Oral Supplementation with Hydrolyzed Fish Cartilage Improves the Morphological and Structural Characteristics of the Skin: A Double-Blind, Placebo-Controlled Clinical Study
Abstract
:1. Introduction
2. Results
2.1. Skin Microrelief
2.2. Dermis Echogenicity and Thickness
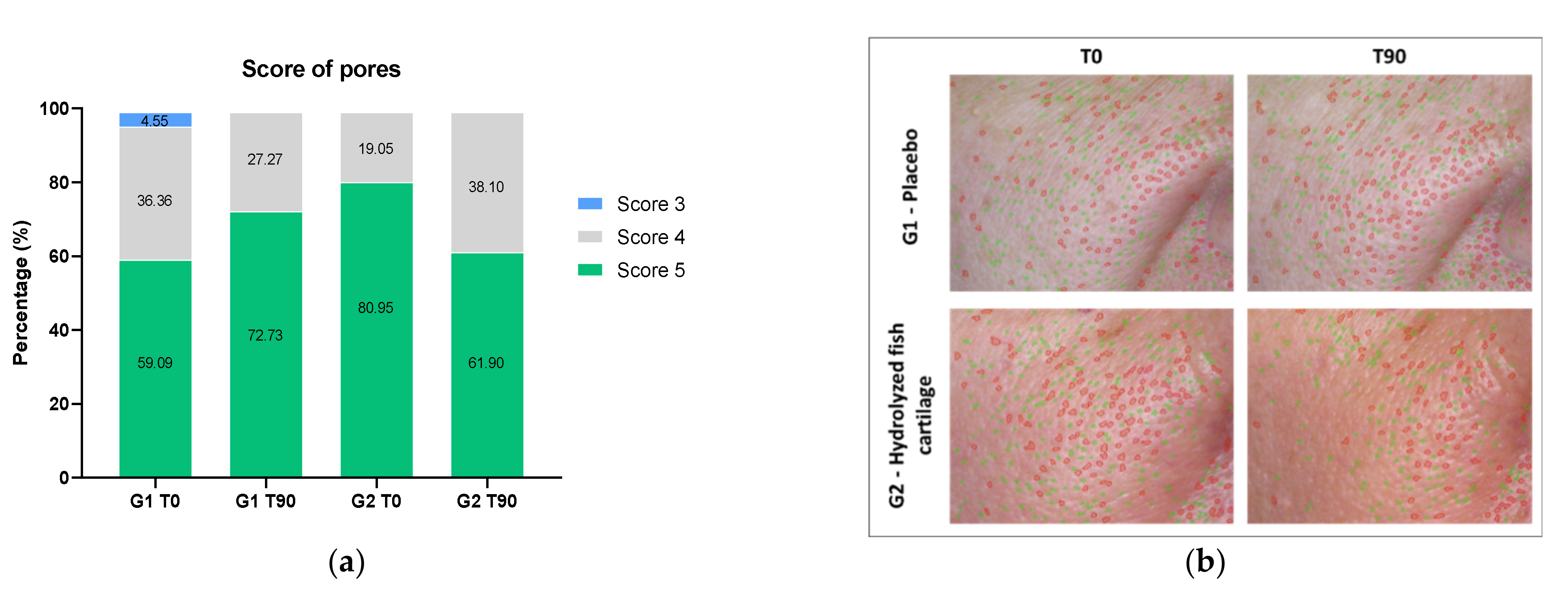
2.3. Skin Wrinkles and Pores Examined by High Definition Imaging Resolution
2.4. Morphological and Structural Skin Characteristics Determined by Reflectance Confocal Microscopy (RCM)
2.5. Perceived Efficacy
3. Discussion
4. Materials and Methods
4.1. Study Design
4.2. Study Participants
4.3. Study Products and Intervention
4.4. Instrumental Measurements
4.4.1. Skin Microrelief
4.4.2. Dermis Echogenicity and Thickness
4.4.3. Skin Wrinkles and Pores Examined by High Definition Imaging Resolution
4.4.4. Morphological and Structural Skin Characteristics Determined by Reflectance Confocal Microscopy
4.5. Perceived Efficacy
4.6. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Krutmann, J.; Bouloc, A.; Sore, G.; Bernard, B.A.; Passeron, T. The skin aging exposome. J. Derm. Sci. 2017, 85, 152–161. [Google Scholar] [CrossRef] [Green Version]
- Mercurio, D.G.; Jdid, R.; Morizot, F.; Masson, P.; Maia Campos, P.M.B.G. Morphological, structural and biophysical properties of French and Brazilian photoaged skin. Br. J. Derm. 2016, 61, 174–553. [Google Scholar] [CrossRef]
- Ali, A.; Khan, H.; Bahadar, R.; Riaz, A.; Asad, M.H.H.B. The impact of airborne pollution and exposure to solar ultraviolet radiation on skin: Mechanistic and physiological insight. Environ. Sci. Pollut. Res. 2020, 27, 28730–28736. [Google Scholar] [CrossRef] [PubMed]
- Rinnerthaler, M.; Bischof, J.; Streubel, M.K.; Trost, A.; Richter, K. Oxidative stress in aging human skin. Biomolecules 2015, 5, 545. [Google Scholar] [CrossRef] [Green Version]
- Fossa Shirata, M.M.; Alves, G.A.D.; Maia Campos, P.M.B.G. Photoaging-related skin changes in different age groups: A clinical evaluation by biophysical and imaging techniques. Int. J. Cosmet. Sci. 2019, 41, 265–273. [Google Scholar] [CrossRef]
- Žmitek, K.; Žmitek, J.; Rogl Butina, M.; Pogačnik, T. Effects of a Combination of Water-Soluble Coenzyme Q10 and Collagen on Skin Parameters and Condition: Results of a Randomised, Placebo-Controlled, Double-Blind Study. Nutrients 2020, 12, 618. [Google Scholar] [CrossRef] [Green Version]
- Czajka, A.; Kania, E.M.; Genovese, L.; Corbo, A.; Merone, G.; Luci, C.; Sibilla, S. Daily oral supplementation with collagen peptides combined with vitamins and other bioactive compounds improves skin elasticity and has a beneficial effect on joint and general wellbeing. Nutr. Res. 2018, 57, 97–108. [Google Scholar] [CrossRef]
