1. Introduction
The inflammasome is one of the pattern recognition receptors responding to pathogen-associated molecular patterns derived from invading pathogens [
1,
2]. The inflammasome is a multi-protein complex that processes the cleavage of pro-interleukin (IL)-1β and pro-IL-18 to mature IL-1β and IL-18 and triggers inflammatory cell death, i.e., pyroptosis [
3]. The NLRP3 (NOD-, LRR-, and pyrin domain-containing protein 3) inflammasome consists of NLRP3, apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC), and pro-caspase-1. NLRP3, acting as a sensor, is composed of an amino-terminal pyrin domain (PYD), a NACHT domain, and a carboxy-terminal leucine-rich repeat (LRR) domain. ASC is composed of an amino-terminal PYD and a carboxy-terminal caspase recruitment domain (CARD). Caspase-1 has an amino-terminal CARD consisting of the catalytic domains p20 and p10. NLRP3 interacts with ASC through PYD–PYD association, while the interaction between ASC and pro-caspase-1 takes place via CARD–CARD interaction [
4,
5]. Activation of caspase-1 is achieved by proximity-induced autocatalytic activation, leading to the cleavage of the pro-forms of IL-1β and IL-18 to biologically active forms [
6]. In addition, active caspase-1 induces the degradation of gasdermin D (GSDMD) to form pores consisting of the N-terminal domain of GSDMD in the plasma membrane, thereby triggering pyroptotic cell death [
7]. The NLRP3 inflammasome is also activated by damage-associated molecular patterns including ATP, K
+ ionophores, bacterial toxins, and particulate matters, in addition to infectious bacteria [
8,
9]. The activation of the NLRP3 inflammasome plays a critical role in the regulation of host immune responses and is closely linked to the pathogenesis of inflammatory diseases such as autoinflammatory diseases, atherosclerosis, non-alcoholic fatty liver disease, diabetes, and gout [
10]. Therefore, the pharmacological inhibition of NLRP3 activation is thought to exert potent therapeutic effects on a wide variety of inflammatory diseases [
11].
Gout is a painful type of inflammatory arthritis, whose incidence has been increasing in modern society due to changes in human living conditions, lifestyle, and eating habits [
12]. The medical treatment of gout includes nonsteroidal anti-inflammatory drugs (NSAIDs), steroids, colchicine, and uric acid-lowering medications. Gout patients may experience relapsing gout flares resulting in the erosion and destruction of a joint. Gout is triggered by the accumulation of monosodium urate (MSU) crystals, the final metabolite of purines, in the big toe or other joints, causing inflammatory arthritis with swelling and intense pain, called gout attacks. It is now well established that MSU crystals induce the activation of the NLRP3 inflammasome, resulting in the production of pro-inflammatory cytokines such as IL-1β and IL-18 and subsequent inflammatory cell death [
13]. Therefore, the NLRP3 inflammasome could be a potential target for gout therapy.
We intended to search and develop a new pharmacological inhibitor of the NLRP3 inflammasome for the prevention of gout inflammation. Loganin is an iridoid glycoside compound (
Figure 1A) with anti-inflammatory activity [
14]. However, it has not been examined whether loganin can regulate the activation of the NLRP3 inflammasome and be effective to treat gout. Therefore, we investigated whether loganin has suppressive effects on the NLRP3 inflammasome activation induced by MSU crystals and may consequently prevent gout inflammation in mouse primary macrophages and mouse acute gout models.
