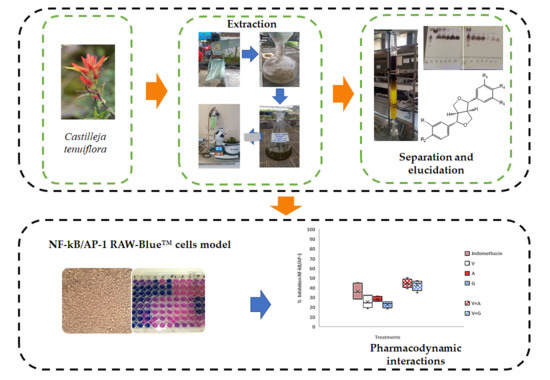
Synergism and Subadditivity of Verbascoside-Lignans and -Iridoids Binary Mixtures Isolated from Castilleja tenuiflora Benth. on NF-κB/AP-1 Inhibition Activity
Abstract
:1. Introduction
2. Results
2.1. Isolation and Identification of Compounds
2.2. EC50 and Emax of the Isolated Compounds
2.3. Pharmacodynamic Screening Using Binary Mixtures
2.4. Pharmacodynamic Interactions
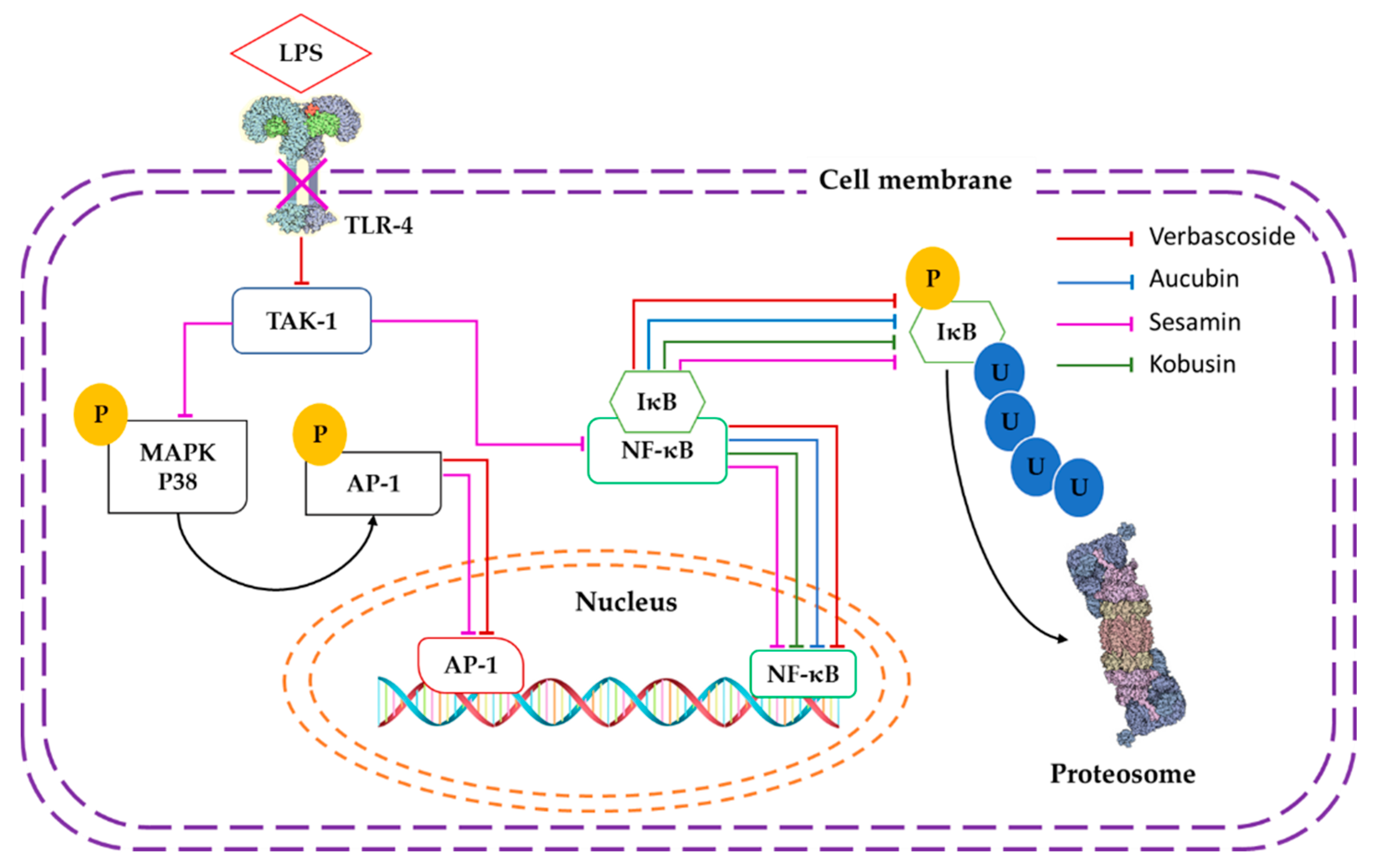
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Extraction and Isolation of Chemical Compounds from Methanolic Extracts
4.3. TLC and HPLC Analysis
4.4. UPLC Analysis
4.5. 1H- and 13C-NMR Experiments
4.6. Raw-Blue™ Cells Culture Experiments
- IA: Inhibition activity (%),
- A: Absorbance of 100% activity,
- B: Absorbance of the treatment.
