Psychrotolerant Mesorhizobium sp. Isolated from Temperate and Cold Desert Regions Solubilizes Potassium and Produces Multiple Plant Growth Promoting Metabolites
Abstract
:1. Introduction
2. Materials and Methods
2.1. Location Description and Collection of Soil Sample
2.2. Isolation of K Solubilizing Rhizobacteria
2.3. Screening of K Solubilizing Rhizobacteria
2.3.1. Qualitative Screening for K Solubilization Potential
2.3.2. Quantitative Screening for K Solubilization Potential
2.4. Production of Plant Growth-Promoting Characteristics
2.4.1. Hydrogen Cyanide
2.4.2. Ammonia Production
2.4.3. Indole Acetic Acid (IAA) Production
2.4.4. Protease Activity
2.4.5. Chitinase Production
2.4.6. Cellulase Activity
2.5. Morphological and Biochemical Characteristics
2.6. Molecular Characterization and Phylogenetic Analysis of KSB
2.7. Statistical Analysis
3. Results and Discussion
3.1. Isolation of K Solubilizing Rhizobacteria
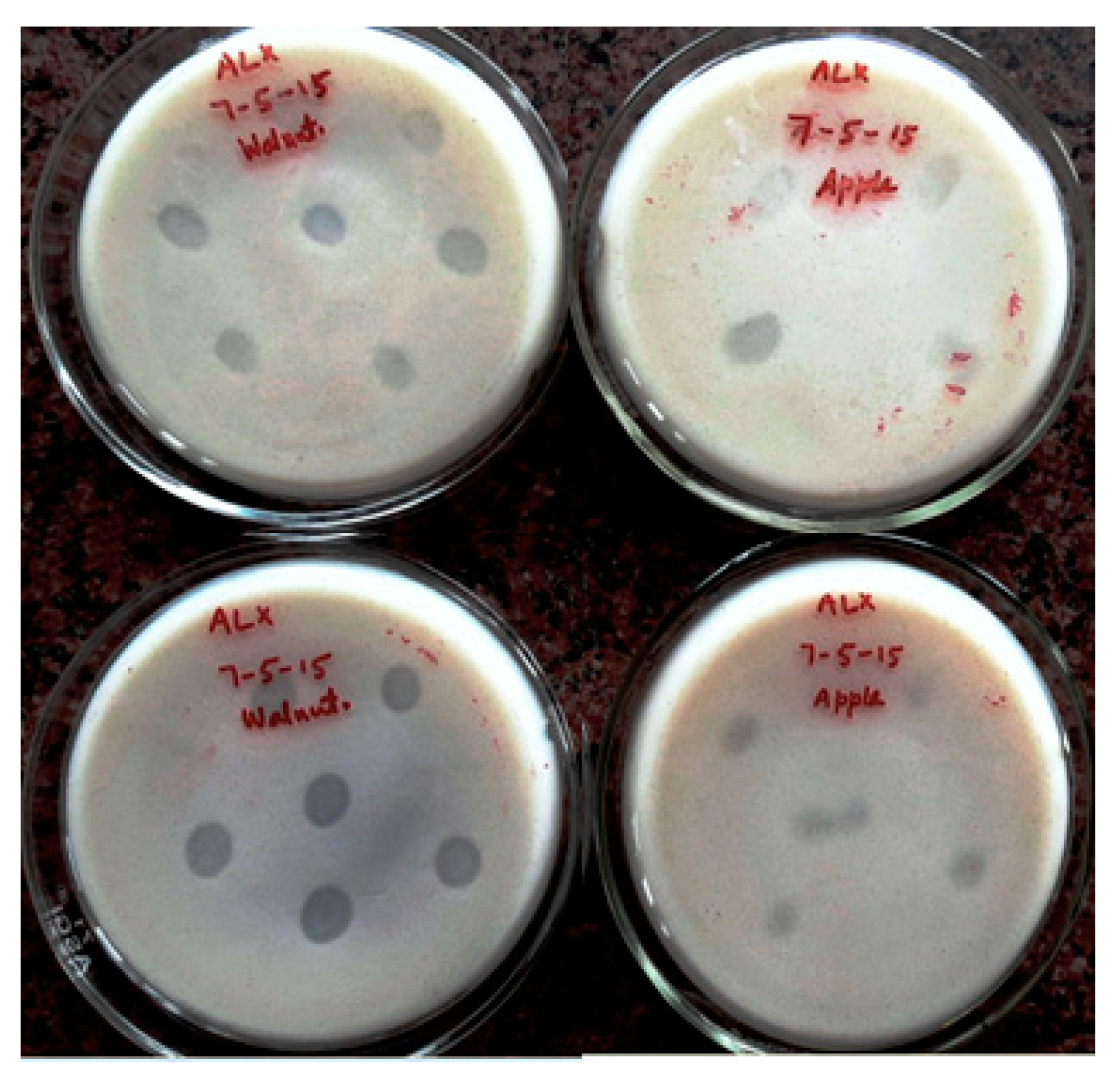
3.2. Qualitative Screening for K Solubilization Potential
3.3. Quantitative Screening for K Solubilization Potential
3.4. Production of Plant Growth-Promoting Substances
3.5. Morphological and Biochemical Characteristics of the Isolate
3.6. Molecular Characterization by 16S rDNA Gene Sequencing Approach
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Gallegos-Cedillo, V.M.; Urrestarazu, M.; Álvaro, J.E. Influence of salinity on transport of nitrates and potassium by means of the xylem sap content between roots and shoots in young tomato plants. J. Soil Sci. Plant Nutr. 2016, 16, 991–998. [Google Scholar] [CrossRef] [Green Version]
- Raji, M.; Thangavelu, M. Isolation and screening of potassium solubilizing bacteria from saxicolous habitat and their impact on tomato growth in different soil types. Arch. Microbiol. 2021, 203, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Pandey, D.; Kehri, H.K.; Zoomi, I.; Singh, U.; Chaudhri, K.L.; Akhtar, O. Potassium solubilizing microbes: Diversity, ecological significances and biotechnological applications. In Sustainable Development and Biodiversity; Springer Science and Business Media: Berlin/Heidelberg, Germany, 2020; pp. 263–286. [Google Scholar]
- Shirale, A.O.; Meena, B.P.; Gurav, P.P.; Srivastava, S.; Biswas, A.K.; Thakur, J.K.; Somasundaram, J.; Patra, A.K.; Rao, A.S. Prospects and challenges in utilization of indigenous rocks and minerals as source of potassium in farming. J. Plant Nutr. 2019, 42, 2682–2701. [Google Scholar] [CrossRef]
- Etesami, H.; Emami, S.; Alikhani, H.A. Potassium solubilizing bacteria (KSB): Mechanisms, promotion of plant growth, and future prospects a review. J. Soil Sci. Plant Nutr. 2017, 17, 897–911. [Google Scholar] [CrossRef]
- Khati, P.; Mishra, P.K.; Parihar, M.; Kumari, A.; Joshi, S.; Bisht, J.K.; Pattanayak, A. Potassium solubilization and mobilization: Functional impact on plant growth for sustainable agriculture. In Microbial Rejuvenation of Polluted Environment; Springer Science and Business Media: Berlin/Heidelberg, Germany, 2020; pp. 21–39. [Google Scholar]
