1. Introduction
Bentonite has been used worldwide as the protein fining agent in winemaking and its use can be dated back more than 80 years ago [
1]. In wine, the charge of proteins is determined by the relative relationship between pH and the isoelectric point (pI) of the protein. Proteins are positively charged when pH is below their pI. Bentonite provides a negatively charged surface to adsorb proteins by cation exchange. It is a mature technology to achieve protein stability of wine by adding sodium (or calcium) bentonite [
2,
3].
There are some drawbacks, however, in the use of bentonite. The separation of bentonite from wine is difficult and time-consuming. It can also cause significant loss of wine because about 3–10% of wine is absorbed by the bentonite slurry due to its significant swelling effect when hydrated in wine [
4]. The use of bentonite can lead to sub-optimal working conditions because of the potential for dust (e.g., silica) generation and create additional downstream processing to remove the solids remaining dispersed in wine [
5]. Due to its low density, bentonite takes 1–2 weeks of time to settle by gravity to the bottom of the tank. Thus, developing environmentally friendly alternatives to reduce both residues and wine loss has become vitally important so that wineries can minimize time and effort spent on removing bentonite residues in downstream processes and not contribute to creating more liquid and solid waste.
Fining agents, which can be regenerated and reused, can be developed to remove wine proteins more efficiently by reducing the time required and minimizing wine loss. Synthetic ion-exchange materials, such as resins developed for bio-tech once-through, packed-bed process operations [
6], have shown reasonable affinity and capacity for wine proteins in model wine solutions at bench-scale [
5,
7,
8]. The physical and chemical principles—those of ion-exchange—are at work in these synthetic materials and in bentonite. The use of an approach based on identical chemical and physical principles would facilitate more seamless integration, in contrast to using membranes, for instance, which can also remove additional large molecules such as polysaccharides and lead to other instabilities after bottling. While synthetic ion-exchange materials can be regenerated and reused in the pharmaceutical applications for which they were designed, data determining the stability of ion-exchange resins after use and regeneration in wine applications are lacking.
Macro-Prep
® High S by Bio-Rad is a non-swelling ion-exchange cellulose particle that has been proposed to be used as a bentonite alternative [
8,
9,
10]. It is a mature bio-tech application for protein purification as proteins can freely bind to and release from the outer surface of the Macro-Prep
® particles especially by changing process conditions between adsorption and desorption. Macro-Prep
® High S is a strong cation exchanger that contains sulfonate functional groups; the working hypothesis is that proteins can be adsorbed onto its surface from wine.
The approach will be to determine adsorption isotherm curve profiles for model wine solutions, which consist of different chemical components that are used to mimic a real wine. Because the sulfonate functional groups of Macro-Prep
® create a fixed number of cation exchange sites, it is anticipated that the adsorption isotherm and the fraction of sites occupied will be consistent with the Langmuir isotherm equation. The Langmuir isotherm explains the equilibrium found in adsorption with a finite number of sites at the monolayer level [
11]. The equilibrium between proteins in model wine solution and Macro-Prep
® is displayed by the Langmuir adsorption isotherm. The adsorption of protein from model wine solution by Macro-Prep
® particles has been shown to be consistent with the assumption of the Langmuir isotherm equation [
8]. Changes in the adsorption isotherms will be interpreted by determining parameters for adsorption affinity and capacity and by comparing to values that characterize adsorption from a simple model wine solution in the absence of additional prototypical wine compounds (e.g., phenolics).
