Selective Esterification of Phosphonic Acids †
Abstract
:1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. General
3.2. General Procedure for Esterification of Phosphonic Acid Using Various Alkoxy Group Donors
3.3. General Procedure for Esterification of Phosphonic Acids Using Triethyl Orthoacetate in Different Solvents
3.4. The Influence of Temperature on the Selectivity of Esterification Reaction of Butylphosphonic acid with Triethyl Orthoacetate
3.5. General Procedure for Selective Monoesterification of Phosphonic Acids 1 Using Triethyl Orthoacetate 2a Leading to Ethyl Hydrogen Phosphonates 3
3.6. General Procedure for Esterification of Phosphonic Acids 1 Using Triethyl Orthoacetate Leading to Diethyl Phosphonic Esters 4
3.6.1. Ethyl Hydrogen Butylphosphonate (3a)
3.6.2. Ethyl Hydrogen Benzylphosphonate (3b)
3.6.3. Ethyl Hydrogen Ethylphosphonate (3c)
3.6.4. Ethyl Hydrogen Vinylphosphonate (3d)
3.6.5. Ethyl Hydrogen Hexylphosphonate (3e)
3.6.6. Ethyl Hydrogen Dodecylphosphonate (3f)
3.6.7. Ethyl Hydrogen Phenylphosphonate (3g)
3.6.8. Ethyl Hydrogen (4-Methoxyphenyl)phosphonate (3h)
3.6.9. Ethyl Hydrogen [(4-Nitrophenyl)methyl]phosphonate (3j)
3.6.10. Ethyl Hydrogen [(4-Bromophenyl)methyl]phosphonate (3k)
3.6.11. Ethyl Hydrogen [(3-Bromophenyl)methyl]phosphonate (3l)
3.6.12. Diethyl Butylphosphonate (4a)
3.6.13. Diethyl Benzylphosphonate (4b)
3.6.14. Diethyl Ethylphosphonate (4c)
3.6.15. Diethyl Hexylphosphonate (4e)
3.6.16. Diethyl Dodecylphosphonate (4f)
3.6.17. Diethyl Phenylphosphonate (4g)
3.6.18. Diethyl (4-Methoxyphenyl)phosphonate (4h)
3.6.19. Diethyl [(4-Hydroxyphenyl)methyl]phosphonate (4i)
3.6.20. Diethyl [(4-Nitrophenyl)methyl]phosphonate (4j)
3.6.21. Diethyl [(4-Bromophenyl)methyl]phosphonate (4k)
3.6.22. Diethyl [(3-Bromophenyl)methyl]phosphonate (4l)
3.6.23. Tetraethyl [1,4-Phenylenebis(methylene)]bis(phosphonate) (4m)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Kafarski, P. Phosphonates: Their natural occurrence and physiological role. In Contemporary Topics about Phosphorus in Biology and Materials; IntechOpen: London, UK, 2019. [Google Scholar]
- Jia, Y.; Lu, Z.; Huang, K.; Herzberg, O.; Dunaway-Mariano, D. Insight into the mechanism of phosphoenolpyruvate mutase catalysis derived from site-directed mutagenesis studies of active site residues. Biochemistry 1999, 38, 14165. [Google Scholar] [CrossRef]
- Maier, L. What are the requirements in the glyphosate molecule in order for it to be herbicidally active? Heteroat. Chem. Int. J. Main Group Elem. 2000, 11, 454. [Google Scholar] [CrossRef]
- Krise, J.P.; Stella, V.J. Prodrugs of phosphates, phosphonates, and phosphinates. Adv. Drug Deliv. Rev. 1996, 19, 287. [Google Scholar] [CrossRef]
