic Membranes
Abstract
1. Introduction
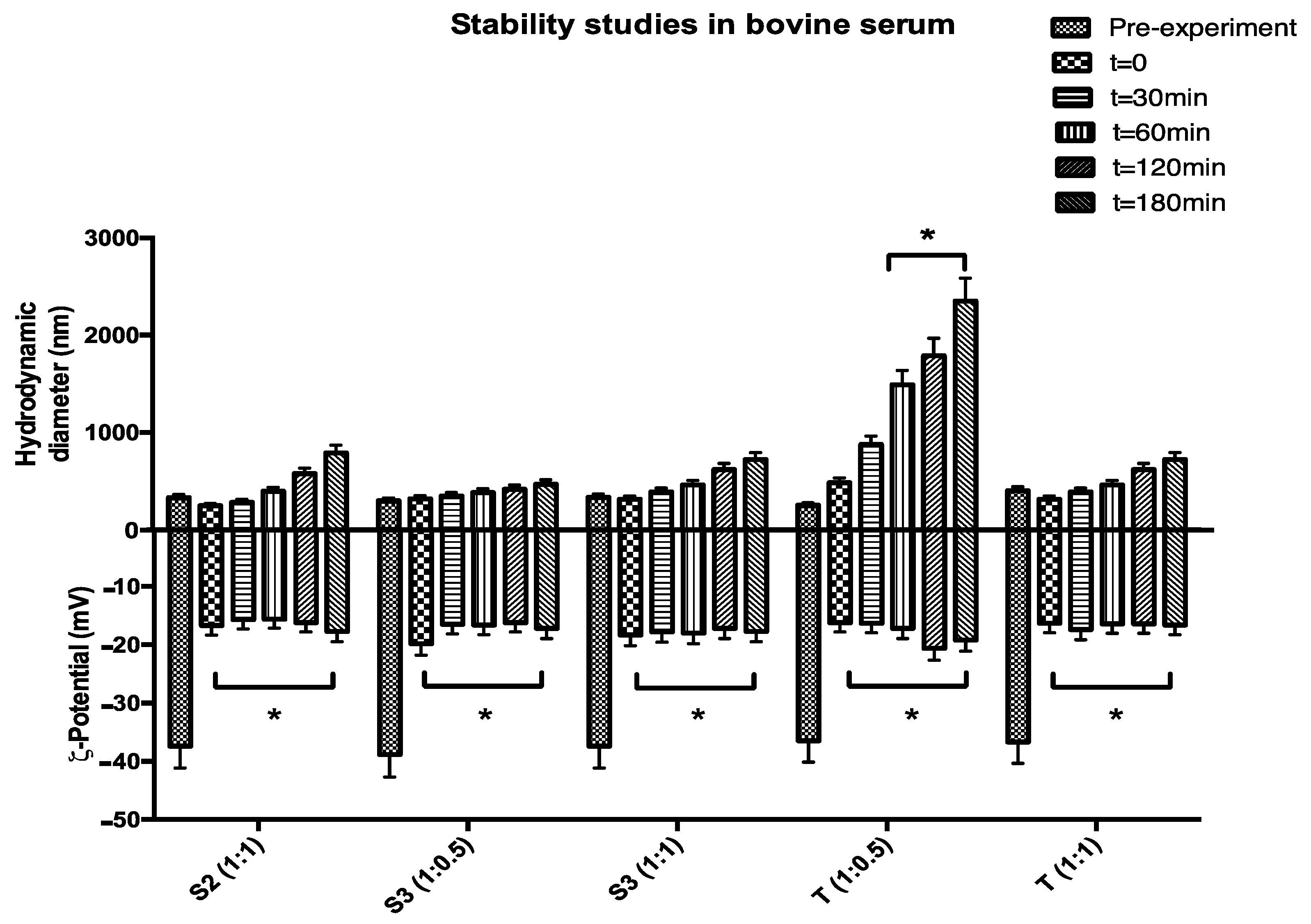
2. Results and Discussion
3. Materials and Methods
3.1. Materials
3.2. Methods
3.2.1. Preparation and Purification of Niosomes
3.2.2. Synthesis of Hyaluronic Acid Derivatized with Cholesterol
3.2.3. Coating of Niosomes with HA-Chol Derivative
3.2.4. Solid State NMR
3.2.5. Dynamic Light Scattering and ζ-Potential Measurements
3.2.6. Transmission Electron Microscopy (TEM)
3.2.7. Stability Studies
3.2.8. Small Angle X-ray Scattering
3.2.9. Bilayer Characterization
3.2.10. Interaction Properties of Nanovesicles with Liposomes
3.2.11. Calcein Release Studies
3.2.12. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Lajevardi, A.; Sadr, M.H.; Yaraki, M.T.; Badiei, A.; Armaghan, M. A pH-responsive and magnetic Fe3O4@ silica@ MIL-100 (Fe)/β-CD nanocomposite as a drug nanocarrier: Loading and release study of cephalexin. New J. Chem. 2018, 42, 9690–9701. [Google Scholar] [CrossRef]
- Coelho, S.C.; Pereira, M.C.; Juzeniene, A.; Juzenas, P.; Coelho, M.A. Supramolecular nanoscale assemblies for cancer diagnosis and therapy. J. Control. Release 2015, 213, 152–167. [Google Scholar] [CrossRef] [PubMed]
- Luque-Michel, E.; Imbuluzqueta, E.; Sebastián, V.; Blanco-Prieto, M.J. Clinical advances of nanocarrier-based cancer therapy and diagnostics. Expert Opin. Drug Deliv. 2017, 14, 75–92. [Google Scholar] [CrossRef] [PubMed]
- Biffi, S.; Voltan, R.; Bortot, B.; Zauli, G.; Secchiero, P. Actively targeted nanocarriers for drug delivery to cancer cells. Expert Opin. Drug Deliv. 2019, 16, 481–496. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, F.; Hanieh, P.N.; Del Favero, E.; Rondelli, V.; Brocca, P.; Pereira, M.C.; Andreev, O.A.; Reshetnyak, Y.K.; Marianecci, C.; Carafa, M. Decoration of nanovesicles with pH (low) insertion peptide (pHLIP) for targeted delivery. Nanoscale Res. Lett. 2018, 13, 1–9. [Google Scholar] [CrossRef]
- Hong, M.; Zhu, S.; Jiang, Y.; Tang, G.; Pei, Y. Efficient tumor targeting of hydroxycamptothecin loaded PEGylated niosomes modified with transferrin. J. Control. Release 2009, 133, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Y.; Wan, J.; Zhou, L.; Ma, W.; Yang, Y.; Luo, W.; Yu, Z.; Wang, H. Stimuli-responsive nanotherapeutics for precision drug delivery and cancer therapy. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2019, 11, e1527. [Google Scholar] [CrossRef]
- Huang, W.-Y.; Lin, J.-N.; Hsieh, J.-T.; Chou, S.-C.; Lai, C.-H.; Yun, E.-J.; Lo, U.-G.; Pong, R.-C.; Lin, J.-H.; Lin, Y.-H. Nanoparticle targeting CD44-positive cancer cells for site-specific drug delivery in prostate cancer therapy. ACS Appl. Mater. Interfaces 2016, 8, 30722–30734. [Google Scholar] [CrossRef]
