Gas Chromatography–Mass Spectrometry Quantification of 1,1-Dimethylhydrazine Transformation Products in Aqueous Solutions: Accelerated Water Sample Preparation
Abstract
:1. Introduction
2. Results and Discussion
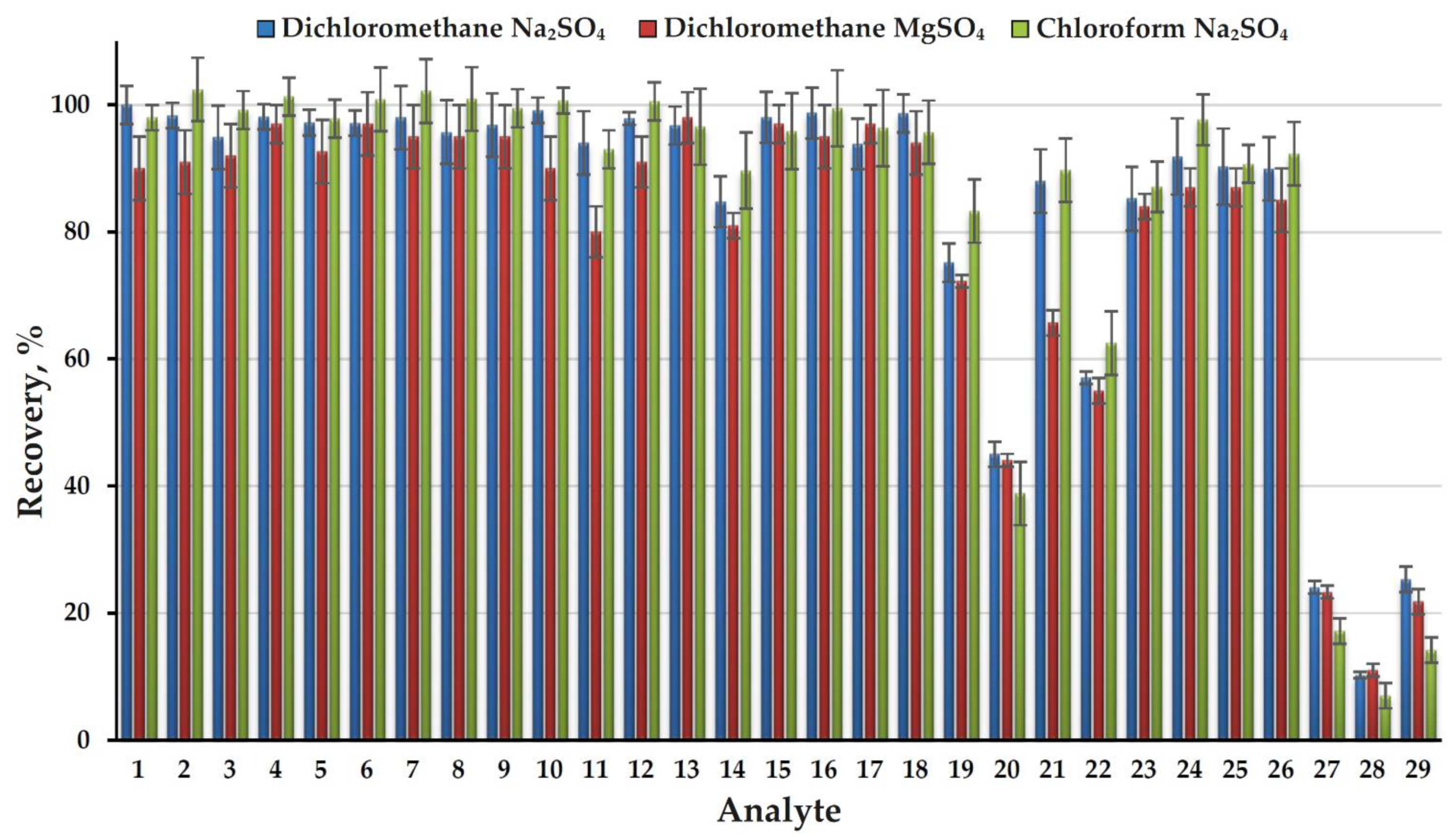
2.1. Extraction Efficiency and Optimization
2.2. Analytical Method and Its Validation
2.3. Analyses of Real Samples
3. Materials and Methods
3.1. Analytes, Reagents and Materials
3.2. Real Objects
- Sample 1. River water (Northern Dvina River) with a salinity of 140 mg L−1 and a dissolved organic carbon content of 13 mg L−1.
