Recent Progress Concerning the N-Arylation of Indoles
Abstract
:1. Introduction
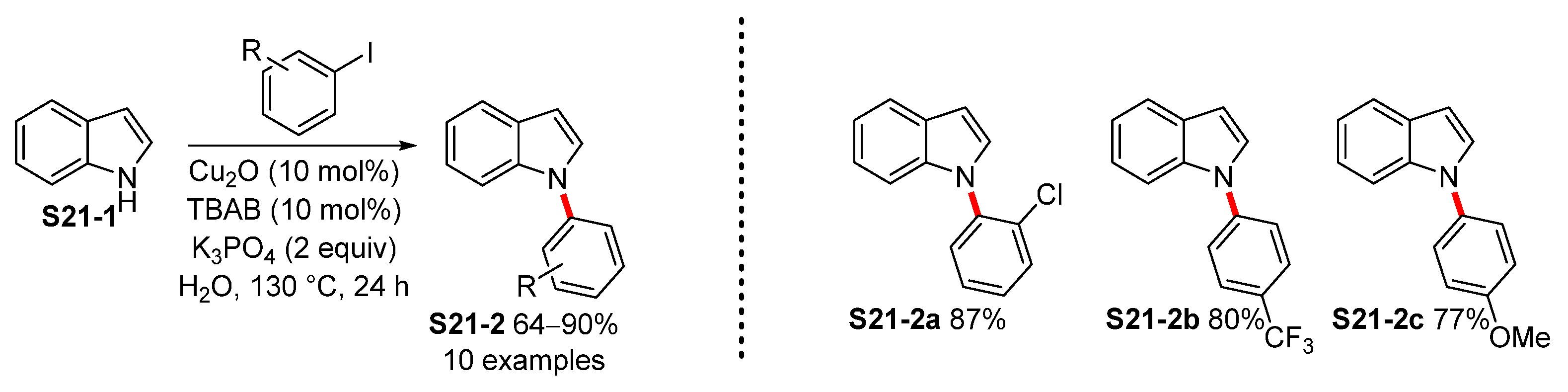
2. Transition-Metal-Free N-Arylation of Indoles
3. Transition-Metal-Catalyzed N-Arylation of Indoles
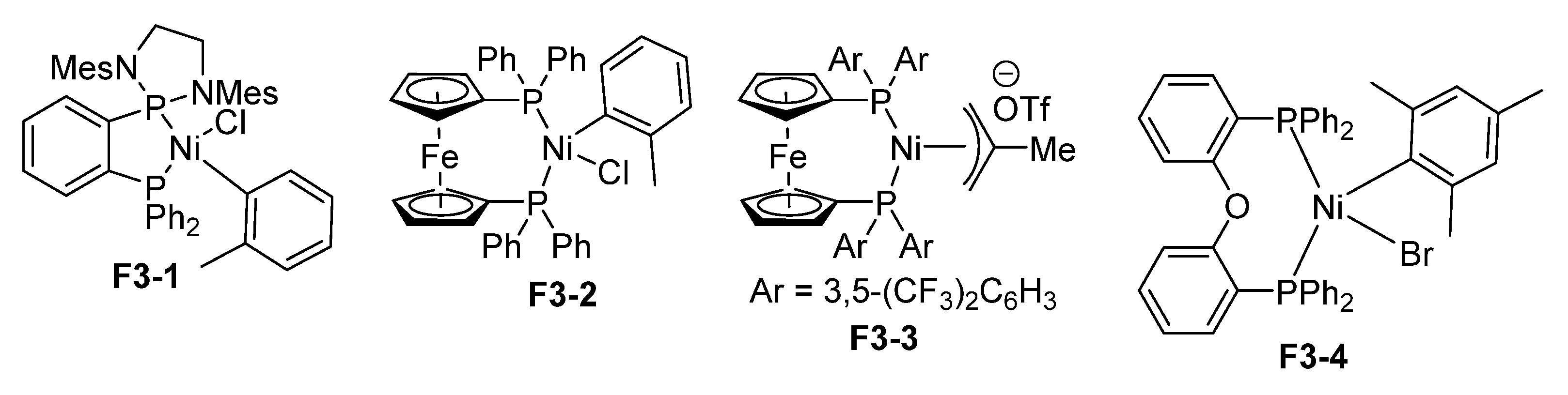
3.1. Nickel-Catalyzed N-Arylation of Indoles
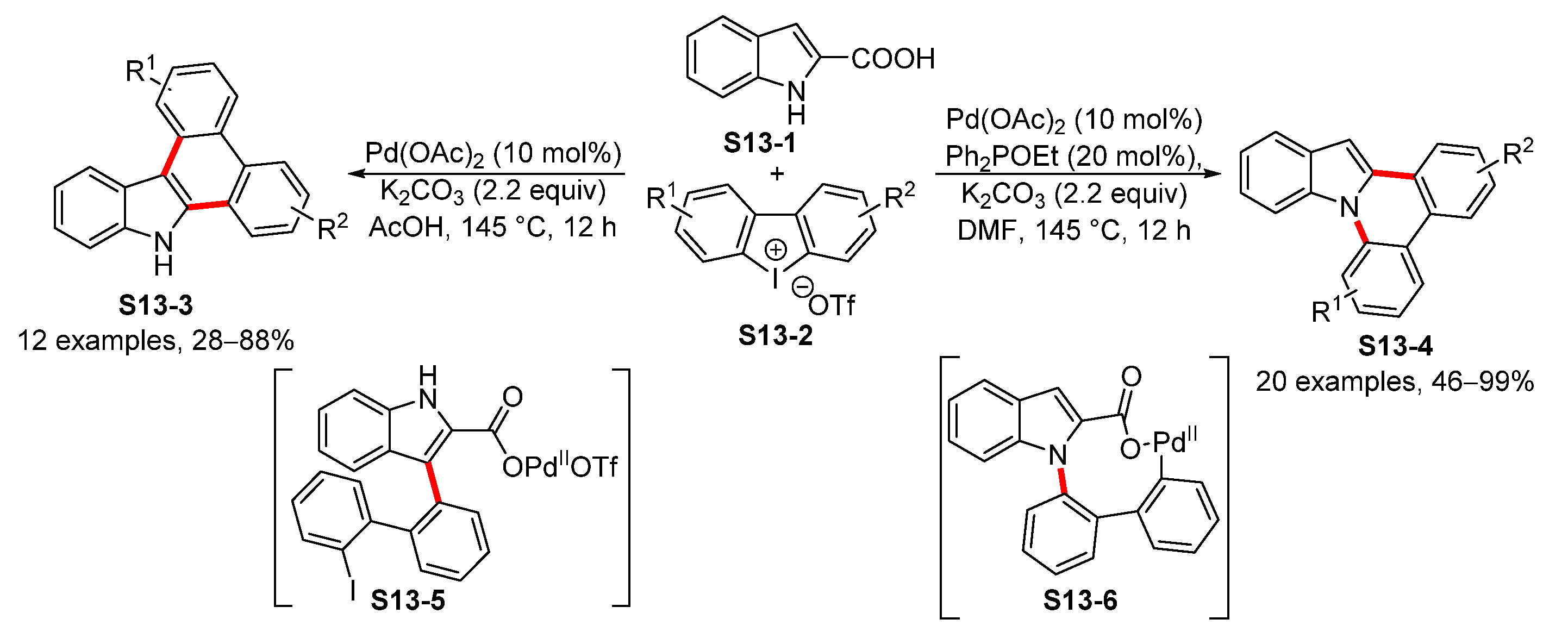
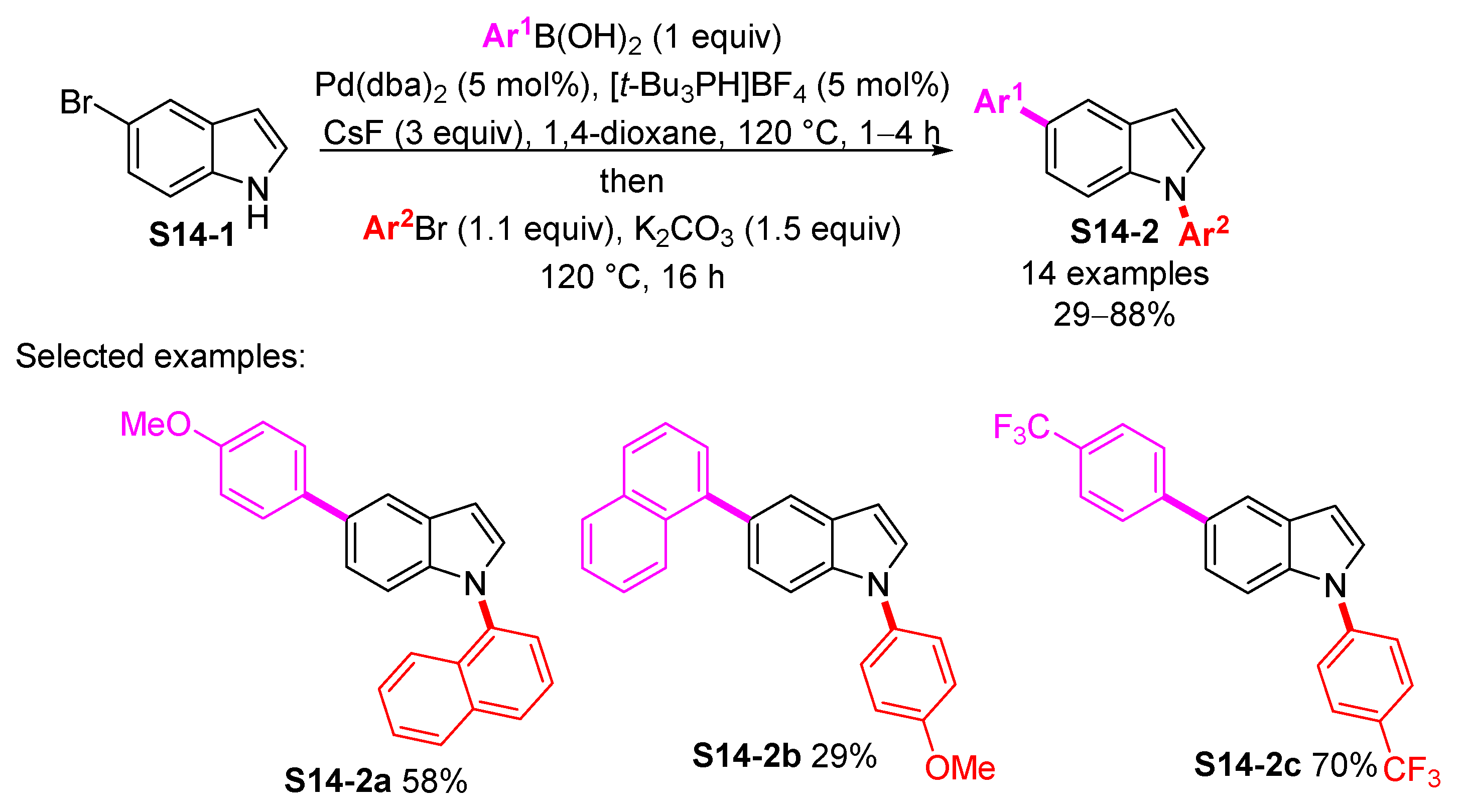
3.2. Palladium-Catalyzed N-Arylation of Indoles
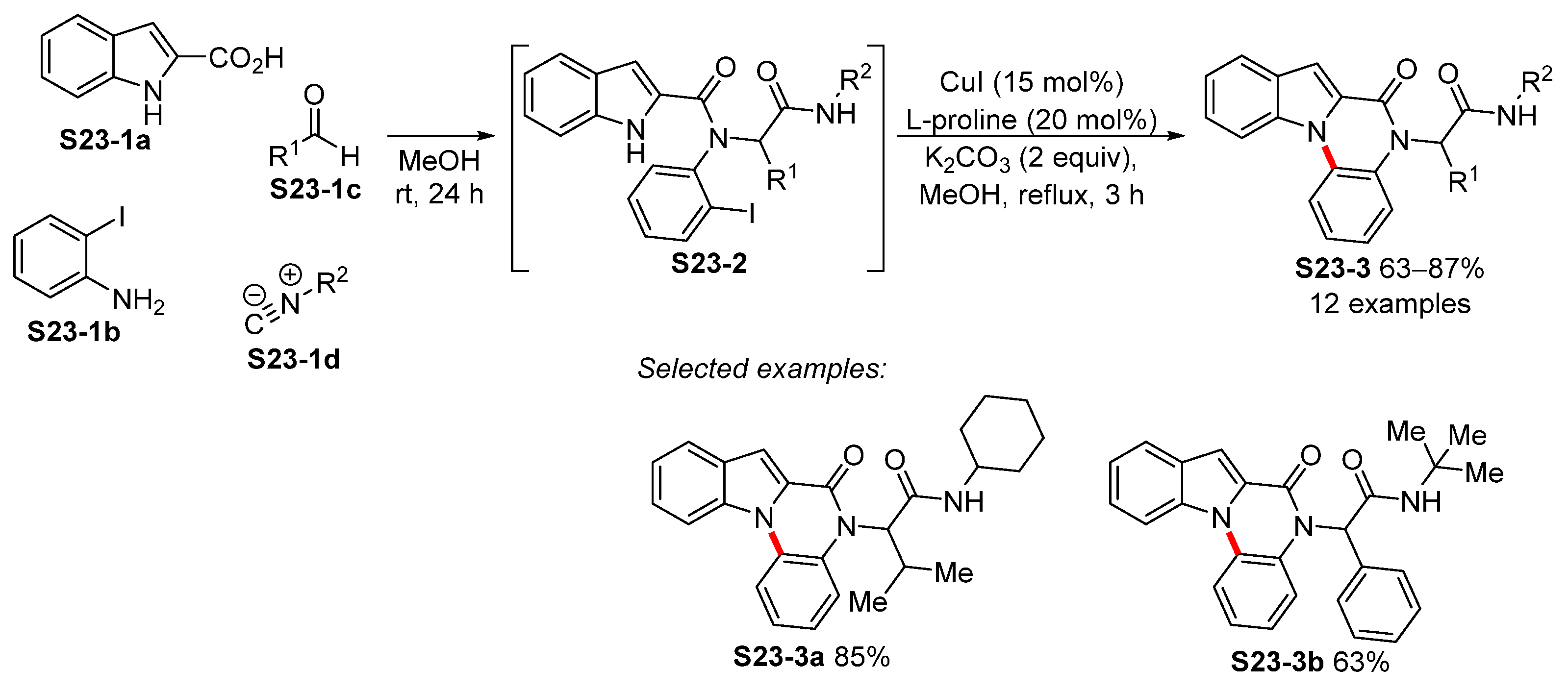
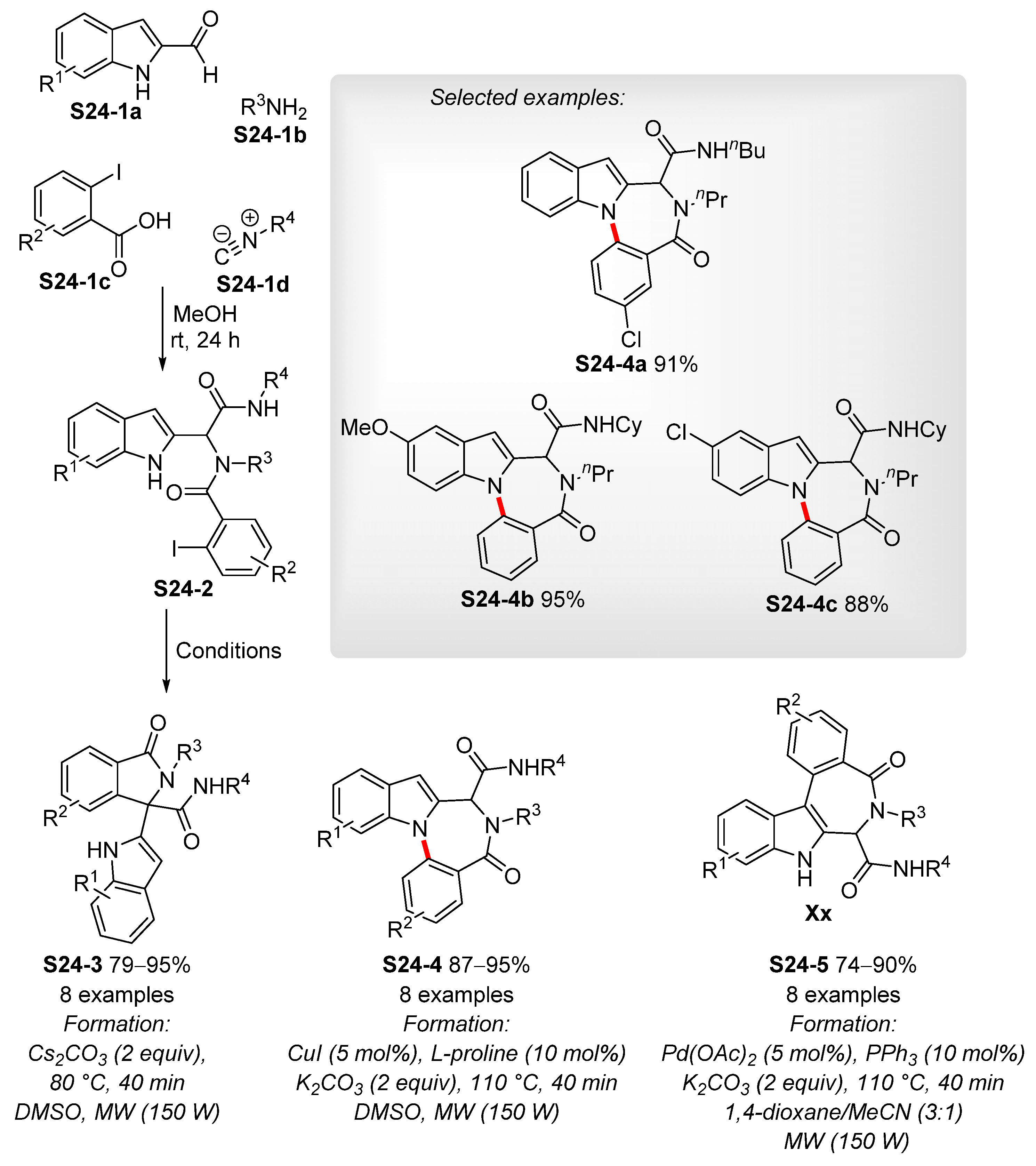
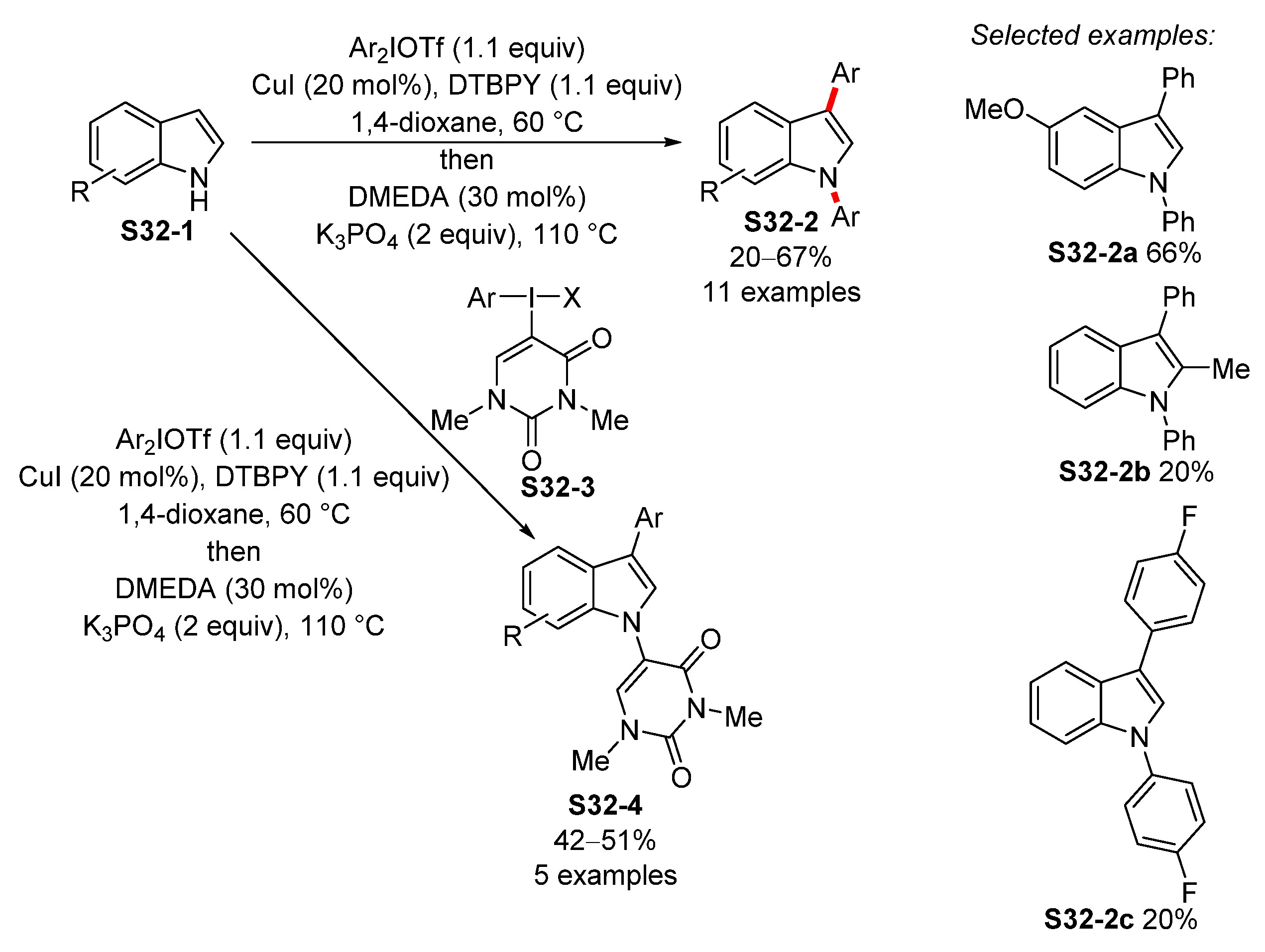
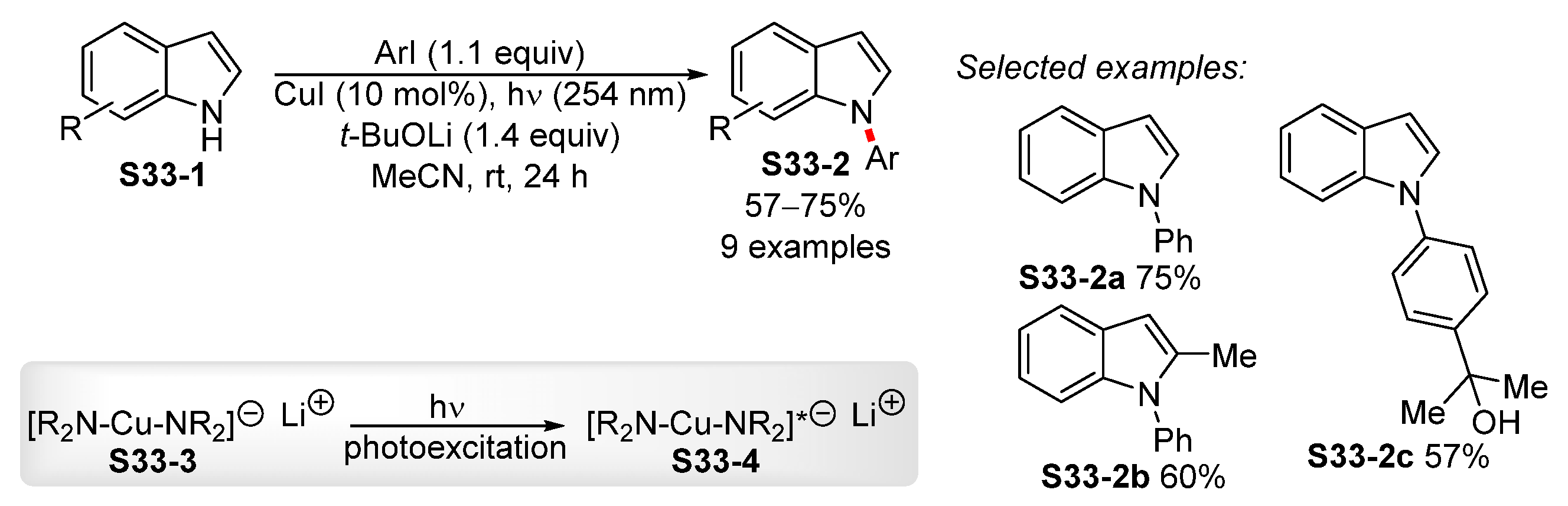
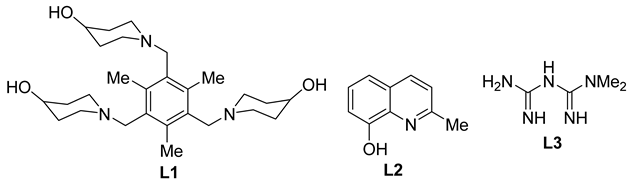
3.3. Copper-Catalyzed N-Arylation of Indoles
3.4. Miscellaneous Transition-Metal-Catalyzed N–Arylation of Indoles
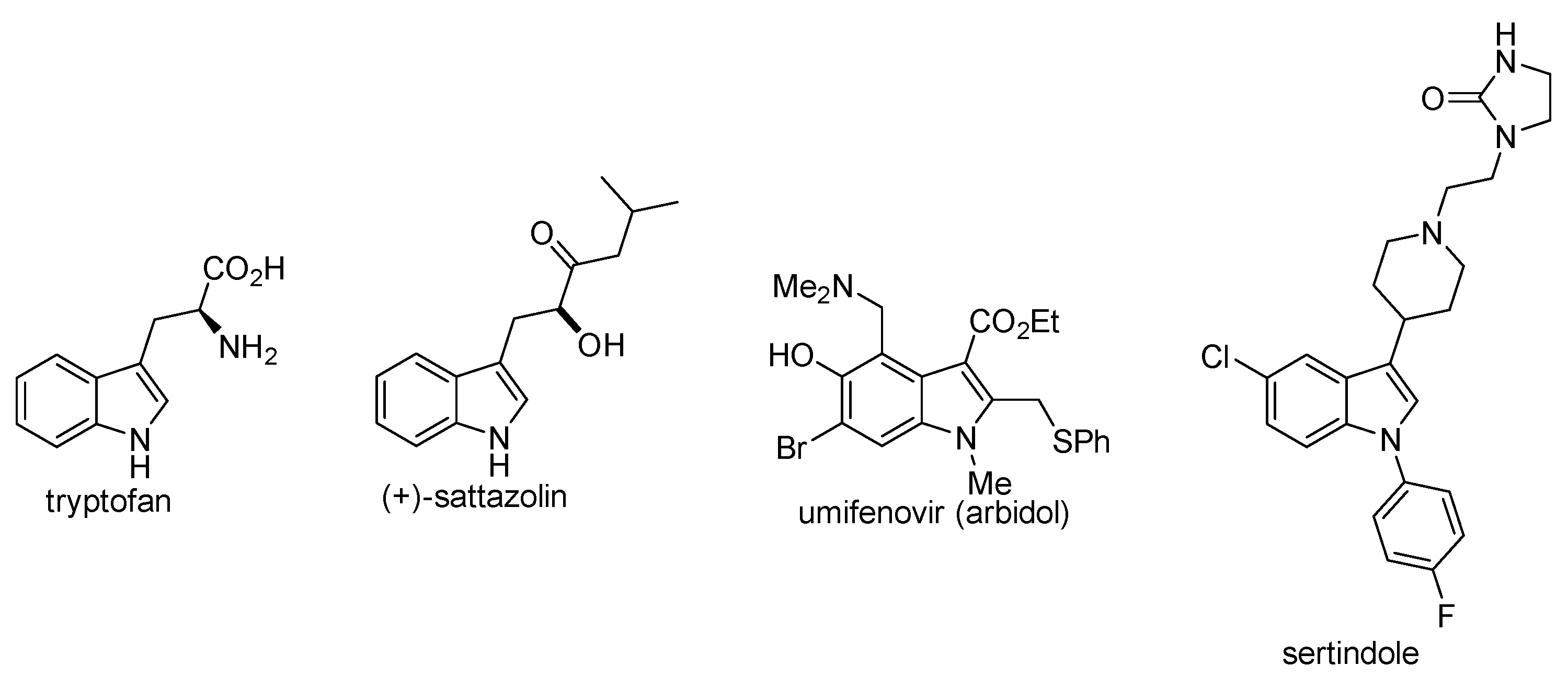
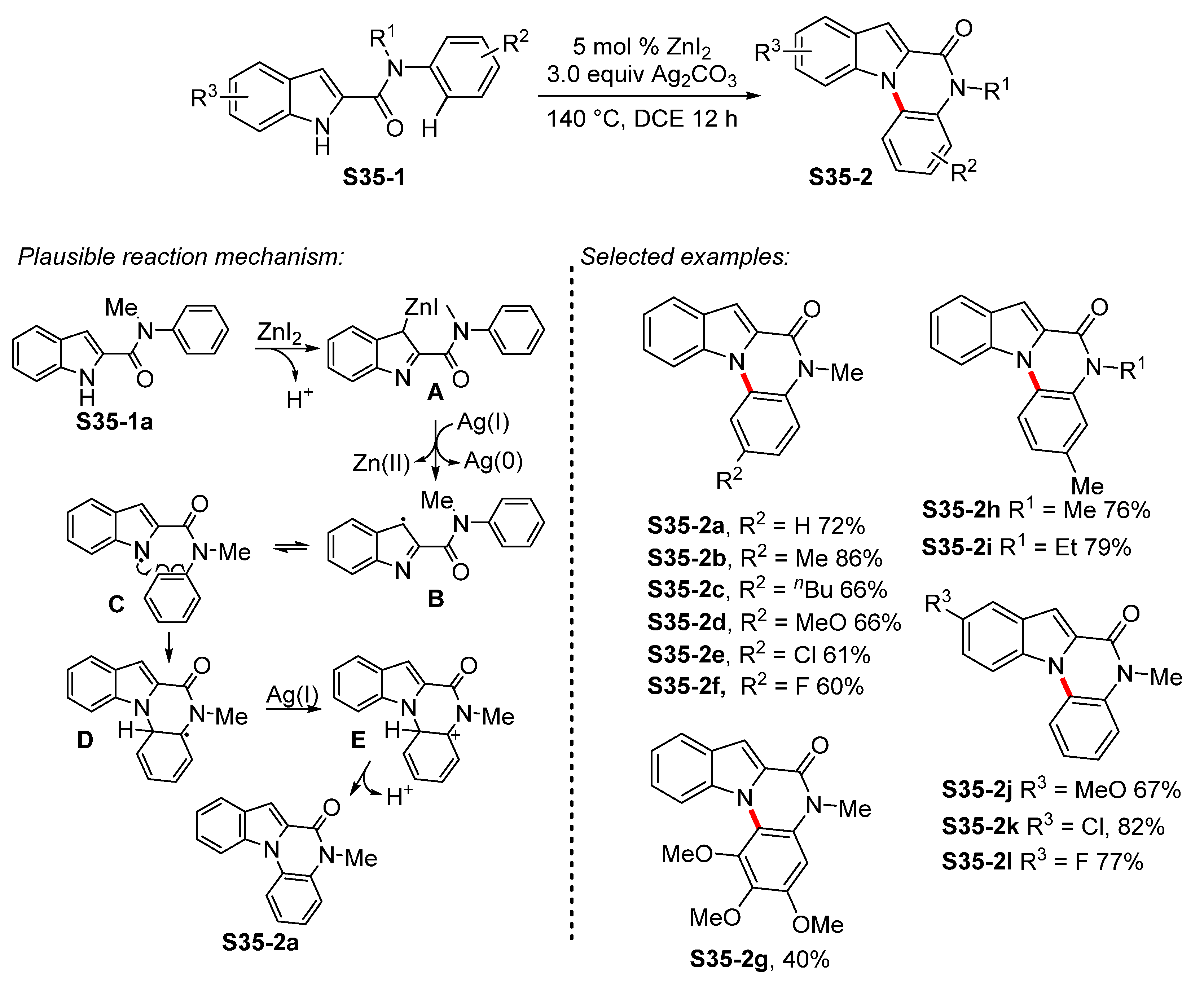
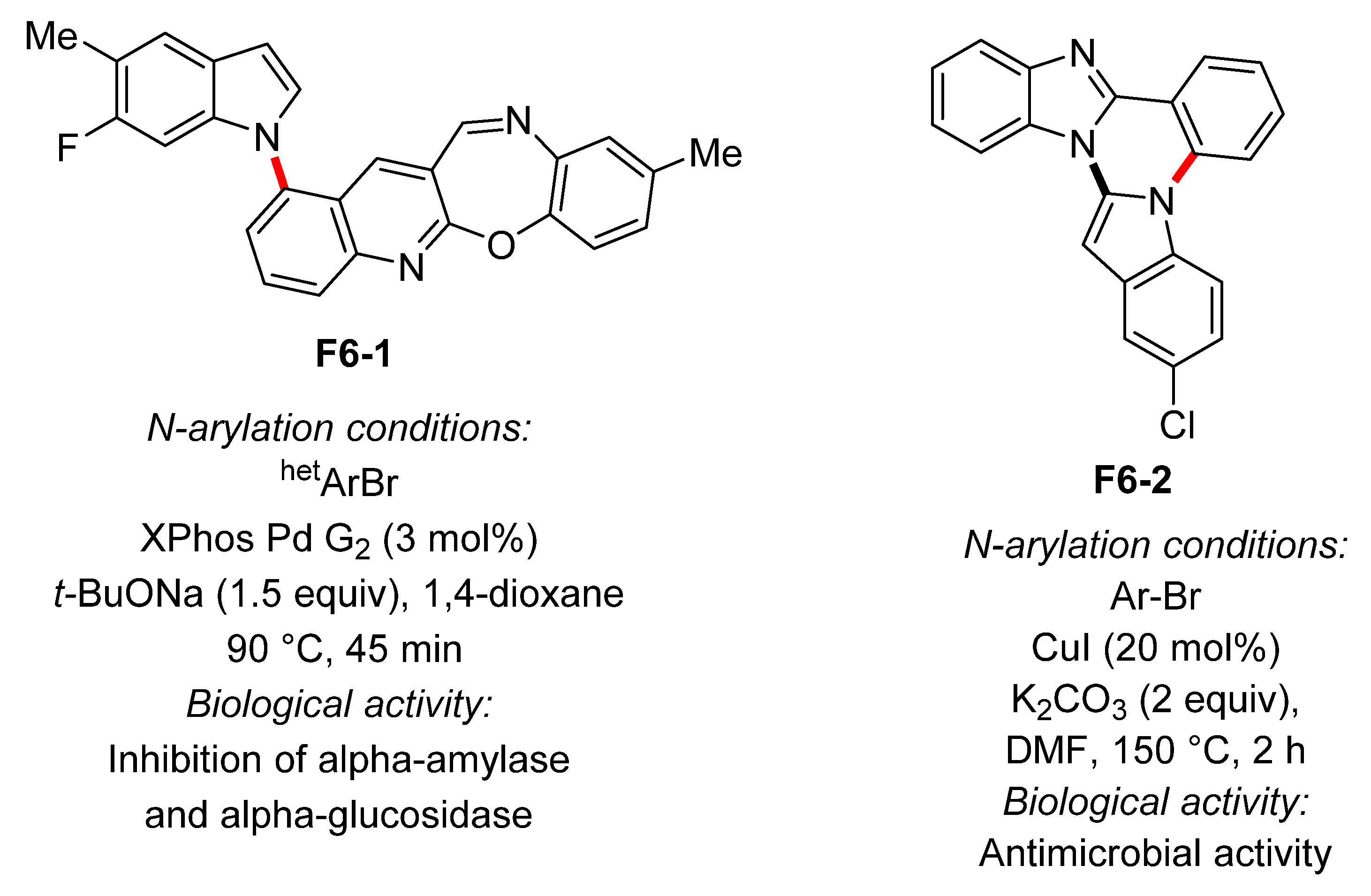
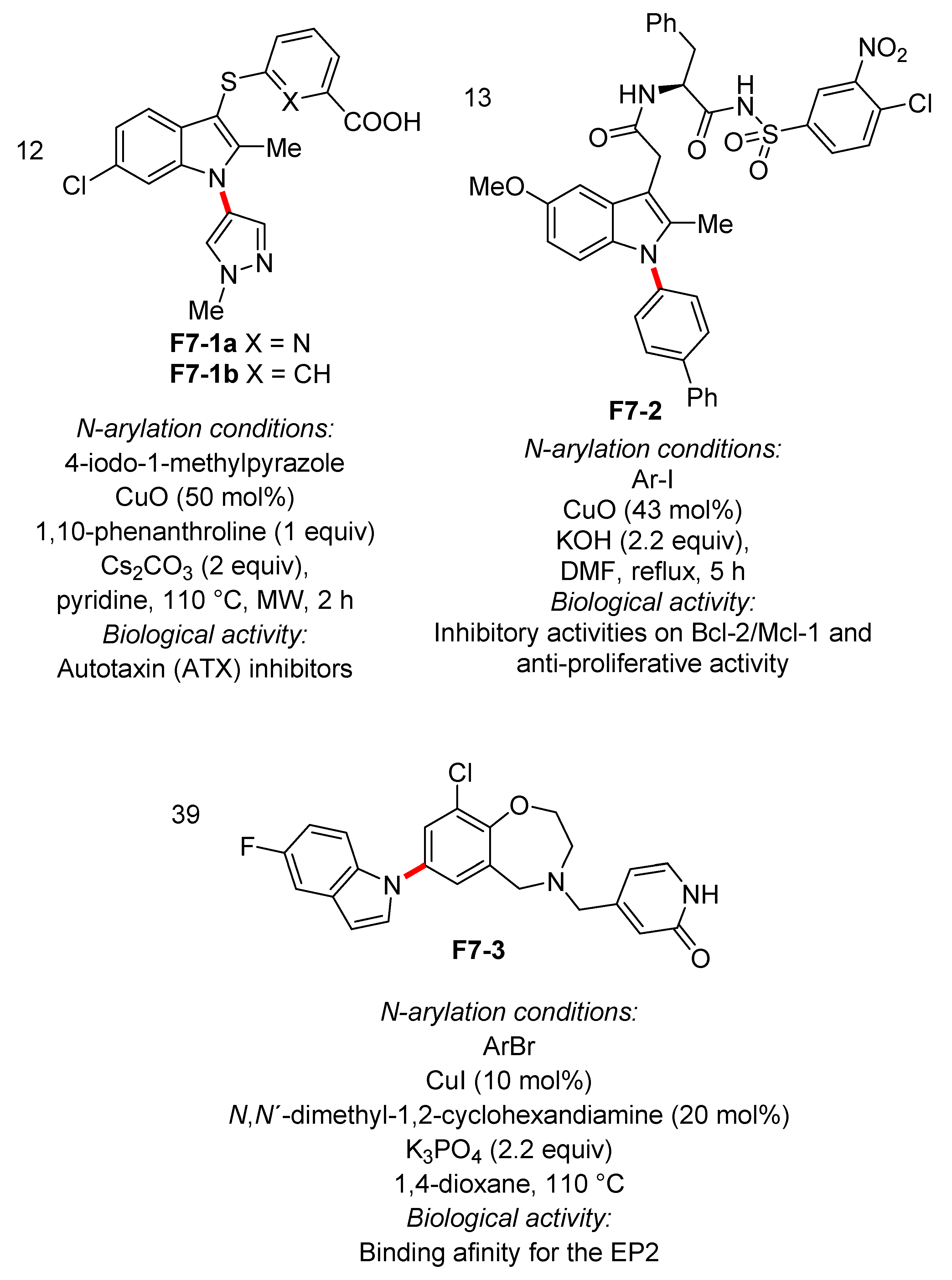
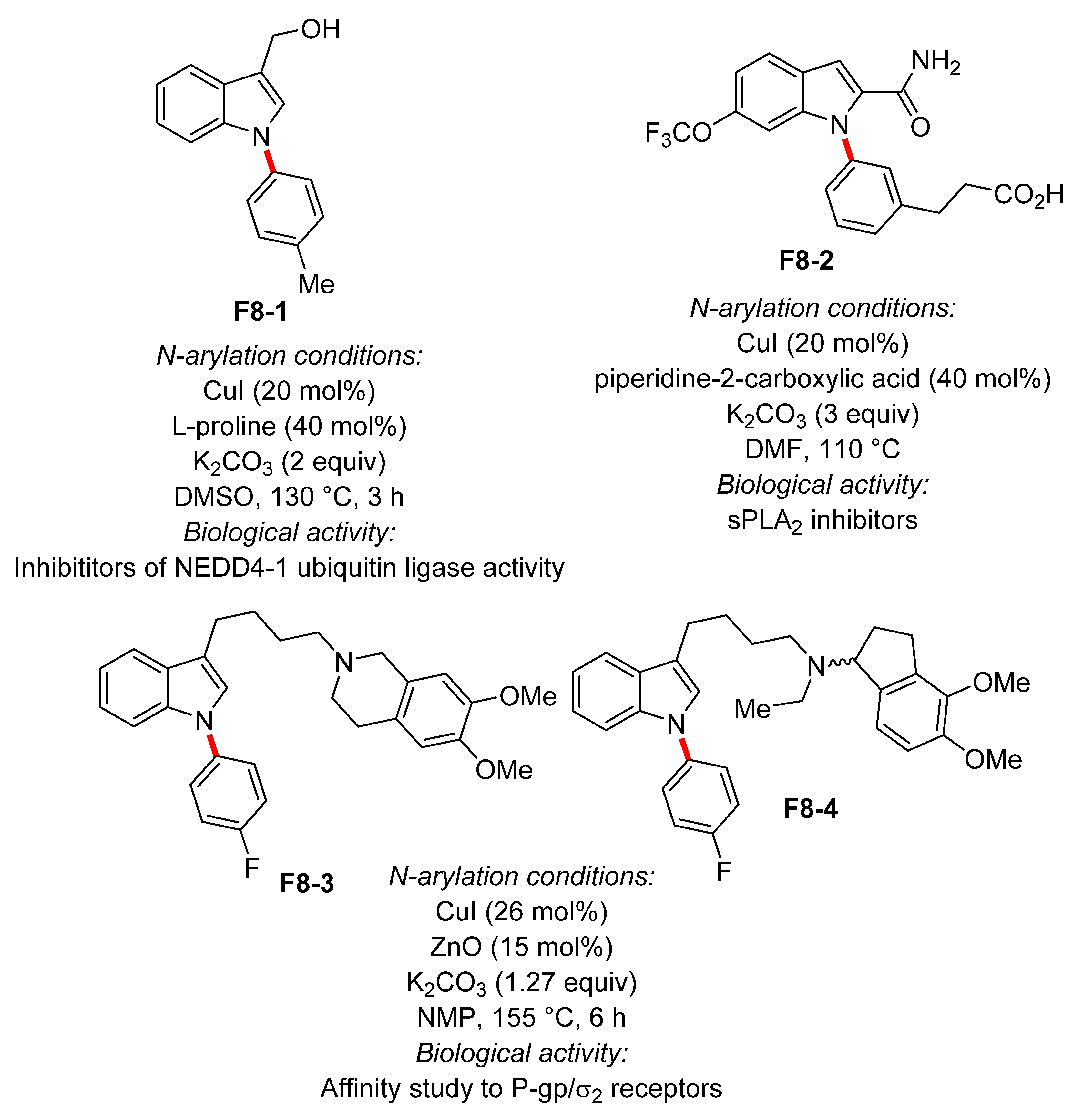
4. N-Arylated Indoles as Biologically Active Substances
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lampis, G.; Deidda, D.; Maullu, C.; Madeddu, M.A.; Pompei, R.; Delle Monachie, F.; Satta, G. Sattabacins and Sattazolins: New Biologically Active Compounds with Antiviral Properties Extracted from a Bacillus sp. J. Antibiot. 1995, 48, 967–972. [Google Scholar] [CrossRef] [Green Version]
- Haviernik, J.; Stefanik, M.; Fojtikova, M.; Kali, S.; Tordo, N.; Rudolf, I.; Hubalek, Z.; Eyer, L.; Ruzek, D. Arbidol (Umifenovir): A Broad-Spectrum Antiviral Drug That Inhibits Medically Important Arthropod-Borne Flaviviruses. Viruses 2018, 10, 184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lewis, R.; Bagnall, A.M.; Leitner, M. Sertindole for schizophrenia. Cochrane Database Syst. Rev. 2005, 3. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, P.; Bora, U. Organocatalytic Dimensions to the C–H Functionalization of the Carbocyclic Core in Indoles: A Review Update. Org. Chem. Front. 2021, 8, 2343–2365. [Google Scholar] [CrossRef]
- Čubiňák, M.; Edlová, T.; Polák, P.; Tobrman, T. Indolylboronic Acids: Preparation and Applications. Molecules 2019, 24, 3523. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trubitsõn, D.; Kanger, T. Enantioselective Catalytic Synthesis of N-Alkylated Indoles. Symmetry 2020, 12, 1184. [Google Scholar] [CrossRef]
- Urbina, K.; Tresp, D.; Sipps, K.; Szostak, M. Recent Advances in Metal-Catalyzed Functionalization of Indoles. Adv. Synth. Catal. 2021, 363, 2723–2739. [Google Scholar] [CrossRef]
- Wen, J.; Shi, Z. From C4 to C7: Innovative Strategies for Site-Selective Functionalization of Indole C-H Bonds. Acc. Chem Res. 2021, 54, 1723–1736. [Google Scholar] [CrossRef]
- Polák, P.; Čejka, J.; Tobrman, T. Formal Transition-Metal-Catalyzed Phosphole C-H Activation for the Synthesis of Pentasubstituted Phospholes. Org. Lett. 2020, 22, 2187–2190. [Google Scholar] [CrossRef]
- Keglevich, G. 1-(2,4,6-Trialkylphenyl)-1H-Phospholes with a Flattened P-Pyramid: Synthesis and Reactivity. In Phosphorus Heterocycles II; Topics in Heterocyclic Chemistry; Bansal, R., Ed.; Springer: Berlin/Heidelberg, Germany, 2010; Volume 21, pp. 149–173. [Google Scholar]
- Quin, L.D. The Continuing Development of the Chemistry of Phospholes. Curr. Org. Chem. 2006, 10, 43–78. [Google Scholar] [CrossRef]
- Romero-Nieto, C.; Baumgartner, T. Dithieno[3,2-b:2′,3′-d]phospholes: A Look Back at the First Decade. Synlett 2013, 24, 920–937. [Google Scholar] [CrossRef]
- Zagidullin, A.A.; Bezkishko, I.A.; Miluykov, V.A.; Sinyashin, O.G. Phospholes—Development and Recent Advances. Mendeleev Commun. 2013, 23, 117–130. [Google Scholar] [CrossRef]
- Ding, Z.; Nie, N.; Chen, T.; Meng, L.; Wang, G.; Chen, Z.; Hu, J. L-Proline N-Oxide Dihydrazides as an Efficient Ligand for Cross-Coupling Reactions of Aryl Iodides and Bromides with Amines and Phenols. Tetrahedron 2021, 79, 131826. [Google Scholar] [CrossRef]
- Antilla, J.C.; Baskin, J.M.; Barder, T.E.; Buchwald, S.L. Copper-Diamine-Catalyzed N-Arylation of Pyrroles, Pyrazoles, Indazoles, Imidazoles, and Triazoles. J. Org. Chem. 2004, 69, 5578–5587. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Li, H.J.; Cheng, Y.F.; Wu, Y.C. Direct C2-Arylation of N-Acyl Pyrroles with Aryl Halides under Palladium Catalysis. Org. Biomol. Chem. 2021, 19, 1555–1564. [Google Scholar] [CrossRef] [PubMed]
- Kaloğlu, M.; Düşünceli, S.D.; Özdemir, İ. The First Used Butylene Linked bis(N-Heterocyclic Carbene)-Palladium-PEPPSI Complexes in the Direct Arylation of Furan and Pyrrole. J. Organomet. Chem. 2020, 915, 121236. [Google Scholar] [CrossRef]
- Chen, D.; Li, J.; Shan, Y.; Cui, P.; Zhao, Y.; Tian, L.; Qiu, G. Halogen-Radical-Promoted Dearomative Aza-Spirocyclization of Alkynylimines: An Efficient Approach to 3-Halo-Spirocyclohexadienones. Synthesis 2020, 52, 609–618. [Google Scholar] [CrossRef]
- Polák, P.; Tobrman, T. Dearomatization Strategy for the Synthesis of Arylated 2H-Pyrroles and 2,3,5-Trisubstituted 1H-Pyrroles. Org. Lett. 2017, 19, 4608–4611. [Google Scholar] [CrossRef]
- Zhuo, C.-X.; Cheng, Q.; Liu, W.-B.; Zhao, Q.; You, S.-L. Enantioselective Synthesis of Pyrrole-Based Spiro- and Polycyclic Derivatives by Iridium-Catalyzed Asymmetric Allylic Dearomatization and Controllable Migration Reactions. Angew. Chem. Int. Ed. 2015, 54, 8475–8479. [Google Scholar] [CrossRef]
- Zhuo, C.-X.; Liu, W.-B.; Wu, Q.-F.; You, S.-L. Asymmetric dearomatization of pyrroles via Ir-catalyzed allylic substitution reaction: Enantioselective synthesis of spiro-2H-pyrroles. Chem. Sci. 2012, 3, 205–208. [Google Scholar] [CrossRef]
- Zhuo, C.-X.; Zhou, Y.; You, S.-L. Highly Regio- and Enantioselective Synthesis of Polysubstituted 2H-Pyrroles via Pd-Catalyzed Intermolecular Asymmetric Allylic Dearomatization of Pyrroles. J. Am. Chem. Soc. 2014, 136, 6590–6593. [Google Scholar] [CrossRef]
- Kunz, K.; Scholz, U.; Ganzer, D. Renaissance of Ullmann and Goldberg Reactions—Progress in Copper Catalyzed C–N-, C–O- and C–S-Coupling. Synlett 2003, 2428–2439. [Google Scholar] [CrossRef] [Green Version]
- Sambiagio, C.; Marsden, S.P.; Blacker, A.J.; McGowan, P.C. Copper Catalysed Ullmann Type Chemistry: From Mechanistic Aspects to Modern Development. Chem. Soc. Rev. 2014, 43, 3525–3550. [Google Scholar] [CrossRef]