- Pradere, U.; Garnier-Amblard, E.C.; Coats, S.J.; Amblard, F.; Schinazi, R.F. Synthesis of nucleoside phosphate and phosphonate prodrugs. Chem. Rev. 2014, 114, 9154. [Google Scholar] [CrossRef] [Green Version]
- Engel, R. Handbook of Organophosphorus Chemistry; CRC Press: Boca Raton, FL, USA, 1992. [Google Scholar]
- Bhattacharya, A.K.; Thyagarajan, G. Michaelis-arbuzov rearrangement. Chem. Rev. 1981, 81, 415. [Google Scholar] [CrossRef]
- Rajeshwaran, G.G.; Nandakumar, M.; Sureshbabu, R.; Mohanakrishnan, A.K. Lewis acid-mediated michaelis–arbuzov reaction at room temperature: A facile preparation of arylmethyl/heteroarylmethyl phosphonates. Org. Lett. 2011, 13, 1270. [Google Scholar] [CrossRef]
- Kem, K.M.; Nguyen, N.V.; Cross, D.J. Phase-transfer-catalyzed Michaelis-Becker reaction. J. Org. Chem. 1981, 46, 5188. [Google Scholar] [CrossRef]
- Cohen, R.J.; Fox, D.L.; Eubank, J.F.; Salvatore, R.N. Mild and efficient Cs2CO3-promoted synthesis of phosphonates. Tetrahedron Lett. 2003, 44, 8617. [Google Scholar] [CrossRef]
- Ilia, G.; Petric, M.; Bálint, E.; Keglevich, G. Synthesis of the mixed alkyl esters of phenylphosphonic acid by two variations of the Atherton–Todd protocol. Heteroat. Chem. 2015, 26, 29. [Google Scholar] [CrossRef]
- Wang, G.; Shen, R.; Xu, Q.; Goto, M.; Zhao, Y.; Han, L.-B. Stereospecific coupling of H-phosphinates and secondary phosphine oxides with amines and alcohols: A general method for the preparation of optically active organophosphorus acid derivatives. J. Org. Chem. 2010, 75, 3890. [Google Scholar] [CrossRef]
- Norlin, R.; Juhlin, L.; Lind, P.; Trogen, L. A-haloenamines as reagents for the conversion of phosphorus oxyacids to their halogenated analogues. Synthesis 2005, 2005, 1765. [Google Scholar] [CrossRef]
- Nowlan, C.; Li, Y.; Hermann, J.C.; Evans, T.; Carpenter, J.; Ghanem, E.; Shoichet, B.K.; Raushel, F.M. Resolution of chiral phosphate, phosphonate, and phosphinate esters by an enantioselective enzyme library. J. Am. Chem. Soc. 2006, 128, 15892. [Google Scholar] [CrossRef]
- Van Helden, H.P.; Benschop, H.P.; Wolthuis, O.L. New simulators in the prophylaxis against soman poisoning: Structural specificity for the depot site (s). J. Pharm. Pharmacol. 1984, 36, 305. [Google Scholar] [CrossRef] [PubMed]
- Roussis, V.; Wiemer, D.F. Synthesis of phosphonates from alpha-hydroxy carbonyl compounds and dialkyl phosphorochloridites. J. Org. Chem. 1989, 54, 627. [Google Scholar] [CrossRef]
- Verbelen, B.; Dehaen, W.; Binnemans, K. Selective substitution of POCl3 with organometallic reagents: Synthesis of phosphinates and phosphonates. Synthesis 2018, 50, 2019. [Google Scholar] [CrossRef] [Green Version]
