Phytochemistry and Biological Activities of Iris Species Growing in Iraqi Kurdistan and Phenolic Constituents of the Traditional Plant Iris postii
Abstract
:1. Introduction
2. Results and Discussion
2.1. Literature Data about Iris germanica and Iris persica
2.1.1. Iris germanica
Isoflavonoids
Other Flavonoids
Miscellaneous Aromatic Compounds
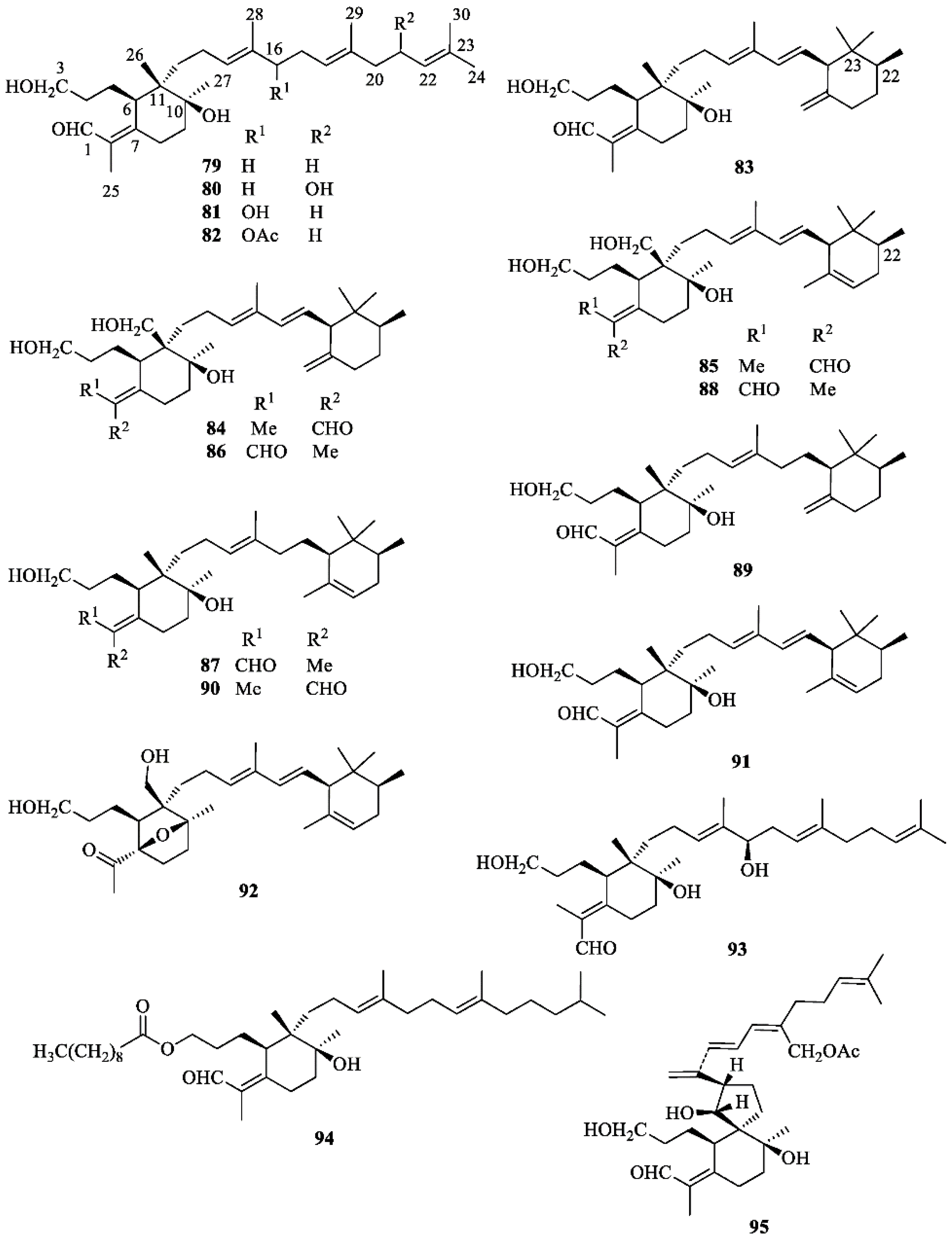
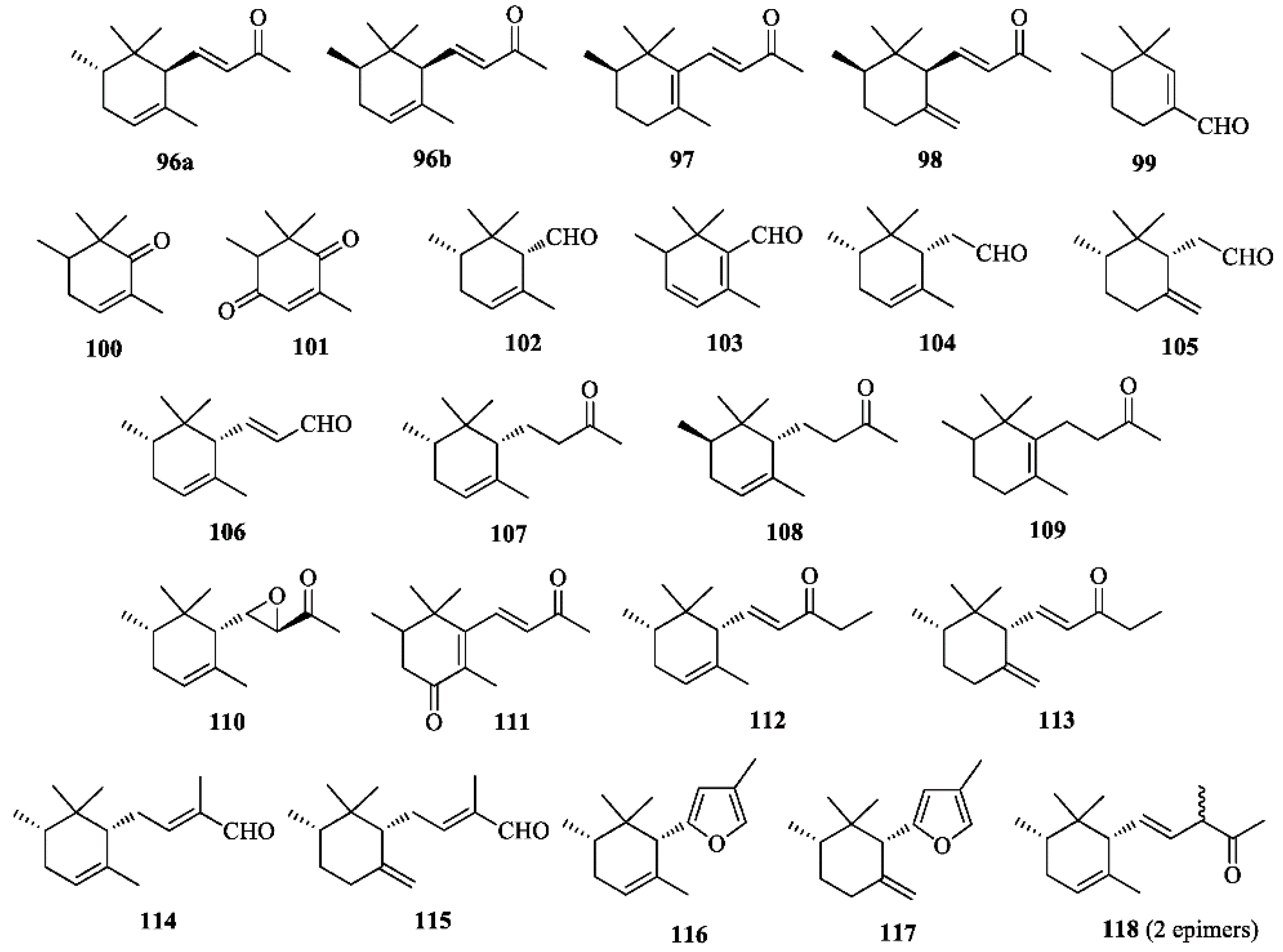
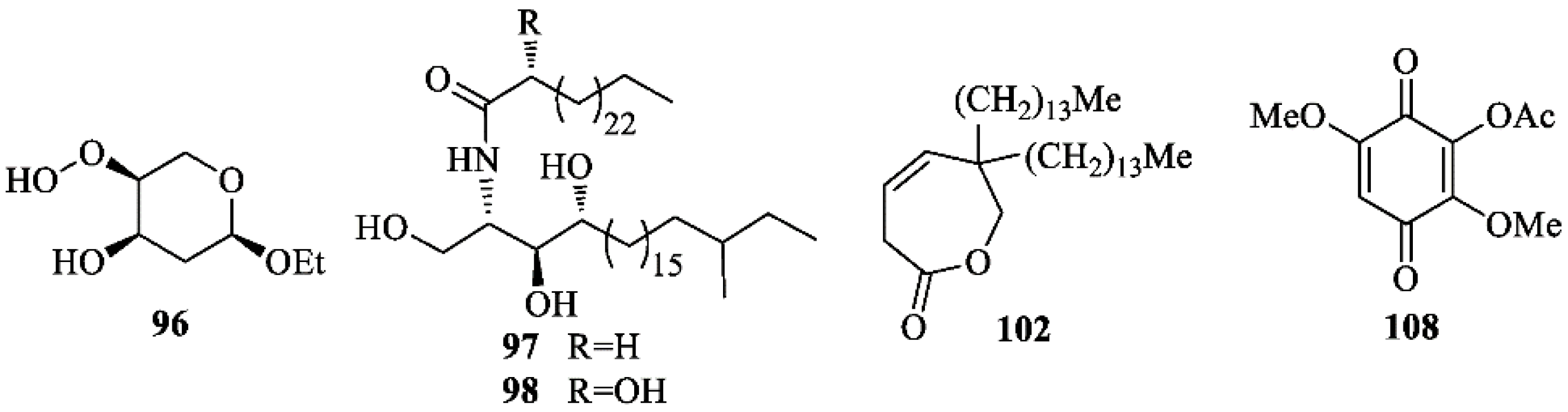
Terpenoids
Steroids and Miscellaneous Compounds
2.1.2. Iris persica
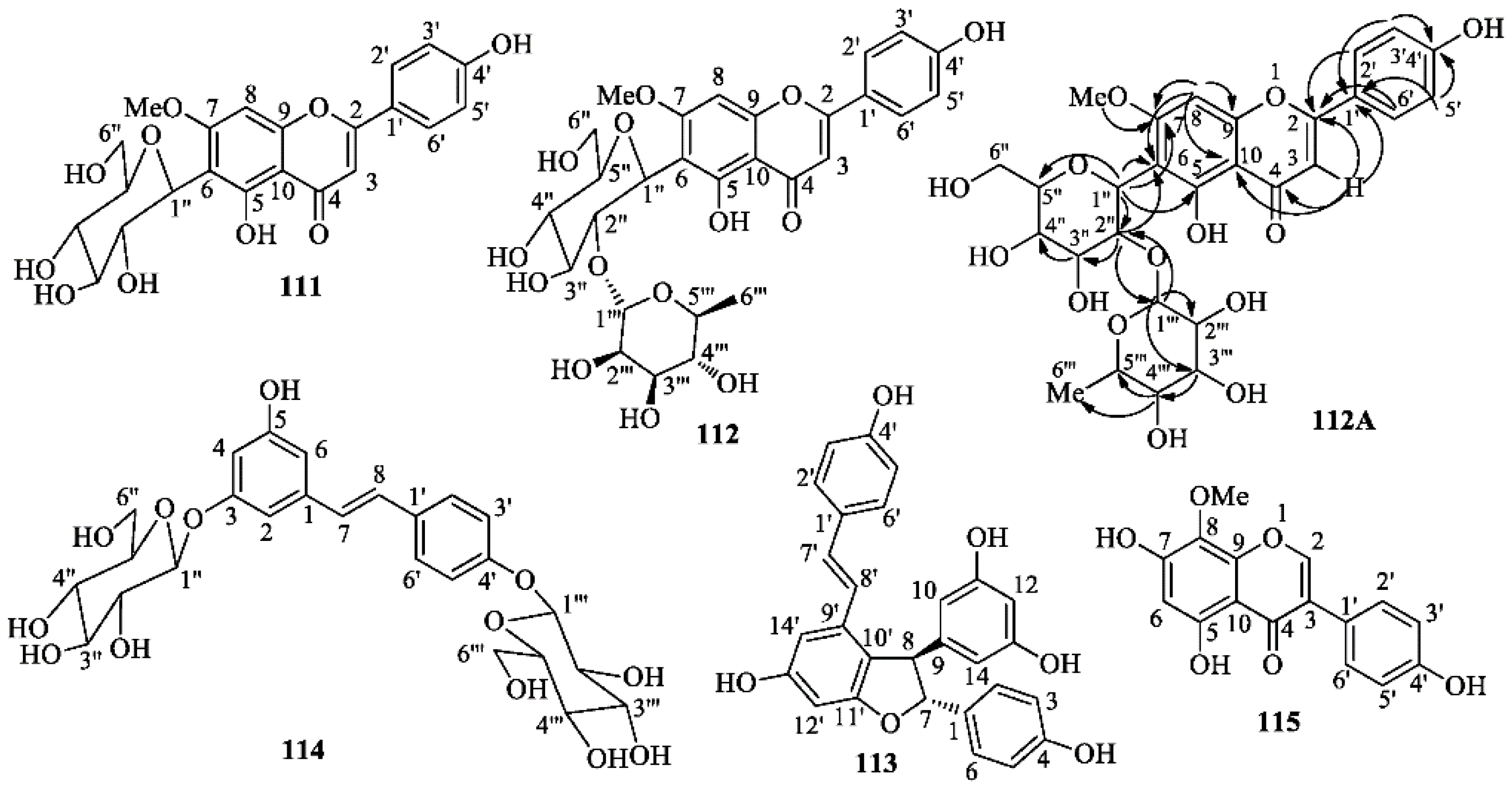
2.2. Phytochemical Studies on Iris postii
3. Material and Methods
3.1. General Experimental Techniques and Procedures
3.2. Plant Material
3.3. Extraction of I. postii and Chromatographic Fractionation of Extracts
3.4. Spectroscopic Data of Isolated Compounds
3.5. Free Radical Scavenging Activity
3.6. Total Antioxidant Capacity (TAOC—Ammonium Phosphomolybdate Assay)
