Removal of Chromium(III) and Cadmium(II) Heavy Metal Ions from Aqueous Solutions Using Treated Date Seeds: An Eco-Friendly Method
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Instrumentation
2.3. Preparation of Standard Solution
2.4. Preparation of Biosorbent
2.5. H2O2 Treatment of Date Powders
2.6. Adsorption and Desorption Studies
2.7. Thermodynamic Studies of Metal Ions Adsorptions
3. Results and Discussion
3.1. Characterization
3.1.1. Analysis of Adsorbent’s Surface and Pore Size
3.1.2. Fourier-Transform Infrared Spectroscopic Analysis (FTIR)
3.2. Adsorption Properties
3.2.1. Effect of pH
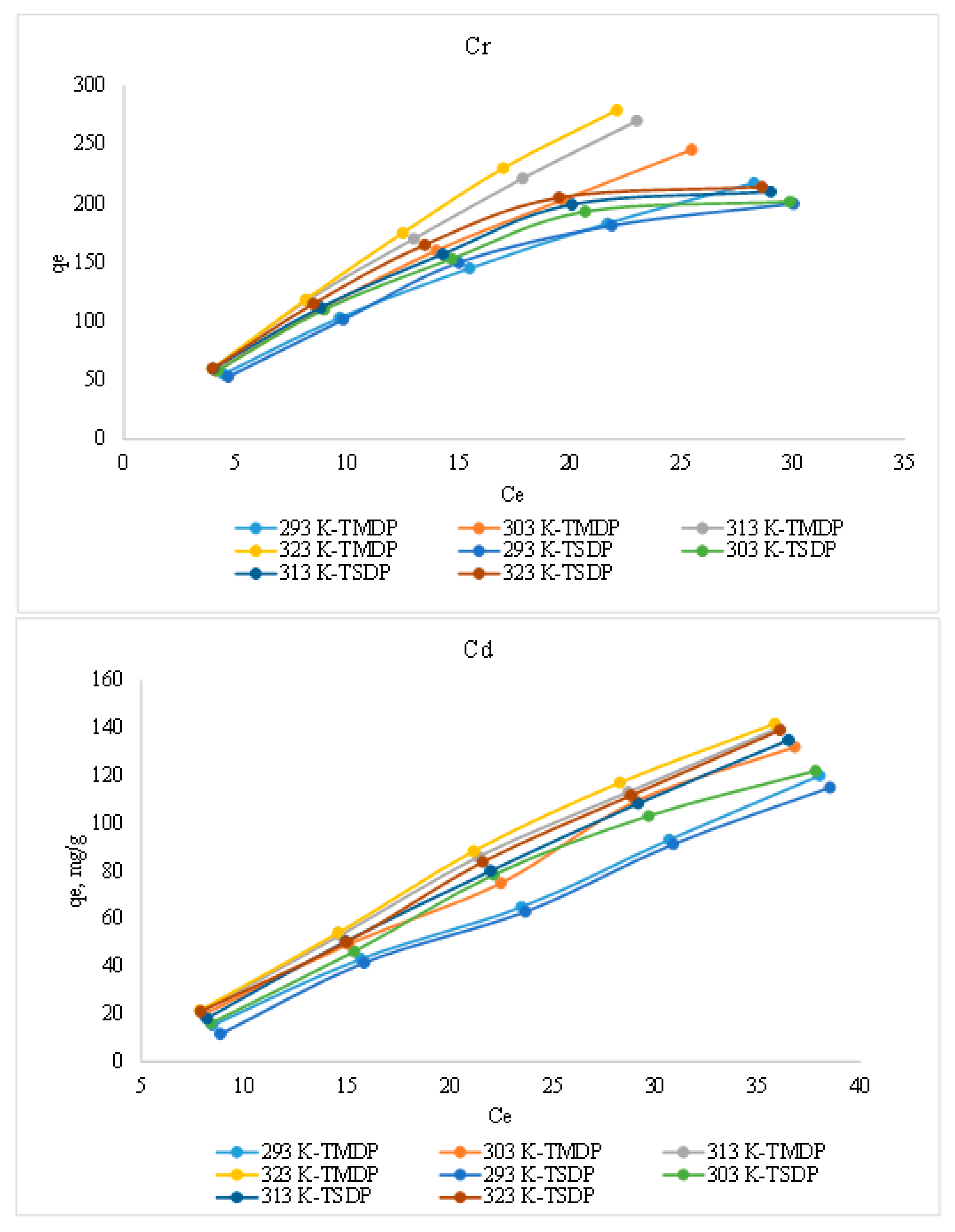
3.2.2. Effect of Initial Concentration and Temperature
3.2.3. Effects of Contact Time
3.2.4. Adsorption Modeling
Equilibrium Isotherm
3.3. Adsorption Kinetics
3.4. Adsorption Thermodynamics
3.5. Elution and Regeneration Studies
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Joseph, L.; Jun, B.M.; Flora, J.R.V.; Park, C.M.; Yoon, Y. Removal of heavy metals from water sources in the developing world using low-cost materials: A review. Chemosphere 2019, 229, 142–159. [Google Scholar] [CrossRef]
- Briffa, J.; Sinagra, E.; Blundell, R. Heavy metal pollution in the environment and their toxicological effects on humans. Heliyon 2020, 8, e04691. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Xi, S. The effects of heavy metals on human metabolism. Toxicol. Mech. Meth. 2020, 30, 167–176. [Google Scholar] [CrossRef]
- Rehman, K.; Fatima, F.; Waheed, I.; Akash, M.S.H. Prevalence of exposure of heavy metals and their impact on health consequences. J. Cell Biochem. 2018, 119, 157–184. [Google Scholar] [CrossRef] [PubMed]
- Ajiboye, T.O.; Oyewo, O.A.; Onwudiwe, D.C. Simultaneous removal of organics and heavy metals from industrial wastewater: A review. Chemosphere 2021, 262, 128379. [Google Scholar] [CrossRef] [PubMed]
- Naveedullah; Hashmi, M.Z.; Yu, C.; Shen, H.; Duan, D.; Shen, C.; Lou, L.; Chen, Y. Risk assessment of heavy metals pollution in agricultural soils of siling reservoir watershed in Zhejiang Province, China. Biomed. Res. Int. 2013, 2013, 590306. [Google Scholar] [CrossRef]
- Milenkovic, B.; Stajic, J.M.; Stojic, N.; Pucarevic, M.; Strbac, S. Evaluation of heavy metals and radionuclides in fish and seafood products. Chemosphere 2019, 229, 324–331. [Google Scholar] [CrossRef]
- Rajaram, T.; Das, A. Water pollution by industrial effluents in India: Discharge scenarios and case for participatory ecosystem specific local regulation. Futures 2008, 40, 56–69. [Google Scholar] [CrossRef]
- Bi, X.; Feng, X.; Yang, Y.; Qiu, G.; Li, G.; Li, F.; Jin, Z. Environmental contamination of heavy metals from zinc smelting areas in Hezhang County, western Guizhou, China. Environ. Int. 2006, 32, 883–890. [Google Scholar] [CrossRef]