To characterize the adsorption affinity and capacity of Macro-Prep
® High S in the presence of prototypical components of wine, a model protein such as Bovine Serum Albumin (BSA) can be used to quantify these adsorption parameters from model wine solutions [
10]. BSA, which has a molecular weight of around 66.5 kDa, is moderately non-reactive and has good solubility in water. It has been shown to have representative protein–phenolics interactions, albeit it under higher pH conditions (e.g., 5–7.5 pH) than present in wine (e.g., 3–4 pH), including with small phenolics such as caffeic acid and catechin [
12,
13,
14]. Interactions between proteins and other wine compounds, such as phenolics, have also been reported under conditions relevant to wine consumption; these often focus on proteins found in saliva or used for wine treatment [
15,
16]. BSA is chosen as a prototypical protein source because its isoelectric point (pI) in water is 4.3–4.8 [
17,
18,
19]; therefore, BSA is positively charged at the pH of wines, ranging from 3.4 to 3.8. The size and pI of BSA is, therefore, characteristic and representative of fractions that are present naturally in wine. BSA has been used as a prototypical protein in wine-like systems [
17,
18].
A potential benefit in using solid ion-exchange materials is that their use can be scaled in continuous processing to achieve efficiencies in time, wine loss, and water usage (for cleaning). The scale-up and commercialization of synthetic ion-exchange materials have been accomplished for non-wine applications. More recently, metal oxides such as zirconium oxide have been evaluated and have also shown promise. However, scaling-up, even for other protein-based applications, to a continuous process has not been successful due to lack of adsorption in pellet-form and difficulties in high-temperature regeneration [
20]. Regeneration protocols for winemaking applications need to be established, however, because wine contains ions and compounds not found in these other applications; work to determine the ability of these materials to be regenerated and reused needs to also be completed. To achieve the full benefits of solid materials, their translations to flow-through processes should be more thoroughly investigated.
In this work, we hypothesize that key classes of compounds found in wine will not have a significantly negative impact on adsorption, particularly under conditions of use for winemaking application. We therefore examine the impact of pH, ethanol concentration, prototypical phenolics and polysaccharide compounds to quantify their impact on adsorption affinity and capacity of Macro-Prep® High S ion-exchange resin. By systematically changing individual chemical components (variables), we generate and compare isotherm plots and quantify adsorption affinity or capacity parameters of Marco-Prep® High S ion-exchange resin.
2. Materials and Methods
2.1. Preparation of Model Wines
Protein-free model wine was prepared as follows in a 1 L flask: add 2 g of Potassium L-tartrate monobasic (KHT) (Sigma-Aldrich, USA) in 600 mL ultrapure deionized water (>18.0 MΩ resistivity) (Millipore, USA); mix the solution for 20 min until the KHT is dissolved; add 127 mL of 95% ethanol into the solution; add ultrapure deionized water into the volumetric flask until the water reaches the 1 L mark. Mix the solution for 20 min.
Model wine containing protein (1 g/L BSA) was prepared as follows in a 1 L Erlenmeyer flask: add 2 g of Potassium L-tartrate monobasic (KHT) (Sigma-Aldrich, USA) in 600 mL ultrapure deionized water; mix the solution for 20 min until KHT is dissolved; add 127 mL of 95% ethanol into the solution. Separately, in a 200 mL beaker, dissolve completely 1 g of OmniPur ® Bovine Serum Albumin (BSA) (Sigma-Aldrich, USA) in 100 mL ultrapure deionized water; transfer the BSA protein solution into the 1 L volumetric flask; add ultrapure deionized water (>18.0 MΩ resistivity) (Millipore, USA) into the volumetric flask until the water reaches the 1 L mark. Mix the solution for 20 min.
Adjustments to pH were made with small amounts of 1 M HCl (Millipore, USA) or 1 M NaOH (Sigma-Aldrich, USA). Ethanol concentrations were adjusted by changing the volume of 95% ethanol added into the solutions. Prototypical compounds representing important classes of compounds found in wines were added at the concentrations specified in
Table 1, including caffeic acid (Sigma-Aldrich, USA), catechin (Sigma-Aldrich, USA) and arabinogalactan (Spectrum Chemical, USA).