- Binner, I.; Dultz, S.; Schellhorn, M.; Schenk, M. Potassium adsorption and release properties of clays in peat-based horticultural substrates for increasing the cultivation safety of plants. Appl. Clay Sci. 2017, 145, 28–36. [Google Scholar] [CrossRef]
- Wen, Y.; You, J.; Zhu, J.; Hu, H.; Gao, J.; Huang, J. Long-term green manure application improves soil K availability in red paddy soil of subtropical China. J. Soils Sediments 2021, 21, 63–72. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Bachani, P.; Jain, D.; Patidar, S.K.; Mishra, S. Extraction of potassium from K-feldspar through potassium solubilization in the halophilic Acinetobacter soli (MTCC 5918) isolated from the experimental salt farm. Int. J. Miner. Process. 2016, 152, 53–57. [Google Scholar] [CrossRef]
- Torabian, S.; Farhangi-Abriz, S.; Qin, R.; Noulas, C.; Sathuvalli, V.; Charlton, B.; Loka, D. Potassium: A vital macronutrient in potato production—A review. Agronomy 2021, 11, 543. [Google Scholar] [CrossRef]
- Madar, R.; Singh, Y.; Meena, M.; Das, T.; Paramesh, V.; Al-Mana, F.; Mattar, M.A.; Elansary, H.O. Residue and potassium management strategies to improve crop productivity, potassium mobilization, and assimilation under zero-till maize–wheat cropping system. Agriculture 2020, 10, 401. [Google Scholar] [CrossRef]
- Rajawat, M.V.S.; Singh, R.; Singh, D.; Yadav, A.N.; Singh, S.; Kumar, M.; Saxena, A.K. Spatial distribution and identification of bacteria in stressed environments capable to weather potassium aluminosilicate mineral. Braz. J. Microbiol. 2020, 51, 751–764. [Google Scholar] [CrossRef]
- Liu, J.N.; Cao, S.Y.; Chang, L.J.; Huang, J.D.; Zhao, X. Summarization of research status of potash feldspar in China. J. Bohai Univ. Nat. Sci. Ed. 2019, 40, 315–320. [Google Scholar]
- Chen, Y.; Ye, J.; Kong, Q. Potassium-solubilizing activity of Bacillus aryabhattai SK1-7 and its growth-promoting effect on Populus alba L. Forests 2020, 11, 1348. [Google Scholar] [CrossRef]
- Rao, C.S.; Srinivas, K. Potassium dynamics and role of non-exchangeable potassium in crop nutrition. Indian J. Fertil. 2017, 13, 80–94. [Google Scholar]
- Tran, A.M. Potassium Fixation by Oxidized and Reduced Forms of Different Phyllo-Silicates. Ph.D. Thesis, Kansas State University, Manhattan, KS, USA, 2012. [Google Scholar]
- Golestanifard, A.; Santner, J.; Aryan, A.; Kaul, H.-P.; Wenzel, W.W. Potassium fixation in northern Iranian paddy soils. Geoderma 2020, 375, 114475. [Google Scholar] [CrossRef]
- Basu, A.; Prasad, P.; Das, S.N.; Kalam, S.; Sayyed, R.Z.; Reddy, M.S.; El Enshasy, H. Plant growth promoting rhizobacteria (PGPR) as green bioinculants: Recent developments, constraints, and prospects. Sustainability 2021, 13, 1140. [Google Scholar] [CrossRef]
- Hamid, B.; Zaman, M.; Farooq, S.; Fatima, S.; Sayyed, R.; Baba, Z.; Sheikh, T.; Reddy, M.; El Enshasy, H.; Gafur, A.; et al. Bacterial plant biostimulants: A sustainable way towards improving growth, productivity, and health of crops. Sustainability 2021, 13, 2856. [Google Scholar] [CrossRef]