- Wickens, J.M.; Alsaab, H.O.; Kesharwani, P.; Bhise, K.; Amin, M.C.I.M.; Tekade, R.K.; Gupta, U.; Iyer, A.K. Recent advances in hyaluronic acid-decorated nanocarriers for targeted cancer therapy. Drug Discov. Today 2017, 22, 665–680. [Google Scholar] [CrossRef]
- Quinones, J.P.; Jokinen, J.; Keinänen, S.; Covas, C.P.; Brüggemann, O.; Ossipov, D. Self-assembled hyaluronic acid-testosterone nanocarriers for delivery of anticancer drugs. Eur. Polym. J. 2018, 99, 384–393. [Google Scholar] [CrossRef]
- Liu, D.; Zhang, Q.; Wang, J.; Guan, S.; Cai, D.; Liu, J. Inhibition of growth and metastasis of breast cancer by targeted delivery of 17-hydroxy-jolkinolide B via hyaluronic acid-coated liposomes. Carbohydr. Polym. 2021, 257, 117572. [Google Scholar] [CrossRef]
- Huang, W.; Xiao, G.; Zhang, Y.; Min, W. Research progress and application opportunities of nanoparticle–protein corona complexes. Biomed. Pharmacother. 2021, 139, 111541. [Google Scholar] [CrossRef] [PubMed]
- Imperlini, E.; Celia, C.; Cevenini, A.; Mandola, A.; Raia, M.; Fresta, M.; Orrù, S.; Di Marzio, L.; Salvatore, F. Nano-bio interface between human plasma and niosomes with different formulations indicates protein corona patterns for nanoparticle cell targeting and uptake. Nanoscale 2021, 13, 5251–5269. [Google Scholar] [CrossRef]
- Almalik, A.; Benabdelkamel, H.; Masood, A.; Alanazi, I.O.; Alradwan, I.; Majrashi, M.A.; Alfadda, A.A.; Alghamdi, W.M.; Alrabiah, H.; Tirelli, N. Hyaluronic acid coated chitosan nanoparticles reduced the immunogenicity of the formed protein corona. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- Montanari, E.; Capece, S.; Di Meo, C.; Meringolo, M.; Coviello, T.; Agostinelli, E.; Matricardi, P. Hyaluronic acid nanohydrogels as a useful tool for BSAO immobilization in the treatment of melanoma cancer cells. Macromol. Biosci. 2013, 13, 1185–1194. [Google Scholar] [CrossRef] [PubMed]
- Nakai, T.; Hirakura, T.; Sakurai, Y.; Shimoboji, T.; Ishigai, M.; Akiyoshi, K. Injectable hydrogel for sustained protein release by salt-induced association of hyaluronic acid nanogel. Macromol. Biosci. 2012, 12, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Di Meo, C.; Montanari, E.; Manzi, L.; Villani, C.; Coviello, T.; Matricardi, P. Highly versatile nanohydrogel platform based on riboflavin-polysaccharide derivatives useful in the development of intrinsically fluorescent and cytocompatible drug carriers. Carbohydr. Polym. 2015, 115, 502–509. [Google Scholar] [CrossRef]
- Manzi, G.; Zoratto, N.; Matano, S.; Sabia, R.; Villani, C.; Coviello, T.; Matricardi, P.; Di Meo, C. “Click” hyaluronan based nanohydrogels as multifunctionalizable carriers for hydrophobic drugs. Carbohydr. Polym. 2017, 174, 706–715. [Google Scholar] [CrossRef]