3.7. Acid Hydrolysis of Compounds 112 and 114
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Nedhal, A.; Luay, Y. A survey of plants used in Iraqi traditional medicine. Jordan J. Pharm. Sci. 2010, 3, 100–107. [Google Scholar]
- Solecki, R.S. Shanidar IV, a Neanderthal Flower Burial in Northern Iraq. Science 1975, 190, 880–881. [Google Scholar] [CrossRef]
- Nahida, A. The most medicinal plants used in Iraq: Traditional knowledge. Adv. Environ. Biol. 2011, 5, 401–406. [Google Scholar]
- Mati, E.; De Boer, H. Ethnobotany and trade of medicinal plants in the Qaysari Market, Kurdish Autonomous Region, Iraq. J. Ethnopharmacol. 2011, 133, 490–510. [Google Scholar] [CrossRef] [PubMed]
- Amin, H.I.M.; Ibrahim, M.F.; Hussain, F.H.S.; Sardar, A.S.; Vidari, G. Phytochemistry and ethnopharmacology of some me-dicinal plants used in the Kurdistan region of Iraq. Nat. Prod. Commun. 2016, 11, 291–296. [Google Scholar] [PubMed] [Green Version]
- WCSP. World Checklist of Selected Plant Families. Available online: Wcsp.science.kew.org (accessed on 2 June 2014).
- Iris. Pacific Bulb Society. Available online: https://www.pacificbulbsociety.org (accessed on 3 March 2012).
- Kaššák, P. Secondary metabolites of the chosen genus Iris species. Acta Univ. Agric. Silvic. Mendel. Brun. 2012, 60, 269–280. [Google Scholar] [CrossRef] [Green Version]
- Kukula-Koch, W.; Sieniawska, E.; Widelski, J.; Urjin, O.; Głowniak, P.; Skalicka-Woźniak, K. Major secondary metabolites of Iris spp. Phytochem. Rev. 2015, 14, 51–80. [Google Scholar] [CrossRef]
- Brenna, E.; Fuganti, C.; Serra, S. From commercial racemic fragrances to odour active enantiopure compounds: The ten isomers of irone. C. R. Chim. 2003, 6, 529–546. [Google Scholar] [CrossRef]
- Townsend, C.; Guest, E. (Eds.) Flora of Iraq, Vol. 8: Monocotyledons; Baghdad-Ministry of Agriculture & Agrarian Reform: Baghdad, Iraq, 1985.