3. Discussion
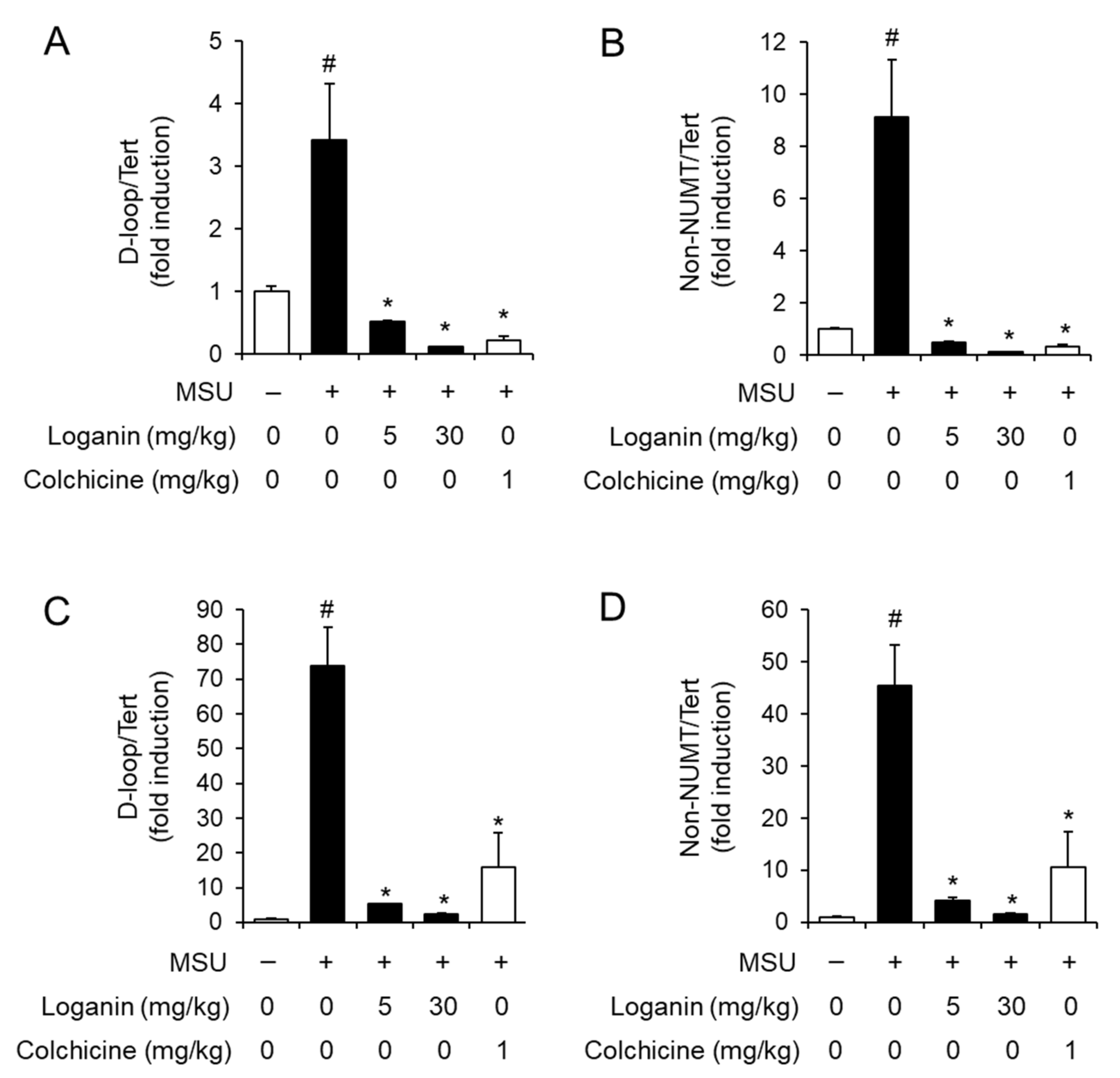
Gout is a type of inflammatory arthritis that is induced by the accumulation of MSU crystals in joints. MSU crystals induce the activation of the NLRP3 inflammasome and subsequent release of mature IL-1β, contributing to the development and progress of gouty arthritis. Our results show that oral administration of loganin prevented gout inflammation induced by MSU crystals via inhibition of NLRP3 inflammasome activation. Loganin reduced the production of mature IL-1β and IL-18 in macrophages and gout tissues stimulated with MSU crystals. The inhibitory effects of loganin on NLRP3 inflammasome activation were consistently observed in both cultured cell system and in vivo animal models.
MSU crystals are phagocytosed by cells, causing lysosomal damage that activates the NLRP3 inflammasome [
13]. Lysosomal damage increases extracellular K
+ levels, cathepsin B release, and production of ROS, which induce NLRP3 inflammasome activation [
13]. While lysosomal damage is the prerequisite for MSU crystals-induced NLRP3 inflammasome, the precise mechanism linking lysosome rupture to NLRP3 inflammasome activation has not been completely understood. Mitochondrial dysfunction has been proposed to play a critical role in NLRP3 inflammasome activation in response to LPS and ATP [
15]. Mitochondrial dysfunction is associated with generation of mitochondrial ROS, decrease in mitochondrial membrane potential, and release of mitochondrial DNA. De novo synthesis of mitochondrial DNA induced by a priming step promotes NLRP3 inflammasome activation [
16]. Our results show that MSU crystals increased the levels of mitochondrial DNA in gouty tissues of mice. Similarly, MSU crystals decreased mitochondrial membrane potential and increased mitochondrial ROS levels in macrophages. These indicate that MSU crystals induce mitochondrial stress in macrophages and inflamed gouty tissues, suggesting the involvement of mitochondria dysfunction and consequent NLRP3 inflammasome activation in the pathogenesis of gout. In contrast, loganin exhibited protective effects on mitochondrial dysfunction by increasing mitochondrial membrane potential and decreasing mitochondrial ROS levels in macrophages. In addition, loganin reduced the levels of de novo synthesized mitochondrial DNA in both macrophages and gouty tissues. A recent study shows that ATP induces mitochondrial dysfunction, leading to cytosolic release of oxidized mitochondrial DNA to bind to and to activate the NLRP3 inflammasome [
17]. Disruption of autophagy induces an increase of dysfunctional mitochondria and the cytosolic release of mitochondrial DNA in response to LPS and ATP in macrophages [
15]. This suggests that loganin may regulate the upstream event of mitochondrial dysfunction such as autophagy, resulting in blockade of NLRP3 inflammasome activation. The effects of loganin on autophagy need to be elucidated in future studies.