- Montanari, E.; Oates, A.; Di Meo, C.; Meade, J.; Cerrone, R.; Francioso, A.; Devine, D.; Coviello, T.; Mancini, P.; Mosca, L. Hyaluronan-based nanohydrogels for targeting intracellular S. aureus in human keratinocytes. Adv. Healthc. Mater. 2018, 7, 1701483. [Google Scholar] [CrossRef] [PubMed]
- Montanari, E.; De Rugeriis, M.C.; Di Meo, C.; Censi, R.; Coviello, T.; Alhaique, F.; Matricardi, P. One-step formation and sterilization of gellan and hyaluronan nanohydrogels using autoclave. J. Mater. Sci. Mater. Med. 2015, 26, 32. [Google Scholar] [CrossRef]
- Bhattacharjee, S. DLS and zeta potential–what they are and what they are not? J. Control. Release 2016, 235, 337–351. [Google Scholar] [CrossRef]
- Pozzi, D.; Caminiti, R.; Marianecci, C.; Carafa, M.; Santucci, E.; De Sanctis, S.C.; Caracciolo, G. Effect of cholesterol on the formation and hydration behavior of solid-supported niosomal membranes. Langmuir 2010, 26, 2268–2273. [Google Scholar] [CrossRef]
- Di Meo, C.; Proietti, N.; Mannina, L.; Capitani, D. NMR methodologies in the study of polysaccharides. In Polysaccharide Hydrogels: Characterization and Biomedical Applications; Pan Stanford Publishing PTE Ltd.: Singapore, 2016; pp. 209–243. [Google Scholar]
- Di Meo, C.; Capitani, D.; Mannina, L.; Brancaleoni, E.; Galesso, D.; De Luca, G.; Crescenzi, V. Synthesis and NMR characterization of new hyaluronan-based NO donors. Biomacromolecules 2006, 7, 1253–1260. [Google Scholar] [CrossRef] [PubMed]
- Crescenzi, V.; Francescangeli, A.; Capitani, D.; Mannina, L.; Renier, D.; Bellini, D. Hyaluronan networking via Ugi’s condensation using lysine as cross-linker diamine. Carbohydr. Polym. 2003, 53, 311–316. [Google Scholar] [CrossRef]
- Ingallina, C.; Rinaldi, F.; Bogni, A.; Ponti, J.; Passeri, D.; Reggente, M.; Rossi, M.; Kinsner-Ovaskainen, A.; Mehn, D.; Rossi, F. Niosomal approach to brain delivery: Development, characterization and in vitro toxicological studies. Int. J. Pharm. 2016, 511, 969–982. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, F.; Del Favero, E.; Moeller, J.; Hanieh, P.N.; Passeri, D.; Rossi, M.; Angeloni, L.; Venditti, I.; Marianecci, C.; Carafa, M. Hydrophilic silver nanoparticles loaded into niosomes: Physical–chemical characterization in view of biological applications. Nanomaterials 2019, 9, 1177. [Google Scholar] [CrossRef]
- Zeng, W.; Li, Q.; Wan, T.; Liu, C.; Pan, W.; Wu, Z.; Zhang, G.; Pan, J.; Qin, M.; Lin, Y. Hyaluronic acid-coated niosomes facilitate tacrolimus ocular delivery: Mucoadhesion, precorneal retention, aqueous humor pharmacokinetics, and transcorneal permeability. Colloids Surf. B Biointerfaces 2016, 141, 28–35. [Google Scholar] [CrossRef]