- Sample 2. Water extract of peat bog soil, typical for landing places of launch vehicle’s spent stages in the European North of Russia. A soil sample weighing 1 g was placed in a 20 mL glass vial and poured with 10 mL of deionized water, then suspended with continuous vigorous stirring on a vortex (1500 rpm) for 20 min. After settling for two days and centrifugation the aqueous solution was separated and stored at 4 °C for no more than one week.
- Sample 3. An aqueous solution of 1,1-dimethylhydrazine with an initial concentration of 7000 mg L−1, which was in contact with air for 4 years and thus underwent significant oxidative conversion. The solution was yellow in color.
- Sample 4. An aqueous solution of 1,1-dimethylhydrazine with an initial concentration of 1000 mg L−1 subjected to pyrolytic gasification in supercritical water for 2 h at 600 °C [27]. This sample represents a product of detoxification of rocket fuel containing wastewater.
3.3. AWASP Procedure
3.4. GC-MS (MS/MS) Analyses
| No | Analyte | tR, min | SIM | MRM | ||||
|---|---|---|---|---|---|---|---|---|
| Quantifier ion, m/z | Qualifier ion m/z | Quantifier | Qualifier | |||||
| m/z | CE, eV | m/z | CE, eV | |||||
| 1 | TMT | 5.91 | 116 | 72 | 116→72 | 6 | 72→44 | 3 |
| 2 | Pyridine | 7.00 | 79 | 52 | 79→52 | 15 | 52→50 | 9 |
| 3 | Pyrazine | 7.54 | 80 | 53 | 80→53 | 12 | 80→51 | 39 |
| 4 | 2-Methylpyridine | 7.75 | 93 | 66 | 93→66 | 15 | 93→78 | 18 |
| 5 | DMAAN | 8.04 | 84 | 58 | 83→42 | 6 | 84→57 | 6 |
| 6 | 2,6-Dimethylpyridine | 8.18 | 107 | 66 | 107→65 | 21 | 107→92 | 18 |
| 7 | 1-Methyl-1H-pyrazole | 8.21 | 82 | 54 | 82→54 | 18 | 82→42 | 21 |
| 8 | 2-Methylpyrazine | 8.50 | 94 | 67 | 94→67 | 12 | 67→40 | 6 |
| 9 | 3-Methylpyridine | 9.00 | 93 | 66 | 93→66 | 15 | 93→39 | 36 |
| 10 | 4-Methylpyridine | 9.38 | 93 | 66 | 93→66 | 15 | 93→39 | 36 |
| 11 | NDMA | 9.51 | 74 | 42 | 74→44 | 6 | 74→42 | 21 |
| 12 | 2,5-Dimethylpyrazine | 9.48 | 108 | 42 | 108→42 | 18 | 108→81 | 9 |
| 13 | 2,6-Dimethylpyrazine | 9.77 | 108 | 42 | 108→42 | 18 | 108→40 | 18 |
| 14 | DMF | 9.82 | 73 | 44 | 73→44 | 6 | 73→58 | 6 |
| 15 | 2,3-Dimethylpyrazine | 9.77 | 108 | 67 | 108→67 | 12 | 67→40 | 6 |
| 16 | 2,4,6-Trimethylpyridine | 10.10 | 121 | 79 | 121→79 | 15 | 79→77 | 12 |
| 17 | 3,5-Dimethylpyridine | 10.78 | 107 | 79 | 107→92 | 15 | 106→77 | 15 |
| 18 | 2,3,5-Trimethylpyridine | 11.70 | 121 | 106 | 121→77 | 33 | 120→77 | 18 |
| 19 | 1-Methyl-1H-1,2,4-triazole | 13.18 | 83 | 56 | 83→56 | 6 | 83→55 | 18 |
| 20 | FADMH | 14.06 | 59 | 43 | 59→44 | 9 | 59→43 | 15 |