- Sattar, A.; Naveed, M.; Ali, M.; Zahir, Z.A.; Nadeem, S.M.; Yaseen, M.; Meena, V.S.; Farooq, M.; Singh, R.; Rahman, M.; et al. Perspectives of potassium solubilizing microbes in sustainable food production system: A review. Appl. Soil Ecol. 2019, 133, 146–159. [Google Scholar] [CrossRef]
- Sun, F.; Ou, Q.; Wang, N.; Guo, Z.X.; Ou, Y.; Li, N.; Peng, C. Isolation and identification of potassium-solubilizing bacteria from Mikania micrantha rhizospheric soil and their effect on M. micrantha plants. Glob. Ecol. Conserv. 2020, 23, e01141. [Google Scholar] [CrossRef]
- Nosheen, S.; Ajmal, I.; Song, Y. Microbes as biofertilizers, a potential approach for sustainable crop production. Sustainability 2021, 13, 1868. [Google Scholar] [CrossRef]
- Rajawat, M.V.S.; Ansari, W.A.; Singh, D.; Singh, R. Potassium solubilizing bacteria (KSB). In Microbial Interventions in Agriculture and Environment; Springer Science and Business Media: Berlin/Heidelberg, Germany, 2019; pp. 189–209. [Google Scholar]
- Bakhshandeh, E.; Pirdashti, H.; Lendeh, K.S. Phosphate and potassium-solubilizing bacteria effect on the growth of rice. Ecol. Eng. 2017, 103, 164–169. [Google Scholar] [CrossRef]
- Khanghahi, M.Y.; Pirdashti, H.; Rahimian, H.; Nematzadeh, G.; Sepanlou, M.G. Potassium solubilizing bacteria (KSB) isolated from rice paddy soil: From isolation, identification to K use efficiency. Symbiosis 2018, 76, 13–23. [Google Scholar] [CrossRef]
- Xiao, Y.; Wang, X.; Chen, W.; Huang, Q. Isolation and identification of three potassium-solubilizing bacteria from rape rhizospheric soil and their effects on ryegrass. Geomicrobiol. J. 2017, 34, 873–880. [Google Scholar] [CrossRef]
- Boubekri, K.; Soumare, A.; Mardad, I.; Lyamlouli, K.; Hafidi, M.; Ouhdouch, Y.; Kouisni, L. The screening of potassium- and phosphate-solubilizing actinobacteria and the assessment of their ability to promote wheat growth parameters. Microorganisms 2021, 9, 470. [Google Scholar] [CrossRef]
- Khan, A.; Upadhayay, V.K.; Panwar, M.; Singh, A.V. Soil microbiota: A key bioagent for revitalization of soil health in hilly regions. In Microbiological Advancements for Higher Altitude Agro-Ecosystems & Sustainability; Springer: Berlin/Heidelberg, Germany, 2020; pp. 183–200. [Google Scholar] [CrossRef]
- Zahid, M.; Abbasi, M.K.; Hameed, S.; Rahim, N. Isolation and identification of indigenous plant growth promoting rhizobacteria from Himalayan region of Kashmir and their effect on improving growth and nutrient contents of maize (Zea mays L.). Front. Microbiol. 2015, 6, 207. [Google Scholar] [CrossRef] [PubMed]
- Joshi, P.; Joshi, G.K.; Mishra, P.K.; Bisht, J.K.; Bhatt, J.C. Diversity of cold-tolerant phosphate solubilizing microorganisms from the northwestern Himalayas. In Bacterial Diversity in Sustainable Agriculture, Sustainable Development and Biodiversity; Maheshwari, D.K., Ed.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 227–264. [Google Scholar]
- Available online: http://www.weatherbase.com (accessed on 30 December 2017).