- Sperotto, E.; van Klink, G.P.M.; van Koten, G.; de Vries, J.G. The Mechanism of the Modified Ullmann Reaction. Dalton Trans. 2010, 39, 10338–10351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dorel, R.; Grugel, C.P.; Haydl, A.M. The Buchwald–Hartwig Amination after 25 Years. Angew. Chem. Int. Ed. 2019, 58, 17118–17129. [Google Scholar] [CrossRef] [PubMed]
- Forero-Cortés, P.A.; Haydl, A.M. The 25th Anniversary of the Buchwald–Hartwig Amination: Development, Applications, and Outlook. Org. Proc. Res. Develop. 2019, 23, 1478–1483. [Google Scholar] [CrossRef]
- Heravi, M.M.; Kheilkordi, Z.; Zadsirjan, V.; Heydari, M.; Malmir, M. Buchwald-Hartwig reaction: An overview. J. Organomet. Chem. 2018, 861, 17–104. [Google Scholar] [CrossRef]
- Chen, J.-Q.; Li, J.-H.; Dong, Z.-B. A Review on the Latest Progress of Chan-Lam Coupling Reaction. Adv. Synth. Catal. 2020, 362, 3311–3331. [Google Scholar] [CrossRef]
- Munir, I.; Zahoor, A.F.; Rasool, N.; Naqvi, S.A.R.; Zia, K.M.; Ahmad, R. Synthetic Applications and Methodology Development of Chan–Lam Coupling: A Review. Mol. Divers. 2019, 23, 215–259. [Google Scholar] [CrossRef] [PubMed]
- West, M.J.; Fyfe, J.W.B.; Vantourout, J.C.; Watson, A.J.B. Mechanistic Development and Recent Applications of the Chan–Lam Amination. Chem. Rev. 2019, 119, 12491–12523. [Google Scholar] [CrossRef]
- Joucla, L.; Djakovitch, L. Transition Metal-Catalysed Direct and Site-Selective N1-, C2- or C3-Arylation of the Indole Nucleus: 20 Years of Improvements. Adv. Synth. Catal. 2009, 351, 673–714. [Google Scholar] [CrossRef]
- Xu, H. Advances on N-Arylation of Indoles by Cross-Coupling Reactions. Mini-Rev. Org. Chem. 2009, 6, 367–377. [Google Scholar] [CrossRef]
- Halder, P.; Roy, T.; Das, P. Recent developments in selective N-arylation of azoles. Chem. Commun. 2021, 57, 5235–5249. [Google Scholar] [CrossRef] [PubMed]
- Chang, D.; Gao, F.; Shi, L. Potassium tert-butoxide-mediated generation of arynes from o-bromoacetophenone derivatives. Tetrahedron 2018, 74, 2428–2434. [Google Scholar] [CrossRef]
- Chen, J.; Wu, J. Transition-Metal-Free C3 Arylation of Indoles with Aryl Halides. Angew. Chem. Int. Ed. 2017, 56, 3951–3955. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, F.; Liu, H.; Jia, J.; Ma, C. Transition-metal-free synthesis of indole-fused dibenzo[b,f][1,4]oxazepines via Smiles rearrangement. Org. Biomol. Chem. 2016, 14, 11076–11079. [Google Scholar] [CrossRef]
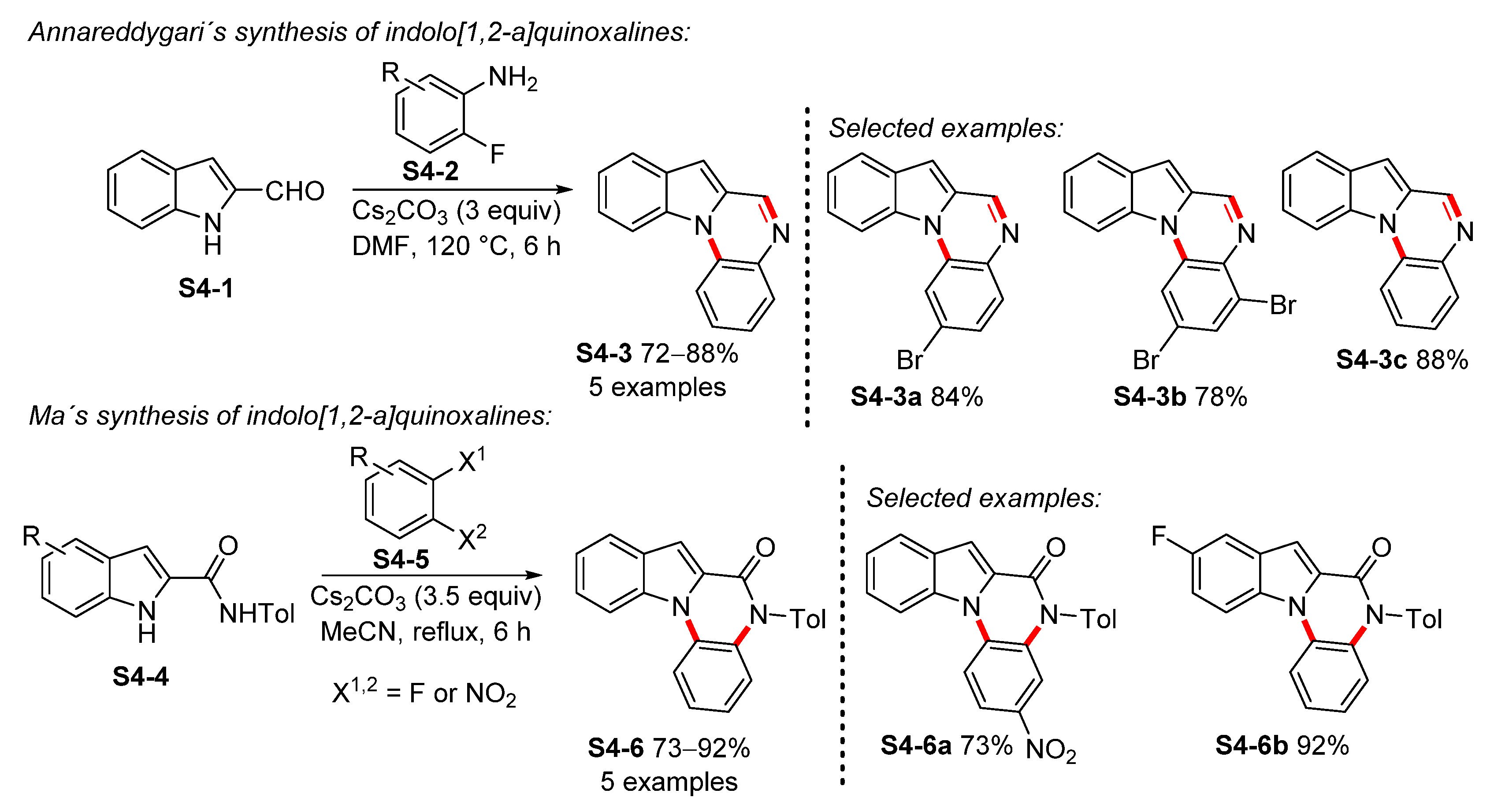
- Annareddygari, S.; Kasireddy, V.R.; Reddy, J. Transition-Metal-Free N-Arylation: A General Approach to Aza-Fused Poly-heteroaromatics. J. Heterocycl. Chem. 2019, 56, 3267–3276. [Google Scholar] [CrossRef]
- Huang, A.; Liu, F.; Zhan, C.; Liu, Y.; Ma, C. One-Pot Synthesis of Pyrrolo[1,2-a]quinoxalines. Org. Biomol. Chem. 2011, 9, 7351–7357. [Google Scholar] [CrossRef]
- Thanetchaiyakup, A.; Rattanarat, H.; Chuanopparat, N.; Ngernmeesri, P. One-Pot Synthesis of Substituted Indolo[1,2-a]quinolines under Transition-Metal-Free Conditions. Tetrahedron Lett. 2018, 59, 1014–1018. [Google Scholar] [CrossRef]
- Xu, H.; Sun, L.; Song, C. Base-Mediated N-Arylation for the Synthesis of 9H-Pyrrolo[1,2-a]indol-9-ones and 10H-Indolo[1,2-a]indol-10-ones. Helv. Chim. Acta 2019, 102, e1800195. [Google Scholar] [CrossRef] [Green Version]
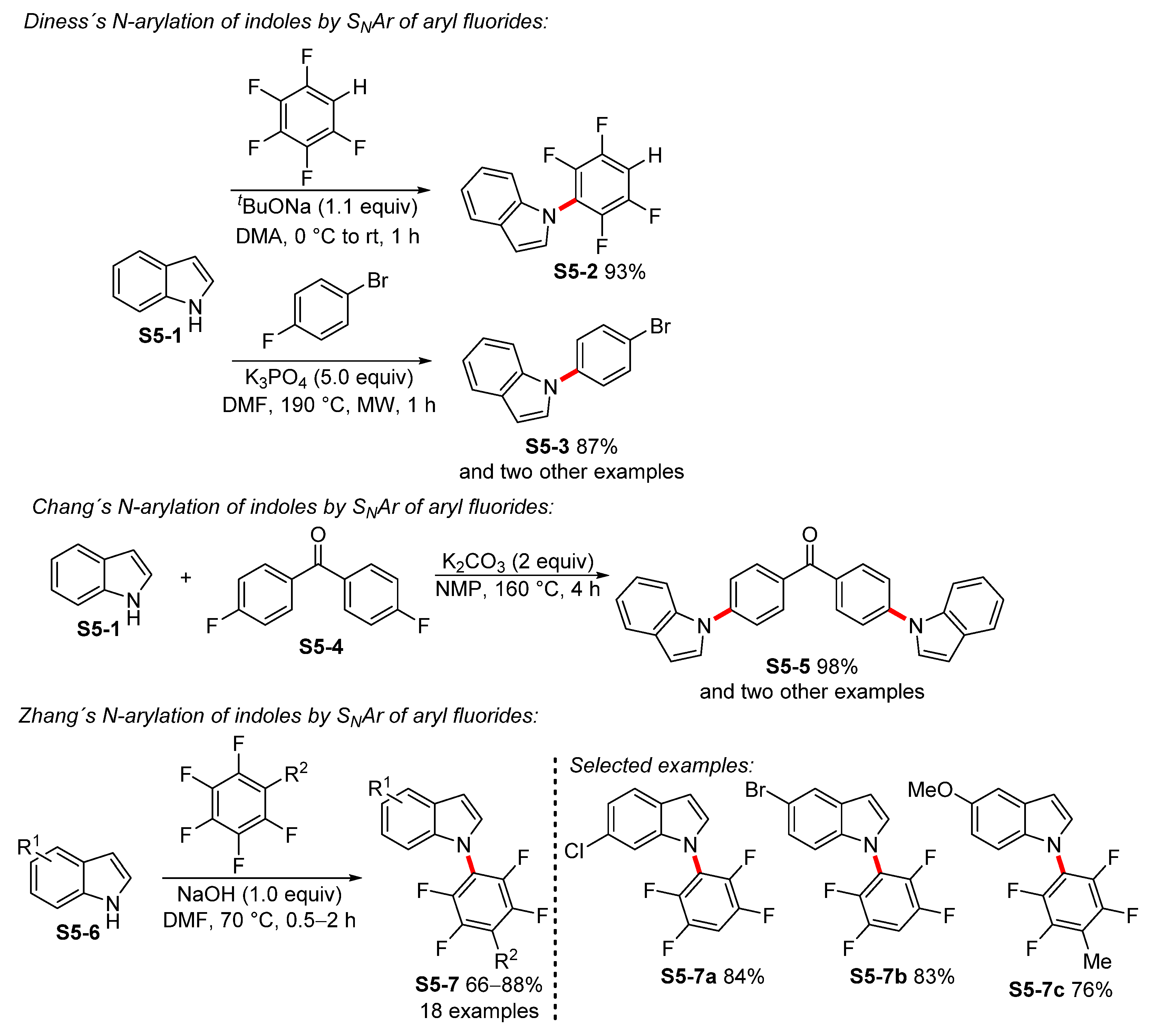
- Diness, F.; Begtrup, M. Sequential Direct SNAr Reactions of Pentafluorobenzenes with Azole or Indole Derivatives. Org. Lett. 2014, 16, 3130–3133. [Google Scholar] [CrossRef]
- Diness, F.; Fairlie, D.P. Catalyst-Free N-Arylation Using Unactivated Fluorobenzenes. Angew. Chem. Int. Ed. 2012, 51, 8012–8016. [Google Scholar] [CrossRef] [PubMed]
- Chang, G.; Yang, L.; Liu, S.; Luo, X.; Lin, R.; Zhang, L. Synthesis of indole-based functional polymers with well-defined structures via a catalyst-free C–N coupling reaction. RSC Adv. 2014, 4, 30630–30637. [Google Scholar] [CrossRef]
- Liu, C.; Wang, H.; Xing, X.; Xu, Y.; Ma, J.-A.; Zhang, B. Selective C4–F bond cleavage of pentafluorobenzene: Synthesis of N-tetrafluoroarylated heterocyclic compounds. Tetrahedron Lett. 2013, 54, 4649–4652. [Google Scholar] [CrossRef]
- Iqbal, M.A.; Mehmood, H.; Lv, J.; Hua, R. Base-Promoted SNAr Reactions of Fluoro- and Chloroarenes as a Route to N-Aryl Indoles and Carbazoles. Molecules 2019, 24, 1145. [Google Scholar] [CrossRef] [Green Version]
- Maiti, B.; Sun, C.-M. Novel Approach Towards the Synthesis of Skeletally Diverse Benzimidazole-pyrrolo[1,2-a]quinoxaline by SNAr/Pictet–Spengler Reaction under Focused Microwave Irradiation. New J. Chem. 2011, 35, 1385–1396. [Google Scholar] [CrossRef]
- Ricci, P.; Krämer, K.; Cambeiro, X.C.; Larrosa, I. Arene–Metal π-Complexation as a Traceless Reactivity Enhancer for C–H Arylation. J. Am. Chem. Soc. 2013, 135, 13258–13261. [Google Scholar] [CrossRef]
- Su, J.; Chen, Q.; Lu, L.; Ma, Y.; Auyoung, G.H.L.; Hua, R. Base-Promoted Nucleophilic Fluoroarenes Substitution of CF Bonds. Tetrahedron 2018, 74, 303–307. [Google Scholar] [CrossRef]
- Tian, Z.-Y.; Ming, X.-X.; Teng, H.-B.; Hu, Y.-T.; Zhang, C.-P. Transition-Metal-Free N-Arylation of Amines by Triarylsulfonium Triflates. Chem. Eur. J. 2018, 24, 13744–13748. [Google Scholar] [CrossRef]
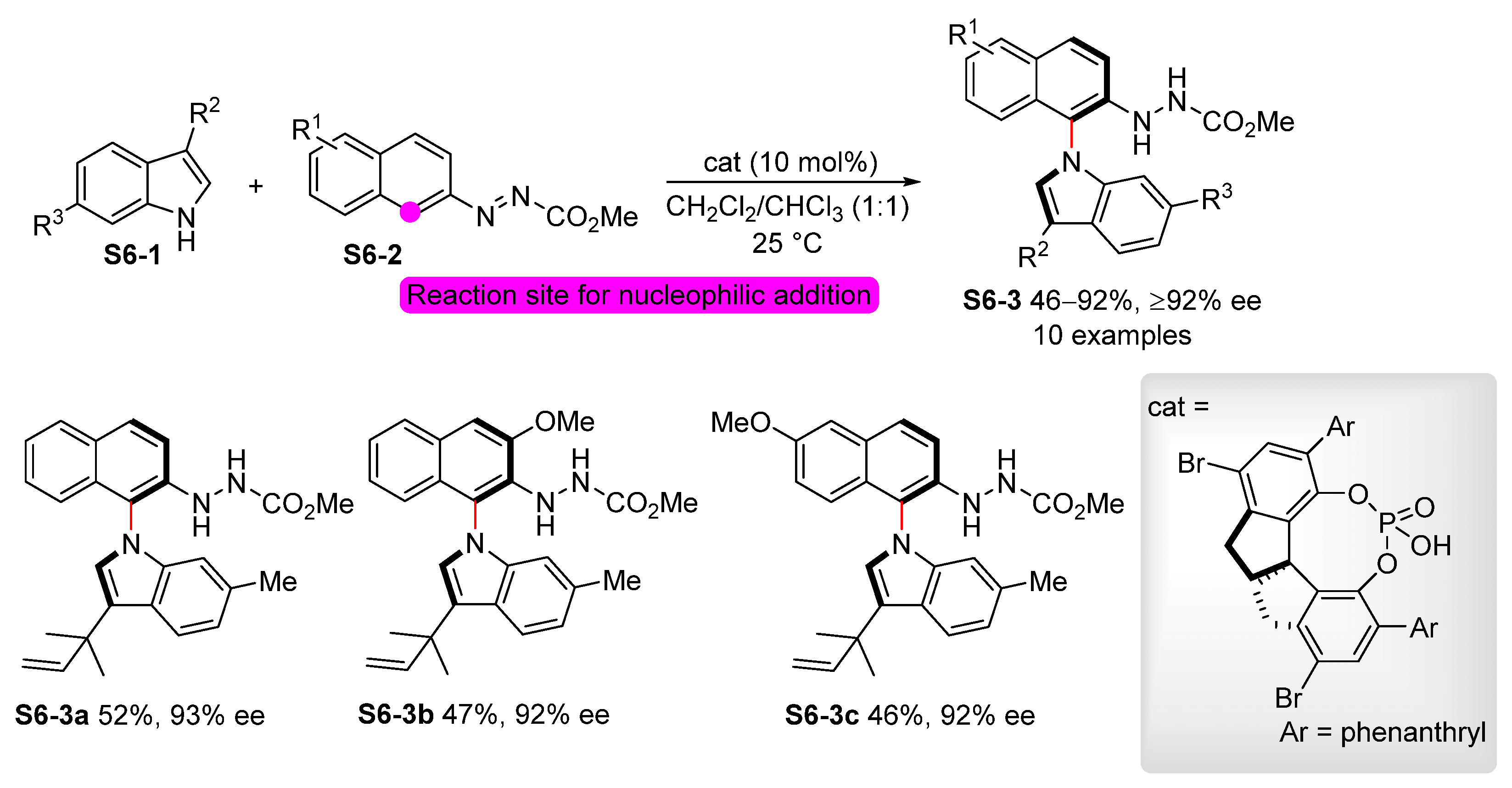
- Xia, W.; An, Q.-J.; Xiang, S.-H.; Li, S.; Wang, Y.-B.; Tan, B. Chiral Phosphoric Acid Catalyzed Atroposelective C−H Amination of Arenes. Angew. Chem. Int. Ed. 2020, 59, 6775–6779. [Google Scholar] [CrossRef] [PubMed]
- Chittimalla, S.K.; Nakka, S.; Koodalingam, M.; Bandi, C. N-Arylation of Heterocycles by a Tandem Aza-Michael Addition Reaction and Aromatization Sequence. Synlett 2018, 29, 57–64. [Google Scholar] [CrossRef] [Green Version]
- Li, S.; Wu, X.-X.; Chen, S. Base-Promoted Direct Synthesis of Functionalized N-Arylindoles via the Cascade Reactions of Allenic Ketones with Indoles. Org. Biomol. Chem. 2019, 17, 789–793. [Google Scholar] [CrossRef] [PubMed]
- Rull, S.G.; Blandez, J.F.; Fructos, M.R.; Belderrain, T.R.; Nicasio, M.C. C–N Coupling of Indoles and Carbazoles with Aromatic Chlorides Catalyzed by a Single-Component NHC-Nickel(0) Precursor. Adv. Synth. Catal. 2015, 357, 907–911. [Google Scholar] [CrossRef]
- Clark, J.S.K.; Voth, C.N.; Ferguson, M.J.; Stradiotto, M. Evaluating 1,1′-Bis(phosphino)ferrocene Ancillary Ligand Variants in the Nickel-Catalyzed C–N Cross-Coupling of (Hetero)aryl Chlorides. Organometallics 2017, 36, 679–686. [Google Scholar] [CrossRef]
