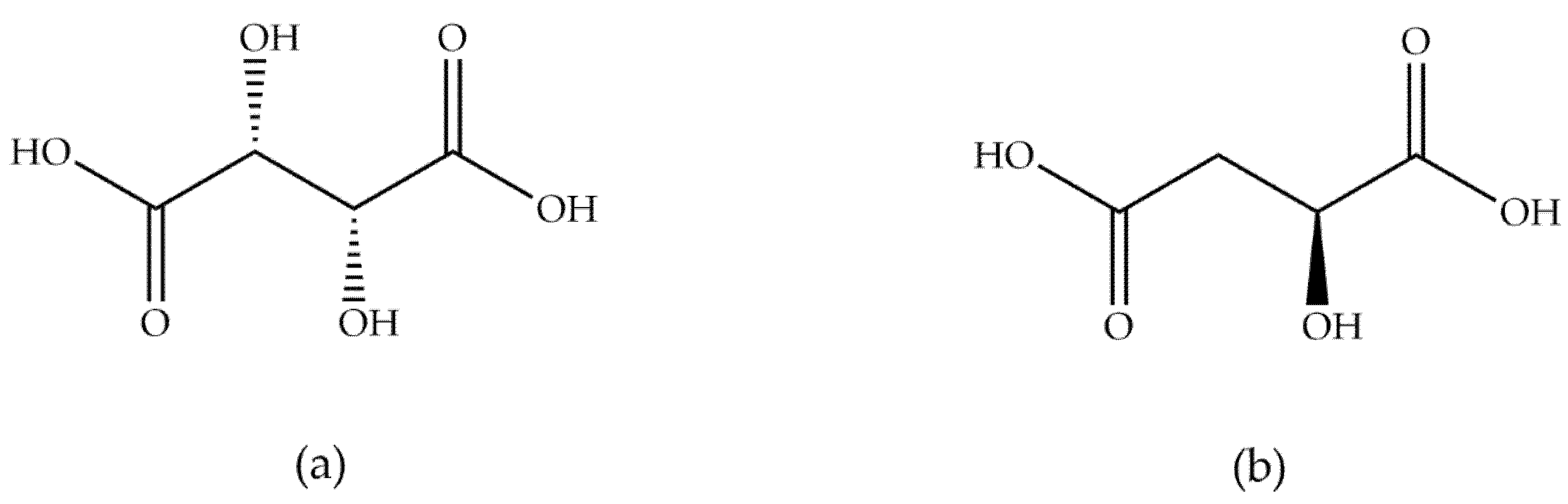2.2. Binary Systems of Copper(II) Ion/α-Hydroxy Acid
In the tested pH range 2.5–11.0, the carboxyl groups of malic and tartaric acids are deprotonated and can be potential coordination centers. The values of the protonation constants of the hydroxyl groups for the α-hydroxy acids were determined by
13C NMR spectroscopy and is from about 11.6 to about 14.0 [
40,
41]. Such a high value causes hydroxyl groups to be ignored in studies of complex formation in aqueous solutions.
Relatively low p
Ka values and protonation constants (
Table 2) of the dissociation of carboxyl groups of the studied α-hydroxy acids indicate that the deprotonation of these groups occurred at low pH values. In biological systems, these compounds can occur in partly or fully deprotonated forms, which allow the formation of complexes with metal ions such as copper(II), cobalt(II), or nickel(II).
Analysis of the obtained p
Ka values for the studied acids confirmed that tartaric acid shows stronger acid properties than malic acid, which contains only one hydroxyl group in the structure. Structural formulae of the studied acids are displayed in
Figure 1.
Computer analysis of the potentiometric data of the binary systems of L-tartaric or L-malic acids has shown the formation of complexes of similar type with copper(II) ions. For these systems, the formations of both monomeric and dimeric forms were found. The presence of two acid molecules in the internal coordination sphere was observed only in systems with an excess amount of ligands. The values of the overall stability constants (log
β) and the equilibrium constants of complex formation (log
Ke) are presented in
Table 1. Equilibrium constants (log
Ke) of binary complexes were determined based on the proposed reaction of their formation oM + pL + qH ⇆ M
oL
pH
q (for simplicity, the ion charges in the potentiometric description of the complexes were omitted) (
Table 2). The results of the presented research will be used in more complicated systems that can be used in medical diagnostics, which is important to maintain exactly the same conditions. The correctness of the selected model was confirmed by the coincidence of experimental and theoretical curves, as well as relatively low values of the standard deviation of log
β.
