Opuntia dillenii (Ker Gawl.) Haw., Seeds Oil Antidiabetic Potential Using In Vivo, In Vitro, In Situ, and Ex Vivo Approaches to Reveal Its Underlying Mechanism of Action
Abstract
:1. Introduction
2. Results
2.1. Yield of Extraction
2.2. Acute Toxicity
2.3. Hypoglycemic Test in Non-Diabetic Rats
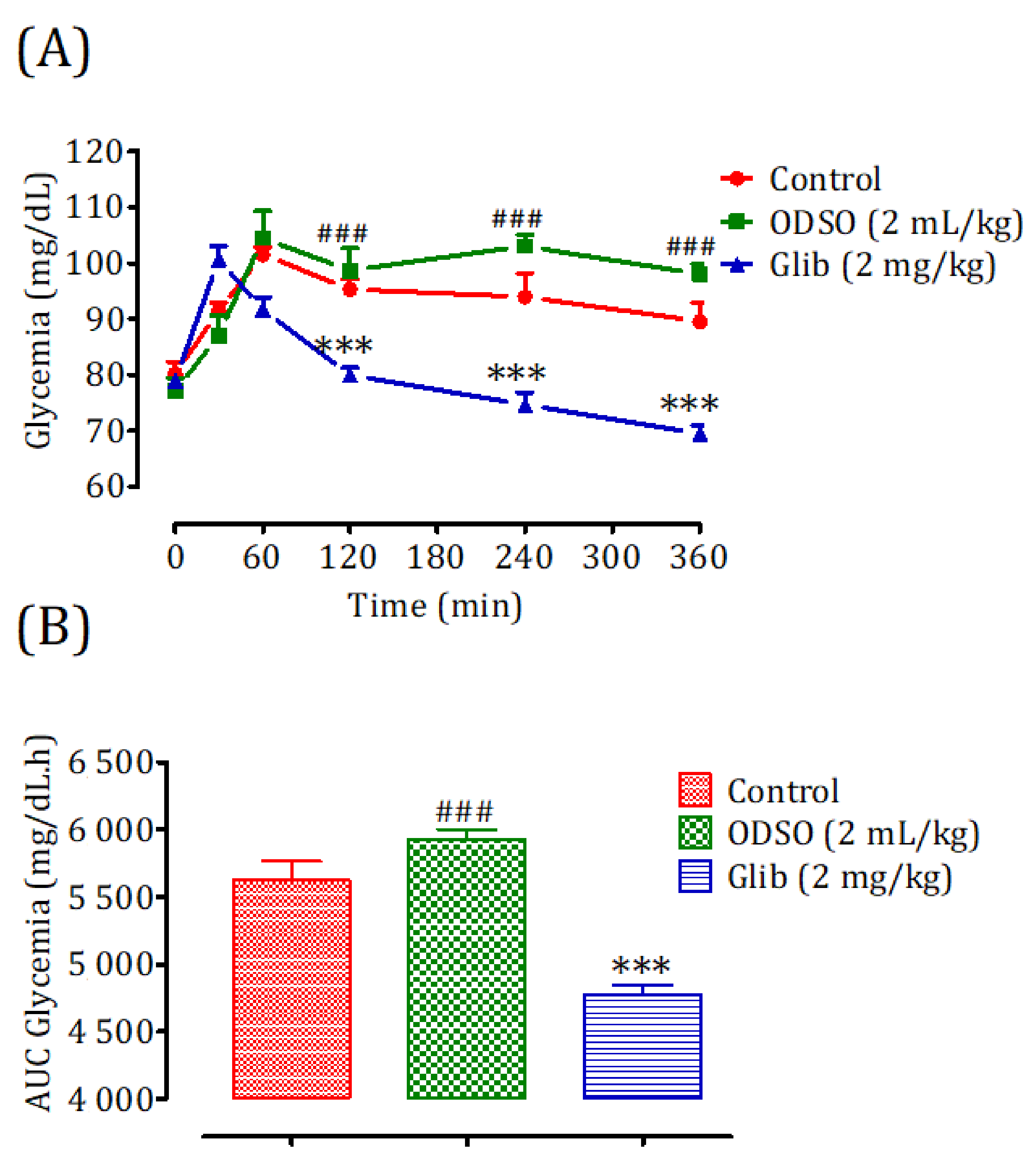
2.4. Antihyperglycemic Study in Nondiabetic and Diabetic Rats
2.4.1. Nondiabetic Rats
2.4.2. Diabetic Rats
2.5. Intestinal Absorption Inhibition of d-Glucose, In Situ
2.6. The ODSO Inhibits the Sodium-Dependent Absorption of d-Glucose
2.7. Inhibition Assay of Pancreatic α-Amylase Activity In Vitro
2.8. Inhibition Assay of Pancreatic α-Amylase Activity in Non-Diabetic and Diabetic Rats
2.8.1. Nondiabetic Rats
2.8.2. Diabetic Rats
2.9. Inhibition Assay of Intestinal α-Glucosidase Activity In Vitro
2.10. Kinetics of Intestinal α-Glucosidase Inhibition In Vitro
2.11. Inhibition Assay of Intestinal α-Glucosidase Activity in Nondiabetic and Diabetic Rats
2.11.1. Nondiabetic Rats
2.11.2. Diabetic Rats
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Opuntia Dillenii Fruit Harvest
