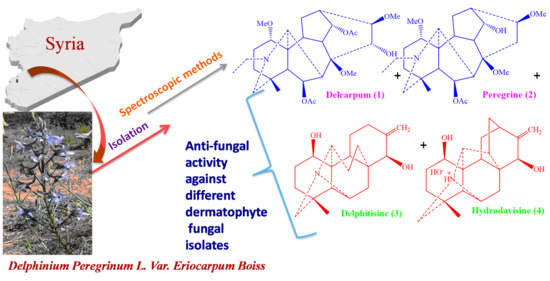
Antifungal Activity of New Diterpenoid Alkaloids Isolated by Different Chromatographic Methods from Delphinium peregrinum L. var. eriocarpum Boiss
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
2.2. Biology
Antifungal Activity
3. Materials and Methods
3.1. Chemistry
3.2. Biology
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Post, G.E. Flore of Syria. Palestine and Sinai; American Press: Reston, VA, USA, 1933; Volume 2, pp. 21–25. [Google Scholar]
- Raza, M.; Shaheen, F.; Choudhary, M.; Sombati, S.; Rafiq, A.; Suria, A.; Rahman, A.-U.; DeLorenzo, R.J. Anticonvulsant activities of ethanolic extract and aqueous fraction isolated from Delphinium denudatum. J. Ethnopharmacol. 2001, 78, 73–78. [Google Scholar] [CrossRef]
- Khare, C.P. Indian Medicinal Plants: An Illustrated Dictionary; Springer: New York, NY, USA, 2007. [Google Scholar]
- Zhang, Z.-T.; Jian, X.-X.; Ding, J.-Y.; Deng, H.-Y.; Chao, R.-B.; Chen, Q.-H.; Chen, D.-L.; Wang, F.-P. Further Studies on Structure-Cardiac Activity Relationships of Diterpenoid Alkaloids. Nat. Prod. Commun. 2015, 10, 2075–2084. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meng, X.-H.; Jiang, Z.-B.; Guo, Q.-L.; Shi, J.-G. A minor arcutine-type C 20-diterpenoid alkaloid iminium constituent of “fu zi. ” Chin. Chem. Lett. 2017, 28, 588–592. [Google Scholar] [CrossRef]
- Liang, X.; Gao, Y.; Luan, S. Two decades of advances in diterpenoid alkaloids with cytotoxicity activities. RSC Adv. 2018, 8, 23937–23946. [Google Scholar] [CrossRef] [Green Version]
- Yin, T.; Cai, L.; Ding, Z. An overview of the chemical constituents from the genus Delphinium reported in the last four decades. RSC Adv. 2020, 10, 13669–13686. [Google Scholar] [CrossRef]
- Ulubelen, A.; Meriçli, A.H.; Meriçli, F.; Kilinçer, N.; Ferizli, A.G.; Emekci, M.; Pelletier, S.W. Insect repellent activity of diterpenoid alkaloids. Phytotherapy Res. 2001, 15, 170–171. [Google Scholar] [CrossRef] [PubMed]
- Rahman, A.-U.; Nasreen, A.; Akhtar, F.; Shekhani, M.S.; Clardy, J.; Parvez, M.; Choudhary, M.I. Antifungal Diterpenoid Alkaloids fromDelphinium denudatum. J. Nat. Prod. 1997, 60, 472–474. [Google Scholar] [CrossRef] [PubMed]
- De La Fuente, G.; Gavín, J.A.; Acosta, R.D.; Morales, J.A. Diterpenoid Alkaloids from Delphinium peregrinum. The Structure of Peregrine. Heterocycles 1988, 27, 1–5. [Google Scholar] [CrossRef]
- De La Fuente, G.; Ruiz-Mesía, L. Norditerpenoid alkaloids from Delphinium peregrinum var. elongatum. Phytochemistry 1995, 39, 1459–1465. [Google Scholar] [CrossRef]
- Ulubelen, A.; Meriçli, A.H.; Meriçli, F.; Ilarslan, R. Diterpene alkaloids from delphinuim peregrinum. J. Phytochem. 1992, 31, 1019–1022. [Google Scholar] [CrossRef]
- Zhou, X.-L.; Chen, D.-L.; Chen, Q.-H.; Wang, F.-P. C20-Diterpenoid Alkaloids fromDelphiniumtrifoliolatum. J. Nat. Prod. 2005, 68, 1076–1079. [Google Scholar] [CrossRef] [PubMed]