4.6.1. Cell Viability on Raw-Blue ™ Cells Using the MTT Method
- C: Absorbance of the control,
- T: Treatment absorbance.
4.6.2. Inhibition of NF-κB/AP-1 by the Effective Concentration 50 (EC50) of Compounds Isolated from C. tenuiflora Using Raw-Blue™ Cells
4.6.3. Inhibition of NF-κB/AP-1 in LPS Stimulated Raw-blue ™ Cells by Binary Mixtures of Iridoid Glycosides and Lignans with Verbascoside
4.6.4. Pharmacodynamic Interaction of Binary Mixtures of Iridoids and Lignans with Verbascoside in RAW Blue™ Cells
- OIA: Observed inhibition activity (%),
- TIA: Expected inhibition activity (%).
4.7. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Martínez, M. Catálogo de Nombres Vulgares y Científicos de Plantas Mexicanas, 3rd ed.; Fondo de Cultura Económica: Ciudad de México, Mexico, 1994. [Google Scholar]
- Alonso-Castro, A.J.; Villarreal, M.L.; Salazar-Olivo, L.A.; Gomez-Sánchez, M.; Dominguez, F.; Garcia-Carranca, A. Mexican medicinal plants used for cancer treatment: Pharmacological, phytochemical and ethnobotanical studies. J. Ethnopharmacol. 2011, 133, 945–972. [Google Scholar] [CrossRef] [PubMed]
- Carrillo-Ocampo, D.; Bazaldúa-Gómez, S.; Bonilla-Barbosa, J.R.; Aburto-Amar, R.; Rodríguez-López, V. Anti-inflammatory activity of iridoids and verbascoside isolated from Castilleja tenuiflora. Molecules 2013, 18, 12109–12118. [Google Scholar] [CrossRef] [PubMed]
- Herrera-Ruiz, M.; López-Rodríguez, R.; Trejo-Tapia, G.; Dominguez-Mendoza, B.E.; González-Cortazar, M.; Tortoriello, J.; Zamilpa, A. A new furofuran lignan diglycoside and other secondary metabolites from the antidepressant extract of Castilleja tenuiflora Benth. Molecules 2015, 20, 13127–13143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gómez-Aguirre, Y.A.; Zamilpa, A.; González-Cortazar, M.; Trejo-Tapia, G. Adventitious root cultures of Castilleja tenuiflora Benth. as a source of phenylethanoid glycosides. Ind. Crops Prod. 2012, 36, 188–195. [Google Scholar]
- Moreno-Escobar, J.A.; Bazaldúa, S.; Villarreal, M.L.; Bonilla-Barbosa, J.R.; Mendoza, S.; Rodríguez-López, V. Cytotoxic and antioxidant activities of selected Lamiales species from Mexico. Pharm. Biol. 2011, 49, 1243–1248. [Google Scholar] [CrossRef]
- López-Laredo, A.; Gómez-Aguirre, Y.; Medina-Pérez, V.; Salcedo-Morales, G.; Sepúlveda-Jiménez, G.; Trejo-Tapia, G. Variation in antioxidant properties and phenolics concentration in different organs of wild growing and greenhouse cultivated Castilleja tenuiflora Benth. Acta Physiol. Plant. 2012, 34, 2435–2442. [Google Scholar] [CrossRef]
- Sanchez, P.M.; Villarreal, M.L.; Herrera-Ruiz, M.; Zamilpa, A.; Jiménez-Ferrer, E.; Trejo-Tapia, G. In vivo anti-inflammatory and anti-ulcerogenic activities of extracts from wild growing and in vitro plants of Castilleja tenuiflora Benth. (Orobanchaceae). J. Ethnopharmacol. 2013, 150, 1032–1037. [Google Scholar] [CrossRef]
- López-Rodríguez, R.; Herrera-Ruiz, M.; Trejo-Tapia, G.; Domínguez-Mendoza, B.E.; González-Cortazar, M.; Zamilpa, A. In vivo gastroprotective and antidepressant effects of iridoids, verbascoside and tenuifloroside from Castilleja tenuiflora Benth. Molecules 2019, 24, 1292. [Google Scholar]