- Tirelli, N.; Almalik, A.; Karimi, S.; Ouasti, S.; Donno, R.; Day, P. Receptor-mediated uptake of hyaluronic acid (HA)-coated nanoparticles: HA presentation modulates affinity and endocytosis kinetics. Biomaterials 2013, 34, 5369–5380. [Google Scholar]
- Marianecci, C.; Rinaldi, F.; Di Marzio, L.; Pozzi, D.; Caracciolo, G.; Manno, D.; Dini, L.; Paolino, D.; Celia, C.; Carafa, M. Interaction of pH-sensitive non-phospholipid liposomes with cellular mimetic membranes. Biomed. Microdev. 2013, 15, 299–309. [Google Scholar] [CrossRef]
- Provencher, S.W. CONTIN: A general purpose constrained regularization program for inverting noisy linear algebraic and integral equations. Comput. Phys. Commun. 1982, 27, 229–242. [Google Scholar] [CrossRef]
- De Vos, C.; Deriemaeker, L.; Finsy, R. Quantitative assessment of the conditioning of the inversion of quasi-elastic and static light scattering data for particle size distributions. Langmuir 1996, 12, 2630–2636. [Google Scholar] [CrossRef]
- Sennato, S.; Bordi, F.; Cametti, C.; Marianecci, C.; Carafa, M.; Cametti, M. Hybrid niosome complexation in the presence of oppositely charged polyions. J. Phys. Chem. B 2008, 112, 3720–3727. [Google Scholar] [CrossRef]
- Rinaldi, F.; Seguella, L.; Gigli, S.; Hanieh, P.; Del Favero, E.; Cantù, L.; Pesce, M.; Sarnelli, G.; Marianecci, C.; Esposito, G. inPentasomes: An innovative nose-to-brain pentamidine delivery blunts MPTP parkinsonism in mice. J. Control. Release 2019, 294, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, F.; Hanieh, P.N.; Imbriano, A.; Passeri, D.; Del Favero, E.; Rossi, M.; Marianecci, C.; De Panfilis, S.; Carafa, M. Different instrumental approaches to understand the chitosan coated niosomes/mucin interaction. J. Drug Deliv. Sci. Technol. 2020, 55, 101339. [Google Scholar] [CrossRef]
- Doucet, M.; Cho, J.H.; Alina, G.; Bakker, J.; Bouwman, W.; Butler, P.; Campbell, K.; Gonzales, M.; Heenan, R.; Jackson, A. SasView Version 4.1; Zenodo. Sasview. Org.: Geneva, Switzerland, 2017. [Google Scholar]
- Lentz, B.R. Membrane “fluidity” as detected by diphenylhexatriene probes. Chem. Phys. Lipids 1989, 50, 171–190. [Google Scholar] [CrossRef]
- Zachariasse, K.A. Intramolecular excimer formation with diarylalkanes as a microfluidity probe for sodium dodecyl sulphate micelles. Chem. Phys. Lett. 1978, 57, 429–432. [Google Scholar] [CrossRef]
- Polozova, A.; Winnik, F.M. Contribution of hydrogen bonding to the association of liposomes and an anionic hydrophobically modified poly (N-isopropylacrylamide). Langmuir 1999, 15, 4222–4229. [Google Scholar] [CrossRef]
- Rinaldi, F.; Hanieh, P.N.; Marianecci, C.; Carafa, M. DLS Characterization of Non-Ionic Surfactant Vesicles for Potential Nose to Brain Application. Nanosci. Nanometr. 2015, 1, 8–14. [Google Scholar]







