Ion Chromatography with Pulsed Amperometric Detection for Determining Cyanide in Urine and Meconium Samples
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Objectives
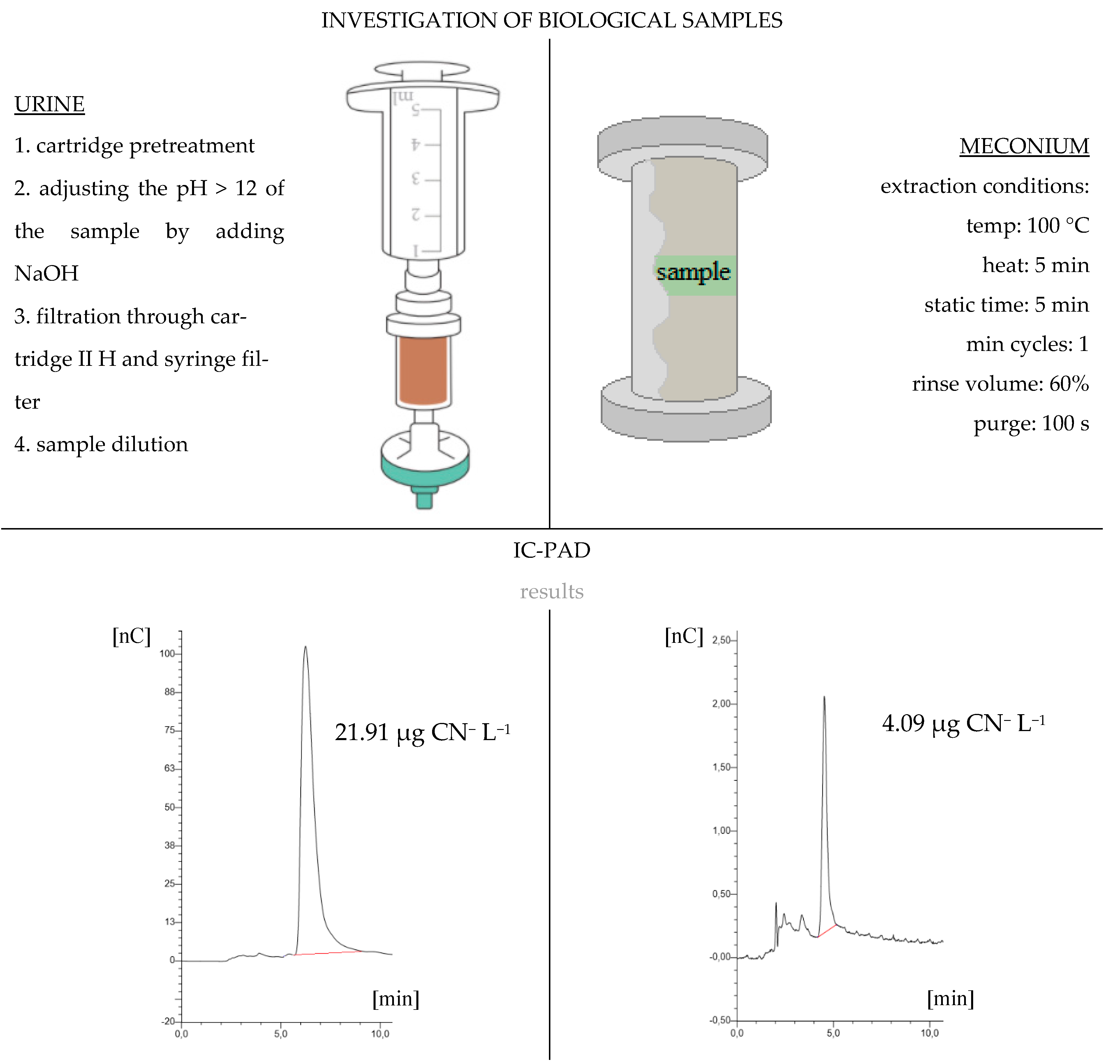
2.2. Sample Collection and Preparation
2.3. IC-PAD Parameters
3. Result and Discussion
Correlations between the Levels of Cyanide in Parents’ and Newborn’s Biological Samples
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Joo, S.H.; Hong, S.; Kim, N.J. Comparative study on Korean and international chemical control regulations of the physical hazards of sodium cyanide and hydrogen cyanide. J. Loss Prev. Process. Ind. 2016, 44, 143–149. [Google Scholar] [CrossRef]
- Brüger, A.; Fafilek, G.; Restrepo B., O.J.; Rojas-Mendoza, L. On the volatilisation and decomposition of cyanide contaminations from gold mining. Sci. Total Environ. 2018, 627, 1167–1173. [Google Scholar] [CrossRef] [PubMed]
- Luque-Almagro, V.M.; Moreno-Vivián, C.; Roldán, M.D. Biodegradation of cyanide wastes from mining and jewellery industries. Curr. Opin. Biotechnol. 2016, 38, 9–13. [Google Scholar] [CrossRef] [Green Version]
- Jaszczak, E.; Polkowska, Ż.; Narkowicz, S.; Namieśnik, J. Cyanides in the environment—analysis—problems and challenges. Environ. Sci. Pollut. Res. 2017, 24, 15929–15948. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bolarinwa, I.F.; Orfila, C.; Morgan, M.R.A. Amygdalin content of seeds, kernels and food products commercially- available in the UK. Food Chem. 2014, 152, 133–139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.; Zhang, G.; Wood, E.; Rogel Castillo, C.; Mitchell, A.E. Quantification of amygdalin in nonbitter, semibitter, and bitter almonds (prunus dulcis) by UHPLC-(ESI)QqQ MS/MS. J. Agric. Food Chem. 2013, 61, 7754–7759. [Google Scholar] [CrossRef]
- Senica, M.; Stampar, F.; Veberic, R.; Mikulic-Petkovsek, M. Fruit Seeds of the Rosaceae Family: A Waste, New Life, or a Danger to Human Health? J. Agric. Food Chem. 2017, 65, 10621–10629. [Google Scholar] [CrossRef] [PubMed]
- Blaheta, R.A.; Nelson, K.; Haferkamp, A.; Juengel, E. Amygdalin, quackery or cure? Phytomedicine 2016, 23, 367–376. [Google Scholar] [CrossRef]
- Napierala, M.; Mazela, J.; Merritt, T.A.; Florek, E. Tobacco smoking and breastfeeding: Effect on the lactation process, breast milk composition and infant development. A critical review. Environ. Res. 2016, 151, 321–338. [Google Scholar] [CrossRef]
- Hernández, A.F.; Lozano-Paniagua, D.; González-Alzaga, B.; Kavvalakis, M.P.; Tzatzarakis, M.N.; López-Flores, I.; Aguilar-Garduño, C.; Caparros-Gonzalez, R.A.; Tsatsakis, A.M.; Lacasaña, M. Biomonitoring of common organophosphate metabolites in hair and urine of children from an agricultural community. Environ. Int. 2019, 131, 104997. [Google Scholar] [CrossRef] [PubMed]