- Meena, V.S.; Maurya, B.R.; Verma, J.P.; Aeron, A.; Kumar, A.; Kim, K.; Bajpai, V.K. Potassium solubilizing rhizobacteria (KSR): Isolation, identification, and K-release dynamics from waste mica. Ecol. Eng. 2015, 81, 340–347. [Google Scholar] [CrossRef]
- Sadiq, H.M.; Jahangir, G.Z.; Nasir, I.A.; Iqtidar, M.; Iqbal, M. Isolation and characterization of phosphate-solubilizing bacteria from rhizosphere soil. Biotechnol. Biotechnol. Equip. 2013, 27, 4248–4255. [Google Scholar] [CrossRef]
- Bagyalakshmi, B.; Ponmurugan, P.; Balamurugan, A. Potassium solubilization, plant growth promoting substances by potassium solubilizing bacteria (KSB) from southern Indian Tea plantation soil. Biocatal. Agric. Biotechnol. 2017, 12, 116–124. [Google Scholar] [CrossRef]
- Passari, A.K.; Mishra, V.K.; Gupta, V.K.; Yadav, M.K.; Saikia, R.; Singh, B.P. In Vitro and In Vivo plant growth promoting activities and DNA fifingerprinting of antagonistic endophytic actinomycetes associates with medicinal plants. PLoS ONE 2015, 10, 0139468. [Google Scholar] [CrossRef] [Green Version]
- Ashfaq, M.; Hassan, H.M.; Ghazali, A.H.A.; Ahmad, M. Halotolerant potassium solubilizing plant growth promoting rhizobacteria may improve potassium availability under saline conditions. Environ. Monit. Assess. 2020, 192, 697. [Google Scholar] [CrossRef]
- Joshi, G.; Kumar, V.; Brahmachari, S.K. Screening and identification of novel halotolerant bacterial strains and assessment for insoluble phosphate solubilization and IAA production. Bull. Natl. Res. Cent. 2021, 45, 1–12. [Google Scholar] [CrossRef]
- Zhou, C.; Qin, H.; Chen, X.; Zhang, Y.; Xue, Y.; Ma, Y. A novel alkaline protease from alkaliphilic Idiomarina sp. C9-1 with potential application for eco-friendly enzymatic dehairing in the leather industry. Sci. Rep. 2018, 8, 16467. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, K.; Sadaiappan, B.; Aruni, W.; Kumarappan, A.; Thirunavukarasu, R.; Srinivasan, G.P.; Subramanian, M. Bioconversion of chitin and concomitant production of chitinase and N-acetylglucosamine by novel Achromobacter xylosoxidans isolated from shrimp waste disposal area. Sci. Rep. 2020, 10, 11898. [Google Scholar] [CrossRef]
- Pramanik, S.K.; Mahmud, S.; Paul, G.K.; Jabin, T.; Naher, K.; Uddin, S.; Zaman, S.; Saleh, A. Fermentation optimization of cellulase production from sugarcane bagasse by Bacillus pseudomycoides and molecular modeling study of cellulase. Curr. Res. Microb. Sci. 2021, 2, 100013. [Google Scholar] [CrossRef]
- Sarikhani, M.R.; Oustan, S.; Ebrahimi, M.; Aliasgharzad, N. Isolation and identification of potassium-releasing bacteria in soil and assessment of their ability to release potassium for plants. Eur. J. Soil Sci. 2018, 69, 1078–1086. [Google Scholar] [CrossRef]
- Yadav, A.N.; Sachan, S.G.; Verma, P.; Saxena, A.K. Bioprospecting of plant growth promoting psychrotrophic Bacilli from the cold desert of north western Indian Himalayas. Indian J. Exp. Biol. 2016, 54, 142–150. [Google Scholar]
- Ahmad, M.S.; Zargar, M.Y. Characterization of potassium solubilizing bacteria (KSB) in rhizospheric soils of apple (Malus domestica borkh.) in temperate Kashmir. J. Appl. Life Sci. Int. 2017, 15, 1–7. [Google Scholar] [CrossRef]