- Iranpoor, N.; Panahi, F. Direct Nickel-Catalyzed Amination of Phenols via C–O Bond Activation using 2,4,6-Trichloro-1,3,5-triazine (TCT) as Reagent. Adv. Synth. Catal. 2014, 356, 3067–3073. [Google Scholar] [CrossRef]
- Morioka, T.; Nakatani, S.; Sakamoto, Y.; Kodama, T.; Ogoshi, S.; Chatani, N.; Tobisu, M. Nickel-Catalyzed Decarbonylation of N-Acylated N-Heteroarenes. Chem. Sci. 2019, 10, 6666–6671. [Google Scholar] [CrossRef] [Green Version]
- Krishnaveni, T.; Lakshmi, K.; Kadirvelu, K.; Kaveri, M.V. Exploration of Catalytic Activity of Quercetin Mediated Hydrothermally Synthesized NiO Nanoparticles Towards C–N Coupling of Nitrogen Heterocycles. Catal. Lett. 2020, 150, 1628–1640. [Google Scholar] [CrossRef]
- Gatien, A.V.; Lavoie, C.M.; Bennett, R.N.; Ferguson, M.J.; McDonald, R.; Johnson, E.R.; Speed, A.W.H.; Stradiotto, M. Application of Diazaphospholidine/Diazaphospholene-Based Bisphosphines in Room-Temperature Nickel-Catalyzed C(sp2)–N Cross-Couplings of Primary Alkylamines with (Hetero)aryl Chlorides and Bromides. ACS Catal. 2018, 8, 5328–5339. [Google Scholar] [CrossRef]
- Park, N.H.; Teverovskiy, G.; Buchwald, S.L. Development of an Air-Stable Nickel Precatalyst for the Amination of Aryl Chlorides, Sulfamates, Mesylates, and Triflates. Org. Lett. 2014, 16, 220–223. [Google Scholar] [CrossRef]
- Liu, R.Y.; Dennis, J.M.; Buchwald, S.L. The Quest for the Ideal Base: Rational Design of a Nickel Precatalyst Enables Mild, Homogeneous C–N Cross-Coupling. J. Am. Chem. Soc. 2020, 142, 4500–4507. [Google Scholar] [CrossRef]
- Sawatzky, R.S.; Ferguson, M.J.; Stradiotto, M. Thieme Chemistry Journals Awardees—Where Are They Now? Efficient Cross-Coupling of Secondary Amines/Azoles and Activated (Hetero)Aryl Chlorides Using an Air-Stable DPEPhos/Nickel Pre-Catalyst. Synlett 2017, 28, 1586–1591. [Google Scholar]
- Panahi, F.; Roozbin, F.; Rahimi, S.; Moayyed, M.; Valaei, A.; Iranpoor, N. A Triazine-Phosphite Polymeric Ligand Bearing Cage-Like P,N-Ligation Sites: An Efficient Ligand in the Nickel-Catalyzed Amination of Aryl Chlorides and Phenols. RSC Adv. 2016, 6, 80670–80678. [Google Scholar] [CrossRef]
- Malapit, C.A.; Borrell, M.; Milbauer, M.W.; Brigham, C.E.; Sanford, M.S. Nickel-Catalyzed Decarbonylative Amination of Carboxylic Acid Esters. J. Am. Chem. Soc. 2020, 142, 5918–5923. [Google Scholar] [CrossRef]
- Lokhande†, S.K.; Vaidya†, G.N.; Satpute, D.P.; Venkatesh, A.; Kumar, S.; Kumar, D. Structure Ligation Relationship of Amino Acids for the Selective Indole C−H Arylation Reaction: L-Aspartic acid as Sustainable Alternative of Phosphine Ligands. Adv. Synth. Catal. 2020, 362, 2857–2863. [Google Scholar] [CrossRef]
- Mohr, Y.; Renom-Carrasco, M.; Demarcy, C.; Quadrelli, E.A.; Camp, C.; Wisser, F.M.; Clot, E.; Thieuleux, C.; Canivet, J. Regiospecificity in Ligand-Free Pd-Catalyzed C–H Arylation of Indoles: LiHMDS as Base and Transient Directing Group. ACS Catal. 2020, 10, 2713–2719. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Suzuki, K.; Sato, Y.; Manabe, K. Palladium-Catalyzed Direct C3-Selective Arylation of N-Unsubstituted Indoles with Aryl Chlorides and Triflates. Org. Lett. 2017, 19, 5388–5391. [Google Scholar] [CrossRef] [PubMed]
- Ye, Z.; Li, Y.; Xu, K.; Chen, N.; Zhang, F. Cascade π-Extended Decarboxylative Annulation Involving Cyclic Diaryliodonium Salts: Site-Selective Synthesis of Phenanthridines and Benzocarbazoles via a Traceless Directing Group Strategy. Org. Lett. 2019, 21, 9869–9873. [Google Scholar] [CrossRef] [PubMed]
- Mayer, L.; Kohlbecher, R.; Müller, T.J.J. Concatenating Suzuki Arylation and Buchwald–Hartwig Amination by a Sequentially Pd-Catalyzed One-Pot Process—Consecutive Three-Component Synthesis of C,N-Diarylated Heterocycles. Chem. Eur. J. 2020, 26, 15130–15134. [Google Scholar] [CrossRef]
- Chen, H.; Yang, H.; Li, N.; Xue, X.; He, Z.; Zeng, Q. Palladium-Catalyzed C–N Cross-Coupling of NH-Heteroarenes and Quaternary Ammonium Salts via C–N Bond Cleavage. Org.Process Res. Develop. 2019, 23, 1679–1685. [Google Scholar] [CrossRef]
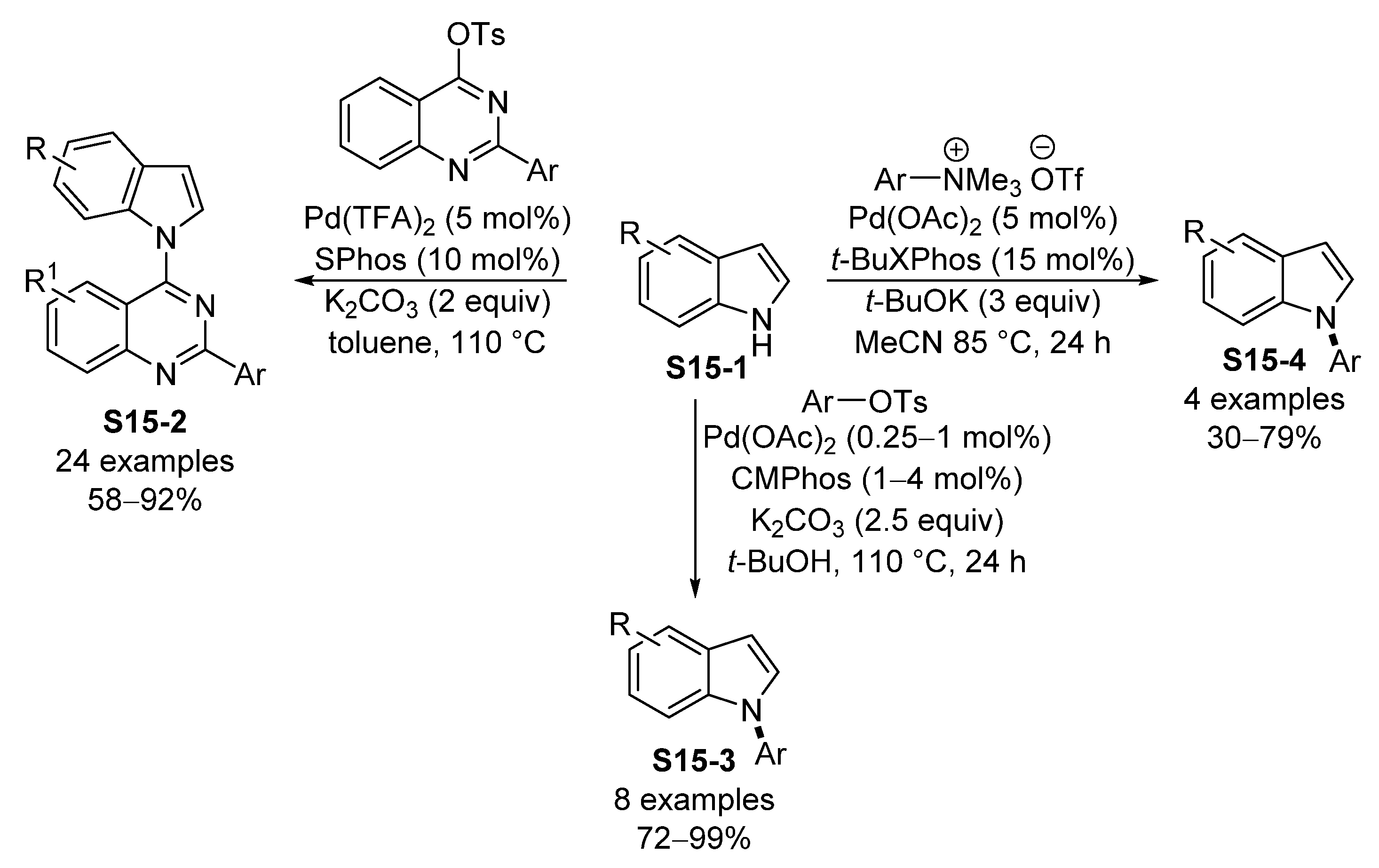
- Ye, X.; Huang, J.; Deng, Z.; Yuan, J.; Peng, Y. Palladium-Catalyzed Cross-Coupling Reactions of 4-Tosyl-oxyquinazolines with Indoles: An Efficient Approach to 4-(1H-Indol-1-yl)quinazolines. Synthesis 2021, 53, 383–390. [Google Scholar]
- Choy, P.Y.; Chung, K.H.; Yang, Q.; So, C.M.; Sun, R.W.-Y.; Kwong, F.Y. A General Palladium–Phosphine Complex to Explore Aryl Tosylates in the N-Arylation of Amines: Scope and Limitations. Chem. Asian J. 2018, 13, 2465–2474. [Google Scholar] [CrossRef] [PubMed]
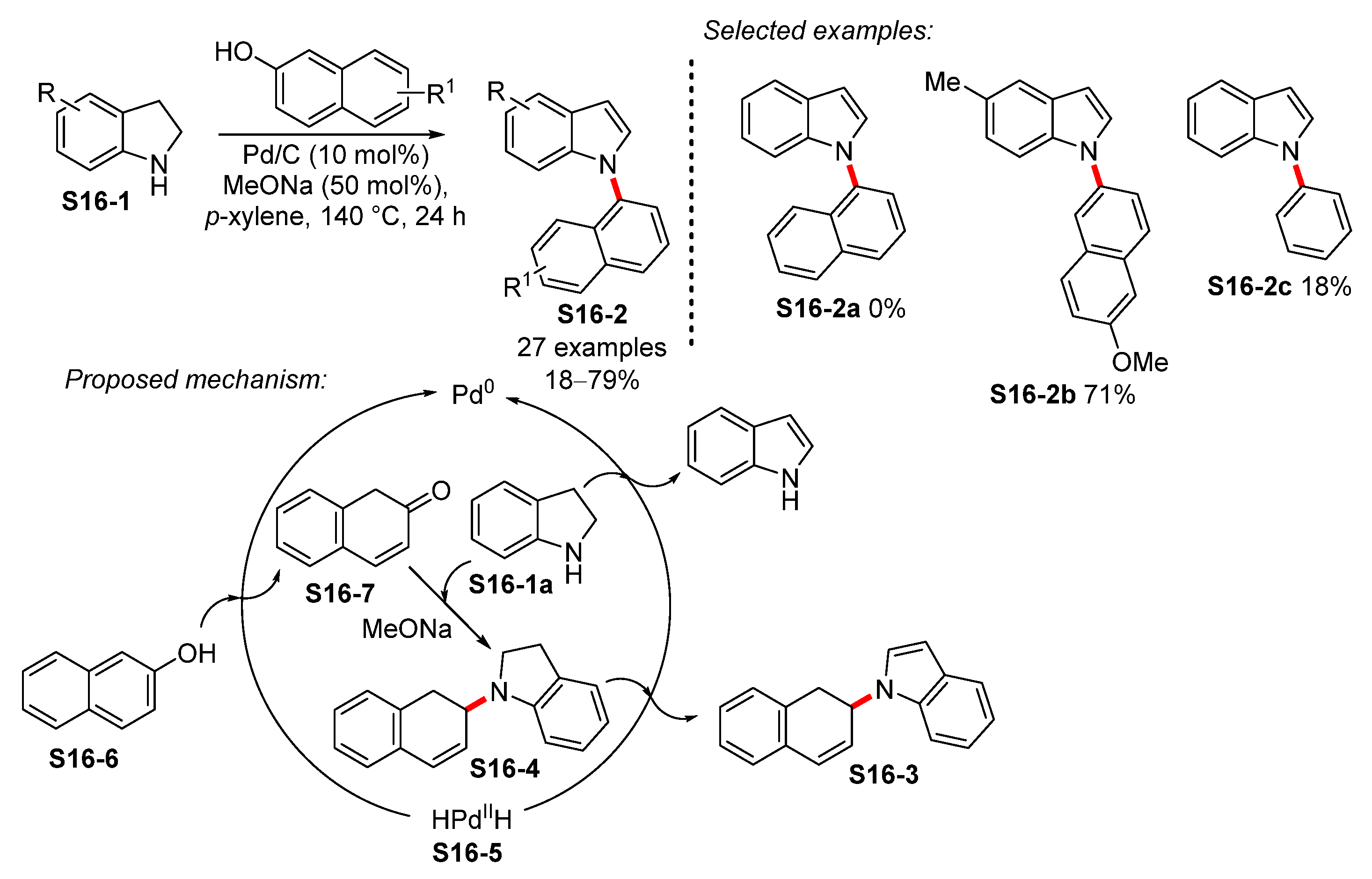
- Chen, X.; Yang, Z.; Chen, X.; Liang, W.; Zhu, Z.; Xie, F.; Li, Y. Hydrogen-Transfer-Mediated N-Arylation of Naphthols Using Indolines as Hydrogen Donors. J. Org. Chem. 2020, 85, 508–514. [Google Scholar] [CrossRef] [PubMed]
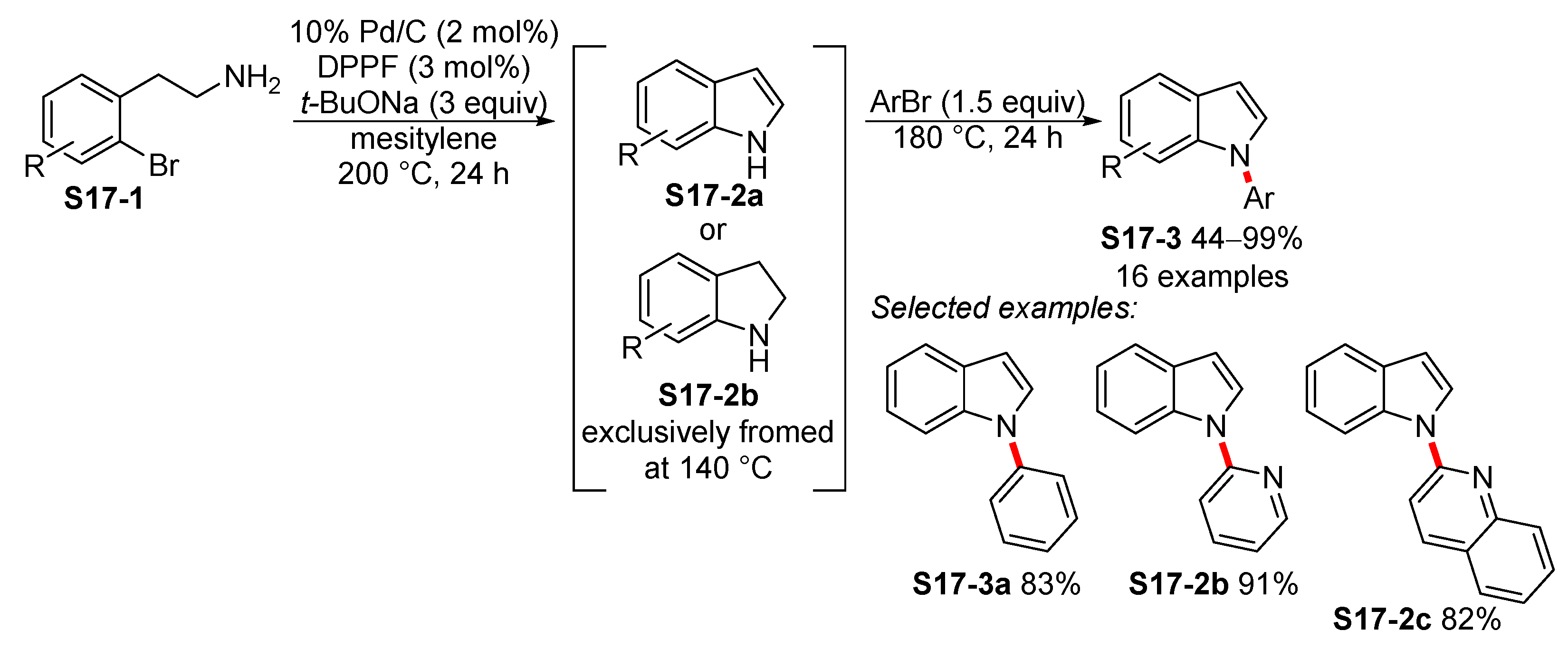
- Monguchi, Y.; Marumoto, T.; Takamatsu, H.; Sawama, Y.; Sajiki, H. Palladium on Carbon-Catalyzed One-Pot N-Arylindole Synthesis: Intramolecular Aromatic Amination, Aromatization, and Intermolecular Aromatic Amination. Adv. Synth. Catal. 2014, 356, 1866–1872. [Google Scholar] [CrossRef]
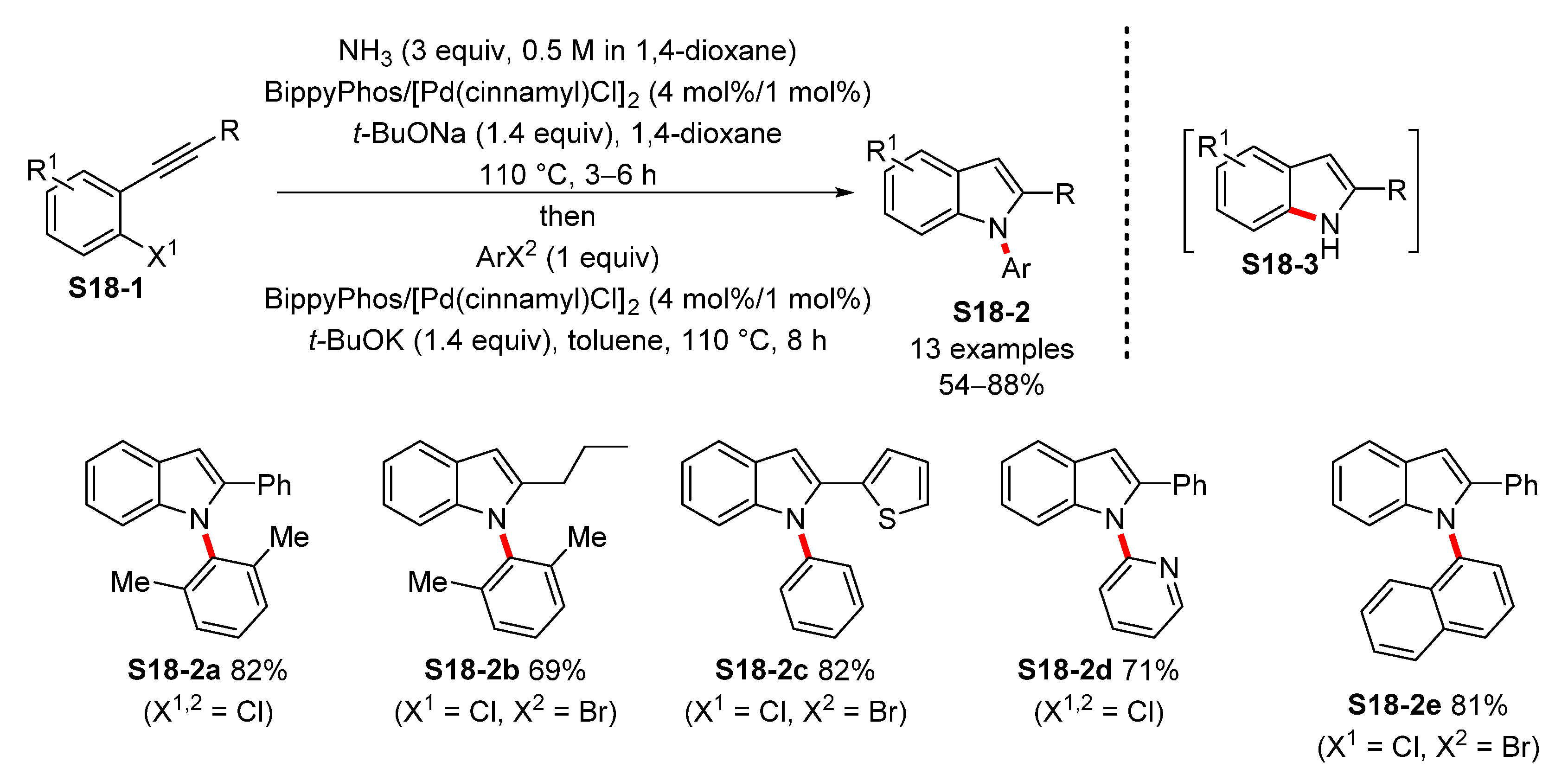
- Crawford, S.M.; Lavery, C.B.; Stradiotto, M. BippyPhos: A Single Ligand With Unprecedented Scope in the Buchwald–Hartwig Amination of (Hetero)aryl Chlorides. Chem. Eur. J. 2013, 19, 16760–16771. [Google Scholar] [CrossRef] [PubMed]
- Ghorbani-Vaghei, R.; Hemmati, S.; Hamelian, M.; Veisi, H. An Efficient, Mild and Selective Ullmann-Type N-Arylation of Indoles Catalysed by Pd Immobilized on Amidoxime-Functionalized Mesoporous SBA-15 as Heterogeneous and Recyclable Nanocatalyst. Appl. Organomet. Chem. 2015, 29, 195–199. [Google Scholar] [CrossRef]
- Veisi, H.; Poor Heravi, M.R.; Hamelian, M. SBA-15-Functionalized Melamine–Pyridine Group-Supported Palladium(0) as an Efficient Heterogeneous and Recyclable Nanocatalyst for N-Arylation of Indoles through Ullmann-Type Coupling Reactions. Appl. Organomet. Chem. 2015, 29, 334–337. [Google Scholar] [CrossRef]
- Veisi, H.; Morakabati, N. Palladium Nanoparticles Supported on Modified Single-Walled Carbon Nanotubes: A Heterogeneous and Reusable Catalyst in the Ullmann-Type N-Arylation of Imidazoles and Indoles. New J. Chem. 2015, 39, 2901–2907. [Google Scholar] [CrossRef]