- Seyfi, D.; Behzad, S.B.; Nabiuni, M.; Parivar, K.; Tahmaseb, M.; Amini, E. Verbascoside attenuates Rac-1 and HIF-1α signaling cascade in colorectal cancer cells. Med. Chem. 2018, 18, 2149–2155. [Google Scholar] [CrossRef]
- Kartini, S.P.; Piyaviriyakul, S.; Thongpraditchote, S.; Siripong, P.; Vallisuta, O. Effects of plantago major extracts and its chemical compounds on proliferation of cancer cells and cytokines production of lipopolysaccharide-activated THP-1 macrophages. Pharmacogn. Mag. 2017, 13, 393–399. [Google Scholar] [CrossRef] [Green Version]
- Niu, J.; Straubinger, R.M.; Mager, D.E. Pharmacodynamic Drug-Drug Interactions. Clin. Pharmacol. Ther. 2019, 105, 1395–1406. [Google Scholar] [CrossRef] [PubMed]
- Cascorbi, I. Drug interactions—Principles, examples and clinical consequences. Deutsches Arzteblatt Int. 2012, 109, 546–556. [Google Scholar]
- Einbond, L.S.; Shimizu, M.; Ma, H.; Wu, H.; Goldsberry, S.; Sicular, S.; Panjikaran, M.; Genovese, G.; Cruz, E. Actein inhibits the Na+-K+-ATPase and enhances the growth inhibitory effect of digitoxin on human breast cancer cells. Biochem. Biophys. Res. Commun. 2008, 375, 608–613. [Google Scholar] [CrossRef] [PubMed]
- Attia, Y.M.; El-Kersh, D.M.; Wagdy, H.A.; Elmazar, M.M. Verbascoside: Identification, quantification, and potential sensitization of colorectal cancer cells to 5-FU by targeting PI3K/AKT pathway. Sci. Rep. 2018, 8, 16939. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luszczki, J.J.; Ratnaraj, N.; Patsalos, P.N.; Czuczwar, S.J. Pharmacodynamic and/or pharmacokinetic characteristics of interactions between loreclezole and four conventional antiepileptic drugs in pentylenetetrazole-induced seizures in mice: An isobolographic analysis. Epilepsy Behav. 2005, 7, 639–651. [Google Scholar] [CrossRef]
- Greten, F.R.; Grivennikov, S.I. Inflammation and Cancer: Triggers, Mechanisms, and Consequences. Immunity 2019, 51, 27–41. [Google Scholar] [CrossRef]
- Kinney, J.W.; Bemiller, S.M.; Murtishaw, A.S.; Leisgang, A.M.; Salazar, A.M.; Lamb, B.T. Inflammation as a central mechanism in Alzheimer’s disease. Alzheimers Dement. 2018, 4, 575–590. [Google Scholar] [CrossRef]
- Bäck, M.; Yurdagul, A.; Tabas, I.; Öörni, K.; Kovanen, P.T. Inflammation and its resolution in atherosclerosis: Mediators and therapeutic opportunities. Nat. Rev. Cardiol. 2019, 16, 389–406. [Google Scholar] [CrossRef]
- Quinton, L.J.; Walkey, A.J.; Mizgerd, J.P. Integrative physiology of pneumonia. Physiol. Rev. 2018, 98, 1417–1464. [Google Scholar] [CrossRef]
- Guo, Q.; Wang, Y.; Xu, D.; Nossent, J.; Pavlos, N.J.; Xu, J. Rheumatoid arthritis: Pathological mechanisms and modern pharmacologic therapies. J. Bone Res. 2018, 6, 15. [Google Scholar] [CrossRef]