- Woźniak, M.K.; Jaszczak, E.; Wiergowski, M.; Polkowska, Ż.; Namieśnik, J.; Biziuk, M. Meconium analysis as a promising diagnostic tool for monitoring fetal exposure to toxic substances: Recent trends and perspectives. TrAC Trends Anal. Chem. 2018, 109, 124–141. [Google Scholar] [CrossRef]
- Zhang, Q.; Maddukuri, N.; Gong, M. A direct and rapid method to determine cyanide in urine by capillary electrophoresis. J. Chromatogr. A 2015, 1414, 158–162. [Google Scholar] [CrossRef] [Green Version]
- Jaszczak, E.; Kozioł, K.; Kiełbratowska, B.; Polkowska, Ż. Application of ion chromatography with pulsed amperometric detection for the determination of trace cyanide in biological samples, including breast milk. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2019, 1110–1111, 36–42. [Google Scholar] [CrossRef]
- Mani, J.; Rutz, J.; Maxeiner, S.; Juengel, E.; Bon, D.; Roos, F.; Chun, F.K.-H.; Blaheta, R.A. Cyanide and lactate levels in patients during chronic oral amygdalin intake followed by intravenous amygdalin administration. Complement. Ther. Med. 2019, 43, 295–299. [Google Scholar] [CrossRef]
- Vinnakota, C.V.; Peetha, N.S.; Perrizo, M.G.; Ferris, D.G.; Oda, R.P.; Rockwood, G.A.; Logue, B.A. Comparison of cyanide exposure markers in the biofluids of smokers and non-smokers. Biomarkers 2012, 17, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Narkowicz, S.; Polkowska, Z.; Kiełbratowska, B.; Namieśnik, J. Meconium samples used to assess infant exposure to the components of ETS during pregnancy. Int. J. Occup. Med. Environ. Health 2015, 28, 955–970. [Google Scholar] [CrossRef]
- Sundermann, T.R.; Schmitt, G.; Bartel, M. Modified congener analysis: Quantification of cyanide in whole blood, other body fluids, and diverse beverages. Drug Test. Anal. 2021, 13, 412–418. [Google Scholar] [CrossRef]
- Tabian, D.; Drochioiu, G.; Damian, S.I.; Girlescu, N.; Toma Gradinaru, O.; Toma, S.I.; Bulgaru Iliescu, D. Toxic blood hydrogen cyanide concentration as a vital sign of a deceased room fire victim-case report. Toxics 2021, 9, 36. [Google Scholar] [CrossRef] [PubMed]
- Wianowska, D.; Gil, M. Critical approach to PLE technique application in the analysis of secondary metabolites in plants. TrAC Trends Anal. Chem. 2019, 114, 314–325. [Google Scholar] [CrossRef]
- Roehsig, M.; De Paula, D.M.L.; Moura, S.; Diniz, E.M.D.A.; Yonamine, M. Determination of eight fatty acid ethyl esters in meconium samples by headspace solidphase microextraction and gas chromatography-Mass spectrometry. J. Sep. Sci. 2010, 33, 2115–2122. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Xu, M.; Alexander, R.R.; Bernert, J.T. Measurement of nicotine, cotinine and trans-3’-hydroxycotinine in meconium by liquid chromatography-tandem mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2011, 879, 2142–2148. [Google Scholar] [CrossRef] [PubMed]
