Methyl Jasmonate Effect on Betulinic Acid Content and Biological Properties of Extract from Senna obtusifolia Transgenic Hairy Roots
Abstract
:1. Introduction
2. Results
2.1. Transgenic Hairy Root Growth Index
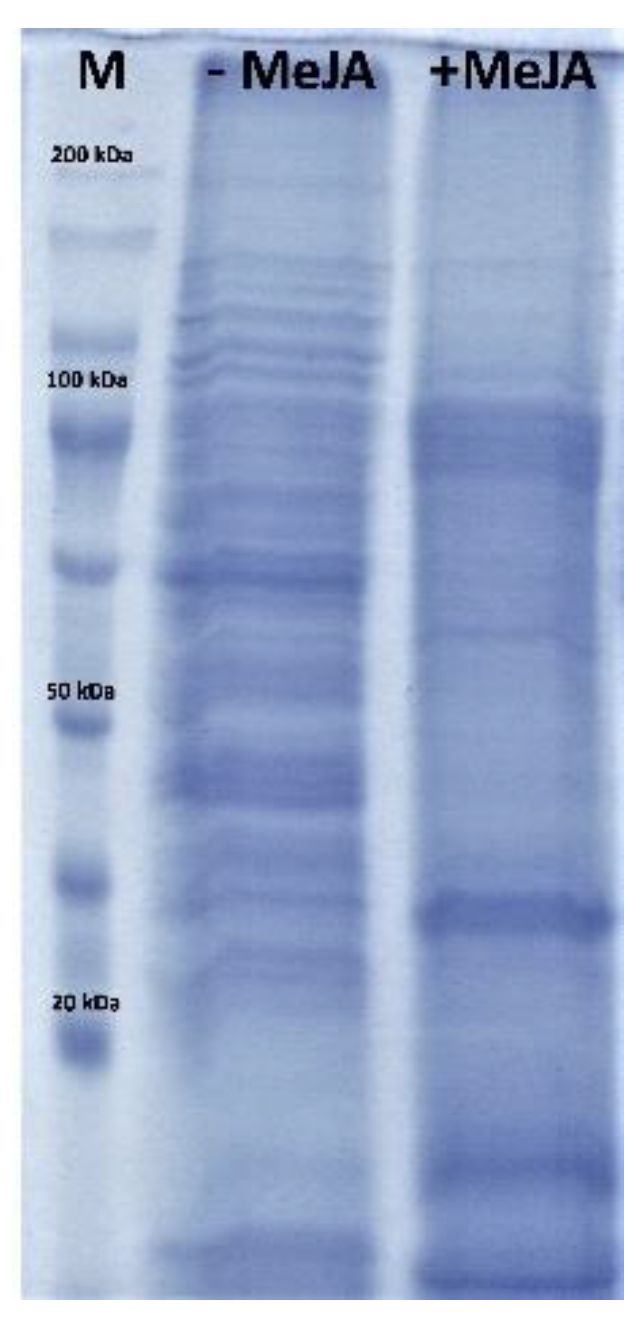
2.2. Effect of Elicitation on Protein Profile of Senna Obtusifolia Transgenic Hairy Roots
2.3. HPLC Analysis
2.4. Effect of Transgenic Hairy Roots of Senna obtusifolia Extracts on U87MG, DU-145 and A549 Cancer Cells Lines
2.5. Apoptosis and Necrosis Determination Using Flow Cytometry
2.6. Loss of Mitochondrial Membrane Potential (MMP)
2.7. Chromosomal DNA Fragmentation
2.8. DNA Damage Measured by Comet Assay
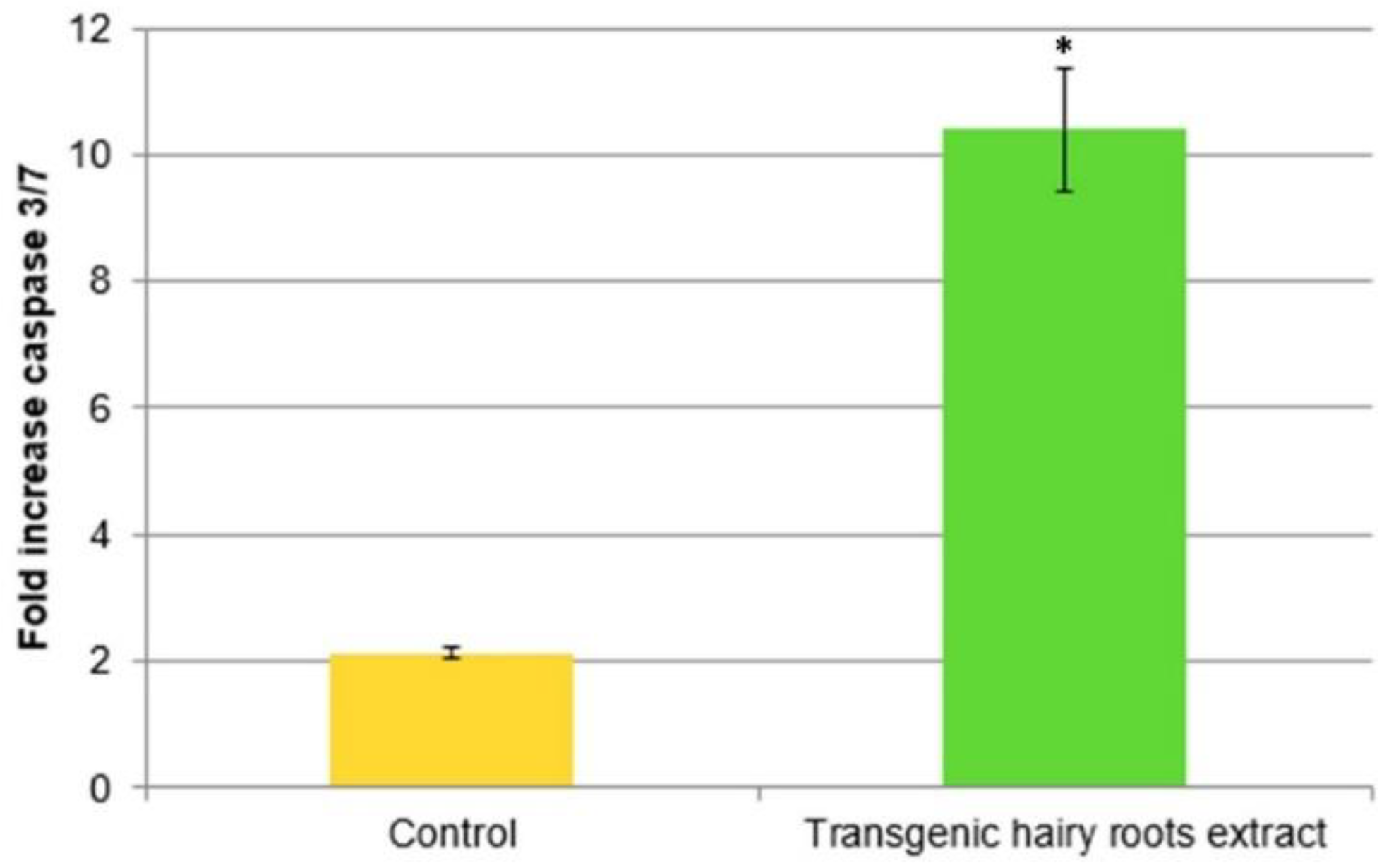
2.9. Influence on the Caspase-3/7 Activity
2.10. Topoisomerase I Inhibition Assay with Hairy Root Extract
3. Discussion
4. Materials and Methods
4.1. Establishment of Hairy Root Cultures
4.2. Optimization of Hairy Root Culture Conditions in the Bioreactor
4.3. Elicitor Treatment
4.4. Hairy Roots Growth Index Determination
4.5. Hairy Root Protein Isolation
4.6. SDS-PAGE Protein Electrophoresis
4.7. Plant Extract Preparation
4.8. HPLC Determination of Betulinic Acid
4.9. Cell Cultures
4.10. Cell Viability
4.11. Apoptosis/Necrosis Detection by Flow Cytometry
4.12. Mitochondrial Membrane Potential (MMP)
4.13. Chromosomal DNA Fragmentation
4.14. DNA Damage Measured by Comet Assay
4.15. Effect on the Activity of Caspase-3/7
4.16. Topoisomerase I Inhibition Assay with Hairy Root Extract
4.17. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Durazzo, A.; Id, L.D.A.; Camilli, E.; Piccinelli, R.; Turrini, A.; Marletta, L.; Marconi, S.; Lucarini, M.; Lisciani, S.; Gabrielli, P.; et al. From Plant Compounds to Botanicals and Back : A Current Snapshot. Molecules 2018, 23, 1844. [Google Scholar] [CrossRef] [Green Version]
- Salehi, B.; Kumar, N.V.A.; Şener, B.; Sharifi-Rad, M.; Kılıç, M.; Mahady, G.B.; Vlaisavljevic, S.; Iriti, M.; Kobarfard, F.; Setzer, W.N.; et al. Medicinal Plants Used in the Treatment of Human Immunodeficiency Virus. Int. J. Mol. Sci. 2018, 19, 1459. [Google Scholar] [CrossRef] [Green Version]
- Elshafie, H.S.; Camele, I. An Overview of the Biological Effects of Some Mediterranean Essential Oils on Human Health. Biomed. Res. Int. 2017, 2017, 9268468. [Google Scholar] [CrossRef]
- Salehi, B.; Ata, A.; Kumar, N.V.A.; Sharopov, F.; Ram, K.; Ruiz-ortega, A.; Ayatollahi, S.A. Antidiabetic Potential of Medicinal Plants and Their Active Components. Biomolecules 2019, 9, 551. [Google Scholar] [CrossRef] [Green Version]