Many phytochemicals and small-molecule inhibitors of the NLRP3 inflammasome have been reported [
10]. We intend to search for new small molecule inhibitors with different inhibitory mechanisms and targets to provide eventually more effective strategies for drug development. Consistently with the protective effects of loganin on mitochondrial dysfunction, epigallocatechin-3-gallate (EGCG) suppressed the de novo synthesis of mitochondrial DNA, which was correlated with its inhibitory effects on NLRP3 inflammasome activation [
18]. This suggests that inhibition of mitochondrial stress by small-molecule inhibitors could be an efficient strategy to suppress the activation of the NLRP3 inflammasome. Mitochondrial dysfunction in connection with NLRP3 inflammasome activation has been reported to play a role in inflammatory diseases such as diabetes, atherosclerosis, neurological disorders, cardiovascular disease, and kidney disease [
19,
20]. Therefore, the beneficial effects of loganin on mitochondrial dysfunction could be applied to other NLRP3-related diseases.
In general, effective concentrations and sensitivity of chemicals could be variable in different experimental conditions, depending, for example, on cell type, treatment time, stimulators, and biomarkers. Cheng et al. showed the inhibitory effects of loganin at 1 μM on high-glucose-induced NLRP3 inflammasome activation [
21]. Cheng et al. performed these experiments in a rat Schwann cell line treating with loganin for 48 h, while we used primary macrophages isolated from mouse bone marrow and treated with loganin for maximum 7 h. Cheng et al. used a high glucose concentration to induce NLRP3 inflammasome activation, whereas we used MSU crystals as an NLRP3 activator. Moreover, Cheng et al. determined the inhibitory effects of loganin on the priming step involving NF-κB activation induced by high glucose levels and showed that loganin decreased the mRNA expression of
Nlrp3,
Asc,
Caspase-1, and
Il-1β, which were upregulated upon NF-κB activation. In contrast, we intended to investigate the effect of loganin on the activation step induced by MSU crystals, not on the priming step induced by LPS, by adding loganin after washing out LPS. Although we used loganin up to 100 μM, its inhibitory effects began at a concentration of 25 μM, as shown in
Figure 1B,C, and
Figure 5A,D and were evident at a concentration of 50 μM. Other studies used similar concentration ranges of loganin, as loganin reduced the production of pro-inflammatory cytokines, TNF-α, IL-6, and monocyte chemoattractant protein-1 (MCP-1) and the activation of NF-κB in apoCIII-stimulated mouse 3T3L1 adipocytes at 16 μM–128 μM [
14]. Loganin at 100 μM was effective to inhibit rat renal mesangial cell proliferation induced by advanced glycation end products [
22].
Since gout is a recurrent inflammatory disease, gout patients suffer relapsing gout flares, despite currently used medications are effective to reduce inflammation and pain. Thus, it is necessary to develop more effective therapeutic strategies for preventing gout inflammation and relapsing gout flares. Inhibitors of IL-1 such as anakinra, rilonacept, and canakinumab have been investigated. However, their use is limited by their high cost, inconvenient treatment routes, and side effects [
23,
24]. Therefore, an effective oral administration of loganin would confer additional advantages, including accessibility, cost, and convenience. To the best of our knowledge, the current in vivo experimental gout models induce an acute gout attack by injecting MSU crystals into air pouches, footpads, ankle joints, or knee joints. Accordingly, we used two acute gout models, an air pouch model and a footpad gout model to study the effects of loganin on gout inflammation. Nevertheless, information on the long-term effects of loganin would be valuable to evaluate the application of loganin for the clinical treatment of gout. Since gout is characterized by recurrent attacks, a long-term model of gout needs to be established, so that the effectiveness of treatments could be evaluated for relapsing gout attacks. The long-term effects of loganin on recurrent gout attacks need to be further investigated in future studies.