- Advancement in Research on Delphinium, sp. (Renunculaceae): A Review. Int. J. Green Herb. Chem. 2018, 7, 251–259. [Google Scholar] [CrossRef]
- Ulubelen, A.; Desai, H.K.; Srivastava, S.K.; Hart, B.P.; Park, J.-C.; Joshi, B.S.; Pelletier, S.W.; Meriçli, A.H.; Meriçli, F.; Ilarslan, R. Diterpenoid Alkaloids fromDelphinium davisii. J. Nat. Prod. 1996, 59, 360–366. [Google Scholar] [CrossRef]
- Alhilal, S.; Alhilal, M. Isolation and Structural Elucidation of a New Alkaloid from Delphinium peregrinum L. var. eriocarpum Boiss. Basic Appl. Sci. J. King Faisal Univ. 2020, 21, 54–59. [Google Scholar] [CrossRef]
- Wang, F.-P.; Liang, X.-T. C20-diterpenoid alkaloids. Alkaloids Chem. Biol. 2002, 59, 1–280. [Google Scholar] [CrossRef] [PubMed]
- Shan, L.-H.; Zhang, J.-F.; Gao, F.; Huang, S.; Zhou, X.-L. Diterpenoid Alkaloids from Delphinium anthriscifolium var. majus. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef]
- Cowan, M.M. Plant Products as Antimicrobial Agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar] [CrossRef] [Green Version]
- Deng, W.; Sung, W.L. Three new C19-diterpenoid alkaloids, delbrunine, delbruline and desbrusine from Delphinium brunonianum royle. Heterocycles 1986, 24, 873–876. [Google Scholar]
- Hemaiswarya, S.; Kruthiventi, A.K.; Doble, M. Synergism between natural products and antibiotics against infectious diseases. Phytomedicine 2008, 15, 639–652. [Google Scholar] [CrossRef] [PubMed]
- Elmegeed, A.S.M.A.; Ouf, S.; Moussa, T.A.; Eltahlawi, S. Dermatophytes and other associated fungi in patients attending to some hospitals in Egypt. Braz. J. Microbiol. 2015, 46, 799–805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clinical and Laboratory Standards Institute (CLSI). Reference Method for Broth Dilution Antifungal Susceptibility Testing of Filamentous Fungi; Approved Standard, 2nd ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2008. [Google Scholar]




| Position | Delcarpum 1 CDCl3 | Peregrine 2 CDCl3 | Delphitisine 3 CDCl3 | Hydrodavisine 4 MeOD | ||||
|---|---|---|---|---|---|---|---|---|
| HMQC δH | HMQC δH | HMQC δH | HMQC δH | |||||
| δC | (mult., J = Hz) | δC | (mult., J = Hz) | δC | (mult., J = Hz) | δC | (mult., J = Hz) | |
| 1 | 82.3 | 3.22 m | 84.1 | 3.11 - | 73.3 | 2.67 s | 72.9 | 3.10 s |
| 2 | 24.6 | 1.87 m | 25.9 | 2.01 - | 25.1 | 1.79 m | 31.5 | 1.38 m |
| 2.15 - | 2.08 - | 1.94 m | ||||||
| 3 | 28.8 | 1.25 m | 36.5 | 1.25 - | 28.1 | 1.29 m | 25.9 | 1.84 m |
| 1.57 - | 1.90 m | |||||||
| 4 | 32.8 | - | 33.9 | - | 35.7 | - | 35.4 | - |
| 5 | 54 | 1.50 s | 55.7 | 1.48 s | 54.1 | 1.99 m | 54 | 2.21 m |
| 6 | 71.4 | 5.29 d (7.53) | 72.8 | 5.25 d (7.28) | 64.2 | 4.23 m | 63.6 | 4.20 s |
| 7 | 35.2 | 3.17 - | 41.7 | 2.75 d (7.29) | 26 | 1.29 m | 25.9 | 1.84 m |
| 1.79 m | ||||||||
| 8 | 80.8 | - | 78.5 | - | 44.1 | - | 36.3 | - |
| 9 | 40.6 | 3.35 - | 44 | 3.05 - | 31.8 | 2.26 d (2.29) | 33.1 | 2.25 m |
| 10 | 44.3 | 2.15 - | 45.6 | 2.01 - | 53.4 | - | 54.2 | - |