- Uritu, C.M.; Mihai, C.T.; Stanciu, G.D.; Dodi, G.; Alexa-Stratulat, T.; Luca, A.; Leon-Constantin, M.M.; Stefanescu, R.; Bild, V.; Melnic, S.; et al. Medicinal Plants of the Family Lamiaceae in Pain Therapy: A Review. Pain Res. Manag. 2018, 2018, 7801543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tanase, C.; Coșarcă, S.; Muntean, D.-L. A Critical Review of Phenolic Compounds Extracted from the Bark of Woody Vascular Plants and Their Potential Biological Activity. Molecules 2019, 24, 1182. [Google Scholar] [CrossRef] [Green Version]
- Reddy, P.R.K.; Elghandour, M.M.M.Y.; Salem, A.Z.M.; Yasaswini, D.; Reddy, P.P.R.; Reddy, A.N.; Nagarjuna, R.; Hyderd, A. Plant secondary metabolites as feed additives in calves for antimicrobial stewardship. Anim. Feed Sci. Technol. 2020, 264, 114469. [Google Scholar] [CrossRef]
- Chiocchio, I.; Mandrone, M.; Tomasi, P.; Marincich, L.; Poli, F. Plant Secondary Metabolites: An Opportunity for Circular Economy. Molecules 2021, 26, 495. [Google Scholar] [CrossRef] [PubMed]
- Birchfield, A.S.; McIntosh, C.A. Metabolic Engineering and Synthetic Biology of Plant Natural Products—A Minireview. Curr. Plant Biol. 2020, 24, 100163. [Google Scholar] [CrossRef]
- Qiu, H.; Su, L.; Wang, H.; Zhang, Z. Chitosan Elicitation of Saponin Accumulation in Psammosilene Tunicoides Hairy Roots by Modulating Antioxidant Activity, Nitric Oxide Production and Differential Gene Expression. Plant Physiol. Biochem. 2021, 166, 115–127. [Google Scholar] [CrossRef] [PubMed]
- Amdoun, R.; Benyoussef, E.H.; Benamghar, A.; Khelifi, L. Prediction of Hyoscyamine Content in Datura Stramonium L. Hairy Roots Using Different Modeling Approaches: Response Surface Methodology (RSM), Artificial Neural Network (ANN) and Kriging. Biochem. Eng. J. 2019, 144, 8–17. [Google Scholar] [CrossRef]
- Nourozi, E.; Hosseini, B.; Maleki, R.; Mandoulakani, B.A. Pharmaceutical Important Phenolic Compounds Overproduction and Gene Expression Analysis in Dracocephalum Kotschyi Hairy Roots Elicited by SiO2 Nanoparticles. Ind. Crop. Prod. 2019, 133, 435–446. [Google Scholar] [CrossRef]
- Akhgari, A.; Laakso, I.; Maaheimo, H.; Choi, Y.H.; Seppänen-Laakso, T.; Oksman-Caldentey, K.M.; Rischer, H. Methyljasmonate Elicitation Increases Terpenoid Indole Alkaloid Accumulation in Rhazya Stricta Hairy Root Cultures. Plants 2019, 8, 534. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wei, T.; Deng, K.; Gao, Y.; Chen, L.; Song, W.; Zhang, Y.; Wang, C.; Chen, C. SmKSL Overexpression Combined with Elicitor Treatment Enhances Tanshinone Production from Salvia Miltiorrhiza Hairy Roots. Biochem. Eng. J. 2020, 158, 107562. [Google Scholar] [CrossRef]