- Cui, L.; Yin, C.; Chen, T.; Quan, G.; Ippolito, J.A.; Liu, B.; Yan, J.; Ding, C.; Hussain, Q.; Umer, M. Remediation of organic halogen-contaminated wetland soils using biochar. Sci. Total Environ. 2019, 696, 134087. [Google Scholar] [CrossRef]
- Poshkus, A.; Herweh, J. The reaction of neutral esters of trivalent phosphorus acids with inorganic acid chlorides. III. The reaction of trialkyl phosphites with thionyl chloride. J. Am. Chem. Soc. 1962, 84, 555. [Google Scholar] [CrossRef]
- Rabinowitz, R. Synthesis of monoesters of phosphonic acids. J. Am. Chem. Soc. 1960, 82, 4564. [Google Scholar] [CrossRef]
- Sathe, M.; Gupta, A.K.; Kaushik, M. An efficient method for the esterification of phosphonic and phosphoric acids using silica chloride. Tetrahedron Lett. 2006, 47, 3107. [Google Scholar] [CrossRef]
- Purohit, A.K.; Pardasani, D.; Tak, V.; Kumar, A.; Jain, R.; Dubey, D. Mild and efficient esterification of alkylphosphonic acids using polymer-bound triphenylphosphine. Tetrahedron Lett. 2012, 53, 3795. [Google Scholar] [CrossRef]
- Kumar, R.; Gupta, A.; Kaushik, M. Surface-mediated synthesis of O-Alkyl 2-methoxyethyl alkylphosphonates under solvent free conditions: Potential marker of nerve agents. Phosphorus Sulfur Silicon 2010, 185, 2064. [Google Scholar] [CrossRef]
- Henyecz, R.; Kiss, A.; Mórocz, V.; Kiss, N.Z.; Keglevich, G. Synthesis of phosphonates from phenylphosphonic acid and its monoesters. Synth. Commun. 2019, 49, 2642. [Google Scholar] [CrossRef] [Green Version]
- Pardasani, D.; Purohit, A.; Kumar, A.; Tak, V.; Goud, D.R.; Gupta, A.K.; Dubey, D.K. Synthesis of O-Alkyl Alkylphosphonates via hydrazine mediated partial dealkylation of phosphonate diesters. ChemistrySelect 2018, 3, 253. [Google Scholar] [CrossRef] [Green Version]
- Timperley, C.M.; Bird, M.; Holden, I.; Black, R.M. Organophosphorus chemistry. Part 1. The synthesis of alkyl methylphosphonic acids. J. Chem. Soc. Perkin Trans. 1 2001, 1, 26–30. [Google Scholar] [CrossRef]
- Crenshaw, M.D.; Cummings, D.B. Preparation, derivatization with trimethylsilyldiazomethane, and GC/MS analysis of a “pool” of alkyl methylphosphonic acids for use as qualitative standards in support of counterterrorism and the chemical weapons convention. Phosphorus Sulfur Silicon 2004, 179, 1009. [Google Scholar] [CrossRef]
- Crenshaw, M.D. Synthesis of alkyl-and arylphosphonic acid monoesters by direct esterification of dibasic phosphonic acids in the presence of an arsonic acid catalyst. Phosphorus Sulfur Silicon 2004, 179, 1509. [Google Scholar] [CrossRef]