- Ghorbani-Vaghei, R.; Hemmati, S.; Hekmati, M. Pd Immobilized on Modified Magnetic Fe3O4 Nanoparticles: Magnetically Recoverable and Reusable Pd Nanocatalyst for Suzuki-Miyaura Coupling Reactions and Ullmann-Type N-Arylation of Indoles. J. Chem. Sci. 2016, 128, 1157–1162. [Google Scholar] [CrossRef] [Green Version]
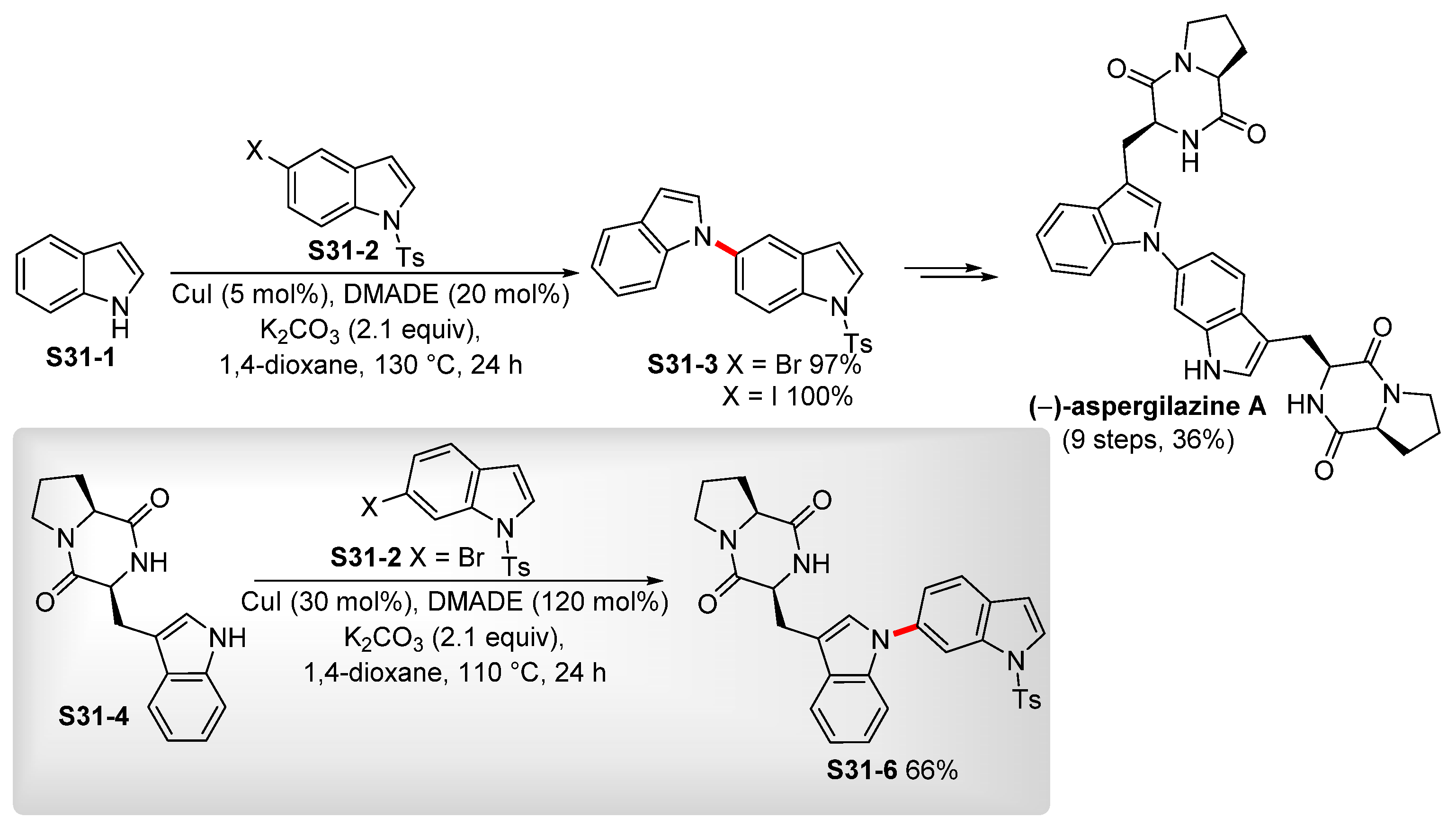
- Boyd, E.M.; Sperry, J. Total Synthesis of (−)-Aspergilazine A. Org. Lett. 2014, 16, 5056–5059. [Google Scholar] [CrossRef]
- Hajipour, A.R.; Dordahan, F.; Rafiee, F. Synthesis of Tertiary Aryl Amines of Various Aryl Halides and Secondary Amines using Ortho-Palladated Complex of Tribenzylamine. Appl. Organomet. Chem. 2013, 27, 704–706. [Google Scholar] [CrossRef]
- Monti, A.; Rama, R.J.; Gómez, B.; Maya, C.; Álvarez, E.; Carmona, E.; Nicasio, M.C. N-Substituted Aminobiphenyl Palladacycles Stabilized by Dialkylterphenyl Phosphanes: Preparation and Applications in CN Cross-Coupling Reactions. Inorg. Chim. Acta 2021, 518, 120214. [Google Scholar] [CrossRef]
- Wagner, P.; Bollenbach, M.; Doebelin, C.; Bihel, F.; Bourguignon, J.-J.; Salomé, C.; Schmitt, M. t-BuXPhos: A Highly Efficient Ligand for Buchwald–Hartwig Coupling in Water. Green Chem. 2014, 16, 4170–4178. [Google Scholar] [CrossRef]
- Izquierdo, J.; Jain, A.D.; Abdulkadir, S.A.; Schiltz, G.E. Palladium-Catalyzed Coupling Reactions on Functionalized 2-Trifluoromethyl-4-chromenone Scaffolds: Synthesis of Highly Functionalized Trifluoromethyl Heterocycles. Synthesis 2019, 51, 1342–1352. [Google Scholar] [CrossRef] [PubMed]
- Grimm, J.B.; Lavis, L.D. Synthesis of Rhodamines from Fluoresceins Using Pd-Catalyzed C–N Cross-Coupling. Org. Lett. 2011, 13, 6354–6357. [Google Scholar] [CrossRef]
- Shimizu, K.; Minami, Y.; Goto, O.; Ikehira, H.; Hiyama, T. Silicon-based C–N Cross-coupling Reaction. Chem. Lett. 2014, 43, 438–440. [Google Scholar] [CrossRef]
- Minami, Y.; Komiyama, T.; Shimizu, K.; Hiyama, T.; Goto, O.; Ikehira, H. Catalytic Carbon–Nitrogen Bond-Forming Cross-Coupling Using N-Trimethylsilylamines. Bull. Chem. Soc. Japan 2015, 88, 1437–1446. [Google Scholar] [CrossRef]
- Hosseini-Sarvari, M.; Razmi, Z. Highly Active Recyclable Heterogeneous Pd/ZnO Nanoparticle Catalyst: Sustainable Developments for the C–O and C–N Bond Cross-Coupling Reactions of Aryl Halides under Ligand-Free Conditions. RSC Adv. 2014, 4, 44105–44116. [Google Scholar] [CrossRef]
- Fareghi-Alamdari, R.; Haqiqi, M.G.; Zekri, N. Immobilized Pd(0) Nanoparticles on Phosphine-Functionalized Graphene as a Highly Active Catalyst for Heck, Suzuki and N-Arylation Reactions. New J. Chem. 2016, 40, 1287–1296. [Google Scholar] [CrossRef]
- Panahi, F.; Daneshgar, F.; Haghighi, F.; Khalafi-Nezhad, A. Immobilized Pd Nanoparticles on Silica-Starch Substrate (PNP-SSS): Efficient Heterogeneous Catalyst in Buchwald–Hartwig C–N Cross-Coupling Reaction. J. Organomet. Chem. 2017, 851, 210–217. [Google Scholar] [CrossRef]
- Yong, F.-F.; Teo, Y.-C.; Tay, S.-H.; Tan, B.Y.-H.; Lim, K.-H. A Ligand-Free Copper(I) Oxide Catalyzed Strategy for the N-Arylation of Azoles in Water. Tetrahedron Lett. 2011, 52, 1161–1164. [Google Scholar] [CrossRef]
- Liu, L.; Wu, F.; Liu, Y.; Xie, J.; Dai, B.; Zhou, Z. Copper-Catalysed N-Arylation of Pyrrole with Aryl Iodides Under Ligand-Free Conditions. J. Chem. Res. 2019, 38, 180–182. [Google Scholar] [CrossRef]
- Khalil, A.; Fihri, A.; Jouiad, M.; Hashaikeh, R. Electrospun Copper Oxide Nanoparticles as an Efficient Heterogeneous Catalyst for N-Arylation of Indole. Tetrahedron Lett. 2014, 55, 5973–5975. [Google Scholar] [CrossRef]
- Amadine, O.; Maati, H.; Abdelouhadi, K.; Fihri, A.; El Kazzouli, S.; Len, C.; El Bouari, A.; Solhy, A. Ceria-Supported Copper Nanoparticles: A Highly Efficient and Recyclable Catalyst for N-Arylation of Indole. J. Mol. Cat. A-Chem. 2014, 395, 409–419. [Google Scholar] [CrossRef]
- Chaudhary, K.; Subodh; Prakash, K.; Mogha, N.K.; Masram, D.T. Fruit Waste (Pulp) Decorated CuO NFs as Promising Platform for Enhanced Catalytic Response and Its Peroxidase Mimics Evaluation. Arab. J. Chem. 2020, 13, 4869–4881. [Google Scholar] [CrossRef]
- Hemmati, S.; Mehrazin, L.; Hekmati, M.; Izadi, M.; Veisi, H. Biosynthesis of CuO Nanoparticles Using Rosa Canina Fruit Extract as a Recyclable and Heterogeneous Nanocatalyst for C-N Ullmann Coupling Reactions. Mater. Chem. Phys. 2018, 214, 527–532. [Google Scholar] [CrossRef]
- Lim, J.; Kim, J.D.; Choi, H.C.; Lee, S. CNT-CuO Catalyzed C–N Bond Formation for N-Arylation of 2-Phenylindoles. J. Organomet. Chem. 2019, 902, 120970. [Google Scholar] [CrossRef]
- Pai, G.; Chattopadhyay, A.P. Ligand-Free Copper Nanoparticle Promoted N-Arylation of Azoles with Aryl and Heteroaryl Iodides. Tetrahedron Lett. 2014, 55, 941–944. [Google Scholar] [CrossRef]
- Suramwar, N.V.; Thakare, S.R.; Karade, N.N.; Khaty, N.T. Green Synthesis of Predominant (111) Facet CuO Nanoparticles: Heterogeneous and Recyclable Catalyst for N-Arylation of Indoles. J. Mol. Catal. A-Chem. 2012, 359, 28–34. [Google Scholar] [CrossRef]
- Talukdar, D.; Das, G.; Thakur, S.; Karak, N.; Thakur, A.J. Copper Nanoparticle Decorated Organically Modified Montmorillonite (OMMT): An Efficient Catalyst for the N-Arylation of Indoles and Similar Heterocycles. Catal. Commun. 2015, 59, 238–243. [Google Scholar] [CrossRef]
- Reddy, K.H.V.; Satish, G.; Ramesh, K.; Karnakar, K.; Nageswar, Y.V.D. An Efficient Synthesis of N-Substituted Indoles from Indoline/Indoline Carboxylic Acid via Aromatization Followed by C–N Cross-Coupling Reaction by Using Nano Copper Oxide as a Recyclable Catalyst. Tetrahedron Lett. 2012, 53, 3061–3065. [Google Scholar] [CrossRef]
- Patil, P.H.; Nallasivam, J.L.; Fernandes, R.A. Unimolecular 4-Hydroxypiperidines: New Ligands for Copper-Catalyzed N-Arylation. Asian J. Org. Chem. 2015, 4, 552–559. [Google Scholar] [CrossRef]
- Yang, X.; Xing, H.; Zhang, Y.; Lai, Y.; Zhang, Y.; Jiang, Y.; Ma, D. CuI/8-Hydroxyquinalidine Promoted N-Arylation of Indole and Azoles. Chin. J. Chem. 2012, 30, 875–880. [Google Scholar] [CrossRef]