| Sample | Span20 (mM) | Tween20 (mM) | Chol (mM) | Hydrodynamic Diameter (nm) ± SD | ζ-Potential (mV) ± SD | PDI |
|---|---|---|---|---|---|---|
| S1 | 15.0 | - | 15.0 | 98.0 ± 2.0 | −34.9 ± 0.1 | 0.2 |
| S2 | 11.2 | 212.0 ± 2.0 | −43.3 ± 2.5 | 0.2 | ||
| S3 | 7.5 | 192.0 ± 2.0 | −42.3 ± 2.5 | 0.2 | ||
| T | - | 15.0 | 15.0 | 194.0 ± 2.0 | −26.7 ± 2.4 | 0.2 |
| Sample with HA-Chol | Hydrodynamic Diameter (nm) ± SD | ζ-Potential (mV) ± SD | PDI |
|---|---|---|---|
| S2 (1:1) | 331.8 ± 6.0 | −37.4 ± 2.3 | 0.6 |
| S3 (1:0.5) | 297.6 ± 19.3 | −36.8 ± 1.7 | 0.6 |
| S3 (1:1) | 333.9 ± 14.1 | −37.4 ± 0.6 | 0.6 |
| T (1:0.5) | 250.5 ± 12.9 | −36.5 ± 0.9 | 0.7 |
| T (1:1) | 401.4 ± 16.8 | −36.7 ± 1.3 | 0.4 |
| Sample with and without HA-Chol | Fluidity (Anisotropy) | Polarity (I1/I3) | Microviscosity (IE/I3) |
|---|---|---|---|
| S2 | 0.11 | 1.14 | 0.38 |
| S2 (1:1) | 0.24 | 1.16 | 0.30 |
| S3 | 0.14 | 1.12 | 0.74 |
| S3 (1:0.5) | 0.15 | 1.23 | 0.36 |
| S3 (1:1) | 0.21 | 1.22 | 0.33 |
| T | 0.20 | 1.58 | 0.18 |
| T (1:0.5) | 0.27 | 1.15 | 0.17 |
| T (1:1) | 0.26 | 1.12 | 0.29 |
| Sample | Span20 (mM) | Tween20 (mM) | Chol (mM) |
|---|---|---|---|
| S1 | 15.0 | - | 15.0 |
| S2 | 11.2 | ||
| S3 | 7.5 | ||
| T | - | 15.0 | 15.0 |
| Sample | Molar Ratio |
|---|---|
| S3 | 1:0.5 |
| 1:1 | |
| T | 1:0.5 |
| Sample | Molar Ratio |
|---|---|
| S2 | 1:1 |
| T | 1:1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hanieh, P.N.; Forte, J.; Di Meo, C.; Ammendolia, M.G.; Del Favero, E.; Cantù, L.; Rinaldi, F.; Marianecci, C.; Carafa, M. Hyaluronic Acid Derivative Effect on Niosomal Coating and Interaction with Cellular Mimetic Membranes. Molecules 2021, 26, 3434. https://doi.org/10.3390/molecules26113434
Hanieh PN, Forte J, Di Meo C, Ammendolia MG, Del Favero E, Cantù L, Rinaldi F, Marianecci C, Carafa M. Hyaluronic Acid Derivative Effect on Niosomal Coating and Interaction with Cellular Mimetic Membranes. Molecules. 2021; 26(11):3434. https://doi.org/10.3390/molecules26113434
Chicago/Turabian StyleHanieh, Patrizia N., Jacopo Forte, Chiara Di Meo, Maria Grazia Ammendolia, Elena Del Favero, Laura Cantù, Federica Rinaldi, Carlotta Marianecci, and Maria Carafa. 2021. "Hyaluronic Acid Derivative Effect on Niosomal Coating and Interaction with Cellular Mimetic Membranes" Molecules 26, no. 11: 3434. https://doi.org/10.3390/molecules26113434
APA StyleHanieh, P. N., Forte, J., Di Meo, C., Ammendolia, M. G., Del Favero, E., Cantù, L., Rinaldi, F., Marianecci, C., & Carafa, M. (2021). Hyaluronic Acid Derivative Effect on Niosomal Coating and Interaction with Cellular Mimetic Membranes. Molecules, 26(11), 3434. https://doi.org/10.3390/molecules26113434