- Masindi, V.; Muedi, K.L. Environmental contamination by heavy metals. Heavy Met. 2018, 10, 115–132. [Google Scholar]
- Tchounwou, P.B.; Yedjou, C.G.; Patlolla, A.K.; Sutton, D.J. Heavy metal toxicity and the environment. Mol. Clin. Environ. Toxicol. 2012, 101, 133–164. [Google Scholar]
- Kim, H.S.; Kim, Y.J.; Seo, Y.R. An overview of carcinogenic heavy metal: Molecular toxicity mechanism and prevention. J. Cancer Prev. 2015, 20, 232. [Google Scholar] [CrossRef]
- Engwa, G.A.; Ferdinand, P.U.; Nweke Nwalo, F.; Unachukwu, M.N. Mechanism and health effects of heavy metal toxicity in humans. In Poisoning in the Modern World-New Tricks for an Old Dog; Intech: London, UK, 2019. [Google Scholar]
- Jaishankar, M.; Tseten, T.; Anbalagan, N.; Mathew, B.B.; Beeregowda, K.N. Toxicity, mechanism and health effects of some heavy metals. Interdiscipl. Toxicol. 2014, 7, 60–72. [Google Scholar] [CrossRef]
- Korkmaz, C.; Ay, Ö.; Ersoysal, Y.; Köroğlu, M.A.; Erdem, C. Heavy metal levels in muscle tissues of some fish species caught from north-east Mediterranean: Evaluation of their effects on human health. J. Food Compos. Anal. 2019, 81, 1–9. [Google Scholar] [CrossRef]
- Akintujoye, J.F.; Anumudu, C.I.; Awobode, H.O. Assessment of heavy metal residues in water, fish tissue and human blood from Ubeji, Warri, Delta State, Nigeria. J. Appl. Sci. Environ. Manag. 2013, 17, 291–297. [Google Scholar] [CrossRef]
- Godt, J.; Scheidig, F.; Grosse-Siestrup, C.; Esche, V.; Brandenburg, P.; Reich, A.; Groneberg, D.A. The toxicity of cadmium and resulting hazards for human health. J. Occup. Med. Toxicol. 2006, 1, 22–27. [Google Scholar] [CrossRef]
- Gabbiani, G.; Baic, D.; Deziel, C. Toxicity of cadmium for the central nervous system. Experiment. Neurol. 1967, 18, 154–160. [Google Scholar] [CrossRef]
- Sridevi Sangeetha, K.S.; UmAmAHeSwAri, S. Human Exposure to Lead, Mechanism of Toxicity and Treatment Strategy—A Review. J. Clin. Diagnos. Res. 2020, 14, 1–5. [Google Scholar]
- Plumlee, G.S.; Durant, J.T.; Morman, S.A.; Neri, A.; Wolf, R.E.; Dooyema, C.A.; Hageman, P.L.; Lowers, H.A.; Fernette, G.L.; Meeker, G.P.; et al. Linking geological and health sciences to assess childhood lead poisoning from artisanal gold mining in Nigeria. Environ. Health Perspect. 2013, 121, 744–750. [Google Scholar] [CrossRef] [PubMed]
- Sah, D.; Verma, P.K.; Kumari, K.M.; Lakhani, A. Chemical partitioning of fine particlebound As, Cd, Cr, Ni, Co, Pb and assessment of associated cancer risk due to inhalation, ingestion and dermal exposure. Inhal. Toxicol. 2017, 29, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Howard, H.; Michael, M.C. (Eds.) Human Health and Heavy Metals Exposure; Life Support: The Environment and Human Health; MIT Press: Massachusetts, MA, USA, 2002. [Google Scholar]