- Tsalamandris, S.; Antonopoulos, A.S.; Oikonomou, E.; Papamikroulis, G.A.; Vogiatzi, G.; Papaioannou, S.; Deftereos, S.; Tousoulis, D. The role of inflammation in diabetes: Current concepts and future perspectives. Eur. Cardiol. 2019, 14, 50–59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, L.; Deng, H.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget 2017, 9, 7204–7218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mu, H.X.; Lin, C.Y.; Huang, L.F.; Yang, D.J.; Lu, A.P.; Han, Q.B.; Bian, Z.X. A novel coumarin, (+)-3′-angeloxyloxy-4′-keto-3′,4′-dihydroseselin, isolated from Bupleurum malconense (Chaihu) inhibited NF-κB activity. Chin. Med. 2016, 11, 5–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iitsuka, H.; Koizumi, K.; Suzaki, M.; Otsuka, Y.; Jo, M.; Shibahara, N. Immunostimulatory effects of cell wall-based nanoparticles in boiled Glycyrrhizae radix water extracts involves TLR4. Biomed. Rep. 2020, 12, 303–308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pesce, M.; Franceschelli, S.; Ferrone, A.; De Lutiis, M.A.; Patruno, A.; Grilli, A.; Felaco, M.; Speranza, L. Verbascoside down-regulates some pro-inflammatory signal transduction pathways by increasing the activity of tyrosine phosphatase SHP-1 in the U937 cell line. J. Mol. Cell Med. 2015, 19, 1548–1556. [Google Scholar] [CrossRef]
- Ramírez-Cisneros, M.A.; Rios, M.; Aguilar-Guadarrama, A.; Rao, P.; Aburto-Amar, R.; Rodríguez-López, V. In vitro COX-1 and COX-2 enzyme inhibitory activities of iridoids from Penstemon barbatus, Castilleja tenuiflora, Cresentia alata and Vitex molli. Bioorg. Med. Chem. Lett. 2015, 25, 4505–4508. [Google Scholar]
- Ersoz, T.; Berkman, M.Z.; Tasdemir, D.; Ireland, C.M.; Calis, I. An iridoid glucoside from Euphrasia pectinata. J. Nat. Prod. 2000, 63, 1449–1450. [Google Scholar] [CrossRef]
- Calle, J. Aislamiento, purificación e identificación de sesamina a partir de lodos de microfiltrado en la fabricación del aceite virgen de Sesamum indicum L. (ajonjolí). Rev. Colomb. Cienc. Quím. Farm. 2007, 36, 5–10. [Google Scholar]
- Chang, W.; Kim, K.; Lee, I.K.; Choi, S.; Lee, K. Phytochemical constituents of Geranum eriostemon. Nat. Prod. Sci. 2009, 15, 151–155. [Google Scholar]
- Hao, L.; Zhi, S.; Da-Guang, L.; Tian-Yi, Z.; Feng, L.; Kai, Z.; Kui, L.; Liang, Y.; Jing, H.; Jian-Ping, L. Anticonvulsant and sedative effects of eudesmin isolated from Acorus tatarinowii on mice and rats. Phytother. Res. 2015, 29, 996–1003. [Google Scholar]
- Miyazawa, M.; Ishikawa, Y.; Kasahara, H.; Yamanaka, J.; Kameoka, H. An insect growth inhibitory lignan from flower buds of Magnolia fargesii. Phytochemistry 1994, 35, 611–613. [Google Scholar] [CrossRef]
- Jain, H.; Dhingra, N.; Narsinghani, T.; Sharma, R. Insights into the mechanism of natural terpenoids as NF-κB inhibitors: An overview on their anticancer potential. Exp. Oncol. 2016, 38, 158–168. [Google Scholar] [CrossRef]
- Shi, Q.; Cao, J.; Fang, L.; Zhao, H.; Liu, Z.; Ran, J.; Zheng, X.; Li, X.; Zhou, Y.; Ge, D.; et al. Geniposide suppresses LPS-induced nitric oxide, PGE2 and inflammatory cytokine by downregulating NF-κB, MAPK and AP-1 signaling pathways in macrophages. Int. Immunopharmacol. 2014, 20, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Du, Z.; Wang, W.; Song, M.; Sanidad, K.; Sukamtoh, E.; Zheng, J.; Tian, L.; Xiao, H.; Liu, Z.; et al. Structure-activity relationship of curcumin: Role of the methoxy group in anti-inflammatory and anticolitis effects of curcumin. J. Agric. Food Chem. 2017, 65, 4509–4515. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Lee, K.S.; Yi, S.H.; Kook, S.H.; Lee, J.C. Acteoside suppresses RANKL-mediated osteoclastogenesis by inhibiting c-Fos induction and NF-κB pathway and attenuating ROS production. PLoS ONE 2013, 8, 80873. [Google Scholar] [CrossRef] [PubMed]