2.2. Experimental Design
The following variables were varied independently in the model wine solutions containing protein Bovine Serum Albumin (BSA): pH, ethanol concentration, caffeic acid (prototypical phenolic compound), catechin (prototypical phenolic compound), and arabinogalactan (prototypical polysaccharide), shown in
Table 1. For pH, ethanol concentration, and arabinogalactan variations, the concentrations of the protein were measured with a UV-Vis spectrometer at 280 nm as well as by using the Bradford protein assay along with a UV-Vis spectrometer at 595 nm. However, due to the phenolic ring structure of catechin and caffeic acid, which also absorb at 280 nm, the concentrations of proteins in experiments with these two compounds could only be quantified by using the Bradford protein assay. Three different values of pH and ethanol were used, while two values of caffeic acid, catechin, and arabinogalactan were investigated. The quantitative values for these independent variables are found in
Table 1.
2.3. Adsorption Isotherms
Macro-Prep® High S resin (Bio-Rad, USA) was dried in an oven at 35 °C for 24 h to enable more accurate quantification of the mass use to prepare the model wine solutions. Before transferring the resin into wine solution, the dried Macro-Prep® High S was weighed and subsequently rehydrated with 10 mL ultrapure deionized water for 10 min so that the rehydration condition of the resin is consistent. The resin was then placed into a 250 mL Erlenmeyer flask along with 150 mL of the previously prepared model wine solution (e.g., with a composition of 0.5 g/L OmniPur ® Bovine Serum Albumin (BSA) (Sigma-Aldrich, USA), 