- Yadav, N.; Yadav, A.N. Biodiversity and biotechnological applications of novel plant growth promoting methylotrophs. J. Appl. Biotechnol. Bioeng. 2018, 5, 342–344. [Google Scholar] [CrossRef]
- Verma, A.; Patidar, Y.; Vaishampayan, A. Isolation and purification of potassium solubilizing bacteria from different regions of India and its effect on crop’s yield. Indian J. Microbiol. Res. 2016, 3, 483–488. [Google Scholar]
- Pandey, A.; Yarzábal, L.A. Bioprospecting cold-adapted plant growth promoting microorganisms from mountain environments. Appl. Microbiol. Biotechnol. 2019, 103, 643–657. [Google Scholar] [CrossRef] [PubMed]
- D’Amico, S.; Collins, T.; Marx, J.-C.; Feller, G.; Gerday, C. Psychrophillic microorganisms: Challenges for life. EMBO Rep. 2006, 7, 385–389. [Google Scholar] [CrossRef]
- Verma, P.; Yadav, A.N.; Shukla, L.; Saxena, A.K.; Suman, A. Alleviation of cold stress in wheat seedlings by Bacillus amyloliquefaciens IARIHHS2-30, an endophytic psychrotolerant K-solubilizing bacterium from NW Indian Himalayas. Natl. J. Life Sci. 2015, 12, 105–110. [Google Scholar]
- Yadav, A.N.; Kour, D.; Sharma, S.; Sachan, S.G.; Singh, B.; Chauhan, V.S.; Sayyed, R.Z.; Kaushik, R.; Saxena, A.K. Psychrotrophic microbes: Biodiversity, mechanisms of adaptation, and biotechnological implications in alleviation of cold stress in plants. In Plant Growth Promoting Rhizobacteria for Sustainable Stress Management; Springer Science and Business Media: Berlin/Heidelberg, Germany, 2019; pp. 219–253. [Google Scholar]
- Rajawat, M.V.S.; Singh, R.; Singh, D.; Saxena, A.K. Psychrotrophs of the genus Janthinobacterium with potential to weather potassium aluminosilicate mineral. 3 Biotech 2019, 9, 142. [Google Scholar] [CrossRef]
- Nirmala, J.L.I.; Surendranatha, R. Essential nutrients solubilization ability of fluorescent pseudomonads and their multinutrient management. Int. J. Sci. Res. 2015, 4, 43–49. [Google Scholar]
- Bahadur, I.; Maurya, B.R.; Meena, V.S.; Saha, M.; Kumar, A.; Aeron, A. Mineral release dynamics of tricalcium phosphate and waste muscovite by mineral-solubilizing rhizobacteria isolated from indo-gangetic plain of India. Geomicrobiol. J. 2016, 34, 454–466. [Google Scholar] [CrossRef]
- Berde, C.V.; Gawde, S.S.; Berde, V.B. Potassium solubilization: Mechanism and functional impact on plant growth. In Sustainable Development and Biodiversity; Springer: Berlin/Heidelberg, Germany, 2021; pp. 133–148. [Google Scholar]
- Yadav, A.N.; Verma, P.; Sachan, S.G.; Kaushik, R.; Saxena, A.K. Psychrotrophic microbiomes: Molecular diversity and beneficial role in plant growth promotion and soil health. In Microorganisms for Green Revolution; Panpatte, D., Jhala, Y., Shelat, H., Vyas, R., Eds.; Springer: Berlin/Heidelberg, Germany, 2018; pp. 197–240. [Google Scholar]
- Mazumdar, D.; Saha, S.P.; Ghosh, S. Isolation, screening and application of a potent PGPR for enhancing growth of chickpea as affected by nitrogen level. Int. J. Veg. Sci. 2020, 26, 333–350. [Google Scholar] [CrossRef]
- Richard, P.O.; Adekanmbi, A.O.; Ogunjobi, A.A. Screening of bacteria isolated from the rhizosphere of maize plant (Zea mays L.) for ammonia production and nitrogen fixation. Afr. J. Microbiol. Res. 2018, 12, 829–834. [Google Scholar]