- Sipahi, H.; Gostner, J.M.; Becker, K.; Charehsaz, M.; Kirmizibekmez, H.; Schennach, H.; Aydin, A.; Fuchs, D. Bioactivites of two common polyphenolic compounds: Verbascoside and catechin. Pharm. Biol. 2016, 54, 712–719. [Google Scholar] [CrossRef]
- Khullar, M.; Sharma, A.; Wani, A.; Sharma, N.; Sharma, N.; Chandan, B.K.; Kumar, A.; Ahmed, Z. Acteoside ameliorates inflammatory responses through NF-kB pathway in alcohol induced hepatic damage. Int. Immunopharmacol. 2019, 69, 109–117. [Google Scholar] [CrossRef]
- Jeong, H.J.; Koo, H.N.; Na, H.J.; Kim, M.S.; Hong, S.H.; Eom, J.W.; Kim, K.S.; Shin, T.Y.; Kim, H.M. Inhibition of TNF-α and Il-6 production by aucubin through blockade of NF-κB activation in RBL-2H3 mast cells. Cytokine 2002, 18, 252–259. [Google Scholar] [CrossRef]
- Kim, J.Y.; Lim, H.J.; Lee, D.Y.; Kim, J.S.; Kim, D.H.; Lee, H.J.; Kim, H.D.; Jeon, R.; Ryu, J.H. In vitro anti-inflammatory activity of lignans isolated from Magnolia fargesii. Bioorg. Med. Chem. Lett. 2009, 19, 937–940. [Google Scholar] [CrossRef]
- Udomruk, S.; Kaewmool, C.; Pothacharoen, P.; Phitak, T.; Kongtawelert, P. Sesamin suppresses LPS-induced microglial activation via regulation of TLR4 expression. J. Funct. Food 2018, 49, 32–43. [Google Scholar] [CrossRef]
- Mason, P.E.; Neilson, G.W.; Enderby, J.E.; Saboungi, M.L.; Brady, J.W. Structure of aqueous glucose solutions as determined by neutron diffraction with isotopic substitution experiments and molecular dynamics calculations. J. Phys. Chem. B 2005, 109, 13104–13111. [Google Scholar] [CrossRef] [PubMed]
- Asensio, J.L.; Ardá, A.; Cañada, F.J.; Jiménez-Barbero, J. Carbohydrate—Aromatic interactions. Acc. Chem. Res. 2013, 46, 946–954. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spiwok, V. CH/π Interactions in Carbohydrate Recognition. Molecules 2017, 22, 1038. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Méndez-Martínez, M.; Trejo-Moreno, C.; Maldonado-Mejía, L.; Ezquivel-Guadarrama, F.; Pedraza-Chaverri, J.; Zamilpa, A.; Medina-Campos, O.; Alarcón-Aguilar, F.; Almanza-Pérez, J.; Contreras-Nuñez, E.; et al. Effect of Cucumis sativus on dysfunctional 3T3-L1 adipocytes. Sci. Rep. 2019, 9, 13372. [Google Scholar]
- Medrano-Jiménez, E.; Jiménez-Ferrer, I.; Pedraza-Escalona, M.; Ramírez-Serrano, C.; Álvarez-Arellano, L.; Cortés-Mendoza, J.; Herrera-Ruiz, M.; Jiménez-Ferrer, E.; Zamilpa, A.; Tortoriello, J.; et al. Malva parviflora extract ameliorates the deleterious effects of a high fat diet on the cognitive deficit in a mouse model of Alzheimer’s disease by restoring microglial function via a PPAR-γ-dependent mechanism. J. Neuroinflamm. 2019, 16, 143. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Frum, Y.; Viljoen, A.M.; Van Heerden, F.R. Verbascoside and luteolin-5-O-β-D-glucoside isolated from Halleria lucida L. exhibit antagonistic anti-oxidant properties in vitro. S. Afr. J. Bot. 2007, 73, 583–587. [Google Scholar] [CrossRef] [Green Version]