4.3. Powder Preparation and Oil Extraction of Opuntia dillenii Seeds
4.4. Experimental Animals
4.5. Acute Oral Toxicity in Mice
4.6. Hypoglycemic Test in Nondiabetic Rats
4.7. Induction of Experimental Diabetes
4.8. Antihyperglycemic Study in Nondiabetic and Diabetic Rats
4.9. Single-Pass Intestinal Perfusion in Rats
4.10. Ussing Chamber Assay
4.11. Inhibition Assay of α-Amylase Activity In Vitro
4.12. Inhibition Assay of α-Amylase Activity in Normal and Diabetic Rats
4.13. Inhibition Assay of Intestinal α-Glucosidase Activity In Vitro
4.14. Kinetics of Intestinal α-Glucosidase Inhibition In Vitro
4.15. Inhibition Assay of Intestinal α-Glucosidase Activity in Normal and Diabetic Rats
4.16. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Fajardo, S.; García-Galvan, R.F.; Barranco, V.; Galvan, J.C.; Batlle, S.F. A Review of the Antidiabetic Activities of Ginge; Intech Open Limited: London, UK, 2016. [Google Scholar]
- Bi, X.; Lim, J.; Henry, C.J. Spices in the management of diabetes mellitus. Food Chem. 2017, 217, 281–293. [Google Scholar] [CrossRef] [PubMed]
- IDF Diabetes Atlas, 9th ed.; International Diabetes Federation: Brussels, Belgium, 2019; ISBN 978-2-930229-87-4.
- Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A.A.; Ogurtsova, K.; et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res. Clin. Pract. 2019, 157, 107843. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pheiffer, C.; Wyk, V.P.-V.; Joubert, J.D.; Levitt, N.; Nglazi, M.D.; Bradshaw, D. The prevalence of type 2 diabetes in South Africa: A systematic review protocol. BMJ Open 2018, 8, 21–29. [Google Scholar] [CrossRef] [Green Version]
- Avogaro, A.; Fadini, G.P. The Effects of Dipeptidyl Peptidase-4 Inhibition on Microvascular Diabetes Complications. Diabetes Care 2014, 37, 2884–2894. [Google Scholar] [CrossRef] [Green Version]
- Le Marchand-Brustel, Y.; Jeanrenaud, B.; Freychet, P. Insulin binding and effects in isolated soleus muscle of lean and obese mice. Am. J. Physiol. Metab. 1978, 234, E348. [Google Scholar] [CrossRef] [PubMed]
- Sheard, N.F.; Clark, N.G.; Brand-Miller, J.C.; Franz, M.J.; Pi-Sunyer, F.X.; Mayer-Davis, E.; Kulkarni, K.; Geil, P. Dietary Carbohydrate (Amount and Type) in the Prevention and Management of Diabetes: A statement by the American Diabetes Association. Diabetes Care 2004, 27, 2266–2271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sales, P.M.; Souza, P.M.; Simeoni, L.A.; Magalhães, P.O.; Silveira, D. α-Amylase Inhibitors: A Review of Raw Material and Isolated Compounds from Plant Source. J. Pharm. Pharm. Sci. 2012, 15, 141–183. [Google Scholar] [CrossRef] [Green Version]
- Olaokun, O.O.; McGaw, L.J.; Eloff, J.N.; Naidoo, V. Evaluation of the Inhibition of Carbohydrate Hydrolysing Enzymes, Antioxidant Activity and Polyphenolic Content of Extracts of Ten African Ficus Species (Moraceae) Used Traditionally to Treat Diabetes. BMC Complementary Altern. Med. 2013, 13, 94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hui, H.; Zhao, X.; Perfetti, R. Structure and function studies of glucagon-like peptide-1 (GLP-1): The designing of a novel pharmacological agent for the treatment of diabetes. Diabetes Metbal Res. Rev. 2005, 21, 313–331. [Google Scholar] [CrossRef]
- Bathaie, S.Z.; Mokarizade, N.; Shirali, S. An Overview of the Mechanisms of Plant Ingredients in the Treatment of Diabetes Mellitus. J. Med. Plants 2012, 11, 1–24. [Google Scholar]
- Kooti, W.; Farokhipour, M.; Asadzadeh, Z.; Ashtary-Larky, D.; Asadi-Samani, M. The role of medicinal plants in the treatment of diabetes: A systematic review. Electron. Phys. 2016, 8, 1832–1842. [Google Scholar] [CrossRef] [Green Version]
- Es-Safi, I.; Mechchate, H.; Amaghnouje, A.; El Moussaoui, A.; Cerruti, P.; Avella, M.; Grafov, A.; Bousta, D. Marketing and legal status of phytomedicines and food supplements in Morocco. J. Complement. Integr. Med. 2020. [Google Scholar] [CrossRef]
- Schlienger, J.-L. Diabète et Phytothérapie: Les Faits. Méd. Mal. Métaboliques 2014, 8, 101–106. [Google Scholar] [CrossRef]
- Koski, R.R. Practical Review of Oral Antihyperglycemic Agents for Type 2 Diabetes Mellitus. Diabetes Educ. 2006, 32, 869–876. [Google Scholar] [CrossRef]
- Furman, B.L.; Candasamy, M.; Bhattamisra, S.K.; Veettil, S.K. Reduction of blood glucose by plant extracts and their use in the treatment of diabetes mellitus; discrepancies in effectiveness between animal and human studies. J. Ethnopharmacol. 2020, 247, 112264. [Google Scholar] [CrossRef] [PubMed]
- Marles, R.J. Plants as Sources of Antidiabetic Agents. Econ. Med. Plant Res. 1994, 6, 149–187. [Google Scholar]
- Chopra, A.; Saluja, M.; Tillu, G. Ayurveda-modern medicine interface: A critical appraisal of studies of Ayurvedic medicines to treat osteoarthritis and rheumatoid arthritis. J. Ayurveda Integr. Med. 2010, 1, 190–198. [Google Scholar] [CrossRef] [Green Version]