| 21 | 1-Methyl-1H-imidazole | 14.44 | 82 | 54 | 82→55 | 12 | 82→42 | 18 |
| 22 | 1H-pyrazole | 15.96 | 68 | 41 | 68→41 | 15 | 67→40 | 12 |
| 23 | 3-Methyl-1H-pyrazole | 16.35 | 81 | 82 | 82→54 | 18 | 81→54 | 6 |
| 24 | 3,5-Dimethyl-1H-pyrazole | 16.80 | 95 | 96 | 95→41 | 15 | 95→68 | 6 |
| 25 | 2,4-Dimethyl-1H-imidazole | 17.49 | 96 | 95 | 95→41 | 18 | 95→68 | 6 |
| 26 | 4-Methyl-1H-pyrazole | 16.93 | 82 | 81 | 82→55 | 12 | 81→54 | 6 |
| 27 | 3,4-Dimethyl-1H-pyrazole | 19.87 | 95 | 96 | 96→54 | 21 | 95→68 | 6 |
| 28 | 1H-imidazole | 20.30 | 68 | 55 | 68→41 | 15 | 67→40 | 6 |
| 29 | 4-Methyl-1H-imidazole | 20.42 | 82 | 81 | 82→54 | 18 | 81→54 | 6 |
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Koroleva, T.V.; Krechetov, P.P.; Semenkov, I.N.; Sharapova, A.V.; Lednev, S.A.; Karpachevskiy, A.M.; Kondratyev, A.D.; Kasimov, N.S. The environmental impact of space transport. Transp. Res. D 2018, 58, 54–69. [Google Scholar] [CrossRef]
- Koroleva, T.V.; Semenkov, I.N.; Sharapova, A.V.; Krechetov, P.P.; Lednev, S.A. Ecological consequences of space rocket accidents in Kazakhstan between 1999 and 2018. Environ. Pollut. 2021, 268A, 115711. [Google Scholar] [CrossRef] [PubMed]
- Ul’yanovskii, N.V.; Kosyakov, D.S.; Pikovskoi, I.I.; Khabarov, Y.G. Characterisation of oxidation products of 1,1-dimethylhydrazine by high-resolution orbitrap mass spectrometry. Chemosphere 2017, 174, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Kosyakov, D.S.; Ul’yanovskii, N.V.; Pikovskoi, I.I.; Kenessov, B.; Bakaikina, N.V.; Zhubatov, Z.; Lebedev, A.T. Effects of oxidant and catalyst on the transformation products of rocket fuel 1,1-dimethylhydrazine in water and soil. Chemosphere 2019, 228, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Kenessov, B.; Alimzhanova, M.; Sailaukhanuly, Y.; Baimatova, N.; Abilev, M.; Batyrbekova, S.; Carlsen, L.; Tulegenov, A.; Nauryzbayev, M. Transformation products of 1,1-dimethylhydrazine and their distribution in soils of fall places of rocket carriers in Central Kazakhstan. Sci. Total Environ. 2012, 427–428, 78–85. [Google Scholar] [CrossRef]
- Rodin, I.A.; Anan’eva, I.A.; Smolenkov, A.D.; Shpigun, O.A. Determination of the products of the oxidative transformation of unsymmetrical dimethylhydrazine in soils by liquid chromatography/mass spectrometry. J. Analyt. Chem. 2010, 65, 1405–1410. [Google Scholar] [CrossRef]
- Smolenkov, A.D.; Shpigun, O.A. Direct liquid chromatographic determination of hydrazines: A review. Talanta 2012, 102, 93–100. [Google Scholar] [CrossRef]