In equimolar systems, the complexation process begins with the formation of the M(HL) species in the system with L-malic acid (
Figure 2c) and the dimeric form M
2L
2 in the reaction with L-tartaric (
Figure 2a). In the Cu(II)/Mal system, the Cu(HMal) complex binds about 60% of the metal ions and is the dominant form at pH around 3.5, while in the system with tartaric acid, this form is not detectable. At the pH of the dominance of Cu(HMal), the dimeric complex Cu
2Mal
2 is formed, while Cu
2Tar
2 is the dominant form in the system at pH 3.9. Compared to the system with tartaric acid, the hydroxyl complex Cu
2Mal
2(OH) is the dominant species in the system. At neutral pH, the dominant complex is Cu
2L
2(OH)
2, binding almost 100% of the metal ions. The presence of complexes in the system disappears at pH 11.0, when copper(II) hydroxide forms.
In the system with L-tartaric acid, only the dimeric forms were observed. The participation of two molecules in the coordination sphere of the central ion was recorded in the monomeric complex of copper(II) ions with L-malic acid Cu(HMal)
2. The formation of protonated complexes was observed only in systems containing an excess of malic acid (
Figure 2d). This complex formed at low pH values binding a maximum of 30% of copper(II) ions. The formation of copper(II) ions complexes with two fully deprotonated malic acid molecules was observed at pH about 3.0. At the pH of his maximum concentration, the dimer Cu
2Mal
2 binds about 40% of the metal ions presented in the system. At pH above 4.0, hydroxy complexes Cu
2Mal
2(OH) and Cu
2Mal
2(OH)
2 were observed. Nearly 100% of copper(II) ions are related in the form of a dihydroxy complex. The increased amount of L-tartaric acid, compared to metal ions, resulted in the formation of a similar set of complexes as in the equimolar system excluding the formation of Cu
2Tar
2 (
Figure 2b). As follows, the distribution that formed hydroxy complexes Cu
2Tar
2(OH) and Cu
2Tar
2(OH)
2 at the pH of their dominance bound about 60% of copper(II) ions. Additionally, along with the increase in pH value, Cu
2Tar
2(OH)
3 was observed as a dominant form, where 100% of the metal ions were involved in the formation of the complex in the pH range of 8.0–11.0.
In systems with cobalt(II) and nickel(II) ions, only monomeric complexes were observed (
Figure 3 and
Figure 4). In systems with cobalt(II) ions with both hydroxy acids (molar ratio is 1:1), complexation began with the formation of the ML complex. Moreover, in the system with malic acid, this was the only one observed forming and binding about 45% of metal ions, as shown in
Figure 3. In the system with tartaric acid at pH = 6.0, 60% of free metal ions were involved in complex CoTar. At the pH of the dominance of the CoTar form, the hydroxy complex CoTar(OH) started to form and was dominant above the pH near to 8.0.
In systems with cobalt(II) ions and an excess of α-hydroxy acid, two molecules of acid were involved in coordination. In the system with malic acid, only one form occurred, CoMal
2 (pH range 4.0–10.0 and binding about 35% of the metal ions). In the tartaric acid system, the first CoTar
2 complex formed and involved about 80% of the free metal ions introduced into the system. At the pH of the dominant CoTar
2 complex, the formation of CoTar
2(OH) was observed. In contrast to the equimolar system, a dihydroxy complex of cobalt ions was detected (
Figure 3b).
L-Tartaric acid with nickel(II) ions in an equimolar system formed at pH 2.5 NiTar complex and a hydroxy complex Ni(Tar)(OH) (
Figure 4a). The hydroxy complex was already dominant in the pH range of about 3.0 to 8.0 and bound nearly 100% of metal ions in the pH range 5.0–7.0. At the dominance of this form, di-hydroxy complex started to form and bound about 80% of nickel(II) ions. In the system with malic acid at low pH values, the protonated form of the complex was observed (
Figure 4c). Increasing of pH value led to full deprotonation of L-Malic acid and NiMal complex was formed. Both forms involved nearly 60% of the metal ions introduced to the system. Moreover, free metal ions were observed almost across the entire studied pH range, such as in all systems with cobalt(II) ions.