- Gupta, A.; Kumar, R.; Gupta, H.; Tak, V.; Dubey, D. DCC—Celite hybrid immobilized solid support as a new, highly efficient reagent for the synthesis of O-alkyl hydrogen alkylphosphonates under solvent-free conditions. Tetrahedron Lett. 2008, 49, 1656. [Google Scholar] [CrossRef]
- Leypold, M.; Wallace, P.W.; Kljajic, M.; Schittmayer, M.; Pletz, J.; Illaszewicz-Trattner, C.; Guebitz, G.M.; Birner-Gruenberger, R.; Breinbauer, R. A robust and simple protocol for the synthesis of arylfluorophosphonates. Tetrahedron Lett. 2015, 56, 5619. [Google Scholar] [CrossRef]
- Barucki, H.; Black, R.M.; Kinnear, K.I.; Holden, I.; Read, R.W.; Timperley, C.M. Solid-phase synthesis of some alkyl hydrogen methylphosphonates. Phosphorus Sulfur Silicon Relat. Elem. 2003, 178, 2279. [Google Scholar] [CrossRef]
- Brodzka, A.; Koszelewski, D.; Zysk, M.; Ostaszewski, R. The mechanistic promiscuity of the enzymatic esterification of chiral carboxylic acids. Catal. Commun. 2018, 106, 82. [Google Scholar] [CrossRef]
- Zysk, M.; Zadlo, A.; Brodzka, A.; Wisniewska, C.; Ostaszewski, R. The unexpected kinetic effect of enzyme mixture: The case of enzymatic esterification. J. Mol. Cat. B Enzym. 2014, 102, 225–229. [Google Scholar] [CrossRef]
- Koszelewski, D.; Brodzka, A.; Zadlo, A.; Paprocki, D.; Trzepizur, D.; Zysk, M.; Ostaszewski, R. Dynamic kinetic resolution of 3-aryl-4-pentenoic acids. ACS Catal. 2016, 6, 3287–3292. [Google Scholar] [CrossRef]
- Brodzka, A.; Koszelewski, D.; Cwiklak, M.; Ostaszewski, R. Studies on the chemoenzymatic synthesis of 3-phenyl-GABA and 4-phenyl-pyrrolid-2-one: The influence of donor of the alkoxy group on enantioselective esterification. Tetrahedron Asymmetry 2013, 24, 427–433. [Google Scholar] [CrossRef]
- Kafarski, P.; Lejczak, B. A facile conversion of aminoalkanephosphonic acids into their diethyl esters. The use of unblocked aminoalkanephosphonic acids in phosphono peptide synthesis. Synthesis 1988, 4, 307–310. [Google Scholar] [CrossRef]
- Lagadic, E.; Bruyneel, F.; Demeyer, N.; Herent, M.-F.; Garcia, Y.; Marchand-Brynaert, J. Phosphonated benzoxazole derivatives: Synthesis and metal-complexing properties. Synlett 2013, 24, 817–822. [Google Scholar]
- Yoshino, T.; Imori, S.; Togo, H. Efficient esterification of carboxylic acids and phosphonic acids with trialkyl orthoacetate in ionic liquid. Tetrahedron 2006, 62, 1309–1317. [Google Scholar] [CrossRef]
- Lagadic, E.; Garcia, Y.; Marchand-Brynaert, J. Selective protection and deprotection of ortho-functionalized arylphosphonates. Synthesis 2012, 44, 93–98. [Google Scholar]
- Fredriksen, K.A.; Amedjkouh, M. Investigation of reactive intermediates and reaction pathways in the coupling agent mediated phos-phonamidation reaction. Eur. J. Org. Chem. 2016, 2016, 474–482. [Google Scholar] [CrossRef]