| 11 | 47.6 | - | 47.6 | - | 31.3 | 1.10 m | 30.9 | 1.70 m |
| 1.79 m | ||||||||
| 12 | 28.2 | 1.97 - | 28 | 1.88 m | 39.8 | 2.07 m | 42.3 | 2.11 m |
| 2.50 dd (3.17, 12.6) | 2.25 dd (5.31) | |||||||
| 13 | 36.4 | 2.32 t (5.54) | 37.9 | 2.35 t (5.67) | 25.4 | 1.79 m | 26.6 | 1.38 m |
| 1.94 m | 1.84 m | |||||||
| 14 | 75.1 | 4.67 t (4.71) | 74.8 | 4.02 q (10.59) | 41.1 | 2.07 m | 41.4 | 2.11 m |
| 15 | 74.1 | 4.18 d (6.91) | 32.5 | 2.08 - | 69.4 | 4.05 s | 69.5 | 3.99 s |
| 16 | 91.4 | 3.11 - | 81.9 | 3.39 - | 154.1 | - | 154.2 | - |
| 17 | 62.8 | 3.22 - | 64.3 | 3.16 d (2.07) | 107.6 | 4.99 d (7.60) | 108.1 | 5.01 d (7.21) |
| 18 | 25.2 | 0.85 s | 25.3 | 0.85 s | 26.7 | 1.08 s | 26.6 | 1.16 s |
| 19 | 56.8 | 2.08 d (5.78) | 56. 9 | 60.1 | 2.50 d (12.45) | 59.4 | 2. 86 q (12.29) | |
| 2.86 d (11.93) | 2.69 d (12.44) | |||||||
| 20 | 48.5 | 2.63 m | 48.7 | 2.47 m | 64.4 | 3.56 m | 66.3 | 3.93 s |
| 2.72 m | ||||||||
| 21 | 11.9 | 1.14 t (6.99) | 13.1 | 1.07 t (7.12) | 30.4 | 1.79 m | - | - |
| 2.07 m | ||||||||
| 1′ | 55 | 3.32 s | 55. 6 | 3.28 s | - | - | - | - |
| 6′ | 170.3 | - | 170.7 | - | - | - | - | |
| 6′′ | 20.6 | 1.97 - | 21.2 | 2.08 - | - | - | - | - |
| 8′ | 48.6 | 3.17 - | 47.7 | 3.11 - | - | - | - | - |
| 14′ | 170 | - | - | - | - | - | - | - |
| 14′′ | 20.2 | 1.97 - | - | - | - | - | - | - |
| 16′ | 56.2 | 3.45 s | 55.9 | 3.39 - | - | - | - | - |
| Alkaloid Derivative | Dermatophytes (Five Isolates Tested for Each Species) | |||||
|---|---|---|---|---|---|---|
| Epidermophyton floccosum | Microsporum canis | Trichophyton rubrum | ||||
| MIC | MFC | MIC | MFC | MIC | MFC | |
| Delcarpum (1) | 265 | 512 | 64 | 128–265 | 32–64 | 256 |
| Peregrine (2) | 128–256 | >512 | 32–64 | 128 | 32 | 128 |
| Delphitisine (3) | 512 | >512 | 128–265 | 512 | 64–128 | 512 |
| Hydrodavisine (4) | 512 | >512 | 128 | 512 | 64–128 | 512 |
| Mixture of the four alkaloids | 64 | 128 | 32 | 128–258 | 16 | 64 |
| Fluconazole | 32–64 | 64 | 16 | 32–64 | 32 | 64 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alhilal, M.; Sulaiman, Y.A.M.; Alhilal, S.; Gomha, S.M.; Ouf, S.A. Antifungal Activity of New Diterpenoid Alkaloids Isolated by Different Chromatographic Methods from Delphinium peregrinum L. var. eriocarpum Boiss. Molecules 2021, 26, 1375. https://doi.org/10.3390/molecules26051375
Alhilal M, Sulaiman YAM, Alhilal S, Gomha SM, Ouf SA. Antifungal Activity of New Diterpenoid Alkaloids Isolated by Different Chromatographic Methods from Delphinium peregrinum L. var. eriocarpum Boiss. Molecules. 2021; 26(5):1375. https://doi.org/10.3390/molecules26051375
Chicago/Turabian StyleAlhilal, Mohammad, Yaser A. M. Sulaiman, Suzan Alhilal, Sobhi M. Gomha, and Salama A. Ouf. 2021. "Antifungal Activity of New Diterpenoid Alkaloids Isolated by Different Chromatographic Methods from Delphinium peregrinum L. var. eriocarpum Boiss" Molecules 26, no. 5: 1375. https://doi.org/10.3390/molecules26051375