- Kosyakov, D.S.; Ul’yanovskii, N.V.; Bogolitsyn, K.G.; Shpigun, O.A. Simultaneous determination of 1,1-dimethylhydrazine and products of its oxidative transformations by liquid chromatography–tandem mass spectrometry. Int. J. Environ. Anal. Chem. 2014, 94, 1254–1263. [Google Scholar] [CrossRef]
- Kosyakov, D.S.; Pikovskoi, I.I.; Ul’yanovskii, N.V.; Kozhevnikov, A.Y. Direct determination of hydrazine, methylhydrazine, and 1,1-dimethylhydrazine by zwitterionic hydrophilic interaction liquid chromatography with amperometric detection. Int. J. Environ. Anal. Chem. 2017, 97, 313–329. [Google Scholar] [CrossRef]
- Ul’yanovskii, N.V.; Kosyakov, D.S.; Popov, M.S.; Pikovskoi, I.I.; Khoroshev, O.Y. Using a stationary phase based on porous graphitized carbon for the determination of 1,1-dimethylhydrazine transformation products by liquid chromatography–mass spectrometry. J. Analyt. Chem. 2020, 75, 510–518. [Google Scholar] [CrossRef]
- Timchenko, Y.V.; Stavrianidi, A.N.; Smolenkov, A.D.; Pirogov, A.V.; Shpigun, O.A. A novel simple and sensitive approach for determination of 1,1-dimethylhydrazine in aqueous samples by high performance liquid chromatography with ultraviolet and tandem mass spectrometric detection after derivatization with unsubstituted aromatic aldehydes. Chemosphere 2021, 280, 130747. [Google Scholar] [CrossRef]
- An, Z.; Li, P.; Zhang, X.; Liu, L. Simultaneous determination of hydrazine, methylhydrazine, and 1,1-dimethylhydrazine in rat plasma by LC–MS/MS. J. Liq. Chromatogr. Rel. Technol. 2014, 37, 1212–1225. [Google Scholar] [CrossRef]
- Kosyakov, D.S.; Amosov, A.S.; Ul’yanovskii., N.V.; Ladesov, A.V.; Khabarov, Y.G.; Shpigun, O.A. Spectrophotometric determination of hydrazine, methylhydrazine, and 1,1-dimethylhydrazine with preliminary derivatization by 5-nitro-2-furaldehyde. J. Analyt. Chem. 2017, 72, 171–177. [Google Scholar] [CrossRef]
- Ul’yanovskii, N.V.; Kosyakov, D.S.; Pokryshkin, S.A.; Bogolitsyn, K.G. Determination of transformation products of 1,1-dimethylhydrazine by gas chromatography–tandem mass spectrometry. J. Analyt. Chem. 2015, 70, 171–177. [Google Scholar] [CrossRef]
- Kosyakov, D.S.; Ul’yanovskii, N.V.; Pokryshkin, S.A.; Lakhmanov, D.E.; Shpigun, O.A. Rapid determination of 1,1-dimethylhydrazine transformation products in soil by accelerated solvent extraction coupled with gas chromatography–tandem mass spectrometry. Int. J. Environ. Anal. Chem. 2015, 95, 1321–1337. [Google Scholar] [CrossRef]