- Choo, R.E.; Murphy, C.M.; Jones, H.E.; Huestis, M.A. Determination of methadone, 2-ethylidene-1,5-dimethyl-3,3- diphenylpyrrolidine, 2-ethyl-5-methyl-3,3-diphenylpyraline and methadol in meconium by liquid chromatography atmospheric pressure chemical ionization tandem mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2005, 814, 369–373. [Google Scholar] [CrossRef]
- Meyer-Monath, M.; Beaumont, J.; Morel, I.; Rouget, F.; Tack, K.; Lestremau, F. Analysis of BTEX and chlorinated solvents in meconium by headspace-solid-phase microextraction gas chromatography coupled with mass spectrometry. Anal. Bioanal. Chem. 2014, 406, 4481–4490. [Google Scholar] [CrossRef] [PubMed]
- Arbuckle, T.E.; Liang, C.L.; Morisset, A.S.; Fisher, M.; Weiler, H.; Cirtiu, C.M.; Legrand, M.; Davis, K.; Ettinger, A.S.; Fraser, W.D. Maternal and fetal exposure to cadmium, lead, manganese and mercury: The MIREC study. Chemosphere 2016, 163, 270–282. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Konieczka, P.; Namieśnik, J. Quality Assurance and Quality Control in the Analytical Chemical Laboratory: A practical Approach; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Jaszczak, E.; Ruman, M.; Narkowicz, S.; Namieśnik, J.; Polkowska, Ż. Development of an analytical protocol for determination of cyanide in human biological samples based on application of Ion Chromatography with Pulsed Amperometric Detection. J. Anal. Methods Chem. 2017, 2017, 7157953. [Google Scholar] [CrossRef]
- Rocklin, R.D.; Pohl, C.A. Determination of Carbohydrates by Anion Exchange Chromatography with Pulsed Amperometric Detection. J. Liq. Chromatogr. 1983, 6, 1577–1590. [Google Scholar] [CrossRef]
- National Research Council. Acute Exposure Guideline Levels for Selected Airborne Chemicals; The National Academies Press: Washington, DC, USA, 2002; Volume 2, ISBN 0309567734. [Google Scholar] [CrossRef]
- Valentín-Blasini, L.; Blount, B.C.; Delinsky, A. Quantification of iodide and sodium-iodide symporter inhibitors in human urine using ion chromatography tandem mass spectrometry. J. Chromatogr. A 2007, 1155, 40–46. [Google Scholar] [CrossRef]
- Błażewicz, A.; Klatka, M.; Dolliver, W.; Kocjan, R. Determination of total iodine in serum and urine samples by ion chromatography with pulsed amperometric detection—Studies on analyte loss, optimization of sample preparation procedures, and validation of analytical method. J. Chromatogr. B 2014, 962, 141–146. [Google Scholar] [CrossRef]