- IARC (International Agency for Research) on Cancer. IARC Monographs on the Evaluation of Carcinogenic Risk to Human: Arsenic, Metals, Fibers, and Dusts, Volume 100C. A Review of Human Carcinogen; World Health Organization: Lyon, France, 2012. [Google Scholar]
- Hansima, M.A.C.K.; Makehelwala, M.; Jinadasa, K.B.S.N.; Wei, Y.; Nanayakkara, K.G.N.; Herath, A.C.; Weerasooriya, R. Fouling of ion exchange membranes used in the electrodialysis reversal advanced water treatment: A review. Chemosphere 2020, 263, 127951. [Google Scholar] [CrossRef]
- Zhu, F.; Zheng, Y.-M.; Zhang, B.-G.; Dai, Y.-R. A critical review on the electrospun nanofibrous membranes for the adsorption of heavy metals in water treatment. J. Hazard. Mater. 2020, 401, 123608. [Google Scholar] [CrossRef]
- Sharshir, S.W.; Algazzar, A.M.; Elmaadawy, K.A.; Kandeal, A.W.; Elkadeem, M.R.; Arunkumar, T.; Zang, J.; Yang, N. New hydrogel materials for improving solar water evaporation, desalination and wastewater treatment: A review. Desalination 2020, 491, 114564. [Google Scholar] [CrossRef]
- Delil, D.A.; Köleli, N.; Dağhan, H.; Bahçeci, G. Recovery of heavy metals from canola (Brassica napus) and soybean (Glycine max) biomasses using electrochemical process. Environ. Technol. Innov. 2020, 17, 100559. [Google Scholar] [CrossRef]
- Shahrokhi-Shahraki, R.; Benally, C.; El-Din, M.G.; Park, J. High efficiency removal of heavy metals using tire-derived activated carbon vs commercial activated carbon: Insights into the adsorption mechanisms. Chemosphere 2021, 264, 128455. [Google Scholar] [CrossRef] [PubMed]
- Panigrahi, T.; Santhoskumar, A.U. Adsorption process for reducing heavy metals in Textile Industrial Effluent with low cost adsorbents. Prog. Chem. Biochem. Res. 2020, 3, 135–139. [Google Scholar] [CrossRef]
- Chakraborty, R.; Asthana, A.; Singh, K.; Jain, B.; Susan, A.B.H. Adsorption of heavy metal ions by various low-cost adsorbents: A review. Int. J. Environ. Anal. Chem. 2020, 1–38. [Google Scholar] [CrossRef]
- Soliman, N.K.; Moustafa, A.F. Industrial solid waste for heavy metals adsorption features and challenges; a review. J. Mater. Res. Technol. 2020, 5, 10235–10253. [Google Scholar] [CrossRef]
- Khan, M.A.; Otero, M.; Kazi, M.; Alqadami, A.A.; Wabaidur, S.M.; Siddiqui, M.R.; Alothmana, Z.A.; Sumbul, S. Unary and binary adsorption studies of lead and malachite green onto a nanomagnetic copper ferrite/drumstick pod biomass composite. J. Hazard. Mater. 2019, 365, 759–770. [Google Scholar] [CrossRef]
- Tran, H.; You, S.; Chao, H. Thermodynamic parameters of cadmium adsorption onto orange peel calculated from various methods: A comparison study. J. Environ. Chem. Eng. 2016, 4, 2671–2682. [Google Scholar] [CrossRef]