- Ul’yanovskii, N.V.; Lakhmanov, D.E.; Pikovskoi, I.I.; Falev, D.I.; Popov, M.S.; Kozhevnikov, A.Y.; Kosyakov, D.S. Migration and transformation of 1,1-dimethylhydrazine in peat bog soil of rocket stage fall site in Russian North. Sci. Total Environ. 2020, 726, 138483. [Google Scholar] [CrossRef]
- Kenessov, B.N.; Koziel, J.A.; Grotenhuis, T.; Carlsen, L. Screening of transformation products in soils contaminated with unsymmetrical dimethylhydrazine using headspace SPME and GC-MS. Anal. Chim. Acta. 2010, 674, 32–39. [Google Scholar] [CrossRef] [Green Version]
- Buryak, A.K.; Serdyuk, T.M. Chromatography-mass spectrometry in aerospace industry. Russ. Chem. Rev. 2013, 82, 369–392. [Google Scholar] [CrossRef]
- Ul’yanovskii, N.V.; Kosyakov, D.S.; Popov, M.S.; Shavrina, I.S.; Ivakhnov, A.D.; Kenessov, B.; Lebedev, A.T. Rapid quantification and screening of nitrogen-containing rocket fuel transformation products by vortex assisted liquid-liquid microextraction and gas chromatography—high-resolution Orbitrap mass spectrometry. Microchem. J. 2021, 106821. [Google Scholar] [CrossRef]
- Orazbayeva, D.; Kenessov, B.; Psillakis, E.; Nassyrova, D.; Bektassov, M. Determination of transformation products of unsymmetrical dimethylhydrazine in water using vacuum-assisted headspace solid-phase microextraction. J. Chromatogr. A 2018, 1555, 30–36. [Google Scholar] [CrossRef]
- Bakaikina, N.V.; Kenessov, B.; Ul’yanovskii, N.V.; Kosyakov, D.S.; Pokryshkin, S.A.; Derbissalin, M.; Zhubatov, Z.K. Quantification of transformation products of unsymmetrical dimethylhydrazine in water using SPME and GC-MS. Chromatographia 2017, 80, 931–940. [Google Scholar] [CrossRef]
- Zhubatov, Z.K.; Kenessov, B.; Bakaikina, N.V.; Bimaganbetova, A.O.; Akynbayev, N.; Bakhytkyzy, I. Fast Determination of 1-methyl-1H-1,2,4-triazole in soils contaminated by rocket fuel using solvent extraction, isotope dilution and GC–MS. Chromatographia 2016, 79, 491–499. [Google Scholar] [CrossRef]
- Polyakova, O.V.; Mazur, D.M.; Artaev, V.B.; Lebedev, A.T. Determination of polycyclic aromatic hydrocarbons in water by gas chromatography/mass spectrometry with accelerated sample preparation. J. Analyt. Chem. 2013, 68, 1099–1103. [Google Scholar] [CrossRef]
- Polyakova, O.V.; Mazur, D.M.; Artaev, V.B.; Lebedev, A.T. Rapid liquid–liquid extraction for the reliable GC/MS analysis of volatile priority pollutants. Environ. Chem. Lett. 2016, 14, 251–257. [Google Scholar] [CrossRef]
- Polyakova, O.V.; Mazur, D.M.; Lebedev, A.T. Improved sample preparation and GC-MS analysis of priority organic pollutants. Environ. Chem. Lett. 2014, 12, 419–427. [Google Scholar] [CrossRef]