- Valdiani, A.; Hansen, O.K.; Nielsen, U.B.; Johannsen, V.K.; Shariat, M.; Georgiev, M.I.; Omidvar, V.; Ebrahimi, M.; Tavakoli Dinanai, E.; Abiri, R. Bioreactor-Based Advances in Plant Tissue and Cell Culture: Challenges and Prospects. Crit. Rev. Biotechnol. 2019, 39, 20–34. [Google Scholar] [CrossRef]
- Hao, X.; Shi, M.; Cui, L.; Xu, C.; Zhang, Y.; Kai, G. Effects of Methyl Jasmonate and Salicylic Acid on Tanshinone Production and Biosynthetic Gene Expression in Transgenic Salvia Miltiorrhiza Hairy Roots. Biotechnol. Appl. Biochem. 2015, 62, 24–31. [Google Scholar] [CrossRef]
- Kowalczyk, T.; Sitarek, P.; Toma, M.; Rijo, P.; Domínguez-Martín, E.; Falcó, I.; Sánchez, G.; Śliwiński, T. Enhanced Accumulation of Betulinic Acid in Transgenic Hairy Roots of Senna Obtusifolia Growing in the Sprinkle Bioreactor and Evaluation of Their Biological Properties in Various Biological Models. Chem. Biodivers. 2021, 18, e2100455. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Fu, J.; Yin, X.; Yang, C.; Zhang, X.; Wang, W.; Du, X.; Wang, Q.; Ni, J. Cassiae Semen: A Review of Its Phytochemistry and Pharmacology (Review). Mol. Med. Rep. 2017, 16, 2331–2346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shabab, S.; Gholamnezhad, Z.; Mahmoudabady, M. Protective Effects of Medicinal Plant against Diabetes Induced Cardiac Disorder: A Review. J. Ethnopharmacol. 2021, 265, 113328. [Google Scholar] [CrossRef]
- Qian, K.; Nakagawa-Goto, K.; Yu, D.; Morris-Natschke, S.L.; Nitz, T.J.; Kilgore, N.; Allaway, G.P.; Lee, K.H. Anti-AIDS Agents 73: Structure-Activity Relationship Study and Asymmetric Synthesis of 3-O-Monomethylsuccinyl-Betulinic Acid Derivatives. Bioorg. Med. Chem. Lett. 2007, 17, 6553–6557. [Google Scholar] [CrossRef] [Green Version]
- Msanne, J.; Kim, H.; Cahoon, E.B. Biotechnology Tools and Applications for Development of Oilseed Crops with Healthy Vegetable Oils. Biochimie 2020, 178, 4–14. [Google Scholar] [CrossRef]
- Park, S.M.; Kim, H.W.; Park, H.J. Callus-Based 3D Printing for Food Exemplified with Carrot Tissues and Its Potential for Innovative Food Production. J. Food Eng. 2020, 271, 109781. [Google Scholar] [CrossRef]
- Nogareda, C.; Moreno, J.A.; Angulo, E.; Sandmann, G.; Portero, M.; Capell, T.; Zhu, C.; Christou, P. Carotenoid-Enriched Transgenic Corn Delivers Bioavailable Carotenoids to Poultry and Protects Them against Coccidiosis. Plant Biotechnol. J. 2016, 14, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.D.; Kumar, S.; Daniell, H. Expression of b -Glucosidase Increases Trichome Density and Artemisinin Content in Transgenic Artemisia Annua Plants. Plant Biotechnol. J. 2016, 14, 1034–1045. [Google Scholar] [CrossRef] [Green Version]