- Maia Campos, P.M.B.G.; Melo, M.O.; Siqueira César, F.C. Topical application and oral supplementation of peptides in the improvement of skin viscoelasticity and density. J. Cosmet. Derm. 2019, 18, 1693–1699. [Google Scholar] [CrossRef]
- AlAli, M.; Alqubaisy, M.; Aljaafari, M.N.; AlAli, A.O.; Baqais, L.; Molouki, A.; Abushelaibi, A.; Lai, K.-S.; Lim, S.-H.E. Nutraceuticals: Transformation of Conventional Foods into Health Promoters/Disease Preventers and Safety Considerations. Molecules 2021, 26, 2540. [Google Scholar] [CrossRef]
- Proksch, E.; Segger, D.; Degwert, J.; Schunck, M.; Zague, V.; Oesser, S. Oral supplementation of specific collagen peptides has beneficial effects on human skin physiology: A double-blind, placebo-controlled study. Ski. Pharm. Physiol. 2014, 27, 47–55. [Google Scholar] [CrossRef]
- Genovese, L.; Corbo, A.; Sibilla, S. An insight into the changes in skin texture and properties following dietary intervention with a nutricosmeceutical containing a blend of collagen bioactive peptides and antioxidants. Ski. Pharm. Physiol. 2017, 30, 146–158. [Google Scholar] [CrossRef] [Green Version]
- Asserin, J.; Lati, E.; Shioya, T.; Prawitt, J. The effect of oral collagen peptide supplementation on skin moisture and the dermal collagen network: Evidence from an ex vivo model and randomized, placebo-controlled clinical trials. J. Cosmet. Derm. 2015, 14, 291–301. [Google Scholar] [CrossRef] [Green Version]
- Vollmer, D.L.; West, V.A.; Lephart, E.D. Enhancing skin health: By oral administration of natural compounds and minerals with implications to the dermal microbiome. Int. J. Mol. Sci. 2018, 19, 3059. [Google Scholar] [CrossRef] [Green Version]
- Aguirre-Cruz, G.; León-López, A.; Cruz-Gómez, V.; Jiménez-Alvarado, R.; Aguirre-Álvarez, G. Collagen Hydrolysates for Skin Protection: Oral Administration and Topical Formulation. Antioxidants 2020, 9, 181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maia Campos, P.M.B.G.; Favaretto, G. Influence of an Oral Supplementation Based on Orthosilicic Acid Choline-Stabilized on Skin, Hair and Nails: A Clinical Study with Objective Approach. Clin. Pharm. Biopharma. 2016, 5, 1–6. [Google Scholar] [CrossRef]
- Manfredini, M.; Mazzaglia, G.; Ciardo, S.; Simonazzi, S.; Farnetani, F.; Longo, C.; Pellacani, G. Does skin hydration influence keratinocyte biology? In vivo evaluation of microscopic skin changes induced by moisturizers by means of Reflectance Confocal Microscopy. Ski. Res. Technol. 2013, 19, 299–307. [Google Scholar]
- Longo, C.; Casari, A.; Pace, B.D.; Simonazzi, S.; Mazzaglia, G.; Pellacani, G. Proposal for an in vivo histopathologic scoring system for skin aging by means of confocal microscopy. Ski. Res. Technol. 2013, 19, 167–173. [Google Scholar] [CrossRef]
- Longo, C.; Casari, A.; Beretti, F.; Cesinaro, A.M.; Pellacani, G. Skin aging: In vivo microscopic assessment of epidermal and dermal changes by means of confocal microscopy. J. Am. Acad. Derm. 2013, 68, 73–82. [Google Scholar] [CrossRef]