- Xie, X.; Backman, D.; Lebedev, A.T.; Artaev, V.B.; Jiang, L.; Ilag, L.L.; Zubarev, R.A. Primordial soup was edible: Abiotically produced Miller-Urey mixture supports bacterial growth. Sci. Rep. 2015, 5, 14338. [Google Scholar] [CrossRef] [Green Version]
- Kosyakov, D.S.; Ul’yanovskii, N.V.; Ivakhnov, A.D.; Pikovskoi, I.I. Transformation of Unsymmetrical Dimethylhydrazine in Supercritical Water. Russ. J. Phys. Chem. B 2019, 13, 1103–1110. [Google Scholar] [CrossRef]
- Yi, L.; Wang, L.; Guo, L.; Cheng, K.; Jin, H.; Chen, Y.; Cao, W. Hydrogen production by supercritical water gasification of methylhydrazine in continuous system. J. Water Process Eng. 2021, 42, 102037. [Google Scholar] [CrossRef]
- Yi, L.; Guo, L.J.; Jin, H.; Kou, J.J.; Zhang, D.M.; Wang, R.Y. Gasification of unsymmetrical dimethylhydrazine in supercritical water: Reaction pathway and kinetics. Int. J. Hydrogen Energy 2018, 43, 8644–8654. [Google Scholar] [CrossRef]
- Milyushkin, A.L.; Birin, K.P.; Matyushin, D.D.; Semeikin, A.V.; Iartsev, S.D.; Karnaeva, A.E.; Uleanov, A.V.; Buryak, A.K. Isomeric derivatives of triazoles as new toxic decomposition products of 1,1-dimethylhydrazine. Chemosphere 2019, 217, 95–99. [Google Scholar] [CrossRef]
- Beltrami, R.T.; Bissell, E.R. Some methylhydrazonium salts; an improved synthesis of tetramethylhydrazine. J. Am. Chem. Soc. 1956, 78, 2467–2468. [Google Scholar] [CrossRef]

| No | Analyte | SIM | MRM | Linear Range | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| a | r2 | LOD | LOQ | a | r2 | LOD | LOQ | |||
| 1 | TMT | 0.477 | 0.999 | 3.1 | 10 | 0.215 | 0.999 | 4.7 | 16 | LOQ–50 |
| 2 | Pyridine | 0.854 | 0.999 | 2.1 | 7.0 | 0.840 | 0.999 | 0.7 | 2.4 | LOQ–58 |
| 3 | Pyrazine | 0.721 | 0.999 | 2.0 | 6.5 | 0.461 | 0.999 | 2.7 | 9.1 | LOQ–58 |
| 4 | 2-methyl-Pyridine | 0.963 | 0.999 | 2.1 | 7.1 | 0.373 | 0.999 | 3.2 | 11 | LOQ–71 |
| 5 | DMAAN | 0.359 | 0.999 | 26 | 88 | 0.227 | 0.999 | 3.7 | 12 | LOQ–54 |
| 6 | 2,6-Dimethylpyridine | 1.16 | 0.999 | 1.0 | 3.2 | 0.273 | 0.999 | 2.4 | 8.1 | LOQ–56 |
| 7 | 1-Methyl-1H-Pyrazole | 0.865 | 0.999 | 2.3 | 7.7 | 0.198 | 0.999 | 2.5 | 8.2 | LOQ–58 |
| 8 | 2-Methylpyrazine | 0.898 | 0.999 | 2.1 | 6.9 | 0.503 | 0.999 | 1.9 | 6.4 | LOQ–59 |
| 9 | 3-methylpyridine | 0.905 | 0.999 | 3.2 | 11 | 0.344 | 0.999 | 4.7 | 15 | LOQ–58 |
| 10 | 4-methylpyridine | 0.980 | 0.999 | 3.1 | 10 | 0.427 | 0.999 | 4.0 | 13 | LOQ–60 |
| 11 | NDMA | 0.507 | 0.999 | 4.6 | 15 | 0.307 | 0.999 | 4.8 | 16 | LOQ–50 |