- Parizi, K.J.; Rahpeyma, S.A. The Novel Paclitaxel-Producing System : Establishment of Corylus Avellana L. Hairy Root Culture. Vitr. Cell. Dev. Biol. Plant 2020, 56, 290–297. [Google Scholar] [CrossRef]
- Srivastava, V.; Mehrotra, S.; Verma, P.K. Biotechnological Interventions for Production of Therapeutic Root Cultures of Medicinal Plants; Elsevier: Amsterdam, The Netherlands, 2017; ISBN 9780444636614. [Google Scholar]
- Mehrotra, S.; Srivastava, V.; Rahman, L.U.; Kukreja, A.K. Hairy Root Biotechnology—Indicative Timeline to Understand Missing Links and Future Outlook. Protoplasma 2015, 252, 1189–1201. [Google Scholar] [CrossRef]
- Ibañez, S.; Talano, M.; Ontañon, O.; Suman, J.; Medina, M.I.; Macek, T.; Agostini, E. Transgenic plants and hairy roots: Exploiting the potential of plant species to remediate contaminants. New Biotechnol. 2016, 33, 625–635. [Google Scholar] [CrossRef]
- Chandra, C.; Mohd, G. Chemical Elicitors versus Secondary Metabolite Production in Vitro Using Plant Cell, Tissue and Organ Cultures : Recent Trends and a Sky Eye View Appraisal. Plant Cell Tissue Organ. Cult. PCTOC 2016, 126, 1–18. [Google Scholar] [CrossRef]
- Rivero-Montejo, S.d.J.; Vargas-Hernandez, M.; Torres-Pacheco, I. Nanoparticles as Novel Elicitors to Imsprove Bioactive Compounds in Plants. Agriculture 2021, 11, 134. [Google Scholar] [CrossRef]
- Bhaskar, R.; Spandhana, L.; Xavier, E.; Udayakumaran, G.; Shree, D. Biotic Elicitors : A Boon for the in Vitro Production of Plant Secondary Metabolites. Plant Cell Tissue Organ. Cult. PCTOC 2021, 1–18. [Google Scholar] [CrossRef]
- Jamiołkowska, A. Natural Compounds as Elicitors of Plant Resistance Against Diseases and New Biocontrol Strategies. Agronomy 2020, 10, 173. [Google Scholar] [CrossRef] [Green Version]
- Habibi, P.; Piri, K.; Deljo, A.; Moghadam, Y.A. Increasing Scopolamine Content in Hairy Roots of Atropa Belladonna Using Bioreactor. Braz. Arch. Biol. Technol. 2015, 58, 166–174. [Google Scholar] [CrossRef] [Green Version]
- Krasteva, G.; Georgiev, V.; Pavlov, A. Recent Applications of Plant Cell Culture Technology in Cosmetics and Foods. Eng. Life Sci. 2021, 21, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Espinosa-Leal, C.A.; Puente-Garza, C.A.; García-Lara, S. In Vitro Plant Tissue Culture: Means for Production of Biological Active Compounds. Planta 2018, 248, 1–18. [Google Scholar] [CrossRef]
- Marchev, A.S.; Yordanova, Z.P.; Georgiev, M.I. Green (Cell) Factories for Advanced Production of Plant Secondary Metabolites. Crit. Rev. Biotechnol. 2020, 40, 443–458. [Google Scholar] [CrossRef]