- Vinay, J.U.; Naik, M.K.; Rangeshwaran, R.; Chennappa, G.; Shaikh, S.S.; Sayyed, R.Z. Detection of antimicrobial traits in fluorescent pseudomonads and molecular characterization of an antibiotic pyoluteorin. 3 Biotech 2016, 6, 227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kusale, S.P.; Attar, Y.C.; Sayyed, R.Z.; Malek, R.A.; Ilyas, N.; Suriani, N.L.; El Enshasy, H.A. Production of plant beneficial and antioxidants metabolites by Klebsiella variicola under salinity stress. Molecules 2021, 26, 1894. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, S.S.; Sayyed, R.Z. Role of plant growth-promoting rhizobacteria and their formulation in biocontrol of plant diseases. In Plant Microbes Symbiosis: Applied Facets; Springer: New Delhi, India, 2015; pp. 337–351. [Google Scholar]
- Jabborova, D.; Wirth, S.; Kannepalli, A.; Narimanov, A.; Desouky, S.; Davranov, K.; Bahkali, A.H. Co-inoculation of rhizobacteria and biochar application improves growth and nutrientsin soybean and enriches soil nutrients and enzymes. Agronomy 2020, 10, 1142. [Google Scholar] [CrossRef]
- Jadhav, H.P.; Sayyed, R.Z.; Shaikh, S.S.; Bhamre, H.M.; Sunita, K.; El Enshasy, H.A. Statistically designed bioprocess for enhanced production of alkaline protease in Bacillus cereus HP_RZ17. J. Sci. Ind. Res. 2020, 79, 491–498. [Google Scholar]
- Kusale, S.P.; Attar, Y.C.; Sayyed, R.Z.; El Enshasy, H.; Hanapi, S.Z.; Ilyas, N.; Marraiki, N. Inoculation of Klebsiella variicola alleviated salt stress and improved growth and nutrients in wheat and maize. Agronomy 2021, 11, 927. [Google Scholar] [CrossRef]
- Islam, S.; Akanda, A.M.; Prova, A.; Islam, T.; Hossain, M. Isolation and identification of plant growth promoting rhizobacteria from cucumber rhizosphere and their effect on plant growth promotion and disease suppression. Front. Microbiol. 2016, 6, 1360. [Google Scholar] [CrossRef] [Green Version]
- Baba, Z.A.; Aziz, M.A.; Sheikh, T.A.; Sheikh, F.A.; Bhat, Z.A.; Khan, S.; Saher, T.; Hamid, B. Studies on soil health and plant growth promoting potential Rhizobium isolates. Emir. J. Food Agric. 2015, 27, 423–429. [Google Scholar] [CrossRef] [Green Version]
- Li, S.; Zhang, B.; Zhu, H.; Zhu, T. Cloning and expression of the chitinase encoded by ChiKJ406136 from Streptomyces sampsonii (Millard & Burr) waksman KJ40 and its antifungal effect. Forests 2018, 9, 699. [Google Scholar] [CrossRef] [Green Version]
- Bhagwat, A.; Collins, C.H.; Dordick, J.S. Selective antimicrobial activity of cellulolytic enzymes in a bacterial consortium. Appl. Microbiol. Biotechnol. 2019, 103, 7041–7054. [Google Scholar] [CrossRef]
- Baba, Z.A.; Sheikh, T.; Hamid, B. Functional diversity of culturable phosphate solubilizing fluorescent pseudomonads. Indian J. Agric. Res. 2014, 48, 472. [Google Scholar] [CrossRef]
- Yadav, A.N.; Sachan, S.G.; Verma, P.; Saxena, A.K. Prospecting cold deserts of north western Himalayas for microbial diversity and plant growth promoting attributes. J. Biosci. Bioeng. 2015, 119, 683–693. [Google Scholar] [CrossRef] [PubMed]

| Isolates | K Solubilization Hallow (cm) | Colony Diameter (cm) | Solubilization Efficiency (%) |