- Elliott, E.-C.; Maggs, J.L.; Park, B.K.; O’Neill, P.M.; Stachulski, A.V. Convenient Syntheses of Halo-dibenz[b,f]azepines and Carbamazepine Analogues via N-Arylindoles. Org. Biomol. Chem. 2013, 11, 8426–8434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vaidya, G.N.; Khan, A.; Verma, H.; Kumar, S.; Kumar, D. Structure Ligation Relationship of Amino Acids for the Amination Cross-Coupling Reactions. J. Org. Chem. 2019, 84, 3004–3010. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Lei, M.; Hu, L. Synthesis of 1-Aryl Indoles via Coupling Reaction of Indoles and Aryl Halides Catalyzed by CuI/metformin. Tetrahedron 2014, 70, 5626–5631. [Google Scholar] [CrossRef]
- Balalaie, S.; Bararjanian, M.; Hosseinzadeh, S.; Rominger, F.; Bijanzadeh, H.R.; Wolf, E. Designing a Sequential Ugi/Ullmann Type Reaction for the Synthesis of Indolo[1,2-a]quinoxalinones Catalyzed by CuI/l-Proline. Tetrahedron 2011, 67, 7294–7300. [Google Scholar] [CrossRef]
- Zhang, L.; Zhao, F.; Zheng, M.; Zhai, Y.; Liu, H. Rapid and Selective Access to Three Distinct Sets of Indole-Based Heterocycles from a Single Set of Ugi-Adducts under Microwave Heating. Chem. Commun. 2013, 49, 2894–2896. [Google Scholar] [CrossRef]
- Zhang, L.; Zheng, M.; Zhao, F.; Zhai, Y.; Liu, H. Rapid Generation of Privileged Substructure-Based Compound Libraries with Structural Diversity and Drug-Likeness. ACS Comb. Sci. 2014, 16, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Choi, J.H.; Shin, S.; Heo, J.-N.; Lim, H.J. N-Arylation of Sterically Hindered NH-Nucleophiles: Copper-Mediated Syntheses of Diverse N-Arylindole-2-carboxylates. Synthesis 2015, 47, 3301–3308. [Google Scholar] [CrossRef]
- Lee, J.-H.; Kim, H.; Kim, T.; Song, J.H.; Kim, W.-S.; Ham, J. Functionalization of Organotrifluoroborates via Cu-Catalyzed C–N Coupling Reaction. Bull. KoreanChem. Soc. 2013, 34, 42–48. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.; Han, L.-Y.; Liu, R.-L.; Xu, L.-G.; Bi, Y.-L. Copper-Catalyzed N-Arylation of 2-Arylindoles with Aryl Halides. Chin. Chem. Lett. 2014, 25, 1240–1243. [Google Scholar] [CrossRef]
- Rodrigues, M.B.; Feitosa, S.C.; Wiethan, C.W.; Rosa, W.C.; da Silveira, C.H.; Pagliari, A.B.; Martins, M.A.P.; Zanatta, N.; Iglesias, B.A.; Bonacorso, H.G. Ullmann-Type Copper-Catalyzed Coupling Amination, Photophysical and DNA/HSA-Binding Properties of New 4-(Trifluoromethyl)quinoline Derivatives. J. Fluor. Chem. 2019, 221, 84–90. [Google Scholar] [CrossRef]
- Ghobrial, M.; Mihovilovic, M.D.; Schnurch, M. Exploration of C-H and N-H-Bond Functionalization Towards 1-(1,2-Diarylindol-3-yl)tetrahydroisoquinolines. Beilstein J. Org. Chem. 2014, 10, 2186–2199. [Google Scholar] [CrossRef] [Green Version]
- Yadav, D.K.T.; Rajak, S.S.; Bhanage, B.M. N-Arylation of Indoles with Aryl Halides Using Copper/Glycerol as a Mild and Highly Efficient Recyclable Catalytic System. Tetrahedron Lett. 2014, 55, 931–935. [Google Scholar] [CrossRef]
- Wei, J.J.; Song, W.B.; Zhu, Y.F.; Wei, B.L.; Xuan, L.J. N,N-Dimethyl-d-glucosamine as an Efficient Ligand for Copper-Catalyzed Ullmann-Type Coupling of N-H Heterocycles with Aryl Halides. Tetrahedron 2018, 74, 19–27. [Google Scholar] [CrossRef]
- Chen, Y.; Du, F.; Chen, F.; Zhou, Q.; Chen, G. Methyl-α-d-glucopyranoside as Green Ligand for Selective Copper-Catalyzed N-Arylation. Synthesis 2019, 51, 4590–4600. [Google Scholar] [CrossRef]
- Ge, X.; Zhang, S.; Chen, X.; Liu, X.; Qian, C. A Designed Bi-Functional Sugar-Based Surfactant: Micellar Catalysis for C–X Coupling Reaction in Water. Green Chem. 2019, 21, 2771–2776. [Google Scholar] [CrossRef]
- Yuan, C.; Zhao, Y.; Zheng, L. α-d-Galacturonic Acid as Natural Ligand for Selective Copper-Catalyzed N-Arylation of N-Containing Heterocycles. Synlett 2019, 30, 2173–2180. [Google Scholar] [CrossRef]
- Zhou, Q.; Du, F.; Chen, Y.; Fu, Y.; Sun, W.; Wu, Y.; Chen, G. l-(−)-Quebrachitol as a Ligand for Selective Copper(0)-Catalyzed N-Arylation of Nitrogen-Containing Heterocycles. J. Org. Chem. 2019, 84, 8160–8167. [Google Scholar] [CrossRef]
- Bollenbach, M.; Aquino, P.G.V.; de Araújo-Júnior, J.X.; Bourguignon, J.-J.; Bihel, F.; Salomé, C.; Wagner, P.; Schmitt, M. Efficient and Mild Ullmann-Type N-Arylation of Amides, Carbamates, and Azoles in Water. Chem. Eur. J. 2017, 23, 13676–13683. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zhou, J. Aqueous Copper-Catalyzed N-Arylation of Indoles: The Surfactant Strategy. New J. Chem. 2013, 37, 2537–2540. [Google Scholar] [CrossRef]
- Malavade, V.; Patil, M.; Patil, M. Scope, Kinetics, and Mechanism of “On Water” Cu Catalysis in the C–N Cross-Coupling Reactions of Indole Derivatives. Eur. J. Org. Chem. 2020, 561–569. [Google Scholar] [CrossRef]
- Molaei, H.; Ghanbari, M.M. Practical Copper-Catalyzed N-Arylation of Amines with 20% Aqueous Solution of n-Bu4NOH. Chin. Chem. Lett. 2012, 23, 301–304. [Google Scholar] [CrossRef]
- Abele, E.; Abele, R. KOH/Adogen 464/Proline System for Highly Effective Cu-Catalyzed “On-Water” N–H Arylation of Heteroaromatic Compounds. Chem. Heterocycl. Com. 2013, 49, 1384–1386. [Google Scholar] [CrossRef]
- Engel-Andreasen, J.; Shimpukade, B.; Ulven, T. Selective Copper Catalysed Aromatic N-Arylation in Water. Green Chem. 2013, 15, 336–340. [Google Scholar] [CrossRef] [Green Version]
- Mukhopadhyay, C.; Tapaswi, P.K. Highly Efficient and Simple Catalytic System for the N-Arylation of Some Hindered Aza-Heterocycles in Water. Synth. Commun. 2012, 42, 2217–2228. [Google Scholar] [CrossRef]
- Salomé, C.; Wagner, P.; Bollenbach, M.; Bihel, F.; Bourguignon, J.-J.; Schmitt, M. Buchwald–Hartwig Reactions in Water Using Surfactants. Tetrahedron 2014, 70, 3413–3421. [Google Scholar] [CrossRef]
- Teo, Y.-C.; Yong, F.-F.; Lim, G.S. A Manganese/Copper Bimetallic Catalyst for C–N Coupling Reactions under Mild Conditions in Water. Tetrahedron Lett. 2011, 52, 7171–7174. [Google Scholar] [CrossRef]
- Zhou, G.; Chen, W.; Zhang, S.; Liu, X.; Yang, Z.; Ge, X.; Fan, H.-J. A Newly Designed Carbohydrate-Derived Alkylamine Promotes Ullmann Type C–N Coupling Catalyzed by Copper in Water. Synlett 2019, 30, 193–198. [Google Scholar]
- Zhou, Q.; Du, F.; Chen, Y.; Fu, Y.; Chen, G. “On Water” Promoted N-Arylation Reactions Using Cu (0)/Myo-inositol Catalytic System. Tetrahedron Lett. 2019, 60, 1938–1941. [Google Scholar] [CrossRef]
- Damkaci, F.; Alawaed, A.; Vik, E. N-Picolinamides as Ligands for Ullmann-type CN Coupling Reactions. Tetrahedron Lett. 2016, 57, 2197–2200. [Google Scholar] [CrossRef]