- Reyes-Díaz, M.; Lobos, T.; Cardemil, L.; Nunes-Nesi, A.; Retamales, J.; Jaakola, L.; Alberdi, M.; Ribera-Fonseca, A. Methyl Jasmonate: An Alternative for Improving the Quality and Health Properties of Fresh Fruits. Molecules 2016, 21, 567. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, P.; Rasool, S.; Gul, A.; Sheikh, S.A.; Akram, N.A.; Ashraf, M.; Kazi, A.M.; Gucel, S. Jasmonates: Multifunctional roles in stress tolerance. Front. Plant Sci. 2016, 7, 813. [Google Scholar] [CrossRef] [Green Version]
- Mangas, S.; Bonfill, M.; Osuna, L.; Moyano, E.; Tortoriello, J.; Cusido, R.M.; Teresa Piñol, M.; Palazón, J. The Effect of Methyl Jasmonate on Triterpene and Sterol Metabolisms of Centella Asiatica, Ruscus Aculeatus and Galphimia Glauca Cultured Plants. Phytochemistry 2006, 67, 2041–2049. [Google Scholar] [CrossRef]
- Hajati, J.R.; Payamnoor, V.; Ahmadian Chashmi, N.; Ghasemi Bezdi, K. Improved Accumulation of Betulin and Betulinic Acid in Cell Suspension Culture of Betula Pendula Roth by Abiotic and Biotic Elicitors. Prep. Biochem. Biotechnol. 2018, 48, 867–876. [Google Scholar] [CrossRef]
- Pandey, H.; Pandey, P.; Singh, S.; Gupta, R.; Banerjee, S. Production of Anti-Cancer Triterpene (Betulinic Acid) from Callus Cultures of Different Ocimum Species and Its Elicitation. Protoplasma 2015, 252, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Zhao, S.; Yeo, H.J.; Park, Y.E.; Baska, T.B.; Arasu, M.V.; Al-Dhabi, N.A.; Park, S.U. Comparison of Different Strains of Agrobacterium Rhizogenes for Hairy Root Induction and Betulin and Betulinic Acid Production in Morus Alba. Nat. Prod. Commun. 2017, 12, 479–482. [Google Scholar] [CrossRef] [Green Version]
- Wielanek, M.; Urbanek, H.; Majorowicz, H. Endogenous Hydrolysis of Glucotropaeolin to Benzyl Isothiocyanate in Hairy Root Cultures of Tropaeolum Majus L. Biotechnologia 2004, 1, 210–220. [Google Scholar]
- Kang, S.M.; Jung, H.Y.; Kang, Y.M.; Yun, D.J.; Bahk, J.D.; Yang, J.K.; Choi, M.S. Effects of Methyl Jasmonate and Salicylic Acid on the Production of Tropane Alkaloids and the Expression of PMT and H6H in Adventitious Root Cultures of Scopolia Parviflora. Plant. Sci. 2004, 166, 745–751. [Google Scholar] [CrossRef]
- Yaozu, Z.; Liu, Y.; Zhao, H.; Peng, P.; Tingbao, Z.; Jincao, C. Betulinic Acid Inhibits Glioma Cell Viability by Downregulation of NF-ΚB and Enhancement of Apoptosis. Trop. J. Pharm. Res. 2020, 19, 2545–2551. [Google Scholar] [CrossRef]