| Compound | Emax (% Inhibition) | EC50 (µM) |
|---|---|---|
| Verbascoside | 55.55 | 10.08 |
| Aucubin | 40.00 | 0.08 |
| Geniposide | 57.47 | 0.01 |
| Tenuifloroside | 52.91 | 0.11 |
| Magnolin | 60.97 | 3.05 |
| Eudesmin | 56.82 | 0.006 |
| Kobusin | 45.45 | 2.37 |
| Sesamin | 52.63 | 0.01 |
| Mixture | Expected NF-κB/AP-1 Inhibition (%) | Observed NF-κB/AP-1 Inhibition (%) | Inhibition Ratio |
|---|---|---|---|
| Indomethacin | 36.31 ± 9.15 a | ||
| Verbascoside-aucubin | 53.65 | 46.16 ± 3.67 a,b | 0.84 |
| Verbascoside-geniposide | 48.58 | 43.04 ± 5.59 a,b | 0.89 |
| Verbascoside-tenuifloroside | 60.18 | 37.99 ± 7.70 a | 0.63 |
| Verbascoside-magnolin | 52.95 | 39.33 ± 7.59 a,b | 0.74 |
| Verbascoside-eudesmin | 52.70 | 51.01 ± 7.99 b | 0.97 |
| Verbascoside-kobusin | 55.60 | 63.06 ± 5.90 b,c | 1.13 |
| Verbascoside-sesamin | 44.94 | 47.40 ± 16.28 b,c | 1.05 |
| Binary Mixture | Expected NF-κB/AP-1 Inhibition (%) | Observed NF-κB/AP-1 Inhibition (%) | Inhibition Ratio |
|---|---|---|---|
| V-A proportion Indomethacin | 33.22 ± 6.53 a | ||
| 1.00/0:00 | 26.54 | 26.10 ± 4.24 a | 1.05 |
| 0.75/0.25 | 27.53 | 34.82 ± 8.37 a | 1.32 |
| 0.50/0.50 | 28.52 | 34.21 ± 4.15 a | 1.21 |
| 0.25/0.75 | 29.52 | 25.47 ± 10.65 a | 0.86 |
| 0.00/1.00 | 29.84 | 30.42 ± 4.20 a | 1.02 |
| V-K proportion | |||
| Indomethacin | 34.07 ± 9.13 b,c | ||
| 1.00/0:00 | 26.54 | 22.77 ± 5.57 a,b | 0.91 |
| 0.75/0.25 | 27.46 | 33.93 ± 4.07 b,c | 1.24 |
| 0.50/0.50 | 28.38 | 43.23 ± 5.17 c | 1.56 |
| 0.25/0.75 | 29.29 | 36.52 ± 4.60 b,c | 1.26 |
| 0.00/1.00 | 30.21 | 29.10 ± 6.63 a | 0.95 |
| V-T proportion | |||
| Indomethacin | 30.77 ± 4.91 b,c | ||
| 1.00/0:00 | 26.54 | 26.45 ± 2.47 b | 0.93 |
| 0.75/0.25 | 28.60 | 20.59 ± 2.70 ª | 0.72 |
| 0.50/0.50 | 30.66 | 14.16 ± 6.07 ª | 0.46 |
| 0.25/0.75 | 32.72 | 28.66 ± 2.82 b | 0.91 |
| 0.00/1.00 | 34.78 | 37.98 ± 5.97 c | 1.07 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arango-De la Pava, L.D.; Zamilpa, A.; Trejo-Espino, J.L.; Domínguez-Mendoza, B.E.; Jiménez-Ferrer, E.; Pérez-Martínez, L.; Trejo-Tapia, G. Synergism and Subadditivity of Verbascoside-Lignans and -Iridoids Binary Mixtures Isolated from Castilleja tenuiflora Benth. on NF-κB/AP-1 Inhibition Activity. Molecules 2021, 26, 547. https://doi.org/10.3390/molecules26030547
Arango-De la Pava LD, Zamilpa A, Trejo-Espino JL, Domínguez-Mendoza BE, Jiménez-Ferrer E, Pérez-Martínez L, Trejo-Tapia G. Synergism and Subadditivity of Verbascoside-Lignans and -Iridoids Binary Mixtures Isolated from Castilleja tenuiflora Benth. on NF-κB/AP-1 Inhibition Activity. Molecules. 2021; 26(3):547. https://doi.org/10.3390/molecules26030547
Chicago/Turabian StyleArango-De la Pava, Luis David, Alejandro Zamilpa, José Luis Trejo-Espino, Blanca Eda Domínguez-Mendoza, Enrique Jiménez-Ferrer, Leonor Pérez-Martínez, and Gabriela Trejo-Tapia. 2021. "Synergism and Subadditivity of Verbascoside-Lignans and -Iridoids Binary Mixtures Isolated from Castilleja tenuiflora Benth. on NF-κB/AP-1 Inhibition Activity" Molecules 26, no. 3: 547. https://doi.org/10.3390/molecules26030547