- El-Hendawy, A.A. Variation in the FTIR spectra of a biomass under impregnation, carbonization and oxidation conditions. J. Anal. Appl. Pyrolysis. 2006, 75, 159–166. [Google Scholar] [CrossRef]
- Reddy, D.H.K.; Seshaiaha, K.; Reddy, A.V.R.; Lee, S.-M. Optimization of Cd(II), Cu(II)and Ni(II) biosorption by chemically modified Moringa oleifera leaves powder, Carbohydr. Polym. 2012, 88, 1077–1086. [Google Scholar]
- Pandey, K.K.; Pitman, A.J. FTIR studies of the changes in wood chemistry following decay by brown-rot and white-rot fungi. Int. Biodeterior. Biodegrad. 2003, 52, 151–160. [Google Scholar] [CrossRef]
- Nurchi, V.A.; Villaescusa, I.; Matovic, D. (Eds.) The Chemistry Behind the Use of Agricultural Biomass as Sorbent for Toxic Metal Ions: pH Influence, Binding Groups, and Complexation Equilibria, Biomass-Detection, Production and Usage; InTech: London, UK, 2011. [Google Scholar] [CrossRef]
- Alqadami, A.A.; Khan, M.A.; Siddiqui, M.R.; Alothman, Z.A. Development of citric anhydride anchored mesoporous MOF through post synthesis modification to sequester potentially toxic lead (II) from water, Micropor. Mesopor. Mater. 2018, 261, 198–206. [Google Scholar] [CrossRef]
- Khan, M.A.; Ngabura, M.; Choong, T.S.Y.; Masood, H.; Chuah, L.A. Biosorption and desorption of nickel on oil cake: Batch and column studies. Bioresour. Technol. 2012, 103, 35–42. [Google Scholar] [CrossRef]
- Hilal, N.M.; Ahmed, I.A.; El-Sayed, R.E. Activated and Nonactivated Date Pits Adsorbents for the Removal of Copper(II) and Cadmium(II) from Aqueous Solutions. Int. Sch. Res. Netw. 2012, 985853. [Google Scholar] [CrossRef]
- Rodembusch, F.S.; Dias, S.L.P. Synthesis and characterization of a novel organicinorganic hybrid clay adsorbent for the removal of acid red 1 and acid green 25 from aqueous solutions. J. Clean. Prod. 2018, 171, 30–44. [Google Scholar]
- Leite, A.J.B.; Lima, E.C.; dos Reis, G.S.; Thue, P.S.; Saucier, C.; Rodembusch, F.S.; Dias, S.L.P.; Umpierres, C.S.; Dott, G.L. Hybrid adsorbents of tannin and APTES (3-aminopropyltriethoxysilane) and their application for the highly efficient removal of acid red 1 dye from aqueous solutions. J. Environ. Chem. Eng. 2017, 5, 4307–4318. [Google Scholar] [CrossRef]
- Ghobadi, M.; Gharabaghi, M.; Abdollahi, H.; Boroumand, Z.; Moradian, M. MnFe2O4-graphene oxide magnetic nanoparticles as a high-performance adsorbent for rare earth elements: Synthesis, isotherms, kinetics, thermodynamics and desorption. J. Hazard. Mater. 2018, 351, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Lima, E.C.; Adebayo, M.A.; Machado, F.M. Kinetic and equilibrium models of adsorption. In Carbon Nanomaterials as Adsorbents for Environmental and Biological Applications; Bergmann, C.P., Machado, F.M., Eds.; Springer: Cham, Switzerland, 2015; pp. 33–69. [Google Scholar] [CrossRef]