|---|---|---|---|
| Bacillus mucilaginosus * | 3.26 | 1.92 | 169.79 h |
| KSB(Smg) | 3.37 | 2.82 | 119.50 l |
| KSB (Drs) | 4.11 | 1.89 | 217.46 e |
| KSBM | 3.10 | 1.98 | 156.56 i |
| KSB(Gmr) | 3.47 | 2.61 | 132.95 k |
| KSB(Mng) | 4.72 | 1.56 | 302.56 c |
| KSB(Pd) | 3.25 | 2.97 | 109.42 m |
| KSB(Sls) | 3.17 | 3.12 | 101.60 n |
| KSB(Trs) | 3.97 | 2.10 | 189.04 f |
| KSB(Kgl) | 4.38 | 1.77 | 247.45 d |
| KSB(Grz) | 5.27 | 1.14 | 462.28 a |
| KSB(Phg) | 3.84 | 2.18 | 176.14 g |
| KSB(Glm) | 4.97 | 1.26 | 394.44 b |
| KSB(Afr) | 3.61 | 2.54 | 142.12 j |
| Isolates | Incubation Temperature | ||||||
|---|---|---|---|---|---|---|---|
| 0 °C | 1 °C | 3 °C | 5 °C | 7 °C | 15 °C | 20 °C | |
| Bacillus mucilaginosus | 0.00 i | 0.00 n | 0.00 h | 0.00 l | 0.89 n | 2.29 i | 4.31 l |
| KSB(Smg) | 1.15 c | 1.53 f | 1.97 bcd | 2.35 f | 3.18 f | 5.11 bcd | 7.41 b |
| KSB(Drs) | 0.89 g | 1.618 d | 2.01 abc | 2.50 e | 3.85 b | 4.93 cde | 5.55 i |
| KSBM | 1.14 cd | 1.48 g | 1.97 bcd | 2.06 h | 2.80 i | 3.94 g | 4.90 k |
| KSB(Gmr) | 1.33 b | 1.67 c | 2.18 ab | 3.10 b | 3.55 c | 4.70 de | 6.51 e |
| KSB(Mng) | 1.09 d | 1.15 k | 2.00 e | 2.70 d | 3.21 e | 5.45 b | 6.55 d |
| KSB(Pd) | 1.10 cd | 1.45 h | 1.75 de | 2.11 g | 3.09 g | 5.23 bc | 7.12 c |
| KSB(Sls) | 0.73 h | 0.98 l | 1.26 f | 1.73 j | 2.07 l | 3.87 g | 4.25 m |
| KSB(Trs) | 0.33 j | 0.59 m | 0.91 g | 1.37 k | 1.49 m | 2.13 i | 4.11 n |
| KSB(Kgl) | 1.30 b | 1.57 e | 1.83 cde | 2.05 h | 2.39 k | 4.19 fg | 5.67 h |
| KSB(Grz) | 1.55 a | 1.90 a | 2.29 a | 3.45 a | 4.20 a | 7.11 a | 10.19 a (136.4%) |
| KSB(Phg) | 1.00 e | 1.26 j | 1.58 e | 2.03 i | 2.91 h | 4.55 ef | 6.15 f |
| KSB(Glm) | 1.15 c | 1.69 b | 2.13 ab | 2.75 c | 3.28 d | 5.24 cde | 6.13 g |
| KSB(Afr) | 0.95 f | 1.36 i | 1.72 de | 2.033 i | 2.46 j | 3.89 h | 5.31 j |
| Isolates | Incubation Temperature | ||||||
|---|---|---|---|---|---|---|---|
| 0 °C | 1 °C | 3 °C | 5 °C | 7 °C | 15 °C | 20 °C | |
| Bacillus mucilaginosus | 0.00 n | 0.00 n | 0.00 n | 0.03 n | 1.64 n | 4.9 n | 8.13 j |
| KSB(Smg) | 1.79 b | 3.31 b | 4.27 a | 3.09 a | 5.03 d | 8.43 b | 11.57 b |
| KSB(Drs) | 1.50 h | 2.25 g | 2.80 g | 3.30 g | 5.09 c | 7.89 f | 9.29 h |
| KSBM | 1.22 l | 2.73 d | 3.51 d | 3.95 d | 4.35 f | 5.48 m | 7.91 n |
| KSB(Gmr) | 1.75 d | 3.21 c | 4.03 c | 4.75 c | 5.11 b | 8.13 c | 11.17 c |
| KSB(Mng) | 1.55 f | 2.67 e | 2.91 f | 3.45 f | 4.33 g | 7.85 g | 10.06 f |
| KSB(Pd) | 1.39 j | 1.87 k | 2.39 i | 3.11 i | 3.89 j | 6.31 i | 8.76 i |
| KSB(Sls) | 1.30 k | 1.67 l | 2.00 l | 2.39 l | 3.19 l | 5.88 j | 8.00 l |
| KSB(Trs) | 1.15 m | 1.43 m | 1.89 m | 2.29 m | 3.17 m | 5.76 l | 7.98 m |
| KSB(Kgl) | 1.77 c | 2.53 f | 3.19 e | 3.90 e | 4.49 e | 8.11 d | 11.00 d |
| KSB(Grz) | 2.15 a | 3.67 a | 4.10 b | 4.79 b | 5.29 a | 9.20 a | 13.33 a (63.9%) |
| KSB(Phg) | 1.50 g | 1.89 i | 2.33 j | 3.01 j | 3.99 i | 7.67 h | 10.01 g |
| KSB(Glm) | 1.61 e | 2.037 h | 2.58 h | 3.27 h | 4.11 h | 8.01 e | 10.66 e |
| KSB(Afr) | 1.43 i | 1.87 j | 2.29 k | 2.60 k | 3.21 k | 5.77 k | 8.11 k |
| Isolates | Incubation Temperature | ||||||
|---|---|---|---|---|---|---|---|
| 0 °C | 1 °C | 3 °C | 5 °C | 7 °C | 15 °C | 20 °C | |