- Bolke, L.; Schlippe, G.; Gerß, J.; Voss, W. A Collagen Supplement Improves Skin Hydration, Elasticity, Roughness, and Density: Results of a Randomized, Placebo-Controlled, Blind Study. Nutrients 2019, 11, 2494. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Wang, Q.; Qian, J.; Liang, Q.; Wang, Z.; Xu, J.; He, S.; Ma, H. Bioavailability and bioavailable forms of collagen after oral administration to rats. J. Agric. Food Chem. 2015, 95, 2712–2717. [Google Scholar] [CrossRef]
- Liu, D.; Nikoo, M.; Boran, G.; Zhou, P.; Regenstein, J.M. Collagen and gelatin. Annu. Rev. Food Sci. Technol. 2015, 6, 527–557. [Google Scholar] [CrossRef] [PubMed]
- Ohara, H.; Matsuoto, H.; Ito, K.; Iwai, K.; Sato, K. Comparison of quantity and structures of hydroxyproline-containing peptides in human blood after oral ingestion of gelatin hydrolysates from different sources. J. Agric. Food Chem. 2007, 55, 1532–1535. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, S.R.; Park, J. Ingestion of BioCell Collagen®, a novel hydrolyzed chicken sternal cartilage extract; enhanced blood microcirculation and reduced facial aging signs. Clin. Interv. Aging 2012, 7, 267–273. [Google Scholar] [PubMed] [Green Version]
- Chen, Q.; Hou, H.; Wang, S.; Zhao, X.; Li, B. Effects of early enteral nutrition supplemented with collagen peptides on post-burn inflammatory responses in a mouse model. Food Func 2017, 8, 1933–1941. [Google Scholar] [CrossRef]
- Song, H.; Zhang, L.; Luo, Y.; Zhang, S.; Li, B. Effects of collagen peptides intake on skin ageing and platelet release in chronologically aged mice revealed by cytokine array analysis. J. Cell Mol. Med. 2018, 22, 277–288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, P.; Alexander, R.A.; Liang, C.H.; Liu, C.; Lin, Y.H.; Lin, Y.H.; Kuan, C.M. Collagen formula with Djulis for improvement of skin hydration, brightness, texture, crow’s feet, and collagen content: A double-blind, randomized, placebo-controlled trial. J. Cosmet. Derm. 2021, 20, 188–194. [Google Scholar] [CrossRef]
- Borumand, M.; Sibilla, S. Daily consumption of the collagen supplement Pure Gold Collagen® reduces visible signs of aging. Clin. Interv. Aging 2014, 9, 1747–1758. [Google Scholar] [CrossRef] [Green Version]
- De Miranda, R.B.; Weimer, P.; Rossi, R.C. Effects of hydrolyzed collagen supplementation on skin aging: A systematic review and meta-analysis. Int J. Derm. 2021. [Google Scholar] [CrossRef]
- The International Conference on Harmonization of Technical Requirements for the Registration of Pharmaceuticals for Human Use (ICH). Integrated addendum to ICH E6(R1): Guideline for good clinical practice E6(R2). 2016. Available online: https://database.ich.org/sites/default/files/E6_R2_Addendum.pdf (accessed on 23 June 2020).