| 12 | 2,5-Dimethylpyrazine | 0.910 | 0.999 | 2.0 | 6.8 | 0.274 | 0.999 | 2.0 | 6.5 | LOQ–57 |
| 13 | 2,6-Dimethylpyrazine | 1.21 | 0.999 | 1.5 | 5.1 | 0.285 | 0.999 | 1.9 | 6.4 | LOQ–53 |
| 14 | DMF | 0.496 | 0.999 | 5.5 | 18 | 0.242 | 0.999 | 3.0 | 9.9 | LOQ–57 |
| 15 | 2,3-Dimethylpyrazine | 0.904 | 0.999 | 1.9 | 6.2 | 0.568 | 0.999 | 1.2 | 4.0 | LOQ–60 |
| 16 | 2,4,6-Trimethylpyridine | 1.38 | 0.999 | 1.0 | 3.2 | 0.352 | 0.999 | 1.3 | 4.4 | LOQ–55 |
| 17 | 3,5-Dimethylpyridine | 1.08 | 0.999 | 1.2 | 4.0 | 0.243 | 0.999 | 2.9 | 9.8 | LOQ–57 |
| 18 | 2,3,5-Trimethylpyridine | 1.13 | 0.999 | 1.2 | 3.8 | 0.160 | 0.999 | 2.0 | 6.7 | LOQ–54 |
| 19 | 1-Methyl-1H-1,2,4-triazole | 0.432 | 0.999 | 5.7 | 19 | 0.492 | 0.998 | 2.1 | 7.2 | LOQ–63 |
| 20 | FADMH | 0.136 | 0.995 | 16 | 54 | 0.095 | 0.994 | 17 | 56 | LOQ–59 |
| 21 | 1-Methyl-1H-imidazole | 0.765 | 0.998 | 3.3 | 11 | 0.285 | 0.998 | 2.1 | 7.1 | LOQ–63 |
| 22 | 1H-pyrazole | 0.500 | 0.998 | 3.3 | 11 | 0.257 | 0.999 | 6.3 | 20 | LOQ–57 |
| 23 | 3-Methyl-1H-pyrazole | 0.411 | 0.999 | 5.0 | 17 | 0.127 | 0.999 | 2.8 | 9.4 | LOQ–59 |
| 24 | 3,5-Dimethyl-1H-pyrazole | 0.650 | 0.999 | 3.9 | 13 | 0.143 | 0.999 | 4.6 | 15 | LOQ–58 |
| 25 | 2,4-Dimethyl-1H-imidazole | 0.640 | 0.999 | 4.0 | 13 | 0.136 | 0.999 | 4.1 | 13 | LOQ–58 |
| 26 | 4-Methyl-1H-pyrazole | 0.490 | 0.999 | 4.1 | 14 | 0.146 | 0.999 | 4.4 | 15 | LOQ–54 |
| 27 | 3,4-Dimethyl-1H-pyrazole | 0.158 | 0.991 | 21 | 68 | 0.120 | 0.990 | 10 | 33 | LOQ–56 |
| 28 | 1H-imidazole | 0.225 | 0.991 | 21 | 71 | 0.302 | 0.992 | 37 | 120 | LOQ–58 |
| 29 | 4-methyl-1H-Imidazole | 0.300 | 0.973 | 11 | 36 | 0.153 | 0.980 | 26 | 87 | LOQ–56 |
| No | Analyte | RSD, % | |
|---|---|---|---|
| Intra-Day | Inter-Day | ||
| 1 | TMT | 5 | 6 |
| 2 | Pyridine | 5 | 8 |
| 3 | Pyrazine | 6 | 8 |
| 4 | 2-Methylpyridine | 7 | 11 |
| 5 | DMAAN | 8 | 10 |
| 6 | 2,6-Dimethylpyridine | 8 | 9 |
| 7 | 1-Methyl-1H-pyrazole | 6 | 7 |
| 8 | 2-Methylpyrazine | 8 | 10 |
| 9 | 3-Methylpyridine | 5 | 11 |
| 10 | 4-Methylpyridine | 5 | 10 |
| 11 | NDMA | 4 | 8 |
| 12 | 2,5-Dimethylpyrazine | 4 | 8 |
| 13 | 2,6-Dimethylpyrazine | 4 | 8 |
| 14 | DMF | 4 | 6 |
| 15 | 2,3-Dimethylpyrazine | 5 | 11 |
| 16 | 2,4,6-Trimethylpyridine | 6 | 13 |
| 17 | 3,5-Dimethylpyridine | 5 | 10 |
| 18 | 2,3,5-Trimethylpyridine | 7 | 9 |
| 19 | 1-Methyl-1H-1,2,4-triazole | 8 | 11 |
| 20 | FADMH | 10 | 15 |
| 21 | 1-Methyl-1H-imidazole | 8 | 12 |
| 22 | 1H-pyrazole | 5 | 9 |
| 23 | 3-Methyl-1H-pyrazole | 6 | 8 |
| 24 | 3,5-Dimethyl-H-pyrazole | 4 | 9 |