4. Materials and Methods
4.1. Animals
C57BL/6 mice (male) were obtained from Raon Bio (Seoul, Korea) and were acclimated in specific pathogen-free conditions in an animal facility for at least one week before experimentation. The mice were housed in a room controlled for optimal temperature (23 ± 3 °C) and relative humidity (40–60%) under specific pathogen-free condition. Animal care and the experimental protocols were carried out in accordance with the guidelines of the Institutional Animal Care and Use Committee (IACUC) of the Catholic University of Korea (permission #, 2014-006, 2017-015). Mice of individual experimental groups in each experiment were of similar age and weight and were randomly allocated to treatment groups. Investigators were blinded for the treatment of mice in all experiments.
4.2. Cell Culture
BMDMs were prepared after bone marrow was isolated from C57BL/6 mice as described previously [
25]. Macrophages were cultured in Dulbecco’s modified eagle medium containing 10% (
v/
v) fetal bovine serum (Corning, Somervile, MA, USA), 10,000 units/mL penicillin, and 10,000 μg/mL streptomycin (Thermo Fisher Scientific, Waltham, MA, USA).
4.3. Reagents
Purified lipopolysaccharides (LPS) from Escherichia coli were obtained from List Biological Laboratory (Santa Clara, CA, USA) and dissolved in endotoxin-free water. Loganin (purity: ≥97.0%) was purchased from Sigma-Aldrich (St. Louis, MI, USA). Monosodium urate crystals were purchased from Invivogen (San Diego, CA, USA). The antibody for caspase-1 was obtained from Santa Cruz Biotechnology (Dallas, TX, USA). The antibody for IL-1β was obtained from R&D Systems (Minneapolis, MN, USA).
4.4. Analysis of Inflammasome Activation
BMDMs were seeded at 2 × 10
6 cells/well in 6-well plates for immunoblot assays and 4 × 10
5 cells/well in 96-well plates for ELISAs. BMDMs were primed with LPS for 4 h. To exclude the effect of loganin on the LPS priming step, loganin was added after washing out LPS with phosphate-buffered saline (PBS). After treatment with loganin, the cells were further stimulated with MSU crystals in a serum-free medium. The cell supernatants and lysates were processed for immunoblot assays as previously described [
26].
4.5. Enzyme-Linked Immunosorbent Assays
IL-1β levels were determined using a DuoSet enzyme-linked immunosorbent assay (ELISA) kit (R&D system, Minneapolis, MN, USA) according to the manufacturer’s instructions. IL-18 levels were determined using an ELISA kit (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions.
4.6. Confocal Microscopy Analysis for ASC Specks
This was performed as described previously [
27]. Briefly, BMDMs were fixed with methanol for 30 min, blocked with 1% bovine serum albumin for 30 min, and incubated with an anti-ASC antibody (Santa Cruz Biotechnology Inc., Dallas, TX, USA) at 4 °C overnight. The cells were further incubated with an anti-rabbit IgG-FITC antibody (Invitrogen, Carlsbad, CA, USA) and co-stained with 4′,6-diamidino-2-phenylindole (DAPI, 1 μg/mL; Invitrogen) for nuclei visualization. Samples were examined with an LSM710 laser scanning confocal microscope (Carl Zeiss, Oberkochen, Germany). Images were obtained and analyzed with ZEN2011 software (Carl Zeiss, Oberkochen, Germany).
4.7. Cell Viability Assay
Cell viability was analyzed by MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay. After BMDMs were treated with loganin for 7 h, MTT (5 mg/mL; Cas# 298-93-1, Merck, Kenilworth, NJ, USA) was added to each well and incubated at 37 °C for 4 h. Media was removed and cells were dissolved in DMSO. The optical density was measured at 540 nm using a plate reader (MAGPIX®, Luminex Corporation, Austin, TX, USA).
4.8. Air Pouch Inflammation Model
An air pouch was formed in the back of C57BL/6 mice (8 weeks old) by subcutaneous injection of sterile air, as previously described [
28]. On day 4, loganin (5 or 30 mg/kg), colchicine (1 mg/kg), or vehicle (0.02% DMSO) in 0.2 mL sterilized water was orally administered to mice. After 1 h, MSU crystals (3 mg/1 mL of sterile, endotoxin-free PBS) or PBS were injected into the pouch. Six hours later, the mice were anesthetized, and the air pouch lavages were collected with 2 mL PBS containing 5 mM EDTA and processed for ELISAs, immunoblot assays, and determination of MPO activity.