- Campbell, D.A. The synthesis of phosphonate esters; an extension of the Mitsunobu reaction. J. Org. Chem. 1992, 57, 6331–6335. [Google Scholar] [CrossRef]
- Gobec, S.; Kikelj, D. Synthesis of ethyl 3-(hydroxyphenoxy) benzyl butylphosphonates as potential antigen 85C inhibitors. Tetrahedron 2007, 63, 10698–10708. [Google Scholar]
- Schrader, T. Strong binding of alkylguanidinium ions by molecular tweezers: An artificial selective arginine receptor molecule with a biomimetic recognition pattern. Chem. Eur. J. 1997, 3, 1537–1541. [Google Scholar] [CrossRef]
- Olagnon-Bourgeot, S.; Chastrette, F.; Wilhelm, D. 31P NMR—Structure correlations for phosphonocarboxylic acids and esters. Magn. Reson. Chem. 1995, 33, 971–976. [Google Scholar] [CrossRef]
- Boutevin, B.; Hervaud, Y.; Jeanmaire, T.; Boulahna, A.; Elasri, M. Monodealkylation des esters phosphoniques synthese de monosels et de monoacides phosphoniques. Phosphorus Sulfur Silicon Relat. Elem. 2001, 174, 1–14. [Google Scholar] [CrossRef]
- Meziane, D.; Hardouin, J.; Elias, A.; Guenin, E.; Lecouvey, M. Microwave Michaelis–Becker synthesis of diethyl phosphonates, tetraethyl diphosphonates, and their total or partial dealkylation. Heteroat. Chem. Int. J. Main Group Elem. 2009, 20, 369–377. [Google Scholar] [CrossRef]
- Ryu, T.; Kim, J.; Park, Y.; Kim, S.; Lee, P.H. Rhodium-catalyzed oxidative cyclization of arylphosphonic acid monoethyl esters with alkenes: Efficient synthesis of benzoxaphosphole 1-oxides. Org. Lett. 2013, 15, 3986–3989. [Google Scholar] [CrossRef]
- Seo, J.; Park, Y.; Jeon, I.; Ryu, T.; Park, S.; Lee, P.H. Synthesis of phosphaisocoumarins through rhodium-catalyzed cyclization using alkynes and arylphosphonic acid monoesters. Org. Lett. 2013, 15, 3358–3361. [Google Scholar] [CrossRef]
- Yu, J.; Li, M.; Yu, Y.; Gao, Y.; Liu, J.; Sun, F. Synthetic strategy and performances of a UV-curable poly acryloyl phosphinate flame retardant by carbene polymerization. Phosphorus Sulfur Silicon Relat. Elem. 2015, 190, 1958–1970. [Google Scholar] [CrossRef]
- Fu, Z.; Sun, S.; Yang, A.; Sun, F.; Xu, J. Transition metal-free access to 3,4-dihydro-1,2-oxaphosphinine-2-oxides from phosphonochloridates and chalcones through tandem Michael addition and nucleophilic substitution. Chem. Commun. 2019, 55, 13124–13127. [Google Scholar] [CrossRef] [PubMed]
- Baumann, A.L.; Schwagerus, S.; Broi, K.; Kemnitz-Hassanin, K.; Stieger, C.E.; Trieloff, N.; Schmieder, P.; Hackenberger, C.P. Chemically induced vinylphosphonothiolate electrophiles for thiol–thiol bioconjugations. J. Am. Chem. Soc. 2020, 142, 9544–9552. [Google Scholar] [CrossRef]