- Brentnall, M.; Rodriguez-menocal, L.; de Guevara, R.L.; Cepero, E.; Boise, L.H. Caspase-9, Caspase-3 and Caspase-7 Have Distinct Roles during Intrinsic Apoptosis. BMC Cell Biol. 2013, 14, 32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Butterick, T.A.; Duffy, C.M.; Lee, R.E.; Billington, C.J.; Kotz, C.M.; Nixon, J.P. Use of a Caspase Multiplexing Assay to Determine Apoptosis in a Hypothalamic Cell Model. JoVE 2014, 86, 51305. [Google Scholar] [CrossRef]
- Wani, B.A.; Ramamoorthy, D.; Rather, M.A.; Arumugam, N.; Qazi, A.K.; Majeed, R.; Hamid, A.; Ganie, S.A.; Ganai, B.A.; Anand, R.; et al. Induction of Apoptosis in Human Pancreatic MiaPaCa-2 Cells through the Loss of Mitochondrial Membrane Potential (ΔΨm) by Gentiana Kurroo Root Extract and LC-ESI-MS Analysis of Its Principal Constituents. Phytomedicine 2013, 20, 723–733. [Google Scholar] [CrossRef]
- Goswami, P.; Paul, S.; Banerjee, R.; Kundu, R.; Mukherjee, A. Betulinic Acid Induces DNA Damage and Apoptosis in SiHa Cells. Mutat. Res. Genet. Toxicol. Environ. Mutagenesis 2018, 828, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Sitarek, P.; Kowalczyk, T.; Santangelo, S.; Białas, A.J.; Toma, M.; Wieczfinska, J.; Śliwiński, T.; Skała, E. The Extract of Leonurus Sibiricus Transgenic Roots with AtPAP1 Transcriptional Factor Induces Apoptosis via DNA Damage and Down Regulation of Selected Epigenetic Factors in Human Cancer Cells. Neurochem. Res. 2018, 43, 1363–1370. [Google Scholar] [CrossRef] [Green Version]
- Cheng, X.; Wang, D.; Jiang, L.; Yang, D. DNA topoisomerase I inhibitory alkaloids from Corydalis saxicola. Chem. Biodivers. 2008, 5, 1335–1344. [Google Scholar] [CrossRef]
- Chen, G.; Guo, M. Screening for Natural Inhibitors of Topoisomerases I from Rhamnus Davurica by Affinity Ultrafiltration and High-Performance Liquid Chromatography—Mass Spectrometry. Front. Plant Sci. 2017, 8, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Baikar, S.; Malpathak, N. Secondary Metabolites as DNA Topoisomerase Inhibitors : A New Era towards Designing of Anticancer Drugs. Pharmacogn. Rev. 2020, 4, 12–26. [Google Scholar] [CrossRef] [Green Version]
- Castelli, S.; Coletta, A.; Annessa, I.D.; Fiorani, P.; Tesauro, C.; Desideri, A. Interaction between Natural Compounds and Human Topoisomerase I. Biol. Chem. 2012, 393, 1327–1340. [Google Scholar] [CrossRef]
- Kowalczyk, T.; Sitarek, P.; Toma, M.; Picot, L.; Wielanek, M.; Skała, E.; Śliwiński, T. An Extract of Transgenic Senna Obtusifolia L. Hairy Roots with Overexpression of PgSS1 Gene in Combination with Chemotherapeutic Agent Induces Apoptosis in the Leukemia Cell Line. Biomolecules 2020, 10, 510. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bradford, M.M. A Rapid and Sensitive Method for the Quantitation of Microgram Quantities of Protein Utilizing the Principle of Protein-Dye Binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of Structural Proteins during the Assembly of the Head of Bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Dyballa, N.; Metzger, S. Fast and Sensitive Colloidal Coomassie G-250 Staining for Proteins in Polyacrylamide Gels. J. Vis. Exp. 2009, 30, 1431. [Google Scholar] [CrossRef]