- Argun, M.E.; Dursun, S.; Ozdemir, C.; Karatas, M. Heavy metal adsorption by modified oak sawdust: Thermodynamics and kinetics, J. Hazard. Mater. 2007, 141, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Maryam, M.; Soltani, M.; Siahpoosh, A.; Mehrjan, M.S. Effects of Date Palm (Phoenix dactylifera) Seed Extract on Heavy Metals Concentrations in Carp (Cyprinus carpio). Pol. J. Environ. Stud. 2016, 2, 1117–1123. [Google Scholar]
- Al-Saad, K.; El-Azazy, M.; Issa, A.A.; Al-Yafie, A.; El-Shafie, A.S.; Al-Sulaiti, M.; Shomar, B. Recycling of Date Pits into a Green Adsorbent for Removal of Heavy Metals: A Fractional Factorial Design-Based Approach. Front. Chem. 2019, 7, 552. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, A.A.J.; Vuai, L.A.; Kombo, M.; Chukwuma, O.J. Removal of selected metal ions using powder of seeds of Ajwaa dates from aqueous solution. J. Anal. Pharm Res. 2019, 8, 228–232. [Google Scholar] [CrossRef]
- Esmael, A.I.; Matta, M.E.; Halim, H.A.; Azziz, F.M.A. Adsorption of heavy metals from industrial wastewater using palm date pits as low cost adsorbent. Int. J. Eng. Adv. Technol. 2014, 3, 71–76. [Google Scholar]






| Element | (keV) | Mass% | |||||||
|---|---|---|---|---|---|---|---|---|---|
| MDP | Error | SDP | Error | TMDP | Error | TSDP | Error | ||
| C··K | 0.277 | 40.11 | 0.10 | 40.10 | 0.11 | 37.22 | 0.10 | 37.80 | 0.11 |
| N··K | 0.392 | 26.64 | 0.28 | 27.60 | 0.25 | 21.4 | 0.09 | 22.30 | 0.06 |
| O··K | 0.525 | 33.26 | 0.08 | 32.30 | 0.07 | 41.38 | 0.06 | 39.90 | 0.09 |
| Total | 100 | 100 | 100 | 100 | |||||
| Isotherm | Temperature, K | |||||||
|---|---|---|---|---|---|---|---|---|
| 293 | 303 | 313 | 323 | |||||
| Cr(III) | Cd(II) | Cr(III) | Cd(II) | Cr(III) | Cd(II) | Cr(III) | Cd(II) | |
| TMDP | ||||||||
| Langmuir | ||||||||
| qm (mg/g) | 400 | 352 | 666.67 | 602 | 1000 | 908 | 1428.5 | 1302 |
| KL (L/mg) | 0.03592 | 0.02920 | 0.03727 | 0.03870 | 0.05744 | 0.04344 | 0.07599 | 0.05494 |
| R2 | 0.9929 | 0.9881 | 0.9952 | 0.9897 | 0.9963 | 0.9901 | 0.9981 | 0.9932 |
| Freundlich | ||||||||
| KF (mg/g) | 18.5642 | 12.5642 | 28.5642 | 19.258 | 46.5142 | 36.2548 | 68.56142 | 56.258 |
| (L/mg)1/n | ||||||||
| n | 1.3821 | 1.0821 | 1.6821 | 1.3021 | 1.8211 | 1.5821 | 1.9821 | 1.7821 |
| R2 | 0.9763 | 0.9485 | 0.9805 | 0.9622 | 0.9884 | 0.9715 | 0.9887 | 0.9810 |
| TSDP | ||||||||
| Langmuir | ||||||||
| qm (mg/g) | 388 | 332 | 606.67 | 552 | 910 | 808 | 1228.5 | 1182 |
| KL (L/mg) | 0.03222 | 0.02520 | 0.03327 | 0.03071 | 0.05044 | 0.04004 | 0.07219 | 0.0509 |