- Su, J.; Qiu, Y.; Jiang, S.; Zhang, D. New Ligands for Copper-Catalyzed C–N Coupling Reactions at Gentle Temperature. Chin. J. Chem. 2014, 32, 685–688. [Google Scholar] [CrossRef]
- Yang, K.; Qiu, Y.; Li, Z.; Wang, Z.; Jiang, S. Ligands for Copper-Catalyzed C−N Bond Forming Reactions with 1 Mol% CuBr as Catalyst. J. Org. Chem. 2011, 76, 3151–3159. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Y.; Yang, B.; Zhang, A.; Yao, Q. N-(1-Oxy-2-picolyl)oxalamic Acids as a New Type of O,O-Ligands for the Cu-Catalyzed N-Arylation of Azoles with Aryl Halides in Water or Organic solvent. Org. Biomol. Chem. 2015, 13, 4101–4114. [Google Scholar] [CrossRef]
- Taywade, A.; Chavan, S.; Ulhe, A.; Berad, B. Unique CuI-Pyridine Based Ligands Catalytic Systems for N-Arylation of Indoles and Other Heterocycles. Synth. Commun. 2018, 48, 1443–1453. [Google Scholar] [CrossRef]
- Echeverry-Gonzalez, C.A.; Ortiz Villamizar, M.C.; Kouznetsov, V.V. The Remarkable Selectivity of the 2-Arylquinoline-Based Acyl Hydrazones Toward Copper Salts: Exploration of Their Catalytic Applications in the Copper Catalysed N-Arylation of Indole Derivatives and C1-Alkynylation of Tetrahydroisoquinolines via the A3 Reaction. New J. Chem. 2021, 45, 243–250. [Google Scholar]
- Abe, T.; Takahashi, Y.; Matsubara, Y.; Yamada, K. An Ullmann N-Arylation/2-Amidation Cascade by Self-Relay Copper Catalysis: One-Pot Synthesis of Indolo[1,2-a]quinazolinones. Org. Chem. Front. 2017, 4, 2124–2127. [Google Scholar] [CrossRef]
- Ghosh, T.; Maity, P.; Ranu, B.C. Cobalt-Copper Catalyzed C(sp2)—N Cross Coupling of Amides or Nitrogenated Heterocycles with Styrenyl or Aryl Halides. ChemistrySelect 2018, 3, 4406–4412. [Google Scholar] [CrossRef]
- Mostafa, M.A.B.; Calder, E.D.D.; Racys, D.T.; Sutherland, A. Intermolecular Aryl C−H Amination through Sequential Iron and Copper Catalysis. Chem. Eur. J. 2017, 23, 1044–1047. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sadhu, P.; Punniyamurthy, T. Copper(ii)-Mediated Regioselective N-Arylation of Pyrroles, Indoles, Pyrazoles and Carbazole via Dehydrogenative Coupling. Chem. Commun. 2016, 52, 2803–2806. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, S.; De, P.B.; Punniyamurthy, T. Copper(II)-Mediated Chelation-Assisted Regioselective N-Naphthylation of Indoles, Pyrazoles and Pyrrole through Dehydrogenative Cross-Coupling. J. Org. Chem. 2017, 82, 4883–4890. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Hu, Z.-Y.; Li, X.-C.; Guo, X.-X. Copper-Catalyzed Decarboxylative N-Arylation of Indole-2-carboxylic Acids. Synthesis 2019, 51, 1803–1808. [Google Scholar] [CrossRef]
- Petiot, P.; Dansereau, J.; Gagnon, A. Copper-Catalyzed N-Arylation of Azoles and Diazoles Using Highly Functionalized Trivalent Organobismuth Reagents. RSC Adv. 2014, 4, 22255–22259. [Google Scholar] [CrossRef]
- Hébert, M.; Petiot, P.; Benoit, E.; Dansereau, J.; Ahmad, T.; Le Roch, A.; Ottenwaelder, X.; Gagnon, A. Synthesis of Highly Functionalized Triarylbismuthines by Functional Group Manipulation and Use in Palladium- and Copper-Catalyzed Arylation Reactions. J. Org. Chem. 2016, 81, 5401–5416. [Google Scholar] [CrossRef]
- Jadhav, B.D.; Pardeshi, S.K. A Facile and Practical Copper Diacetate Mediated, Ligand Free C–N Cross Coupling of Trivalent Organobismuth Compounds with Amines and N-heteroarenes. RSC Adv. 2016, 6, 14531–14537. [Google Scholar] [CrossRef]
- Le Roch, A.; Hébert, M.; Gagnon, A. Copper-Promoted O-Arylation of the Phenol Side Chain of Tyrosine Using Triarylbismuthines. Eur. J. Org. Chem. 2020, 5363–5367. [Google Scholar] [CrossRef]
- Le Roch, A.; Chan, H.-C.; Gagnon, A. Copper-Promoted N-Arylation of the Indole Side Chain of Tryptophan Using Triarylbismuthines. Eur. J. Org. Chem. 2020, 5815–5819. [Google Scholar] [CrossRef]
- Alonso, I.; Alvarez, R.; de Lera, Á.R. Indole–Indole Ullmann Cross-Coupling for CAr–N Bond Formation: Total Synthesis of (–)-Aspergilazine A. Eur. J. Org. Chem. 2017, 4948–4954. [Google Scholar] [CrossRef]
- Modha, S.G.; Greaney, M.F. Atom-Economical Transformation of Diaryliodonium Salts: Tandem C−H and N−H Arylation of Indoles. J. Am. Chem. Soc. 2015, 137, 1416–1419. [Google Scholar] [CrossRef]
- Ziegler, D.T.; Choi, J.; Muñoz-Molina, J.M.; Bissember, A.C.; Peters, J.C.; Fu, G.C. A Versatile Approach to Ullmann C−N Couplings at Room Temperature: New Families of Nucleophiles and Electrophiles for Photoinduced, Copper-Catalyzed Processes. J. Am. Chem. Soc. 2013, 135, 13107–13112. [Google Scholar] [CrossRef] [Green Version]
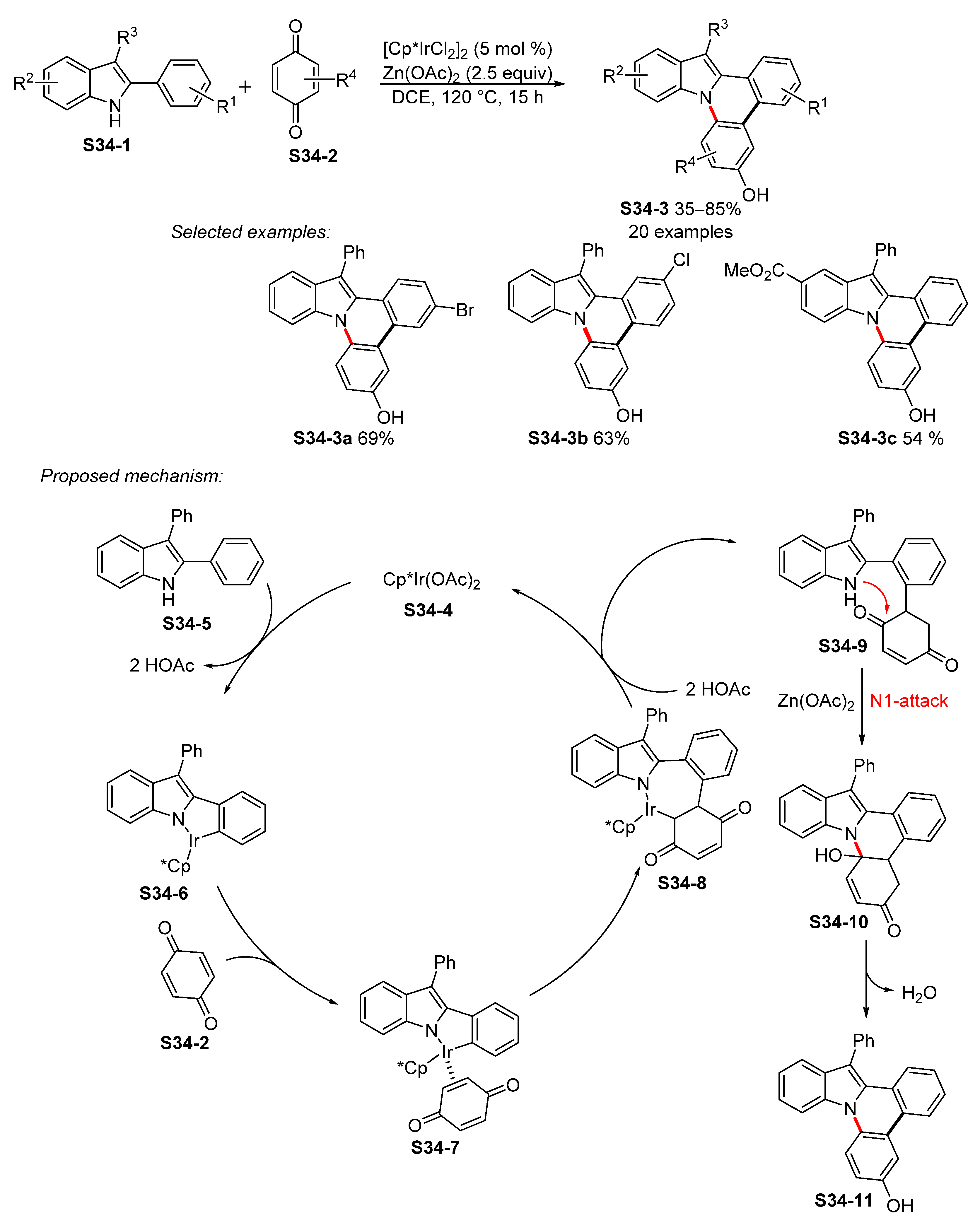
- Guo, S.; Liu, Y.; Zhang, X.; Fan, X. Iridium-Catalyzed Oxidative Annulation of 2-Arylindoles with Benzoquinone Leading to Indolo[1,2-f]phenanthridin-6-ols. Adv. Synth. Catal. 2020, 362, 3011–3020. [Google Scholar] [CrossRef]
- Kong, L.; Sun, Y.; Zheng, Z.; Tang, R.; Wang, M.; Li, Y. Chemoselective N–H or C-2 Arylation of Indole-2-carboxamides: Controllable Synthesis of Indolo[1,2-a]quinoxalin-6-ones and 2,3′-Spirobi[indolin]-2′-ones. Org. Lett. 2018, 20, 5251–5255. [Google Scholar] [CrossRef]
- Liu, X.; Cao, Z.; Huang, H.; Liu, X.; Tan, Y.; Chen, H.; Pei, Y.; Tan, S. Novel D–D–π-A Organic Dyes Based on Triphenylamine and Indole-Derivatives for High Performance Dye-Sensitized Solar Cells. J. Power Sources 2014, 248, 400–406. [Google Scholar] [CrossRef]
- Keruckas, J.; Grazulevicius, J.V.; Volyniuk, D.; Cherpak, V.; Stakhira, P. 3,6-Bis(indol-1-yl)-9-phenylcarbazoles as Electroactive Materials for Electrophosphorescent Diodes. Dye. Pigment. 2014, 100, 66–72. [Google Scholar] [CrossRef]
- Hussain, F.; Wang, X.; Wang, S. Impact of Bidentate N,C-Chelate Ligands on the Performance of Phosphorescent Pt(II) Complexes as Oxygen Sensors. J. Organomet. Chem. 2019, 880, 300–311. [Google Scholar] [CrossRef]
- Xiang, N.; Gao, Z.; Tian, G.; Chen, Y.; Liang, W.; Huang, J.; Dong, Q.; Wong, W.-Y.; Su, J. Novel Fluorene/Indole-Based Hole Transport Materials with High Thermal Stability for Efficient OLEDs. Dye. Pigment. 2017, 137, 36–42. [Google Scholar] [CrossRef]
- Jia, B.; Lian, H.; Chen, Z.; Chen, Y.; Huang, J.; Dong, Q. Novel Carbazole/Indole/Thiazole-Based Host Materials with High Thermal Stability for Efficient Phosphorescent Organic Light-Emitting Diodes. Dye. Pigment. 2017, 147, 552–559. [Google Scholar] [CrossRef]