- Besse, V.; Le Pluart, L.; Cook, W.D.; Pham, T.N.; Madec, P.J. Synthesis and polymerization kinetics of acrylamide phosphonic acids and esters as new dentine adhesives. J. Polym. Sci. Part A Polym. Chem. 2013, 51, 149–157. [Google Scholar] [CrossRef]
- Hu, G.; Chen, W.; Fu, T.; Peng, Z.; Qiao, H.; Gao, Y.; Zhao, Y. Nickel-catalyzed C–P cross-coupling of arylboronic acids with P (O) H compounds. Org. Lett. 2013, 15, 5362–5365. [Google Scholar] [CrossRef]
- Luo, H.; Liu, H.; Chen, X.; Wang, K.; Luo, X.; Wang, K. Ar–P bond construction by the Pd-catalyzed oxidative cross-coupling of arylsilanes with H-phosphonates via C–Si bond cleavage. Chem. Commun. 2017, 53, 956–958. [Google Scholar] [CrossRef]
- Ma, X.; Xu, Q.; Li, H.; Su, C.; Yu, L.; Zhang, X.; Cao, H.; Han, L.-B. Alcohol-based Michaelis–Arbuzov reaction: An efficient and environmentally-benign method for C–P (O) bond formation. Green Chem. 2018, 20, 3408–3413. [Google Scholar] [CrossRef]
- Al-Riyami, L.; Pineda, M.A.; Rzepecka, J.; Huggan, J.K.; Khalaf, A.I.; Suckling, C.J.; Scott, F.J.; Rodgers, D.T.; Harnett, M.M.; Harnett, W. Designing anti-inflammatory drugs from parasitic worms: A synthetic small molecule analogue of the Acanthocheilonema viteae product ES-62 prevents development of collagen-induced arthritis. J. Med. Chem. 2013, 56, 9982–10002. [Google Scholar] [CrossRef]
- Huang, H.; Denne, J.; Yang, C.H.; Wang, H.; Kang, J.Y. Direct aryloxylation/alkyloxylation of dialkyl phosphonates for the synthesis of mixed phosphonates. Angew. Chem. 2018, 130, 6734–6738. [Google Scholar] [CrossRef]
- Huang, T.; Chen, T.; Han, L.-B. Oxidative dephosphorylation of benzylic phosphonates with dioxygen generating symmetrical trans-stilbenes. J. Org. Chem. 2018, 83, 2959–2965. [Google Scholar] [CrossRef]

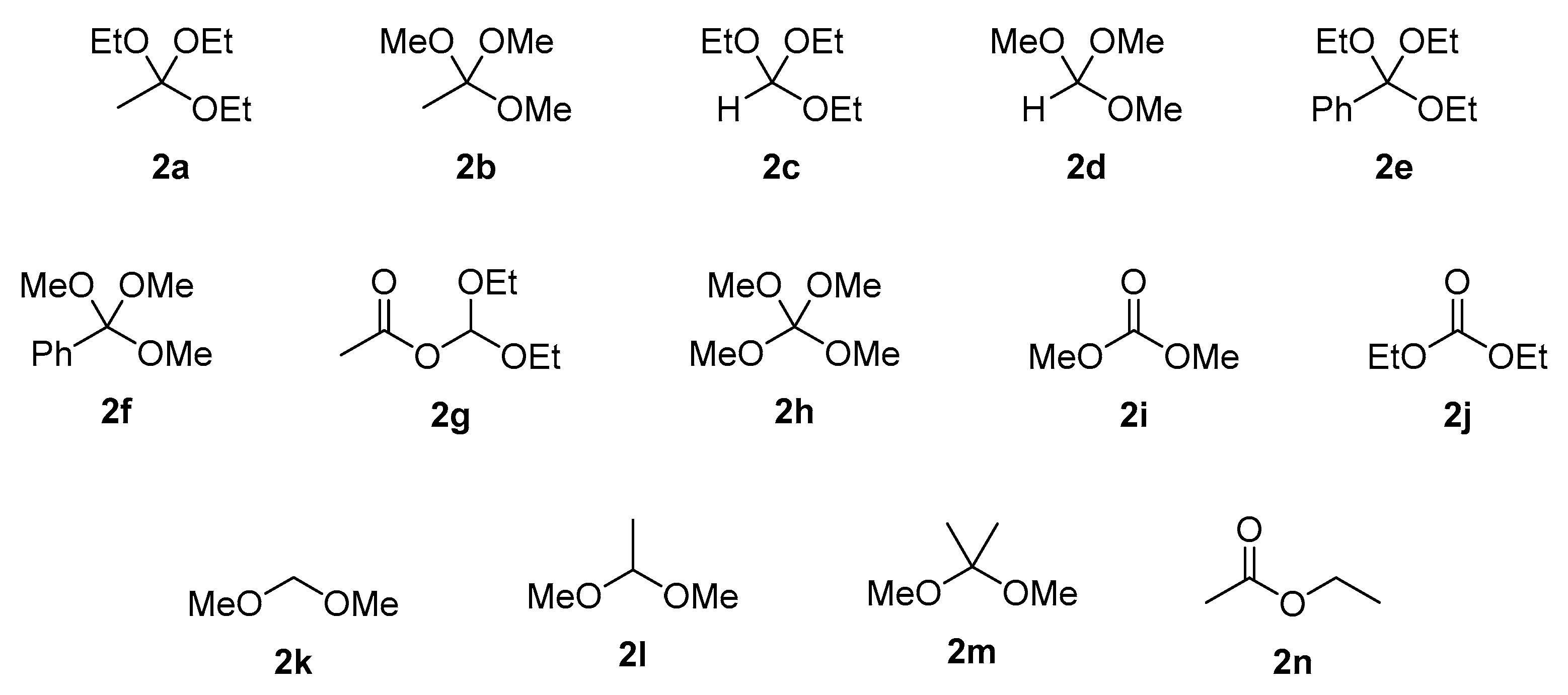




| Entry | Substrate | Donor | Conversion of 1 [%] 2 | Product Yield [%] 2 | |
|---|---|---|---|---|---|
| 3 | 4 | ||||
| 1 | 1a | 2a | 98 | 84 | 3 |
| 2 | 2b | 25 | 25 | 0 | |
| 3 | 2c | 0 | 0 | 0 | |
| 4 | 2d | 0 | 0 | 0 | |
| 5 | 2e | 0 | 0 | 0 | |
| 6 | 2f | 0 | 0 | 0 | |
| 7 | 2g | 39 | 34 | 0 | |
| 8 | 2h | 24 | 23 | 1 | |
| 9 | 2i | 0 | 0 | 0 | |
| 10 | 2j | 0 | 0 | 0 | |
| 11 | 2k | 0 | 0 | 0 | |
| 12 | 2l | 0 | 0 | 0 | |
| 13 | 2m | 0 | 0 | 0 | |
| 14 | 2n | 0 | 0 | 0 | |
| 15 | 1b | 2a | 89 | 83 | 1 |
| 16 | 2b | 50 | 49 | 1 | |
| 17 | 2c | 0 | 0 | 0 | |
| 18 | 2d | 0 | 0 | 0 | |
| 19 | 2e | 0 | 0 | 0 | |
| 20 | 2f | 0 | 0 | 0 | |
| 21 | 2g | 89 | 83 | 6 | |
| 22 | 2h | 52 | 44 | 8 | |
| 23 | 2i | 0 | 0 | 0 | |
| 24 | 2j | 0 | 0 | 0 | |
| 25 | 2k | 0 | 0 | 0 | |
| 26 | 2l | 0 | 0 | 0 | |
| 27 | 2m | 0 | 0 | 0 | |
| 28 | 2n | 0 | 0 | 0 | |
| Entry | Solvent | Substrate 1a Conversion [%] 2 | Product Yield [%] 2 | Substrate 1b Conversion [%] 2 | Product Yield [%] 2 | ||