| R2 | 0.9909 | 0.9289 | 0.9921 | 0.9611 | 0.9938 | 0.9822 | 0.9941 | 0.9938 |
| Freundlich | ||||||||
| KF (mg/g) | 14.2125 | 8.2314 | 22.0213 | 15.1254 | 37.8541 | 28. 8254 | 56.2242 | 42.1258 |
| (L/mg)1/n | ||||||||
| n | 1.0132 | 1.0021 | 1.2168 | 1.1021 | 1.6211 | 1.3021 | 1.7621 | 1.2154 |
| R2 | 0.9521 | 0.8257 | 0.9644 | 0.8666 | 0.9788 | 0.8801 | 0.9801 | 0.9002 |
| Kinetics Model | MDP | TMDP | SDP | TSDP | ||||
|---|---|---|---|---|---|---|---|---|
| Cr(III) | Cd(II) | Cr(III) | Cd(II) | Cr(III) | Cd(II) | Cr(III) | Cd(II) | |
| qe,exp (mg/g) | 159 | 108 | 191 | 121 | 140 | 109 | 190 | 120 |
| Pseudo-first-order | ||||||||
| qe1,cal (mg/g) | 132 | 85 | 151 | 101 | 109 | 79 | 148 | 101 |
| K1(1/min) | 0.0064 | 0.0059 | 0.0062 | 0.0061 | 0.0060 | 0.0058 | 0.0068 | 0.0063 |
| R2 | 0.9787 | 0.9688 | 0.9822 | 0.9588 | 0.9666 | 0.9589 | 0.9787 | 0.9689 |
| Pseudo-second-order | ||||||||
| qe2,cal (mg/g) | 162 | 110 | 194 | 123 | 144 | 111 | 195 | 122 |
| k2 (g/mg.min) | 0.0004 | 0.0009 | 0.0002 | 0.0006 | 0.0006 | 0.0005 | 0.0008 | 0.0007 |
| R2 | 0.9907 | 0.9818 | 0.9877 | 0.9900 | 0.9811 | 0.9901 | 0.9878 | 0.9719 |
| Adsorbents ↓ | ΔH° (kJ/mol) | ΔS° (J/mol-K) | ΔG° (kJ/mol) | |||
|---|---|---|---|---|---|---|
| Temperature → | 293 K | 303 K | 313 K | 323 K | ||
| MDP | 6154 | 20.77 | −113.79 | −430.13 | −644.08 | −802.54 |
| TMDP | 8511 | 29.77 | −153.79 | −539.13 | −877.08 | −912.54 |
| SDP | 4254 | 15.22 | −103.79 | −295.13 | −523.08 | −912.54 |
| TSDP | 7588 | 27.66 | −143.79 | −509.13 | −697.08 | −832.54 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azam, M.; Wabaidur, S.M.; Khan, M.R.; Al-Resayes, S.I.; Islam, M.S. Removal of Chromium(III) and Cadmium(II) Heavy Metal Ions from Aqueous Solutions Using Treated Date Seeds: An Eco-Friendly Method. Molecules 2021, 26, 3718. https://doi.org/10.3390/molecules26123718
Azam M, Wabaidur SM, Khan MR, Al-Resayes SI, Islam MS. Removal of Chromium(III) and Cadmium(II) Heavy Metal Ions from Aqueous Solutions Using Treated Date Seeds: An Eco-Friendly Method. Molecules. 2021; 26(12):3718. https://doi.org/10.3390/molecules26123718
Chicago/Turabian StyleAzam, Mohammad, Saikh Mohammad Wabaidur, Mohammad Rizwan Khan, Saud I. Al-Resayes, and Mohammad Shahidul Islam. 2021. "Removal of Chromium(III) and Cadmium(II) Heavy Metal Ions from Aqueous Solutions Using Treated Date Seeds: An Eco-Friendly Method" Molecules 26, no. 12: 3718. https://doi.org/10.3390/molecules26123718
APA StyleAzam, M., Wabaidur, S. M., Khan, M. R., Al-Resayes, S. I., & Islam, M. S. (2021). Removal of Chromium(III) and Cadmium(II) Heavy Metal Ions from Aqueous Solutions Using Treated Date Seeds: An Eco-Friendly Method. Molecules, 26(12), 3718. https://doi.org/10.3390/molecules26123718