- Selvam, R.; Subramanian, K. Benzimidazole-Indole-Chalcone Connected Methacrylate-Based Side Chain D-π-A Polymer and Its Application in Organic Photovoltaics. J. Polym. Sci. A Polym. Chem. 2017, 55, 997–1007. [Google Scholar] [CrossRef]
- Chen, Y.; Xie, J.; Wang, Z.; Cao, J.; Chen, H.; Huang, J.; Zhang, J.; Su, J. Highly Efficient Bipolar Host Material Based-on Indole and Triazine Moiety for Red Phosphorescent Light-Emitting Diodes. Dye. Pigment. 2016, 124, 188–195. [Google Scholar] [CrossRef]
- Crocetti, L.; Schepetkin, I.A.; Ciciani, G.; Giovannoni, M.P.; Guerrini, G.; Iacovone, A.; Khlebnikov, A.I.; Kirpotina, L.N.; Quinn, M.T.; Vergelli, C. Synthesis and Pharmacological Evaluation of Indole Derivatives as Deaza Analogues of Potent Human Neutrophil Elastase Inhibitors. Drug Develop. Res. 2016, 77, 285–299. [Google Scholar] [CrossRef] [Green Version]
- Hirayama, T.; Okaniwa, M.; Imada, T.; Ohashi, A.; Ohori, M.; Iwai, K.; Mori, K.; Kawamoto, T.; Yokota, A.; Tanaka, T.; et al. Synthetic Studies of Centromere-Associated Protein-E (CENP-E) Inhibitors: 1. Exploration of Fused Bicyclic Core Scaffolds Using Electrostatic Potential Map. Bioorg. Med. Chem. 2013, 21, 5488–5502. [Google Scholar] [CrossRef]
- Alonso, J.A.; Andrés, M.; Bravo, M.; Buil, M.A.; Calbet, M.; Castro, J.; Eastwood, P.R.; Eichhorn, P.; Esteve, C.; Gómez, E.; et al. Structure–Activity Relationships (SAR) and Structure–Kinetic Relationships (SKR) of Bicyclic Heteroaromatic Acetic Acids as Potent CRTh2 Antagonists I. Bioorg. Med. Chem. Lett. 2014, 24, 5118–5122. [Google Scholar] [CrossRef] [PubMed]
- Bzeih, T.; Lamaa, D.; Frison, G.; Hachem, A.; Jaber, N.; Bignon, J.; Retailleau, P.; Alami, M.; Hamze, A. Csp2–Csp2 and Csp2–N Bond Formation in a One-Pot Reaction between N-Tosylhydrazones and Bromonitrobenzenes: An Unexpected Cyclization to Substituted Indole Derivatives. Org. Lett. 2017, 19, 6700–6703. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhong, Y.; Yan, L.-N.; Sun, X.; Gong, T.; Zhang, Z.-R. Synthesis and Preliminary Evaluation of Curcumin Analogues as Cytotoxic Agents. Bioorg. Med. Chem. Lett. 2011, 21, 1010–1014. [Google Scholar] [CrossRef]
- Bao, X.; Zhu, W.; Yuan, W.; Zhu, X.; Yan, Y.; Tang, H.; Chen, Z. Design, Synthesis and Evaluation of Novel Potent Angiotensin II Receptor 1 Antagonists. Eur. J. Med. Chem. 2016, 123, 115–127. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Anh, N.T.P.; Yan, Y.-J.; Xia, M.-B.; Wang, Y.-H.; Qiu, Y.; Chen, Z.-L. Design, Synthesis and Biological Evaluation of AT1 Receptor Blockers Derived from 6-Substituted Aminocarbonyl Benzimidazoles. Eur. J. Med. Chem. 2019, 181, 111553. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Bao, X.; Ren, H.; Da, Y.; Wu, D.; Li, F.; Yan, Y.; Wang, L.; Chen, Z. N-Phenyl Indole Derivatives as AT1 Antagonists with Anti-Hypertension Activities: Design, Synthesis and Biological Evaluation. Eur. J. Med. Chem. 2016, 115, 161–178. [Google Scholar] [CrossRef]
- Zhu, W.; Bao, X.; Ren, H.; Liao, P.; Zhu, W.; Yan, Y.; Wang, L.; Chen, Z. Design, Synthesis, and Pharmacological Evaluation of 5-oxo-1,2,4-oxadiazole Derivatives as AT1 Antagonists with Antihypertension Activities. Clin. Exp. Hypertens. 2016, 38, 435–442. [Google Scholar] [CrossRef]
- Zhu, W.; Da, Y.; Wu, D.; Zheng, H.; Zhu, L.; Wang, L.; Yan, Y.; Chen, Z. Design, Synthesis and Biological Evaluation of New 5-Nitro Benzimidazole Derivatives as AT1 Antagonists with Anti-Hypertension Activities. Bioorg. Med. Chem. 2014, 22, 2294–2302. [Google Scholar] [CrossRef]
- Thiyagamurthy, P.; Teja, C.; Naresh, K.; Gomathi, K.; Nawaz Khan, F.-R. Design, Synthesis and in Silico Evaluation of Benzoxazepino(7,6-b)quinolines as Potential Antidiabetic Agents. Med. Chem. Res. 2020, 29, 1882–1901. [Google Scholar] [CrossRef]
- Nandwana, N.K.; Singh, R.P.; Patel, O.P.S.; Dhiman, S.; Saini, H.K.; Jha, P.N.; Kumar, A. Design and Synthesis of Imidazo/Benzimidazo[1,2-c]quinazoline Derivatives and Evaluation of Their Antimicrobial Activity. ACS Omega 2018, 3, 16338–16346. [Google Scholar] [CrossRef] [Green Version]
- Miller, L.M.; Keune, W.-J.; Castagna, D.; Young, L.C.; Duffy, E.L.; Potjewyd, F.; Salgado-Polo, F.; Engel García, P.; Semaan, D.; Pritchard, J.M.; et al. Structure–Activity Relationships of Small Molecule Autotaxin Inhibitors with a Discrete Binding Mode. J. Med. Chem. 2017, 60, 722–748. [Google Scholar] [CrossRef] [Green Version]
- Xu, G.; Liu, T.; Zhou, Y.; Yang, X.; Fang, H. 1-Phenyl-1H-indole Derivatives as a New Class of Bcl-2/Mcl-1 Dual Inhibitors: Design, Synthesis, and Preliminary Biological Evaluation. Bioorg. Med. Chem. 2017, 25, 5548–5556. [Google Scholar] [CrossRef]
- Fox, B.M.; Beck, H.P.; Roveto, P.M.; Kayser, F.; Cheng, Q.; Dou, H.; Williamson, T.; Treanor, J.; Liu, H.; Jin, L.; et al. A Selective Prostaglandin E2 Receptor Subtype 2 (EP2) Antagonist Increases the Macrophage-Mediated Clearance of Amyloid-Beta Plaques. J. Med. Chem. 2015, 58, 5256–5273. [Google Scholar] [CrossRef] [PubMed]
- Quirit, J.G.; Lavrenov, S.N.; Poindexter, K.; Xu, J.; Kyauk, C.; Durkin, K.A.; Aronchik, I.; Tomasiak, T.; Solomatin, Y.A.; Preobrazhenskaya, M.N.; et al. Indole-3-carbinol (I3C) Analogues are Potent Small Molecule Inhibitors of NEDD4-1 Ubiquitin Ligase Activity that Disrupt Proliferation of Human Melanoma Cells. Biochem. Pharmacol. 2017, 127, 13–27. [Google Scholar] [CrossRef]
- Giordanetto, F.; Knerr, L.; Nordberg, P.; Pettersen, D.; Selmi, N.; Beisel, H.-G.; de la Motte, H.; Månsson, Å.; Dahlström, M.; Broddefalk, J.; et al. Design of Selective sPLA2-X Inhibitor (−)-2-{2-[Carbamoyl-6-(trifluoromethoxy)-1H-indol-1-yl]pyridine-2-yl}propanoic Acid. ACS Med. Chem. Lett. 2018, 9, 600–605. [Google Scholar] [CrossRef] [PubMed]
- Knerr, L.; Giordanetto, F.; Nordberg, P.; Pettersen, D.; Selmi, N.; Beisel, H.-G.; de la Motte, H.; Olsson, T.; Perkins, T.D.J.; Herslöf, M.; et al. Discovery of a Series of Indole-2-Carboxamides as Selective Secreted Phospholipase A2 Type X (sPLA2-X) Inhibitors. ACS Med. Chem. Lett. 2018, 9, 594–599. [Google Scholar] [CrossRef] [PubMed]
- Abate, C.; Pati, M.L.; Contino, M.; Colabufo, N.A.; Perrone, R.; Niso, M.; Berardi, F. From Mixed Sigma-2 Receptor/P-Glycoprotein Targeting Agents to Selective P-Glycoprotein Modulators: Small Structural Changes Address the Mechanism of Interaction at the Efflux Pump. Eur. J. Med. Chem. 2015, 89, 606–615. [Google Scholar] [CrossRef] [PubMed]