|---|---|---|---|---|---|---|---|
| 3a | 4a | 3b | 4b | ||||
| 1 | MTBE | 89 | 87 | 2 | 90 | 85 | 5 |
| 2 | THF | 66 | 62 | 1 | 89 | 80 | 1 |
| 3 | DCM | 88 | 78 | 1 | 98 | 84 | 3 |
| 4 | EtOAc | 76 | 75 | 1 | 68 | 66 | 2 |
| 5 | MeCN | 71 | 65 | 2 | 50 | 41 | 2 |
| 6 | Me2CO | 15 | 10 | 0 | 3 | 2 | 1 |
| 7 | PhMe | 76 | 74 | 2 | 89 | 81 | 3 |
| 8 | Neat 3 | 95 | 89 | 5 | 95 | 88 | 7 |
| 9 | DMSO | 0 | 0 | 0 | 0 | 0 | 0 |
| 10 | DMF | 0 | 0 | 0 | 0 | 0 | 0 |
| Entry | Temperature [°C] | Substrate 1a Conversion [%] 2 | Product Yield [%] 2 | |
|---|---|---|---|---|
| 3a | 4a | |||
| 1 | 30 | 97 | 92 | 5 |
| 2 | 40 | >99 | 94 | 6 |
| 3 | 50 | 87 | 13 | |
| 4 | 60 | 73 | 27 | |
| 5 | 70 | 29 | 66 | |
| 6 | 80 | 21 | 79 | |
| 7 | 90 | 1 | 96 | |
| 8 | 100 | 0 | 95 | |
| Entry | R | Product | Conditions | Yield [%] 2 | 31P NMR [ppm] |
|---|---|---|---|---|---|
| 1 | n-Butyl | 3a | 30 °C, 24 h | 98 | 29.1 |
| 2 | 4a | 90 °C, 24 h | 98 | 32.0 | |
| 3 | Benzyl | 3b | 30 °C, 48 h | 76 | 23.3 |
| 4 | 4b | 90 °C, 24 h | 98 | 26.5 | |
| 5 | Ethyl | 3c | 30 °C, 96 h | 89 | 30.2 |
| 6 | 4c | 90 °C, 24 h | 97 | 33.1 | |
| 7 | Vinyl | 3d | 25 °C, 24 h | 83 | 14.0 |
| 8 3 | 4d | 90 °C, 24 h | <1 | 16.9 | |
| 9 | n-Hexyl | 3e | 30 °C, 48 h | 93 | 28.9 |
| 10 | 4e | 90 °C, 24 h | 91 | 31.9 | |
| 11 | n-Dodecyl | 3f | 30 °C, 72 h | 89 | 28.9 |
| 12 | 4f | 90 °C, 24 h | 89 | 31.9 | |
| 13 | Phenyl | 3g | 30 °C, 24 h | 56 | 15.0 |
| 14 | 4g | 90 °C, 24 h | 98 | 17.8 | |
| 15 | 4-methoxyphenyl | 3h | 30 °C, 96 h | 65 | 15.5 |
| 16 | 4h | 90 °C, 24 h | 99 | 18.8 | |
| 17 4 | 4-hydroxybenzyl | 3i | 30 °C, 96 h | <1 | - |
| 18 | 4i | 90 °C, 24 h | 79 | 27.1 | |
| 19 | 4-nitrobenzyl | 3j | 40 °C, 24 h | 29 | 21.4 |
| 20 | 4j | 90 °C, 24 h | 99 | 24.9 | |
| 21 | 4-bromobenzyl | 3k | 30 °C, 24 h | 78 | 22.4 |
| 22 | 4k | 90 °C, 24 h | 75 | 25.8 | |
| 23 | 3-bromobenzyl | 3l | 30 °C, 48 h | 49 | 22.7 |
| 24 | 4l | 90 °C, 24 h | 74 | 25.9 | |
| 25 4 | p-xylylene | 3m | 30 °C, 48 h | <1 | - |
| 26 | 4m | 145 °C, 24 h | 99 | 26.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trzepizur, D.; Brodzka, A.; Koszelewski, D.; Ostaszewski, R. Selective Esterification of Phosphonic Acids. Molecules 2021, 26, 5637. https://doi.org/10.3390/molecules26185637
Trzepizur D, Brodzka A, Koszelewski D, Ostaszewski R. Selective Esterification of Phosphonic Acids. Molecules. 2021; 26(18):5637. https://doi.org/10.3390/molecules26185637
Chicago/Turabian StyleTrzepizur, Damian, Anna Brodzka, Dominik Koszelewski, and Ryszard Ostaszewski. 2021. "Selective Esterification of Phosphonic Acids" Molecules 26, no. 18: 5637. https://doi.org/10.3390/molecules26185637