4.9. Mouse Foot Gout Model
C57BL/6 mice (8 weeks old) were orally administered loganin (5 or 30 mg/kg) colchicine (1 mg/kg), or vehicle (0.02% DMSO) in 0.2 mL sterilized water. After 1 h, MSU crystals (2 mg/0.1 mL of sterile, endotoxin-free PBS) or PBS were administered via subcutaneous injection on plantar surface of the hind foot [
28]. Changes in foot thickness were measured for 24 h. The foot tissues were homogenized in RIPA buffer (50 mM Tris-HCl, pH 7.4, 1% NP-40, 0.25% sodium deoxycholate, 150 mM NaCl, 1 mM EGTA, 1 mM PMSF, 1 mM Na
3VO
4, 10 μg/mL leupeptin), and the homogenates were centrifuged at 12,000×
g for 10 min. The supernatants were used for MPO assays, ELISAs, and immunoblot assays.
4.10. Myeloperoxidase Activity Assay
The air pouch lavages and the supernatants of foot tissue homogenates were prepared as described in
Section 4.8 and
Section 4.9. MPO, a representative marker for the activation and infiltration of neutrophils, was measured with a MPO Colorimetric activity assay kit (Catalog# K744, Bio Vision, Milpitas, CA, USA) according to the manufacturer’s protocol.
4.11. Quantitative Real-Time Polymerase Chain Reaction Analysis
Total DNA was isolated with the G-spin
TM DNA Mini Kit (iNtRON Biotechnology, Seongnam, Korea) according to the manufacturer’s instruction. Quantitative real-time polymerase chain reaction was performed as previously described [
18]. Mitochondrial DNA was quantified by qPCR using primers specific for the mitochondrial D-loop region or not inserted in nuclear DNA (non-NUMT). Nuclear DNA encoding
Tert was used for normalization. Primer sequences were as follows: D-loop F: 5′-AATCTACCATCCTCCGTGAAACC-3′, D-loop R: 5′-TCAGTTTAGCTACCCCCAAGTTTAA-3′; non-NUMT F: 5′-CTAGAAACCCCGAAACCAAA-3′, non-NUMT R: 5′-CCAGCTATCACCAAGCTCGT-3′;
Tert F: 5′-CTAGCTCATGTGTCAAGACCCTCTT-3′,
Tert R: 5′-GCCAGCACGTTTCTCTCGTT-3′. Specificity of the amplified PCR products was assessed by melting curve analysis.
4.12. Measurement of Mitochondrial Membrane Potential
The MitoProbeTM JC-1 assay kit (Thermo Fisher Scientific, Waltham, MA, USA) was used for the measurement of mitochondrial membrane potential. After LPS-primed BMDMs were treated with loganin, the cells were further stimulated with MSU crystals in serum-free medium. After incubating the JC-1 probe with the cells at 37 °C for 30 min, the cells were washed twice with PBS. Fluorescence intensity was measured at 514/529 (red/green) and 590 nm with a plate reader (Synergy H1, Biotek, Winooski, VT, USA). To obtain fluorescent images by confocal microscopy, after cell incubation with JC-1 at 37 °C for 30 min, the cells were washed and mounted on glass slides. JC-1 monomers were detected by green fluorescence, while JC-1 aggregates were detected by red fluorescence. Images were analyzed by an LSM710 laser scanning confocal microscope (Carl Zeiss, Oberkochen, Germany) with ZEN2011 software (Carl Zeiss).
4.13. Determination of Mitochondrial Reactive Oxygen Species
The measurement of mitochondrial reactive oxygen species was performed using MitoSOXTM (Thermo Fisher Scientific, Waltham, MA, USA). After LPS-primed BMDMs were treated with loganin, the cells were further stimulated with MSU crystals. The cells were incubated with MitoSOX-Red (5 μM) at 37 °C for 30 min and washed gently with PBS. MitoSOX-Red fluorescence was measured at 580 nm after excitation at 510 nm, with a plate reader (Synergy H1, Biotek, Winooski, VT, USA). For confocal microscopy imaging, the cells were incubated with MitoSOX-Red and DAPI. After incubation for 30 min, the cells were washed with PBS and mounted with mounting solution (VECTASHIELD Antifade Mounting Medium, LSBio, Seattle, WA, USA). Confocal Microscopy images were analyzed with ZEN2011 software (Carl Zeiss) by LSM710 laser scanning confocal microscope (Carl Zeiss, Oberkochen, Germany).
4.14. Statistical Analysis
Statistical analysis was performed by the software GraphPad Prism 5 (GraphPad Software, San Diego, CA, USA). All data were analyzed by one-way ANOVA followed by Tukey’s multiple comparison test and expressed as means ± SEM. Values of p < 0.05 were considered significant. The graphs or pictures are representative of at least two or three independent experiments.