- Pati, M.L.; Abate, C.; Contino, M.; Ferorelli, S.; Luisi, R.; Carroccia, L.; Niso, M.; Berardi, F. Deconstruction of 6,7-Dimethoxy-1,2,3,4-tetrahydroisoquinoline Moiety to Separate P-Glycoprotein (P-gp) Activity from σ2 Receptor Affinity in Mixed P-gp/σ2 Receptor Agents. Eur. J. Med. Chem. 2015, 89, 691–700. [Google Scholar] [CrossRef] [PubMed]
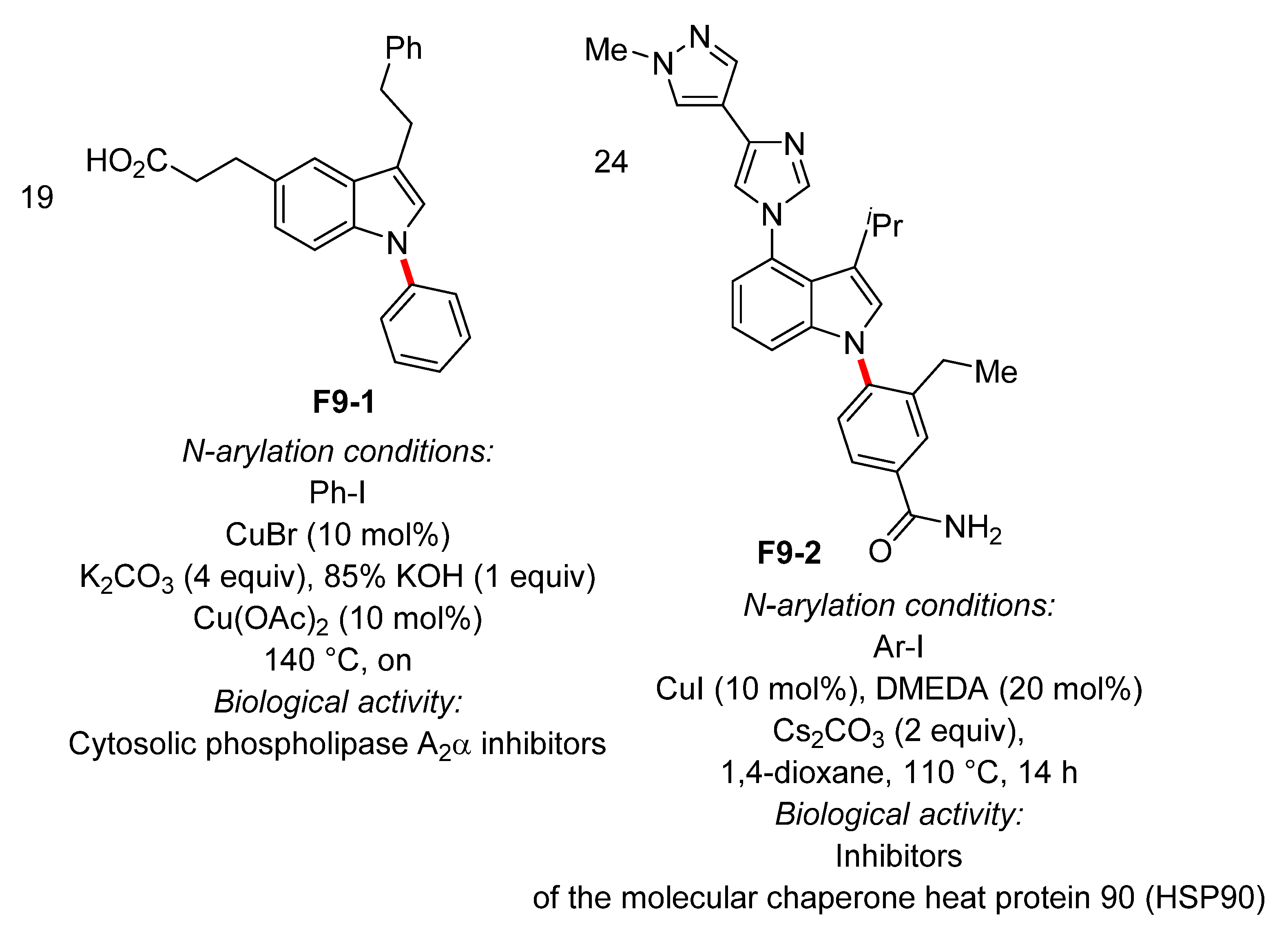
- Tomoo, T.; Nakatsuka, T.; Katayama, T.; Hayashi, Y.; Fujieda, Y.; Terakawa, M.; Nagahira, K. Design, Synthesis, and Biological Evaluation of 3-(1-Aryl-1H-indol-5-yl)propanoic Acids as New Indole-Based Cytosolic Phospholipase A2α Inhibitors. J. Med. Chem 2014, 57, 7244–7262. [Google Scholar] [CrossRef]
- Uno, T.; Kawai, Y.; Yamashita, S.; Oshiumi, H.; Yoshimura, C.; Mizutani, T.; Suzuki, T.; Chong, K.T.; Shigeno, K.; Ohkubo, M.; et al. Discovery of 3-Ethyl-4-(3-isopropyl-4-(4-(1-methyl-1H-pyrazol-4-yl)-1H-imidazol-1-yl)-1H-pyrazolo[3,4-b]pyridin-1-yl)benzamide (TAS-116) as a Potent, Selective, and Orally Available HSP90 Inhibitor. J. Med. Chem. 2019, 62, 531–551. [Google Scholar] [CrossRef] [PubMed]



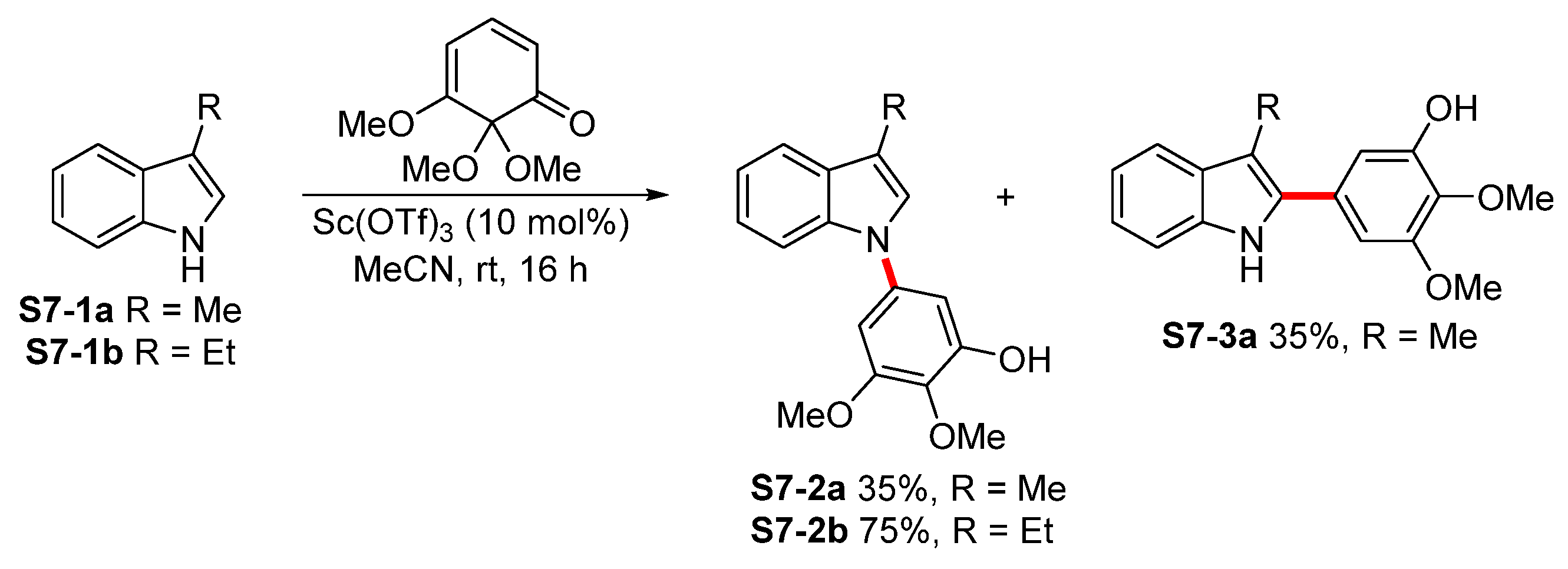
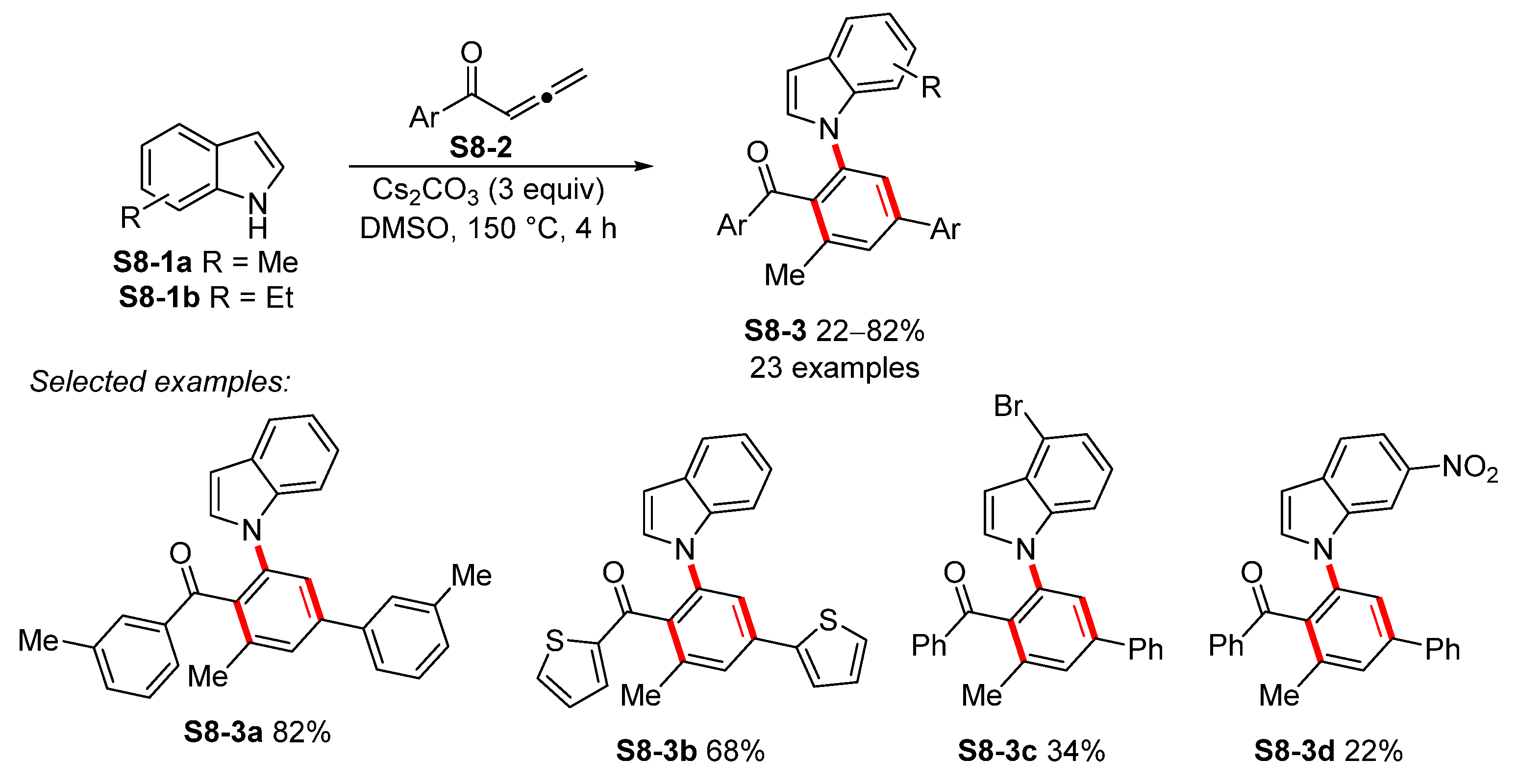


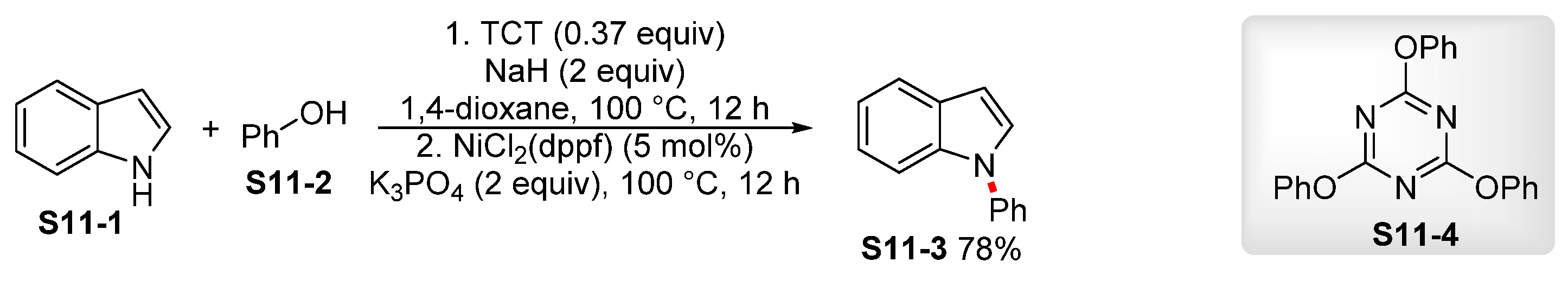
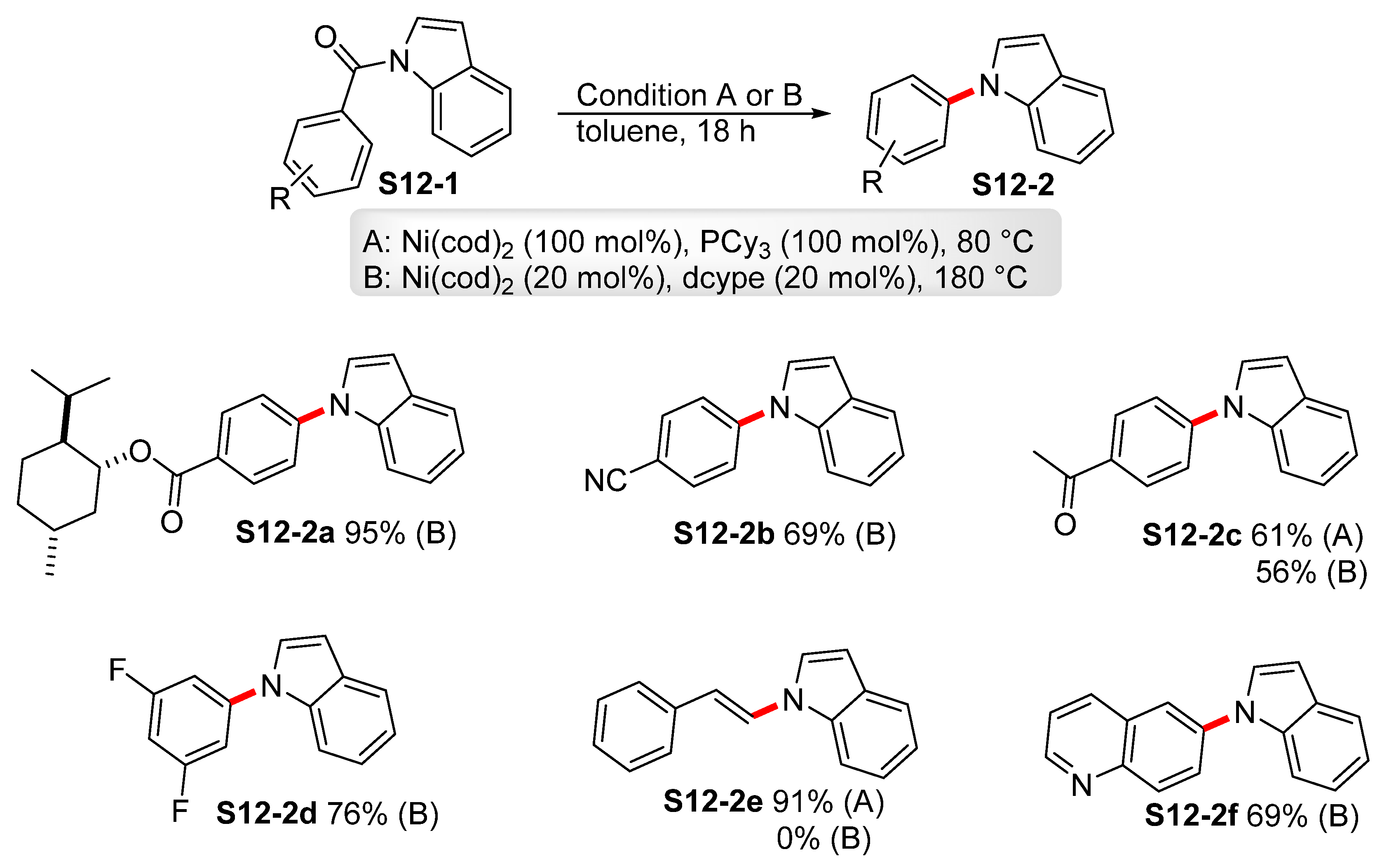


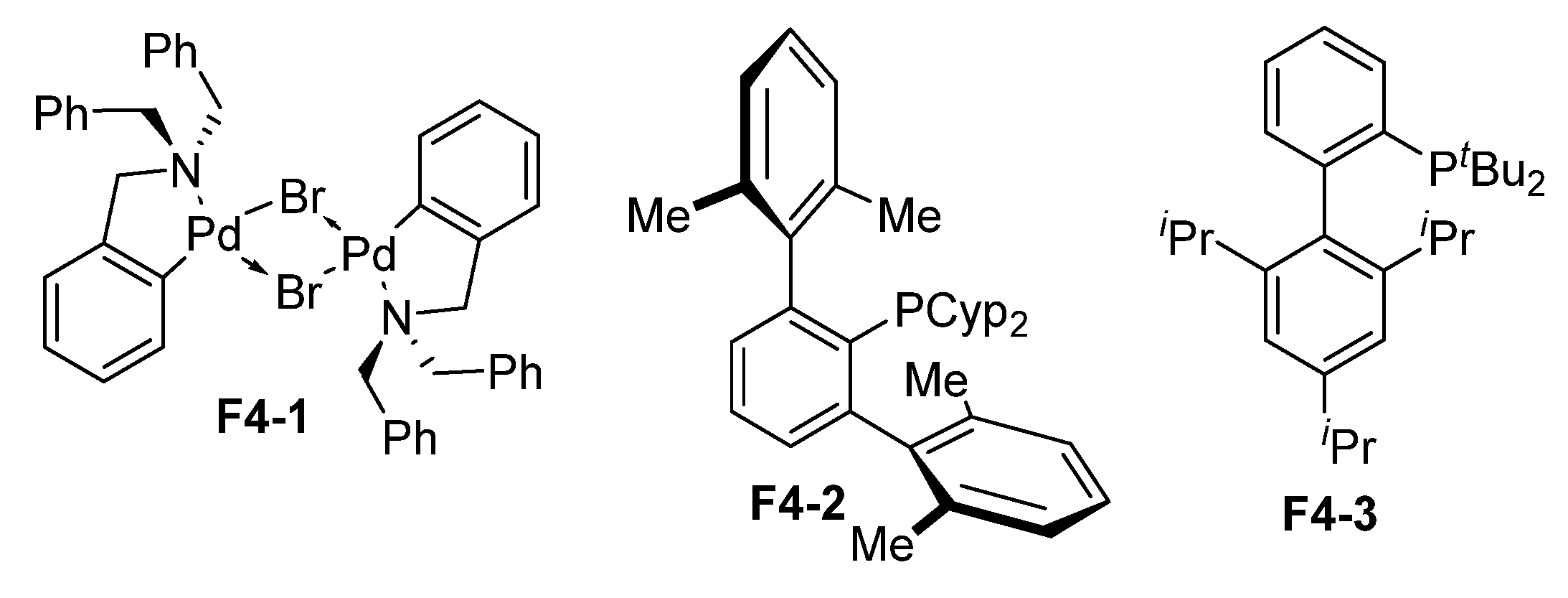

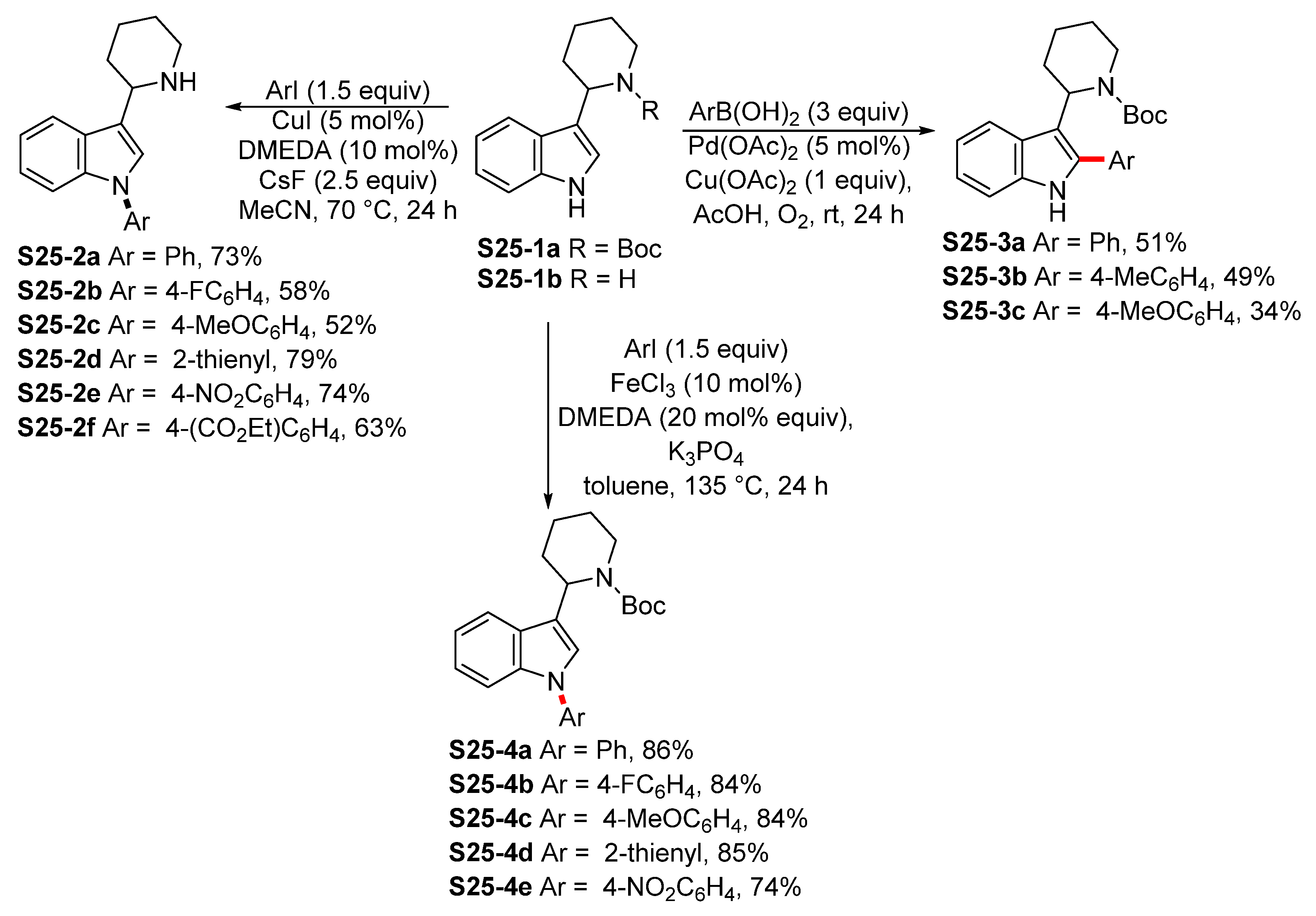
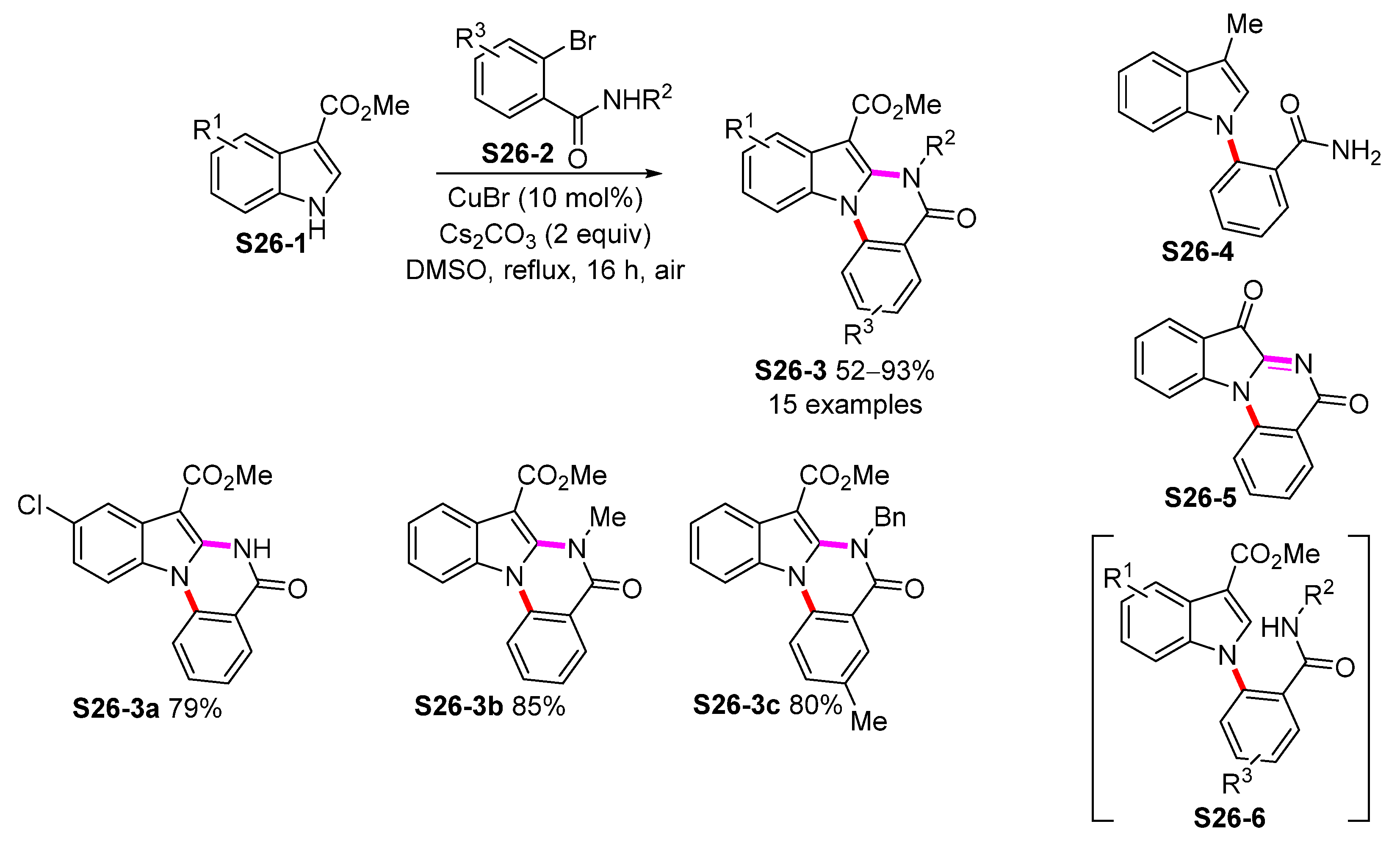
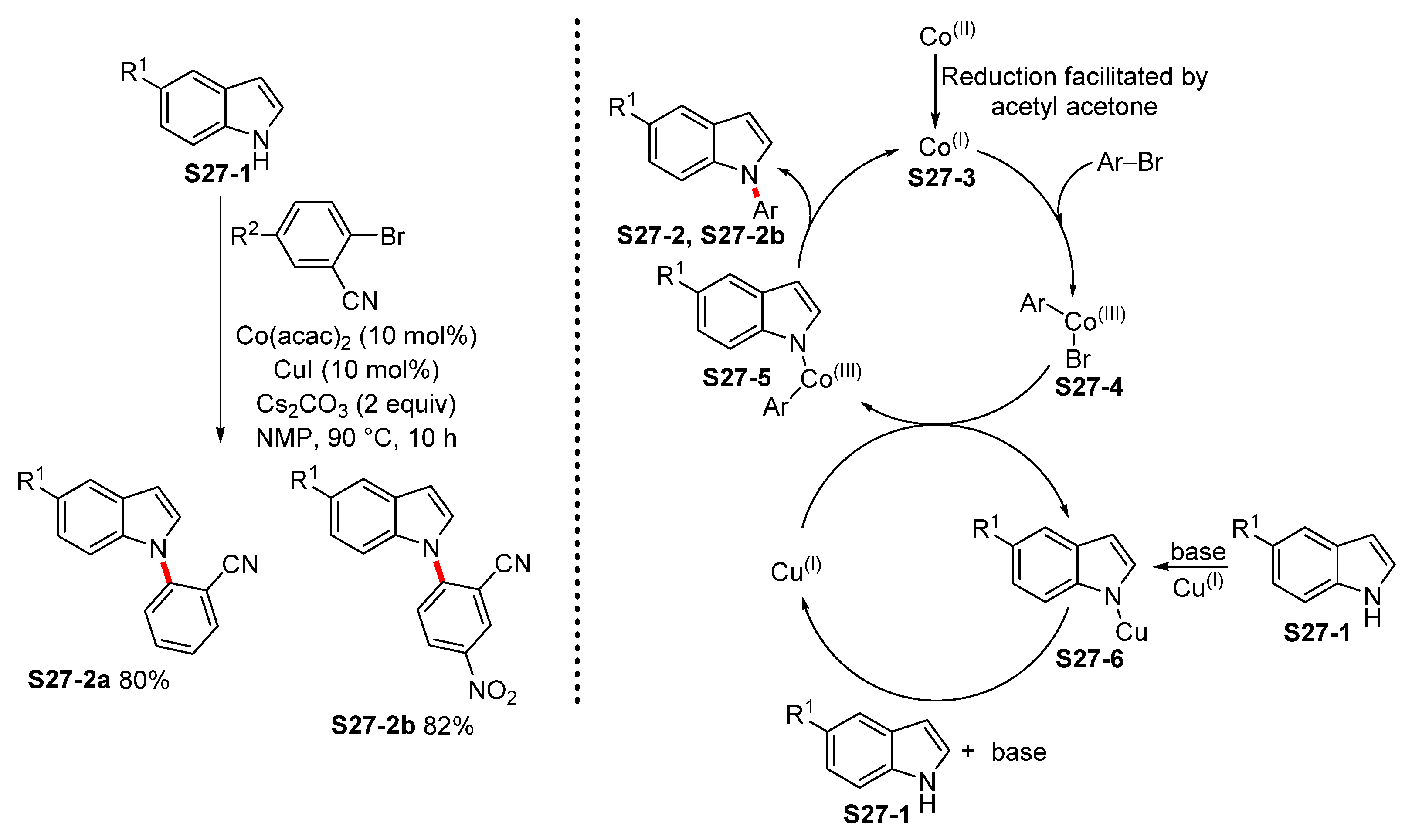


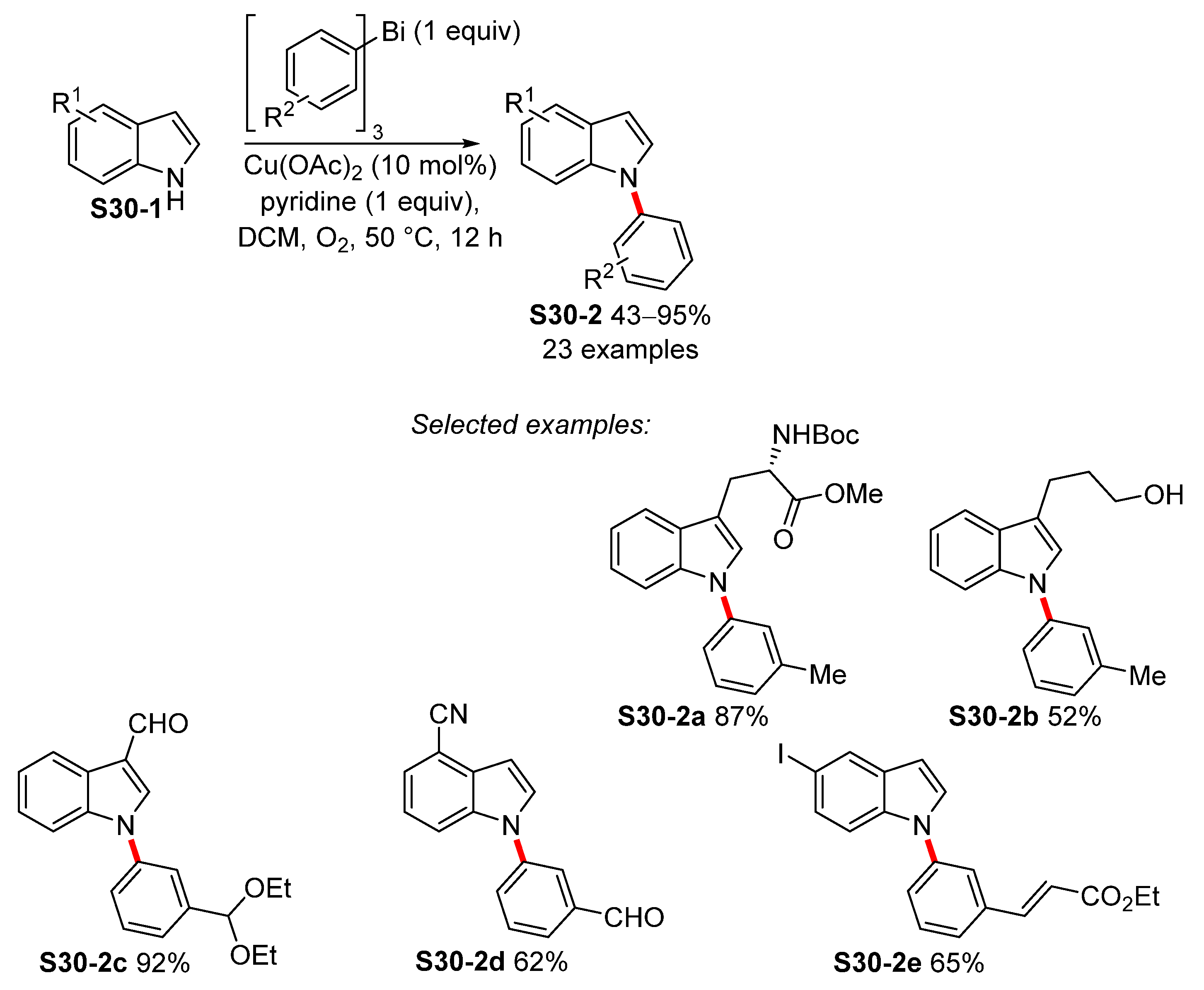

 | ||||
|---|---|---|---|---|
| Entry | Conditions | No. of Examples | indoleFG | Ar/hetArFG |
| 1 | CuO nano (5 mol%), 1,10-phen (50 mol%) KOH (2 equiv), DMSO, reflux, 24 h | 4 56–80% | – | Cl |
| 2 | Cu@CeO2 (5 mol%), 1,10-phen (50 mol%) KOH (2 equiv), DMSO, reflux, 24 h | 6 82–89% | – | Cl |
| 3 | PhI, CuO NFs@MP (15 mg/mmol) K2CO3 (1.5 equiv), DMF, 100 °C, 6 h | 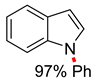 | ||
| 4 | CuO NPs (10 mg/1 mmol of indole), TEA (2 equiv), DMF, reflux, 2 h | 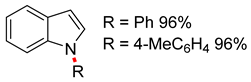 | ||
| 5 | CNT–CuO (5 mol%), tBuONa (3 equiv), DMSO, 120 °C, 12 h | 21 42–95% | Cl, F | Cl, F, COMe, CO2Me, CN, CF3, 3,4-(CH2OCH2), NO2, SMe |
| 6 | CuO nano (5 mol%), K2CO3 (1 equiv), DMF, reflux, 8 h | 15 62–98% | Br, CN, NO2, OMe | NO2 |
| 7 | Copper-decorated OMMT (5 mol%), K2CO3 (2 equiv), DMSO, 130 °C, 6 h | 8 73–97% | CN | OH, NH2, |
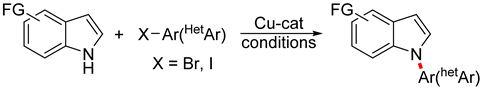 | |||||
|---|---|---|---|---|---|
| Entry | Conditions | No. of Examples | indoleFG | Ar/hetArFG | |
| 1 | CuI (15 mol%)/L1 (15 mol%) tBuOK (2 equiv), DMSO, 120 °C, 36–48 h | 25 50–93% | CH=O, OMe | NO2, OMe, | |
| 2 | CuI (2 mol%)/L2 (4 mol%) K2CO3 (2.5 equiv), DMSO, 90 °C | 19 31–98% | – | Cl, CF3, CN, COCH3, CO2H, F, NH2, NO2 | |
| 3 | CuI (5 mol%)/L-proline (10 mol%), K2CO3 (5 equiv) DMSO, 80–90 °C, 24 h | 18 51–97% | Br, Cl, F, MeO, | Br, Cl, F, OMe, OH | |
| 4 | CuI (5 mol%), L-methionine (10 mol%), K2CO3 (2 equiv), DMF, 100 °C, 24 h | 16 72–92% | Br, F, NO2, OBn, OMe, | F, CF3, NO2, | |
| 5 | CuI (10 mol%), L3 (20 mol%), Cs2CO3 (2 equiv), DMF, 130 °C, 3–24 h | 29 62–91% | CHO, NO2, OMe | Br, Cl, NO2, OMe | |
 | |||||
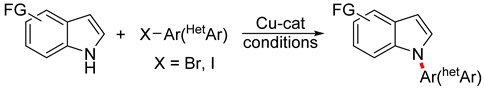 | |||||
|---|---|---|---|---|---|
| Entry | Conditions | No. of Examples | indoleFG | Ar/hetArFG | |
| 1 | CuO (2 equiv), K2CO3 (2 equiv), pyridine, reflux, 2–3 d | 11 15–96% | Cl, F, CO2Et, OBn | CF3, F, OMe, | |
| 2 | CuI (10 mol%), DMEDA (20 mol%), Cs2CO3 (1 equiv) DMSO, 120 °C, 4 h | 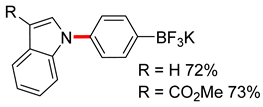 | |||
| 3 | CuI (10 mol%), DMEDA (20 mol%), K3PO4 (2 equiv), toluene, 110 °C, 24 h | 13 74–91% | Ph | Cl, CO2Et, F, OMe | |
| 4 | CuI (10 mol%), K3PO4 (3 equiv), DMEDA (20 mol%), toluene, 110 °C, 12 h |  | |||
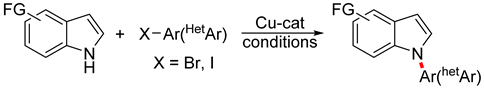 | ||||
|---|---|---|---|---|
| Entry | Conditions | No. of Examples | indoleFG | Ar/hetArFG |
| 1 | CuI (10 mol%), DMSO (1 equiv), K2CO3 (2 equiv), glycerol, 120 °C, 24 h | 12 65–92% | Br, NO2, OMe | Br, F, NO2, OMe |
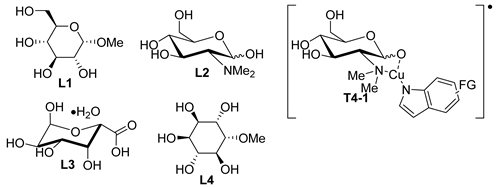
| 2 | Cu (10 mol%), L1 (20 mol%), Cs2CO3 (3 equiv) DMSO-H2O (1:1), 100 °C, 12–24 h | 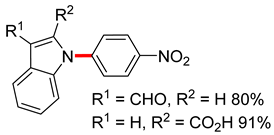 | ||
| 3 | Cu2O (10 mol%), L2 (20 mol%), Cs2CO3 (2 equiv), DMSO, 120 °C, 3–24 h | 15 86–97% | CH3, CHO | Cl, COMe, CO2Me, COCH2CO2Et OMe, NO2 |
 | ||||
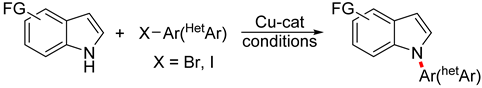 | ||||
|---|---|---|---|---|
| Entry | Conditions | No. of Examples | indoleFG | Ar/hetArFG |
| 1 | CuBr2 (10 mol%), L1 (10 mol%), D-glucose (10 mol%), tBuONa (2 equiv), TPGS-750-M (5 wt%), 50 °C, 24 h | 11 43–95% | Br, Cl, F, OMOM, CHO, OMe, CH2CN, Cl, NO2 OMe | NO2, OMe |
| 2 | CuI (10 mol%), L2 (20 mol%), K3PO4 (2 equiv), betaine (2 equiv.), water, 90 °C, 10 h | 10 57–95% | – | Br, Cl, COMe, F, NO2, OMe |
| 3 | CuI (5 mol%), L3 (10 mol%), KOH (2 equiv), DME-H2O (3:7), 95 °C, 20 h | 23 42–95% | Br, CN, COMe, NO2 | Br, Cl, I, OMe |
 | ||||
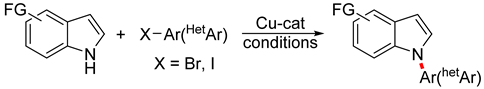 | ||||
|---|---|---|---|---|
| Entry | Conditions | No. of Examples | indoleFG | Ar/hetArFG |
| 1 | Cu2O (10 mol%), L1 (4 mol%), Cs2CO3 (2 equiv), DMSO, 120 °C, 20 h | 6 72–95% | – | NO2, OMe |
| 2 | CuI (10 mol%), L2 (10 mol%), K2CO3 (2 equiv), DMSO, 120 °C, 4–16 h | 7 84–97% | – | NO2, OMe |
| 3 | CuI (10 mol%), L3 (10 mol%), K2CO3 (1.4 equiv), DMSO, 110 °C, 24 h | 4 69–99 | CHO | OMe |
| 4 | CuCl (8 mol%), L4 (16 mol%), NaOH (1 equiv), DMSO, 120 °C, 24 h | 8 25–98% | – | Br, Cl, OEt, OMe |
 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oeser, P.; Koudelka, J.; Petrenko, A.; Tobrman, T. Recent Progress Concerning the N-Arylation of Indoles. Molecules 2021, 26, 5079. https://doi.org/10.3390/molecules26165079
Oeser P, Koudelka J, Petrenko A, Tobrman T. Recent Progress Concerning the N-Arylation of Indoles. Molecules. 2021; 26(16):5079. https://doi.org/10.3390/molecules26165079
Chicago/Turabian StyleOeser, Petr, Jakub Koudelka, Artem Petrenko, and Tomáš Tobrman. 2021. "Recent Progress Concerning the N-Arylation of Indoles" Molecules 26, no. 16: 5079. https://doi.org/10.3390/molecules26165079
APA StyleOeser, P., Koudelka, J., Petrenko, A., & Tobrman, T. (2021). Recent Progress Concerning the N-Arylation of Indoles. Molecules, 26(16), 5079. https://doi.org/10.3390/molecules26165079