| n = 22 | |||
|---|---|---|---|
| Mothers | Fathers | ||
| Age | 21–30 | 50% | 18% |
| 31–40 | 45% | 68% | |
| 41< | 5% | 14% | |
| Education | secondary education | 9% | 32% |
| higher education | 91% | 68% | |
| Smoking status before pregnancy | active | 23% | 27% |
| non-smoking | 77% | 73% | |
| Smoking status during pregnancy | active | 5% | 5% (in the presence of pregnant women) |
| passive (at home) | 14% | 5% | |
| non-smoking | 95% | 95% | |
| Diet rich in almonds | yes | 68% | 36% |
| n = 18 | Newborn babies | ||
| Gender | Male | 8 | |
| Female | 10 | ||
| Mass [cm]/Weight [g] | Min | 49 | 2550 |
| Max | 67 | 3910 | |
| Average | 54.4 | 3333.89 | |
| Method of delivery | C-section | 50% | |
| Normal birth | 50% | ||
| Method of feeding | Breast milk | 39% | |
| Mixed | 61% | ||
| Breast milk volume | Large | 28% | |
| Sufficient | 61% | ||
| Small | 11% | ||
| Sample | Linear Range [µg L−1] | Curve Equation | R | SD | CV [%] | Recovery [%] | LOD [µg L−1] |
|---|---|---|---|---|---|---|---|
| Urine | 1–100 | y = 0.169 x − 0.304 | 0.992 | 0.003 | 1.63 | 80 | 1.8 |
| Meconium | 1–50 | y = 0.200 x − 0.165 | 0.986 | 0.033 | 0.37 | 79–83 | 0.58 |
| Type of Sample | Mean conc. [µg L−1] | SD [µg L−1] | Min conc. [µg L−1] | Max conc. [µg L−1] | |
|---|---|---|---|---|---|
| Mother’s urine (n = 11) | Active smoker (n = 4) | 14.3 | 9.19 | 8.42 | 25.2 |
| Passive smoker (n = 3) | 6.57 | 2.82 | 1.99 | 8.69 | |
| Control group (n = 4) | 3.12 | 1.37 | <LOD | 4.85 | |
| Father’s urine (n = 13) | Active smoker (n = 5) | 20.0 | 1.37 | 18.7 | 21.9 |
| Passive smoker (n = 3) | 5.71 | 1.66 | 4.11 | 8.00 | |
| Control group (n = 5) | 1.71 | 0.571 | <LOD | 2.61 | |
| Meconium (n = 11) | Smoking or passive smoking parents (n = 4) | 12.6 | 6.41 | 6.78 | 24.6 |
| Control group (n = 7) | 2.74 | 0.985 | <LOD | 4.09 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jaszczak-Wilke, E.; Kozioł, K.; Kiełbratowska, B.; Polkowska, Ż. Ion Chromatography with Pulsed Amperometric Detection for Determining Cyanide in Urine and Meconium Samples. Molecules 2021, 26, 4672. https://doi.org/10.3390/molecules26154672
Jaszczak-Wilke E, Kozioł K, Kiełbratowska B, Polkowska Ż. Ion Chromatography with Pulsed Amperometric Detection for Determining Cyanide in Urine and Meconium Samples. Molecules. 2021; 26(15):4672. https://doi.org/10.3390/molecules26154672
Chicago/Turabian StyleJaszczak-Wilke, Ewa, Krystyna Kozioł, Bogumiła Kiełbratowska, and Żaneta Polkowska. 2021. "Ion Chromatography with Pulsed Amperometric Detection for Determining Cyanide in Urine and Meconium Samples" Molecules 26, no. 15: 4672. https://doi.org/10.3390/molecules26154672