- Pai, S.R.; Nimbalkar, M.S.; Pawar, N.V.; Dixit, G.B. Optimization of extraction techniques and quantification of betulinic acid (BA) by RP-HPLC method from Ancistrocladus heyneanus wall. Ex Grah. Ind. Crops Prod. 2011, 34, 1458–1464. [Google Scholar] [CrossRef]
- Kowalczyk, T.; Sitarek, P.; Skała, E.; Toma, M.; Wielanek, M.; Pytel, D.; Wieczfińska, J.; Szemraj, J.; Śliwiński, T. Induction of Apoptosis by in Vitro and in Vivo Plant Extracts Derived from Menyanthes Trifoliata L. in Human Cancer Cells. Cytotechnology 2019, 71, 165–180. [Google Scholar] [CrossRef] [Green Version]
- Kowalczyk, T.; Merecz-Sadowska, A.; Rijo, P.; Isca, V.M.S.; Picot, L.; Wielanek, M.; Śliwiński, T.; Sitarek, P. Preliminary Phytochemical Analysis and Evaluation of the Biological Activity of Leonotis Nepetifolia (L.) R. Br Transformed Roots Extracts Obtained through Rhizobium Rhizogenes-Mediated Transformation. Cells 2021, 10, 1242. [Google Scholar] [CrossRef]
- Sappal, D.S.; McClendon, A.K.; Fleming, J.A.; Thoroddsen, V.; Connolly, K.; Reimer, C.; Blackman, R.K.; Bulawa, C.E.; Osheroff, N.; Charlton, P.; et al. Biological characterization of MLN944: A potent DNA binding agent. Mol. Cancer Ther. 2004, 3, 47–58. [Google Scholar] [PubMed]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kowalczyk, T.; Sitarek, P.; Merecz-Sadowska, A.; Szyposzyńska, M.; Spławska, A.; Gorniak, L.; Bijak, M.; Śliwiński, T. Methyl Jasmonate Effect on Betulinic Acid Content and Biological Properties of Extract from Senna obtusifolia Transgenic Hairy Roots. Molecules 2021, 26, 6208. https://doi.org/10.3390/molecules26206208
Kowalczyk T, Sitarek P, Merecz-Sadowska A, Szyposzyńska M, Spławska A, Gorniak L, Bijak M, Śliwiński T. Methyl Jasmonate Effect on Betulinic Acid Content and Biological Properties of Extract from Senna obtusifolia Transgenic Hairy Roots. Molecules. 2021; 26(20):6208. https://doi.org/10.3390/molecules26206208
Chicago/Turabian StyleKowalczyk, Tomasz, Przemysław Sitarek, Anna Merecz-Sadowska, Monika Szyposzyńska, Aleksandra Spławska, Leslaw Gorniak, Michał Bijak, and Tomasz Śliwiński. 2021. "Methyl Jasmonate Effect on Betulinic Acid Content and Biological Properties of Extract from Senna obtusifolia Transgenic Hairy Roots" Molecules 26, no. 20: 6208. https://doi.org/10.3390/molecules26206208