- Amin, H.I.M.; Amin, A.A.; Tosi, S.; Mellerio, G.G.; Hussain, F.H.S.; Picco, A.M.; Vidari, G. Chemical composition and anti-fungal activity of essential oils from flowers, leaves, rhizomes, and bulbs of the wild Iraqi Kurdish plant Iris persica. Nat. Prod. Commun. 2017, 12, 441–444. [Google Scholar]
- Amin, H.I.M.; Hussain, F.H.S.; Maggiolini, M.; Vidari, G. Bioactive constituents from the traditional Kurdish plant Iris persica. Nat. Prod. Commun. 2018, 13, 1127–1128. [Google Scholar] [CrossRef] [Green Version]
- Rahman, A.-U.; Nasim, S.; Baig, I.; Jalil, S.; Orhan, I.; Sener, B.; Choudary, M.I. Antiinflammatory isoflavonoids from rhizomes of Iris germanica. J. Ethnopharmacol. 2003, 86, 177–180. [Google Scholar] [CrossRef]
- Schütz, C.; Quitschau, M.; Hamburger, M.; Potterat, O. Profiling of isoflavonoids in Iris germanica rhizome extracts by microprobe NMR and HPLC–PDA–MS analysis. Fitoterapia 2011, 82, 1021–1026. [Google Scholar] [CrossRef] [PubMed]
- Shojaii, A.; Haghjoo, E.; Parvizi, M.M. Efficacy of topical herbal remedies for insomnia in Iranian traditional medicine. Pharmacogn. Res. 2019, 11, 188. [Google Scholar] [CrossRef]
- Rahman, A.-U.; Nasim, S.; Baig, I.; Orhan, I.E.; Şener, B.; Ayanoglu, F.; Choudhary, M.I. Isoflavonoid Glycosides from the Rhizomes of Iris germanica. Helvetica Chim. Acta 2003, 86, 3354–3362. [Google Scholar] [CrossRef]
- Abu-Dahab, R. Antiproliferative activity of selected medicinal plants of Jordan against a breast adenocarcinoma cell line (MCF7). Sci. Pharm. 2007, 75, 121–136. [Google Scholar] [CrossRef] [Green Version]
- Halpert, M.; Abu-Abied, M.; Avisar, O.; Moskovitz, Y.; Altshuler, O.; Cohen, A.; Weissberg, M.; Riov, J.; Gottlieb, H.E.; Perl, A.; et al. Rac-dependent doubling of HeLa cell area and impairment of cell migration and cell cycle by compounds from Iris germanica. Protoplasma 2011, 248, 785–797. [Google Scholar] [CrossRef]
- Nadaroğlu, H.; Demir, Y.; Demir, N. Antioxidant and radical scavenging properties of Iris germanica. Pharm. Chem. J. 2007, 41, 409–415. [Google Scholar] [CrossRef]
- Ullah, F.; Ayaz, M.; Sadiq, A.; Hussain, A.; Ahmad, S.; Imran, M.; Zeb, A. Phenolic, flavonoid contents, anticholinesterase and antioxidant evaluation of Iris germanica var; florentina. Nat. Prod. Res. 2015, 30, 1440–1444. [Google Scholar] [CrossRef]
- Basgedik, B.; Ugur, A.; Sarac, N.; Burcu, B.; Aysel, U.; Nurdan, S. Antimicrobial, antioxidant, antimutagenic activities, and phenolic compounds of Iris germanica. Ind. Crop. Prod. 2014, 61, 526–530. [Google Scholar] [CrossRef]
- Asghar, S.F.; Aziz, S.; Habib-Ur-Rehman; Ahmed, I.; Hussain, H.; Rahman, A.-U.; Choudhary, M.I. Secondary metabolites isolated from Iris germanica. Rec. Nat. Prod. 2009, 3, 139–152. [Google Scholar]
- Ibrahim, S.R.M.; Mohamed, G.A.A.; Al Musayeib, N.M. New Constituents from the Rhizomes of Egyptian Iris germanica L. Molecules 2012, 17, 2587–2598. [Google Scholar] [CrossRef] [PubMed]
- Orhan, I.E.; Nasim, S.; Tener, B.; Ayanoglu, F.; Özgüven, M.; Choudhary, M.I.; Rahman, A.-U. Two isoflavones and bioactivity spectrum of the crude extracts of Iris germanica rhizomes. Phytotherapy Res. 2003, 17, 575–577. [Google Scholar] [CrossRef] [PubMed]
- Uzair, A.; Bakht, J.; Iqbal, A.; Naveed, K.; Ali, N. In vitro antimicrobial activities of different solvent extracted samples from Iris germanica. Pak. J. Pharm. Sci. 2016, 29, 145–150. [Google Scholar] [PubMed]
- Sala, E.; Guasch, L.; Iwaszkiewicz, J.; Mulero, M.; Salvadó, M.J.; Bladé, C.; Ceballos, M.; Valls, C.; Zoete, V.; Grosdidier, A.; et al. Identification of human IKK-2 inhibitors of natural origin (Part II): In Silico prediction of IKK-2 inhibitors in natural extracts with known anti-inflammatory activity. Eur. J. Med. Chem. 2011, 46, 6098–6103. [Google Scholar] [CrossRef]
- Hoang, L.; Beneš, F.; Fenclova, M.; Kronusova, O.; Švarcová, V.; Řehořová, K.; Švecová, E.B.; Vosátka, M.; Hajslova, J.; Kaštánek, P.; et al. Phytochemical Composition and In Vitro Biological Activity of Iris spp. (Iridaceae): A New Source of Bioactive Constituents for the Inhibition of Oral Bacterial Biofilms. Antibiot. 2020, 9, 403. [Google Scholar] [CrossRef]
- Muto, Y.; Ichikawa, H.; Kitagawa, O.; Kumagai, K.; Watanabe, M.; Ogawa, E.; Seiki, M.; Shirataki, Y.; Yokoe, I.; Komatsu, M.; et al. Studies on Antiulcer Agents. I. The Effects of Various Methanol and Aqueous Extracts of Crude Drugs on Antiulcer Activity. Yakugaku Zasshi 1994, 114, 980–994. [Google Scholar] [CrossRef] [Green Version]
- Choudhary, M.I.; Naheed, S.; Jalil, S.; Alam, J.M.; Rahman, A.-U. Effects of ethanolic extract of Iris germanica on lipid profile of rats fed on a high-fat diet. J. Ethnopharmacol. 2005, 98, 217–220. [Google Scholar] [CrossRef]