| 25 | 2,4-Dimethyl-1H-imidazole | 8 | 10 |
| 26 | 4-Methyl-1H-pyrazole | 6 | 10 |
| 27 | 3,4-Dimethyl-1H-pyrazole | 5 | 10 |
| 28 | 1H-imidazole | 8 | 13 |
| 29 | 4-Methyl-1H-imidazole | 10 | 15 |
| No | Analyte | Concentration, µg L−1 | |
|---|---|---|---|
| Sample 3 | Sample 4 | ||
| 1 | TMT | n.d.* | 19 ± 1 |
| 2 | Pyridine | 3.2 ± 0.8 | 403 ± 27 |
| 3 | Pyrazine | 5.4 ± 0.1 | 31 ± 2 |
| 4 | 2-Methylpyridine | n.d. | 125 ± 14 |
| 5 | DMAAN | n.d. | n.d. |
| 6 | 2,6-Dimethylpyridine | n.d. | 7.4 ± 1.9 |
| 7 | 1-Methyl-1H-pyrazole | 95 ± 9 | 2180 ± 120 |
| 8 | 2-Methylpyrazine | 5.9 ± 0.2 | 122 ± 5 |
| 9 | 3-Methylpyridine | n.d. | 274 ± 21 |
| 10 | 4-Methylpyridine | n.d. | 11 ± 1 |
| 11 | NDMA | 31,200 ± 1800 | 3590 ± 140 |
| 12 | 2,5-Dimethylpyrazine | n.d. | 4.5 ± 0.2 |
| 13 | 2,6-Dimethylpyrazine | n.d. | 15 ± 1 |
| 14 | DMF | 220 ± 50 | 5920 ± 200 |
| 15 | 2,3-Dimethylpyrazine | n.d. | 8.0 ± 1.0 |
| 16 | 2,4,6-Trimethylpyridine | n.d. | 3.4 ± 1.1 |
| 17 | 3,5-Dimethylpyridine | n.d. | 9.7 ± 1.4 |
| 18 | 2,3,5-Trimethylpyridine | n.d. | 5.2 ± 1.9 |
| 19 | 1-Methyl-1H-1,2,4-triazole | 48,700 ± 7400 | 10,500 ± 220 |
| 20 | FADMH | n.d. | 25,100 ± 2100 |
| 21 | 1-Methyl-1H-imidazole | n.d. | 430 ± 35 |
| 22 | 1H-pyrazole | 25 ± 7 | 30 ± 3 |
| 23 | 3-Methyl-1H-pyrazole | n.d. | 8.8 ± 3.7 |
| 24 | 3,5-Dimethyl-1H-pyrazole | n.d. | n.d. |
| 25 | 2,4-Dimethyl-1H-imidazole | n.d. | n.d. |
| 26 | 4-Methyl-1H-pyrazole | n.d. | n.d. |
| 27 | 3,4-Dimethyl-1H-pyrazole | n.d. | n.d. |
| 28 | 1H-imidazole | n.d. | n.d. |
| 29 | 4-Methyl-1H-imidazole | n.d. | n.d. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Popov, M.S.; Ul’yanovskii, N.V.; Kosyakov, D.S. Gas Chromatography–Mass Spectrometry Quantification of 1,1-Dimethylhydrazine Transformation Products in Aqueous Solutions: Accelerated Water Sample Preparation. Molecules 2021, 26, 5743. https://doi.org/10.3390/molecules26195743
Popov MS, Ul’yanovskii NV, Kosyakov DS. Gas Chromatography–Mass Spectrometry Quantification of 1,1-Dimethylhydrazine Transformation Products in Aqueous Solutions: Accelerated Water Sample Preparation. Molecules. 2021; 26(19):5743. https://doi.org/10.3390/molecules26195743
Chicago/Turabian StylePopov, Mark S., Nikolay V. Ul’yanovskii, and Dmitry S. Kosyakov. 2021. "Gas Chromatography–Mass Spectrometry Quantification of 1,1-Dimethylhydrazine Transformation Products in Aqueous Solutions: Accelerated Water Sample Preparation" Molecules 26, no. 19: 5743. https://doi.org/10.3390/molecules26195743