In the systems with double excess of ligands, two molecules of acid were observed in the internal coordination sphere. More than 80% of the metal ions were involved in the formation of NiTar
2 complex. At higher pH values, the hydroxy complexes formed were: NiTar
2(OH) and NiTar
2(OH)
2 (
Figure 4b). The computer analysis of potentiometric data collected for the system with a double excess of malic acid showed the formation of three types of complexes. From pH 2.5, the presence of Ni(HMal)
2 and Ni(HMal)Mal was detected and 20% of the metal ions introduced into the system were involved in coordination (
Figure 4d). Fully deprotonated molecules of α-hydroxy acid appeared at pH about 4.0 and NiMal
2 complex started forming (dominant in the pH range 4.5–9.0).
In binary systems of α-hydroxy acid with d-electron metal ions, higher values of overall stability constants (log
β) of analogous complex forms for tartaric acid compared to malic acid were observed. In addition, the differences in the acidic properties of these compounds have an influence on the types of complexes formed in the systems. The degree of their deprotonation is especially noticeable at low pH values, where the formation of protonated forms is observed for systems with malic acid. Additionally, the maximum concentration of analogous complexes was shifted to a higher pH value for malic acid and at pH 11.0 only copper(II) hydroxide was observed where with tartaric acid, hydroxy complexes are dominant. The analysis of the obtained results indicates a higher affinity of metal(II) ions in order: copper(II) ions > nickel(II) ions > cobalt(II) ions for the coordination with oxygen atoms of the carboxyl groups of the studied ligands. Similar to our previous study with citric acid [
42], it was observed that copper(II) ions in the equimolar ratio and with double excess of ligands showing a tendency to form dimeric complexes. In contrast to these ions, cobalt(II) and nickel(II) ions, in systems with excess amounts of studied acids, formed complex compounds in which two ligand molecules were located in the inner coordination sphere of the central atom.
2.3. Spectroscopic Study
2.3.1. UV-Vis Spectroscopy and EPR Spectra
The formation of complex compounds in systems containing d-electron metal ions was confirmed using spectroscopic methods. By selecting the optimal pH values, ensuring the highest possible percentage of the form of a given complex (based on distribution diagrams), UV-Vis measurements were performed for the studied systems. The spectral data obtained for the complexes are presented in
Table 3.
In systems with copper(II) ions, the shift of the absorbance toward lower wavelengths indicates a change in the internal coordination sphere of the metal ions (
Figure 5). By the attachment of an additional oxygen atom of the α-hydroxy acid molecule to copper(II) ions, changes in spectral parameters were observed. Differences in the obtained spectral data for analogous complex forms followed from differences in the percentage of copper ions that are involved in the formation of a complex compound.
The occurrence of both dimeric and monomeric forms in the copper(II) ion systems was confirmed by EPR measurements. At low pH values, monomeric forms are dominant, and characteristic spectra for copper(II) ions were observed (
Figure 6). For the protonated complex compound Cu(HMal), the spectral parameters indicate the involvement of one oxygen atom from partially deprotonated L-malic acid molecule. In the formation of Cu(HMal)
2, the EPR parameters: g
II = 2.37 and A
II: 138.59 × 10
−4 cm
−1, imply the coordination two oxygen atoms. The extinction of the EPR spectrum signal observed for at pH value of dominance dimeric forms confirms the presence of complexes containing two copper(II) ions in the molecule (the characteristic signal for copper(II) ions disappears resulting from spin coupling). EPR silence informs that the dinuclear species are an antiferromagnetic exchange interaction and that two cations have coplanarity of their d
xy orbitals.