- Singab, A.N.B.; Ahmed, A.H.; Sinkkonen, J.; Ovcharenko, V.; Pihlaja, K. Molluscicidal Activity and New Flavonoids from Egyptian Iris germanica L. (var. alba). Z. Naturforsch. C 2006, 61, 57–63. [Google Scholar] [CrossRef]
- Borhani, M.; Sharifzadeh, M.; Farzaei, M.H.; Narimani, Z.; Sabbaghziarani, F.; Gholami, M.; Rahimi, R. Protective effect of Iris germanica L. in β-amyloid-induced animal model of alzheimer’s disease. Afr. J. Tradit. Complement. Altern. Med. 2017, 14, 140–148. [Google Scholar] [CrossRef] [Green Version]
- Rahman, A.-U.; Nasim, S.; Baig, I.; Jahan, I.A.; Sener, B.; Orhan, I.; Choudhary, M.I. Isoflavonoid glycosides from the rhizomes of Iris germanica. Chem. Pharm. Bull. 2002, 50, 1100–1102. [Google Scholar] [CrossRef] [Green Version]
- Xie, G.-Y.; Qin, X.-Y.; Liu, R.; Wang, Q.; Lin, B.-B.; Wang, G.-K.; Xu, G.-K.; Wen, R.; Qin, M. New isoflavones with cytotoxic activity from the rhizomes of Iris germanica L. Nat. Prod. Res. 2013, 27, 2173–2177. [Google Scholar] [CrossRef] [PubMed]
- Xie, G.-Y.; Chen, Y.-J.; Wen, R.; Xu, J.-Y.; Wu, S.-S.; Qin, M.-J. Chemical constituents from rhizomes of Iris germanica. China J. Chin. Mater. Medica 2014, 39, 846–850. [Google Scholar]
- Kawase, A.; Ohta, N.; Yagishita, K. On the Chemical Structure of a New Isoflavone Glucoside, Homotectoridin, Isolated Together with Tectoridin from the Rhizomes of Iris germanica Linnaeus. Agric. Biol. Chem. 1973, 37, 145–150. [Google Scholar] [CrossRef]
- Ali, A.; Elemary, N.; Elmoghazi, M.; Darwish, F.; Frahm, A. Three isoflavonoids from Iris germanica. Phytochemistry 1983, 22, 2061–2063. [Google Scholar] [CrossRef]
- Ibrahim, S.R.M.; Mohamed, G.A.; Zayed, M.F.; Ross, S.A. 8-Hydroxyirilone 5-methyl ether and 8-hydroxyirilone, new antioxidant and α-amylase inhibitors isoflavonoids from Iris germanica rhizomes. Bioorganic Chem. 2017, 70, 192–198. [Google Scholar] [CrossRef]
- Akashi, T.; Ishizaki, M.; Aoki, T.; Ayabe, S.-I. Isoflavonoid production by adventitious root cultures of Iris germanica (Iridaceae). Plant. Biotechnol. 2005, 22, 207–215. [Google Scholar] [CrossRef]
- Ibrahim, S.R.M.; Al-Ahdal, A.; Khedr, A.; Mohamed, G.A.A. Antioxidant α-amylase inhibitors flavonoids from Iris germanica rhizomes. Rev. Bras. Farm. 2017, 27, 170–174. [Google Scholar] [CrossRef] [Green Version]
- Mohamed, G.A.; Ibrahim, S.R.; Ross, S.A. New ceramides and isoflavone from the Egyptian Iris germanica L. rhizomes. Phytochem. Lett. 2013, 6, 340–344. [Google Scholar] [CrossRef]
- Wollenweber, E.; Stevens, J.F.; Klimo, K.; Knauft, J.; Frank, N.; Gerhäuser, C. Cancer Chemopreventive in vitro Activities of Isoflavones Isolated from Iris germanica. Planta Medica 2003, 69, 15–20. [Google Scholar] [CrossRef]
- Nazir, N.; Koul, S.; Qurishi, M.A.; Taneja, S.C.; Ahmad, S.F.; Khan, B.; Bani, S.; Qazi, G.N. Immunomodulatory activity of isoflavones isolated from Iris germanica (Iridaceae) on T-lymphocytes and cytokines. Phytotherapy Res. 2009, 23, 428–433. [Google Scholar] [CrossRef]
- Roger, B.; Jeannot, V.; Fernandez, X.; Cerantola, S.; Chahboun, J. Characterisation and Quantification of Flavonoids in Iris germanica L. and Iris pallida Lam. Resinoids from Morocco. Phytochem. Anal. 2012, 23, 450–455. [Google Scholar] [CrossRef]
- Pailer, M.; Franke, F. Constituents of Iris germanica. Monatsh. Chem. 1973, 104, 1394–1408. [Google Scholar] [CrossRef]
- Asghar, S.F.; Rehman, H.-U.; Rahman, A.-U.; Choudhary, M.I. Phytochemical investigations on Iris germanica. Nat. Prod. Res. 2010, 24, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Alam, A.; Verma, M.; Naik, K.K.; Choudhary, D.; Kumar, S. Anti-osteoporotic activity of isoflavones from Iris germanica targeting NF-kappaB. Int. J. Pharm. Investig. 2019, 8, 122–129. [Google Scholar] [CrossRef]
- Xin, R.-h.; Zheng, J.-f.; Cheng, L.; Peng, W.-j.; Luo, Y.-j. Belamcanda chinensis (L.) DC: Ethnopharmacology, phytochemistry and pharmacology of an important traditional Chinese medicine. Afr. J. Tradit. Complement. Altern. Med. 2015, 12, 39–70. [Google Scholar]
- Wuttke, W.; Jarry, H.; Popp, M.; Christoffel, V.; Spengler, B. Use of Extracts and Preparation from Iris Plants and Tectorigenin as Medicaments. U.S. Patent 20040176310, 15 May 2002. [Google Scholar]
- Choudhary, D.; Alam, A. Pharmacology and phytochemistry of isoflavonoids from Iris species. J. Pharm. Clin. Res. 2017, 3, 555609. [Google Scholar]
- Kang, K.A.; Zhang, R.; Piao, M.J.; Ko, D.O.; Wang, Z.H.; Kim, B.J.; Park, J.W.; Kim, H.S.; Kime, D.H.; Hyun, J.W. Protective ef-fect of irisolidone, a metabolite of kakkalide, against hydrogen peroxide induced cell damage via antioxidant effect. Bioorg. Med. Chem. 2008, 16, 1133–1141. [Google Scholar] [CrossRef] [PubMed]
- Kawase, A.; Yagishita, K. On the structure of a new C-Glycosyl flavone, embinin, isolated from the petals of Iris germanica Linnaeous. Agric. Biol. Chem. 1968, 32, 537–538. [Google Scholar] [CrossRef]