- World Medical Association. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef] [Green Version]
- Maia Campos, P.M.B.G.; Melo, M.O.; Mercurio, D.G. Use of advanced imaging techniques for the characterization of oily skin. Front. Physiol. 2019, 10, 1–9. [Google Scholar] [CrossRef]
- Mercúrio, D.G.; Segura, J.H.; Maia Campos, P.M.B.G. Clinical scoring and instrumental analysis to evaluate skin types. Clin. Exp. Derm. 2013, 38, 302–309. [Google Scholar] [CrossRef]
- Melo, M.O.; Maia Campos, P.M.B.G. Characterization of oily mature skin by biophysical and skin imaging techniques. Ski. Res. Technol. 2018, 24, 1–10. [Google Scholar] [CrossRef] [PubMed]










| Placebo Group (n = 6) | Hydrolyzed Fish Cartilage (n = 6) | |||||
|---|---|---|---|---|---|---|
| 1 (µm) | T0 | T90 | p Value | T0 | T90 | p Value |
| Stratum Corneum Thickness | 14.83 ± 1.86 | 16.33 ± 2.40 | 0.1362 | 15.00 ± 1.76 | 17.67 ± 1.33 | 0.0086 * |
| Granular Layer Thickness | 17.00 ± 2.00 | 18.50 ± 2.35 | 0.3943 | 17.83 ± 1.83 | 20.50 ± 2.74 | 0.1055 |
| Dermal Papilla Depth | 17.50 ± 5.05 | 16.83 ± 3.37 | 1.0000 | 13.83 ± 5.00 | 13.47 ± 4.44 | 0.8287 |
| Minimum Epidermis Thickness | 21.17 ± 2.99 | 24.17 ± 2.71 | 0.0756 | 25.00 ± 5.97 | 28.50 ± 3.21 | 0.3072 |
| Maximum Epidermis Thickness | 38.67 ± 7.45 | 41.00 ± 4.94 | 0.3968 | 38.83 ± 7.88 | 41.97 ± 6.68 | 0.2777 |
| Mean Epidermis Thickness | 29.92 ± 5.08 | 32.58 ± 3.61 | 0.2296 | 31.92 ± 6.53 | 35.24 ± 4.75 | 0.2887 |
| Total Epidermis Thickness | 53.50 ± 8.58 | 57.33 ± 4.31 | 0.2647 | 53.83 ± 8.98 | 59.64 ± 7.33 | 0.0916 |
| Placebo (n = 6) | Hydrolyzed Fish Cartilage (n = 6) | |||||
|---|---|---|---|---|---|---|
| RCM Descriptors | T0 | T90 | p Value | T0 | T90 | p Value |
| Irregular honeycomb pattern | 67 | 33 | 0.0001 * | 83 | 33 | 0.0001 * |
| Low interkeratinocyte reflectance | 33 | 17 | 0.0138 * | 33 | 0 | 0.0001 * |
| Polycyclic papillary contours | 83 | 83 | 1.0000 | 67 | 67 | 1.0000 |
| Effacement of rete ridges | 17 | 17 | 1.0000 | 50 | 50 | 1.0000 |
| Coarse collagen structures | 67 | 67 | 1.0000 | 83 | 100 | 0.0001 * |
| Huddled collagen structures | 33 | 33 | 1.0000 | 17 | 0 | 0.0001 * |
| Curled bright structures Elastosis | 50 | 50 | 1.0000 | 83 | 67 | 0.0138 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maia Campos, P.M.B.G.; Franco, R.S.B.; Kakuda, L.; Cadioli, G.F.; Costa, G.M.D.; Bouvret, E. Oral Supplementation with Hydrolyzed Fish Cartilage Improves the Morphological and Structural Characteristics of the Skin: A Double-Blind, Placebo-Controlled Clinical Study. Molecules 2021, 26, 4880. https://doi.org/10.3390/molecules26164880
Maia Campos PMBG, Franco RSB, Kakuda L, Cadioli GF, Costa GMD, Bouvret E. Oral Supplementation with Hydrolyzed Fish Cartilage Improves the Morphological and Structural Characteristics of the Skin: A Double-Blind, Placebo-Controlled Clinical Study. Molecules. 2021; 26(16):4880. https://doi.org/10.3390/molecules26164880
Chicago/Turabian StyleMaia Campos, Patrícia Maria Berardo Gonçalves, Rodolfo Scarpino Barboza Franco, Letícia Kakuda, Gabriel Fernandes Cadioli, Gabriela Maria D’Angelo Costa, and Elodie Bouvret. 2021. "Oral Supplementation with Hydrolyzed Fish Cartilage Improves the Morphological and Structural Characteristics of the Skin: A Double-Blind, Placebo-Controlled Clinical Study" Molecules 26, no. 16: 4880. https://doi.org/10.3390/molecules26164880