2 g/L Potassium L-tartrate monobasic (KHT) (Sigma-Aldrich, USA), 12 v/v % ethanol). Erlenmeyer flasks were placed onto a shaker table with the rotation speed set to 220 rpm for 4 h at room temperature. To collect samples for characterization, syringes and syringe filters (HPLV, 0.45 μm, VWR International, USA) were used to remove solid particles and clarify the model wine solution before subsequent characterization by UV-Vis and the Bradford assay. Samples from each Erlenmeyer flask were collected separately into a 15 mL centrifuge tube. Concentrations of BSA were determined using a UV-Vis spectrometer (ThermoScientific Genesys 10s UV-VIS, USA).
To prepare samples for characterization by the Bradford assay, a sample volume of 4.5 mL from each centrifuge tube was mixed with 0.9 mL Coomassie PlusTM Protein Assay Reagent (ThermoScientific, USA) in another 15 mL centrifuge tube. After 10 min at room temperature, samples were characterized by using the UV-Vis spectrometer at 595 nm.
4. Conclusions
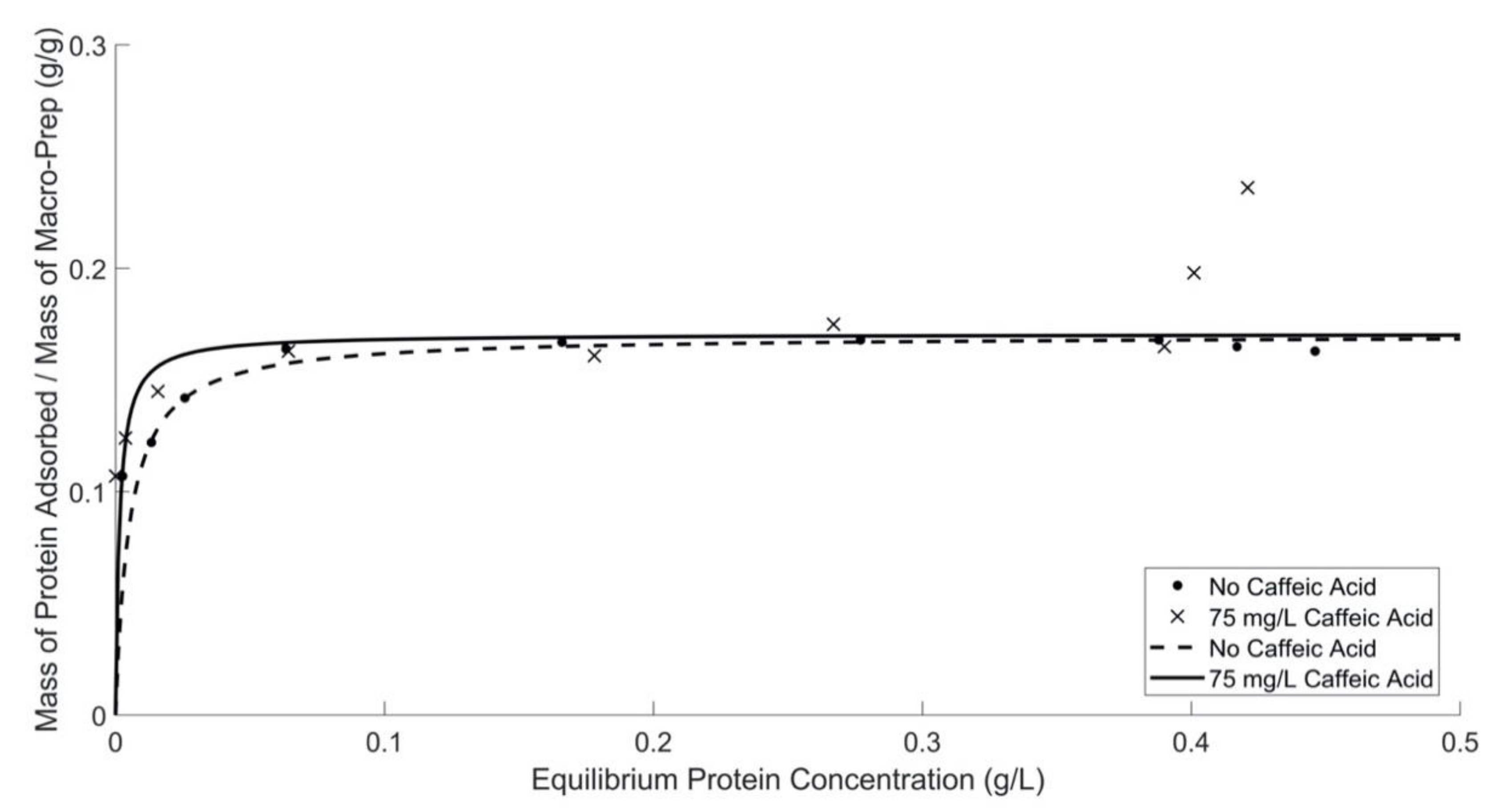
In conclusion, the Macro-Prep® High S resin has a stable performance (i.e., adsorption capacity and affinity) in different wine-like conditions. It is consistent with its potential use as a good replacement for bentonite as a protein fining agent. The protein adsorption capacity of Macro-Prep® High S resin at low protein equilibrium concentrations is negligibly affected by changing pH, changing ethanol concentration, or adding chemicals that represent compound classes commonly found in wine, such as caffeic acid and the model polysaccharide compound arabinogalactan. Typically, the protein adsorption behavior of Macro-Prep® High S follows the model of a Langmuir adsorption isotherm under these conditions.