- Hilsenbeck, R.A.; Mabry, T.J. C-glycosylflavones from Siphonoglossa sessilis. Phytochemistry 1983, 22, 2215–2217. [Google Scholar] [CrossRef]
- Harborne, J.B. Comparative Biochemistry of the Flavonoids; Academic Press: London, UK, 1967; p. 383. ISBN 01-232-4650-4. [Google Scholar]
- Xie, G.; Qin, X.; Chen, Y.; Wen, R.; Wu, S. Alkaloids from the Rhizomes of Iris germanica. Chem. Nat. Compd. 2017, 53, 196–198. [Google Scholar] [CrossRef]
- Jaenicke, L.; Marner, F.-J. The irones and their origin. Pure Appl. Chem. 1990, 65, 1365–1368. [Google Scholar] [CrossRef]
- Benoit-Vical, F.; Imbert, C.; Bonfils, J.-P.; Sauvaire, Y. Antiplasmodial and antifungal activities of iridal, a plant triterpenoid. Phytochemistry 2003, 62, 747–751. [Google Scholar] [CrossRef]
- Bonfils, J.-P.; Sauvaire, Y. Localization of iridals in Iris germanica rhizomes. Phytochemistry 1996, 41, 1281–1285. [Google Scholar] [CrossRef]
- Bonfils, J.-P.; Pinguet, F.; Culine, S.; Sauvaire, Y. Cytotoxicity of Iridals, Triterpenoids from Iris, on Human Tumor Cell Lines A2780 and K562. Planta Medica 2001, 67, 79–81. [Google Scholar] [CrossRef] [PubMed]
- Miyake, Y.; Ito, H.; Yoshida, T. Identification of iridals as piscicidal components of Iridaceous plants and their conformations associated with CD spectra. Can. J. Chem. 1997, 75, 734–741. [Google Scholar] [CrossRef]
- Marner, F.J.; Krick, W.; Gellrich, B.; Jaenicke, L.; Winter, W. Irigermanal and iridogermanal: Two new triterpenoids from rhizomes of Iris germanica L. J. Org. Chem. 1982, 47, 2531–2536. [Google Scholar] [CrossRef]
- Ito, H.; Miyake, Y.; Yoshida, T. New piscicidal triterpenes from Iris germanica. Chem. Pharm. Bull. 1995, 43, 1260–1262. [Google Scholar] [CrossRef] [Green Version]
- Potterat, O.; Herzog, C.; Raith, M.; Ebrahimi, S.N.; Hamburger, M. Irigermanone, a Noriridal with Unprecedented Methylketone Function, from Iris germanica. Helvetica Chim. Acta 2014, 97, 32–38. [Google Scholar] [CrossRef]
- Orhan, I.; Sener, B.; Hashimoto, T.; Asakawa, Y.; Ozgüven, M.; Ayanoğlu, F. Iristectorone K, a novel monocyclic triterpene ester from Iris germanica rhizomes growing in Turkey. Fitoterapia 2002, 73, 316–319. [Google Scholar] [CrossRef]
- Muto, Y.; Ichikawa, H.; Seiki, M.; Shirataki, Y.; Yokoe, I.; Komatsu, M. Studies on Antiulcer Agents. II. Synthesis and Antiulcer Activity of Phenylpropanol Derivatives. Yakugaku Zasshi 1994, 114, 995–1004. [Google Scholar] [CrossRef] [Green Version]
- Maurer, B.; Hauser, A.; Froidevaux, J.-C. New Irone-Related Constituents from the essential oil of Iris germanica L. Helvetica Chim. Acta 1989, 72, 1400–1415. [Google Scholar] [CrossRef]
- Bonfils, J.-P.; Bonfils, C.; Larroque, C.; Surjus, A.; Gleize, D.; Sauvaire, Y. Lipid composition of microsomes of Iris germanica rhizomes. Phytochemistry 1995, 38, 585–587. [Google Scholar] [CrossRef]
- Davoust, D.; Massias, M.; Molho, D. 13C-NMR investigation of flavonoid C-β-d-glucosides. Detection of a conformational equilibrium. Magn. Reson. Chem. 1980, 13, 218–219. [Google Scholar] [CrossRef]
- Cheng, G.; Bai, Y.; Zhao, Y.; Tao, J.; Liu, Y.; Tu, G.; Ma, L.; Liao, N.; Xu, X. Flavonoids from Ziziphus jujuba Mill var. spinosa. Tetrahedron 2000, 56, 8915–8920. [Google Scholar] [CrossRef]
- Bjorøy, Ø.; Rayyan, S.; Fossen, T.; Kalberg, K.; Andersen, Ø.M. C-glycosyl anthocyanidins synthesized from C-glycosylflavones. Phytochemistry 2009, 70, 278–287. [Google Scholar] [CrossRef]
- Quintão, N.L.M.; Meyre-Silva, C.; Silva, G.F.; Antonialli, C.S.; Rocha, L.W.; Lucinda-Silva, R.M.; Malheiros, A.; Souza, M.M.; Filho, V.C.; Bresolin, T. Aleurites moluccana (L.) Willd. Leaves: Mechanical Antinociceptive Properties of a Standardized Dried Extract and Its Chemical Markers. Evid. Based Complement. Altern. Med. 2011, 2011, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Frank, J.H.; Powder-George, Y.L.; Ramsewak, R.S.; Reynolds, W.F. Variable Temperature 1H-NMR Studies on Two C-Glycosylflavones. Molecules 2012, 17, 7914–7926. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.-S.; Lee, I.-J.; Lin, Y.-L. Flavone glycosides from commercially available Lophatheri herba and their chromatographic fingerprinting and quantitation. J. Food Drug Anal. 2015, 23, 821–827. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shirane, S.; Ohya, S.; Matsuo, T.; Hirose, R.; Koga, D.; Ide, A.; Yagishita, K. C-Glycosyl compounds in the leaves of Gemmingia chinensis O. KUNTZE. Agric. Biol. Chem. 1982, 46, 2595–2597. [Google Scholar] [CrossRef] [Green Version]
- Ames, B.N. Dietary carcinogens and anticarcinogens. Oxygen radicals and degenerative diseases. Science 1983, 221, 1256–1264. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free Radicals Biol. Med. 2010, 49, 1603–1616. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Odunola, O.; Olugbami, J.O.; Gbadegesin, M.A. In vitro free radical scavenging and antioxidant properties of ethanol extract of Terminalia glaucescens. Pharmacogn. Res. 2015, 7, 49–56. [Google Scholar] [CrossRef] [Green Version]