In systems containing cobalt(II) ions in complexes with fully deprotonated acids, the d-d parameters suggest similar coordination in CoL and CoL
2 forms. Small shifts of the wavelength at maximum absorption for CoTar
2(OH) to longer wavelengths were observed (
Figure 7). In pH values in which the species CoTar(OH) and CoTar
2(OH)
2 are dominant, it was impossible to record UV-Vis spectra because of precipitate formation.
For the complexes of nickel(II) ions with L-tartaric acid and L-malic acid, small blue-shift is noted (
Figure 8). The molar absorption coefficient for the maximum absorption wavelength increased along with increasing pH values. Based on the spectra obtained for systems with nickel ions, the largest changes were observed in wavelength range 600–800 nm. Overall, these results suggest a change in the internal coordination sphere of metal ion with an increase in pH [
43].
2.3.2. IR Spectroscopy
The involvement of carboxyl groups of L-tartaric and L-malic acids in the coordination of complex compounds with the studied d-electron metal ions was confirmed using IR spectroscopy. The IR spectrum was recorded at the dominant of the pH of complex compounds. Comparative spectra were made for the tested ligands and metal ions under the same pH conditions. In all systems studied, the characteristic asymmetric stretching vibration of the C=O bonds of the carboxyl group (ν
asC=O near 1700 cm
−1) disappeared for the spectra of the complex forms, compared to the spectrum of the free ligands. Interpretation of the spectroscopic information shows the involvement of carboxyl groups in complex formation with metal ions. Furthermore, in the range of symmetric stretching, vibration of the C–O bonds (ν
asC–O 1450–1350 cm
−1) are observed. The shifts observed in this range, compared to the spectra of free ligands and solutions of metal salts, confirm the formation of coordination bonds between metal ions and donor groups of alfa hydroxy acids. The participation of carboxyl groups in the formation of the complex forms in the equimolar systems is presented in
Figure 9.
2.3.3. CD Spectroscopy
The CD and corresponding UV spectra were recorded for all systems with (
R,
R)-tartaric and (
S)-malic acid in water in the range of 185–350 nm at a concentration of 0.001 M. Using a sample cell with 0.5 mm optical pathlength allowed measurements of all samples at relatively high concentration, which allowed a direct comparison with the results of titration measurements. In two cases of samples that were not fully soluble, CD spectra were scaled to UV spectra (values in italics in
Table 3). All series of samples were measured at pH, determined on the basis of the results obtained by computer analysis of potentiometric data.
Tartaric and malic acids do not have aromatic groups incorporated in their structures, which is commonly used in the analysis of conformational preferences of molecules by CD spectroscopy. The analysis of their conformation, and thus the interactions with ions were conducted on the basis of observations of Cotton effects derived from the n-π* electron transitions of carboxylic groups in these molecules. This limited the range of observations between 185 and 250 nm.
In this analysis, the natural conformational preferences of tartaric and malic acids must be considered. It is known that in protonated and deprotonated forms, optically active tartaric acid mainly adopts a trans conformation of its carbon backbone, but as a result of ionization and rearrangement of intramolecular hydrogen bonds, its CD spectra change its shape at different pH. (R,R)-Tartaric acid (L-tartaric acid) in aqueous solution exhibits a single negative Cotton effect Δε = −4 around 210–215 nm and its simple (Na, K, Li) divalent salts show two negative Cotton effects Δε = −4 at 193 nm and −2.5 at around 211 nm. In the case of non-racemic malic acid, the change in its CD spectra also depends on the pH of the solution. (S)-malic acid (L-malic acid), used in experiments in aqueous solutions, exhibits a single positive Cotton effect Δε = 1.3 at 211 nm and its simple (Na, K, Li) divalent salts show positive Cotton effect Δε = 3.8 at 208 nm. All the above-mentioned changes were considered during the analysis process.
In the CD spectrum of a solution of copper and tartaric acid (in a 1:1 ratio) at pH 3.5, a negative Cotton effect Δε = −2.77 at 213 nm can be observed, which indicates that most of the acid remains uncomplexed and adopts the trans conformation. At pH 4.7 (for Cu(II):Tar ratio 1:1) and 4.2 (for Cu(II):Tar ratio 1:2) in both samples, positive Cotton effects in CD spectra can be observed at about 235 nm, indicating the adoption of a gauche conformation by the molecule. The Cotton effect at 207 nm present in CD spectrum for a sample with 1:2 ratio indicates that the second equivalent of the tartaric acid molecules remains in the solution in the trans conformation. Raising the pH to the ones in which the Cu2Tar2OH form dominates in the solutions does not change the pattern of the Cotton effects in CD spectra, which means that the conformation of the carbon skeleton of tartaric acid does not change significantly.