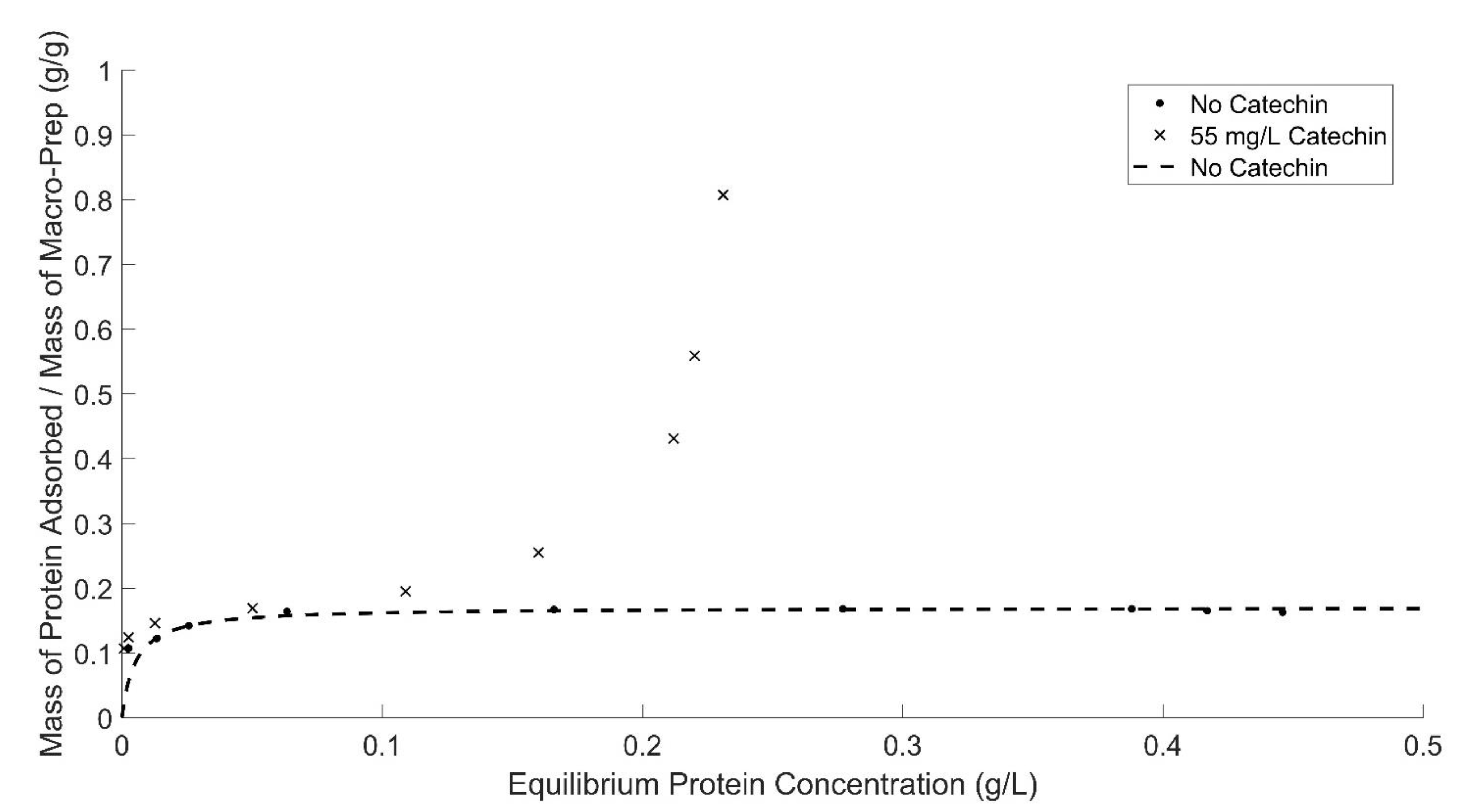
At high equilibrium protein concentrations (>0.3 (g BSA)/L), the protein adsorption behavior by Macro-Prep® changes with the addition of caffeic acid and polysaccharides and at higher ethanol concentrations. Macro-Prep® High S behaves differently under these conditions, likely due to Macro-Prep® acting as an unrestricted multilayer adsorbent. When the Macro-Prep® resin is in contact with a wine solution with low equilibrium protein concentration, Macro-Prep® will generally act as a monolayer adsorbent; however, when catechin is present, the adsorption behavior deviates from a Langmuir isotherm at low equilibrium protein concentrations of less than 0.2 (g BSA)/L. If the protein concentration in the wine solution is high under equilibrated conditions with the Macro-Prep®, the adsorption on the surface of the Macro-Prep® solid can reach saturation and the monolayer coverage reaches a maximum. This point is a critical point where Macro-Prep® can be taken as a mono-multilayer adsorbent. Above this critical point, Macro-Prep® solids, in the presence of wine compounds including caffeic acid and the model polysaccharide arabinogalactan, act as multilayer protein adsorbents. In this situation, both inter-molecule and intra-molecule adsorption can be observed simultaneously.
Differences in adsorption capacity between Macro-Prep® High S and bentonite, as well as any deviations from Langmuir isotherm behavior, can be used to inform design processes that use packed bed adsorption column for removing proteins from wines.