- Sun, L.; Zhang, J.; Lu, X.; Zhang, L.; Zhang, Y. Evaluation to the antioxidant activity of total flavonoids extract from persimmon (Diospyros kaki L.) leaves. Food Chem. Toxicol. 2011, 9, 2689–2696. [Google Scholar] [CrossRef]
- Umamaheswari, M.; Chatterjee, T.K. In vitro antioxidant activities of the fractions of Coccinia grandis L. leaf extract. Afr. J. Tradit. Complement. Altern. Med. 2008, 5, 61–73. [Google Scholar] [CrossRef] [Green Version]
- Gilardoni, G.; Chiriboga, X.; Finzi, P.V.; Vidari, G. New 3,4-Secocycloartane and 3,4-Secodammarane Triterpenes from the Ecuadorian Plant Coussarea macrophylla. Chem. Biodivers. 2015, 12, 946–954. [Google Scholar] [CrossRef] [PubMed]
- De Rosa, S.; De Giulio, A.; Tommonaro, G. Aliphatic and aromatic glycosides from the cell cultures of Lycopersicon esculentum. Phytochemistry 1996, 42, 1031–1034. [Google Scholar] [CrossRef]
- Ersöz, T.; Harput, Ü.Ş.; Saragoğlu, İ.; Çaliş, I.; Ogihara, Y. Phenolic compounds from Scutellaria pontica. Turk. J. Chem. 2002, 26, 581–588. [Google Scholar]
- Zhou, G.; Yan, R.; Wang, X.; Li, S.; Lin, J.; Liu, J.; Zhao, Z. The overlooked rotational isomerism of C-glycosyl flavonoids. Phytochem. Rev. 2019, 18, 443–461. [Google Scholar] [CrossRef]
- Liu, W.-B.; Hu, L.; Hu, Q.; Chen, N.-N.; Yang, Q.-S.; Wang, F.-F. New Resveratrol Oligomer Derivatives from the Roots of Rheum ihasaense. Molecules 2013, 18, 7093–7102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ngoc, T.M.; Hung, T.M.; Thuong, P.T.; Na, M.; Kim, H.; Ha, D.T.; Min, B.-S.; Minh, P.T.H.; Bae, K. Inhibition of human low density lipoprotein and high density lipoprotein oxidation by oligostilbenes from rhubarb. Biol. Pharm. Bull. 2008, 31, 1809–1812. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanchanapoom, T.; Suga, K.; Kasai, R.; Yamasaki, K.; Kamel, M.S.; Mohamed, M.H. Stilbene and 2-arylbenzofuran glucosides from the rhizomes of Schoenocaulon officinale. Chem. Pharm. Bull. 2002, 50, 863–865. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bai, X.; Qu, J.-L.; Liu, J.; Sun, J.-H.; Yuan, D. Isolation and identification of urinary metabolites of tectoridin in rats. J. Asian Nat. Prod. Res. 2011, 13, 604–610. [Google Scholar] [CrossRef]
- He, M.; Min, J.-W.; Kong, W.-L.; He, X.-H.; Li, J.-X.; Peng, B.-W. A review on the pharmacological effects of vitexin and iso-vitexin. Fitoterapia 2016, 115, 74–85. [Google Scholar] [CrossRef]
- Ganesan, K.; Xu, B. Molecular targets of vitexin and isovitexin in cancer therapy: A critical review. Ann. N. Y. Acad. Sci. 2017, 1401, 102–113. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.E.; Jeon, S.J.; Ryu, B.; Park, S.J.; Ko, S.Y.; Lee, Y.; Kim, E.; Lee, S.; Kim, H.; Jang, D.; et al. Swertisin, a C-glucosylflavone, ameliorates scopolamine-induced memory impairment in mice with its adenosine A1 receptor antagonistic property. Behav. Brain Res. 2016, 306, 137–145. [Google Scholar] [CrossRef]
- Wu, C.; Shen, J.; He, P.; Chen, Y.; Li, L.; Zhang, L.; Li, Y.; Fu, Y.; Dai, R.; Meng, W.; et al. The α-glucosidase inhibiting isoflavones isolated from Belamcanda chinensis leaf extract. Rec. Nat. Prod. 2012, 6, 110–120. [Google Scholar]
- Choi, C.W.; Cha, M.-R.; Park, J.H.; Kim, Y.S.; Yon, G.H.; Hong, K.S.; Ryu, S.Y. Glucosidase Inhibitors from Seed Extract of Paeonia lactiflora. J. Korean Soc. Appl. Biol. Chem. 2009, 52, 638–642. [Google Scholar] [CrossRef]
- Wu, Y.; Li, S.; Liu, J.; Liu, X.; Ruan, W.; Lu, J.-W.; Liu, Y.; Lawson, T.; Shimoni, O.; Lovejoy, D.B.; et al. Stilbenes from Veratrum maackii Regel Protect against Ethanol-Induced DNA Damage in Mouse Cerebellum and Cerebral Cortex. ACS Chem. Neurosci. 2018, 9, 1616–1624. [Google Scholar] [CrossRef]
- Falomir, E.; Lucas, R.; Peñalver, P.; Martí-Centelles, R.; Dupont, A.; Zafra-Gómez, A.; Carda, M.; Morales, J.C. Cytotoxic, Antiangiogenic and Antitelomerase Activity of Glucosyl- and Acyl- Resveratrol Prodrugs and Resveratrol Sulfate Metabolites. ChemBioChem 2016, 17, 1343–1348. [Google Scholar] [CrossRef] [Green Version]






| Compound | Bioactivity | Reference |
|---|---|---|
| Germanaism A (1) | Cytotoxic activity IC50 = 43.9 ± 1.5 μM (MTT); 4.5 ± 0.4 μM (ATP) | [34] |
| Tectoridin (9) | Free-radical scavenger, antioxidant, anti-inflammatory, antiproliferative, oestrogenic, anti-alcohol injury, hepatoprotective effects | [48] |
| Iridin (10) | Potent anti-inflammatory effects (induced paw edema test) | [24] |
| Iridin A (11) | High antioxidant activity; α-amylase inhibitory activity | [38,40] |
| Irisolidone 7-O-β-d-glucopyranoside (14) | CyP1A inhibitor; QR inhibitor; DPPH scavenger | |