The decrease in Cotton effect in the long-wavelength region of pH in which Cu
2Tar
2(OH)
2 form dominates, indicates a conformational change in which certain tartrates change its structure to a trans conformation. This is most likely due to the high concentration of negative charge in the resulting structures (
Figure 10a,b).
The CD spectra of copper-malic acid complexes for a pH close to 3.5 indicate partial complexation of the central atom by the acid. The positive Cotton effect at about 210 nm suggests that acid remained uncomplexed in the trans conformation. This effect disappears for measurements performed at pH 4.3 (1:2 ratio), 5.0 (1:2 ratio) and 5.25 (1:1 ratio), which is associated with the formation of a complex in which malic acid changes the conformation from trans to gauche. Possibly due to the larger volume of binary complexes of copper, unlike in cobalt and nickel complexes, the CD spectra do not change significantly when the hydroxyl groups are attached to the complex at pH 7.2 (
Figure 10c,d).
Both measurements of circular dichroism spectra of nickel-tartaric acid complexes in the ratio 1:1 and 1:2 show a similar course of complexation depending on the pH. For an equimolar nickel-tartaric acid complex measured at a pH of 3.7, the CD spectrum indicates the presence of a predominantly non-ionized trans acid. Measurements at the pH of about 6 indicate an increase in ionization while maintaining the trans conformation. Positive Cotton effect at ca 218 nm of Δε = 2.68 and Δε = 3.74 for 1:1 and 1:2 complexes at pH 9.25 and 9.0 respectively, are connected with conformation changes of ionized tartaric acid. This effect disappears for the measurements of the complexes at a pH of 11.0. This is due to the second hydroxylic group complexing the central nickel atom, thus changing the conformation of the acid. In this case, the return to the trans conformation can also be caused by the repulsion of the negative charges of the hydroxyl groups and of the tartaric acid anions.
The CD spectra of the nickel-malic acid 1:1 and 1:2 complexes measured at a pH of 3.7 (Δε = 0.62 at 215nm, Δε = 0.65 at 210 nm for 1:1 ratio and Δε = 1.69 at 210 nm and Δε = 0.85 at 194nm for 1:2 ratio) indicate that, at this pH, most of the malic acid remains unbound in solution and adopts a trans conformation. Comparison of the CD spectra of nickel complexes at pH 8.0, with one and two acid equivalents exhibits a decrease of the positive Cotton effect at 210 nm observed in both cases and indicates the formation of complexes with one acid molecule for an equimolar mixture and two acid molecules for a mixture with double excess of malic acid.
Comparison of CD measurement results of the 1:1 and 1:2 cobalt to tartaric acid molar ratio implies that only one equivalent of acid participates in the coordination process while the other does not. The contribution of the second tartaric acid molecule to the CD spectrum is almost identical to the CD spectrum of its simple salts. The decrease in the intensity of the Cotton effects at the shortest wavelengths indicates that, as in the case of nickel complexes, the introduction of hydroxyl groups into the complexes involves a change in the conformation of tartaric acid. As a result of the low solubility of cobalt complexes of tartaric acid at higher pH, it was necessary to scale the measurement results to the UV spectrum.
CD spectra of malic acid complexes with cobalt exhibited the Cotton effects Δε = 0.68 (210 nm) and Δε = 1.25 (195 nm) for equimolar system and Δε = 2.23 (209 nm), Δε = 2.54 (195 nm) for 1:2 ratio and present relatively small decrease of Cotton effects in comparison to uncomplexed malic acid at corresponding pH. This indicates that a significant part of malic acid stays unbound in solution, which is in agreement with potentiometric studies.
The results of the circular dichroism measurements containing the Cotton effects maxima and the corresponding wavelengths are summarized in
Table 4 for tartaric acid and
Table 5 for malic acid.