| Bacillus mucilaginosus | 0.00 l | 0.00 n | 0.00 m | 0.04 n | 2.97 n | 10.76 m | 15.33 f |
| KSB(Smg) | 2.11 b | 5.21 b | 6.89 b | 8.41 b | 10.58 c | 19.73 b | 19.72 b |
| KSB(Drs) | 1.90 f | 4.91 e | 6.10 e | 7.01 e | 9.13 e | 17.30 e | 17.31 e |
| KSBM | 1.80 h | 4.10 f | 5.92 f | 6.97 f | 8.23 f | 12.10 i | 14.90 g |
| KSB(Gmr) | 2.05 c | 5.04 c | 6.67 c | 7.25 c | 10.27 d | 18.34 c | 18.34 c |
| KSB(Mng) | 1.95 d | 4.97 d | 6.43 d | 7.11 d | 10.89 b | 17.83 d | 17.84 d |
| KSB(Pd) | 1.84 h | 3.70 g | 5.05 i | 6.08 g | 6.78 g | 13.09 g | 13.10 i |
| KSB(Sls) | 1.67 j | 2.78 l | 4.09 k | 4.90 l | 5.55 l | 10.97 k | 10.97 m |
| KSB(Trs) | 1.50 k | 2.47 m | 3.85 l | 4.44 m | 5.13 m | 9.50 l | 9.80 n |
| KSB(Kgl) | 1.93 e | 3.57 i | 5.19 g | 5.90 h | 6.43 h | 12.07 j | 12.10 k |
| KSB(Grz) | 3.17 a | 6.35 a | 8.88 a | 10.00 a | 12.32 a | 23.35 a | 23.38 a (52.5%) |
| KSB(Phg) | 1.79 i | 3.39 k | 5.00 j | 5.817 j | 6.19 j | 12.00 j | 12.00 l |
| KSB(Glm) | 1.87 g | 3.59 h | 5.11 h | 5.87 i | 6.40 i | 13.23 f | 13.24 h |
| KSB(Afr) | 1.80 h | 3.49 j | 5.01 i | 5.70 k | 6.013 k | 12.87 h | 12.89 j |
| Isolates | HCN | Ammonia | IAA (µM/mL) | Protease (µM/mL) | Chitinase (µM/mL) | Cellulase (µM/mL) |
|---|---|---|---|---|---|---|
| Bacillus mucilaginosus * | ++ | ++ | 27.00 g | 525.12 a | 5.10 a | 250.55 bc |
| KSB(Smg) | + | + | 27.22 e | 325.74 e | 3.70 cd | 130 e |
| KSB(Drs) | + | + | 31.00 b | 417.24 b | 2.90 e | 121 e |
| KSBM | 21.37 k | 324.19 f | 2.70 e | 206 cd | ||
| KSB(Gmr) | + | + | 17.09 m | 313.09 g | 5.20 a | 200 e |
| KSB(Mng) | - | + | 23.11 j | 310.23 h | 3.80 cd | 101 e |
| KSB(Pd) | + | + | 27.18 f | 297.12 j | 4.70 b | 150 e |
| KSB(Sls) | + | - | 29.32 d | 281.18 k | 3.90 c | 185 d |
| KSB(Trs) | - | + | 31.44 a | 330.72 d | 4.00 c | 230 bcd |
| KSB(Kgl) | + | + | 25.71 i | 297.88 i | 2.90 e | 137 e |
| KSB(Grz) | + | + | 31.54 a | 397.20 c | 3.80 cd | 390 a |
| KSB(Phg) | + | - | 26.78 h | 267.93 m | 3.60 d | 229 bcd |
| KSB(Glm) | + | + | 30.31 c | 226.19 n | 4.00 c | 115 e |
| KSB(Afr) | - | + | 19.32 l | 278.62 l | 3.50 d | 270 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baba, Z.A.; Hamid, B.; Sheikh, T.A.; Alotaibi, S.H.; El Enshasy, H.A.; Ansari, M.J.; Zuan, A.T.K.; Sayyed, R.Z. Psychrotolerant Mesorhizobium sp. Isolated from Temperate and Cold Desert Regions Solubilizes Potassium and Produces Multiple Plant Growth Promoting Metabolites. Molecules 2021, 26, 5758. https://doi.org/10.3390/molecules26195758
Baba ZA, Hamid B, Sheikh TA, Alotaibi SH, El Enshasy HA, Ansari MJ, Zuan ATK, Sayyed RZ. Psychrotolerant Mesorhizobium sp. Isolated from Temperate and Cold Desert Regions Solubilizes Potassium and Produces Multiple Plant Growth Promoting Metabolites. Molecules. 2021; 26(19):5758. https://doi.org/10.3390/molecules26195758
Chicago/Turabian StyleBaba, Zahoor Ahmad, Basharat Hamid, Tahir Ahmad Sheikh, Saad H. Alotaibi, Hesham A. El Enshasy, Mohammad Javed Ansari, Ali Tan Kee Zuan, and R. Z. Sayyed. 2021. "Psychrotolerant Mesorhizobium sp. Isolated from Temperate and Cold Desert Regions Solubilizes Potassium and Produces Multiple Plant Growth Promoting Metabolites" Molecules 26, no. 19: 5758. https://doi.org/10.3390/molecules26195758