| Irilone (17) | Cytotoxic activity IC50 = 47.7 ± 3.5 μM (MTT), 17.7 ± 1.4 μM (ATP); potent anti-inflammatory effects (induced paw edema test): high antioxidant activity; α-amylase inhibitory activity; potent inhibitor of cytochrome P450 1A activity (IC50 = 0.3 ± 0.1 μM); immunomodulatory activity; CyP1A inhibitor; moderate QR inhibitor; DPPH scavenger | [34] [24] [38,40] [42] [43] |
| Irilone 4′-O-β-d-glucopyranoside (19) | Potent anti-inflammatory effects (induced paw edema test) | [24] |
| 8-Hydroxyirilone (21) | High antioxidant activity; α-amylase inhibitory activity | [38,40] |
| 8-Hydroxyirilone 5-methyl ether (22) | High antioxidant activity; α-amylase inhibitory activity | [38,40] |
| Iriflogenin (23) | Potent inhibitor of cytochrome P450 1A activity (IC50 = 1.4 ± 0.6 μM); CyP1A inhibitor; weak DPPH scavenger | [42] |
| Irifloside (25) | Cytotoxic activity IC50 = 21.5 ± 4.4 μM (MTT); 19.4 ± 1.3 μM (ATP) | [34] |
| Irisolone (nigricin) (28) | High anti-inflammatory activity | [14] |
| Nigricanin (iriskashmirianin) (29) | CyP1A inhibitor; moderate QR inhibitor; weak DPPH scavenger | [42] |
| Iriskashmirianin A (30) | Cytotoxic activity IC50 = 20.9 ± 2.7 μM (MTT); 4.3 ± 0.9 μM (ATP) | [34] |
| 5,7-Dihydroxy-3-(3′-hydroxy-4′,5′-dimethoxy)-8-methoxy-4H-1-benzopyran-4-one (33) | Significant anti-inflammatory activity | [14] |
| Tectorigenin (35) | Antifungal, free radical scavenger, antioxidant, anti-inflammatory, anti-angiogenic, antiproliferative, antineoplastic, hypoglycaemic, oestrogenic, hepatoprotectiv, antithrombotic, cardiovascular, anti-alcohol injury activities; in patented pharmaceutical compositions for the treatment of hormone-related diseases | [48,49] |
| Irigenin (37) | Potent anti-inflammatory effects (induced paw edema test and inhibition against superoxide); α-amylase inhibitory activity; potent inhibitor of cytochrome P450 1A activity (IC50 = 1.2 ± 0.3 μM); CyP1A inhibitor; moderate QR inhibitor, DPPH scavenger | [14,24] [38,40] [42] |
| Irigenin S (38) | Potent anti-inflammatory effects (induced paw edema test) | [24] |
| Irisolidone (42) | Potent anti-inflammatory effects (induced paw edema test); α-amylase inhibitory activity; immunomodulatory activity; CyP1A inhibitor; QR inhibitor; DPPH scavenger; antiproliferative activity against amelanotic melanoma and large lung carcinoma cells; antioxidant properties | [24] [38,40] [43] [50] [51] |
| Iristectorigenin A (43) | Weak anti-inflammatory activity | [14] |
| Isoflavone (44) | Moderate anti-inflammatory activity | [14] |
| 5,7,8-Trihydroxy-3-(4-methoxyphenyl)-2-methyl-4H-chromen-4-one (47) | Significant inhibition of TRAP in NF-kB ligand-induced osteoclastic RAW 264.7 cells (66.67 ± 2.71%) | [47] |
| 6,7-Dihydroxy-3-(4-methoxyphenyl)-2-methyl-4H-chromen-4-one (48) | Significant inhibition of TRAP in NF-kB ligand-induced osteoclastic RAW 264.7 cells (57.32 ± 2.46%) | [47] |
| Sample | DPPH Scavenging Activity | Total Antioxidant Capacity (TAOC) b | ||
|---|---|---|---|---|
| EC50 (μg/mL) | EC50 (μM/L) | AAE a | ||
| Androsin (66) | 48.94 ± 0.09 | 149.21 | 0.48 | - |
| Isovitexin (109) | 50.97 ± 1.11 | 117.99 | 0.46 | - |
| Swertisin (111) | 37.35 ± 0.13 | 83.74 | 0.63 | - |
| 2″-O-α-l-Rhamnosyl swertisin (112) | 26.52 ± 0.11 | 44.79 | 0.89 | - |
| ε-Viniferin (113) | 26.06 ± 0.01 | 57.39 | 0.90 | - |
| Trans-resveratrol 3,4′-O-di-β-d-glucopyranoside (114) | 22.91 ± 0.05 | 41.50 | 1.03 | - |
| Isotectorigenin (115) | 34.87 ± 0.13 | 116.23 | 0.67 | - |
| Ascorbic acid | 23.52 ± 0.22 | 133.63 | 1.00 | - |
| IPA c | 19.21 ± 0.01 | - | 1.22 | 0.39 |
| IPAD d | 62.79 ± 0.03 | - | 0.37 | 0.21 |
| IPR e | 46.28 ± 0.12 | - | 0.51 | 0.29 |
| IPRB f | 39.11 ± 0.10 | - | 0.60 | 0.46 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amin, H.I.M.; Hussain, F.H.S.; Najmaldin, S.K.; Thu, Z.M.; Ibrahim, M.F.; Gilardoni, G.; Vidari, G. Phytochemistry and Biological Activities of Iris Species Growing in Iraqi Kurdistan and Phenolic Constituents of the Traditional Plant Iris postii. Molecules 2021, 26, 264. https://doi.org/10.3390/molecules26020264
Amin HIM, Hussain FHS, Najmaldin SK, Thu ZM, Ibrahim MF, Gilardoni G, Vidari G. Phytochemistry and Biological Activities of Iris Species Growing in Iraqi Kurdistan and Phenolic Constituents of the Traditional Plant Iris postii. Molecules. 2021; 26(2):264. https://doi.org/10.3390/molecules26020264
Chicago/Turabian StyleAmin, Hawraz Ibrahim M., Faiq H. S. Hussain, Soran K. Najmaldin, Zaw Min Thu, Mohammed Farhad Ibrahim, Gianluca Gilardoni, and Giovanni Vidari. 2021. "Phytochemistry and Biological Activities of Iris Species Growing in Iraqi Kurdistan and Phenolic Constituents of the Traditional Plant Iris postii" Molecules 26, no. 2: 264. https://doi.org/10.3390/molecules26020264