- Reyes-Agüero, J.A.; Aguirre-Rivera, J.R.; Hernández, H.M. Notas Sistemáticas y Una Descripción Detallada de Opuntia Ficus-Indica (L.) Mill. (Cactaceae). Agrociencia 2005, 39, 395–408. (In Spanish) [Google Scholar]
- Griffith, M.P. The origins of an important cactus crop, Opuntia ficus-indica (Cactaceae): New molecular evidence. Am. J. Bot. 2004, 91, 1915–1921. [Google Scholar] [CrossRef] [Green Version]
- Hegwood, D.A. Human Health Discoveries with Opuntia sp. (Prickly Pear). HortScience 1990, 25, 1515–1516. [Google Scholar] [CrossRef] [Green Version]
- Enigbokan, M.A.; Felder, T.B.; Thompson, J.O.; Kuti, J.O.; Ekpenyong, K.I. Hypoglycaemic Effects of Opuntia ficus-indica Mill., Opuntia lindheimeri Engelm and Opuntia robusta Wendl. in Streptozotocin-induced Diabetic Rats. Phytother. Res. 1996, 10, 379–382. [Google Scholar] [CrossRef]
- Barbera, G.; Inglese, P.; Pimienta-Barrios, E. Agro-Ecology, Cultivation and Uses of Cactus Pear; FAO: Rome, Italy, 1995; Volume 132. [Google Scholar]
- Keter, L.K.; Mutiso, P.C. Ethnobotanical studies of medicinal plants used by Traditional Health Practitioners in the management of diabetes in Lower Eastern Province, Kenya. J. Ethnopharmacol. 2012, 139, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Raj, V. Plant Opuntia Dillenii: A Review on Its Traditional Uses, Phytochemical and Pharmacological Properties. EC Pharm. Sci. 2015, 1, 29–43. [Google Scholar]
- Zhao, X.; Yang, J.; Tong, Z.; Zhou, Y.; Zhang, W.; Qi, S.; Yuan, G. Clinical Observation of Opuntia Dillenii Tablet in Treating Type 2 Diabetes Mellitus. Chin. J. Integr. Tradit. West. Med. 2002, 8, 215–218. [Google Scholar]
- Qiu, Y.K.; Zhao, Y.Y.; Dou, D.Q.; Xu, B.X.; Liu, K. Two new α-pyrones and other components from the cladodes of Opuntia dillenii. Arch. Pharmacal Res. 2007, 30, 665–669. [Google Scholar] [CrossRef]
- De Paz, P.L.P.; Medina, I.M. Catalogo de Las Plantas Medicinales de La Flora Canaria: Applicaciones Populares; Instituto de Estudios canaries: Tenerife, Spain, 1988; ISBN 84-404-1458-7. (In Spanish) [Google Scholar]
- Khan, M.P.Z.; Ahmad, M.; Zafar, M.; Sultana, S.; Ali, M.I.; Sun, H. Ethnomedicinal uses of Edible Wild Fruits (EWFs) in Swat Valley, Northern Pakistan. J. Ethnopharmacol. 2015, 173, 191–203. [Google Scholar] [CrossRef] [PubMed]
- Perfumi, M.; Tacconi, R. Antihyperglycemic Effect of Fresh Opuntia dillenii Fruit from Tenerife (Canary Islands). Int. J. Pharmacogn. 1996, 34, 41–47. [Google Scholar] [CrossRef]
- Berraaouan, A.; Abderrahim, Z.; Hassane, M.; AbdelKhaleq, L.; Mohammed, A.; Mohamed, B. Evaluation of protective effect of cactus pear seed oil (Opuntia ficus-indica L. MILL.) against alloxan-induced diabetes in mice. Asian Pac. J. Trop. Med. 2015, 8, 532–537. [Google Scholar] [CrossRef] [Green Version]
- Loukili, E.H.; Abrigach, F.; Bouhrim, M.; Bnouham, M.; Fauconnier, M.-L.; Ramdani, M. Chemical Composition and Physicochemical Analysis of Opuntia dillenii Extracts Grown in Morocco. J. Chem. 2021, 2021, 1–11. [Google Scholar] [CrossRef]
- Berraaouan, A.; Ziyyat, A.; Mekhfi, H.; Legssyer, A.; Sindic, M.; Aziz, M.; Bnouham, M. Evaluation of antidiabetic properties of cactus pear seed oil in rats. Pharm. Biol. 2014, 52, 1286–1290. [Google Scholar] [CrossRef]
- Haidara, M.A.; Ibrahim, I.M.; Al-Tuwaijri, A.S.; Awadalla, S.A.; Yaseen, H. Effect of α-Tocopherol on Glucose Uptake and Contractility in Rat Skeletal Muscle. Med Sci. Monit. 2003, 9, BR174–BR177. [Google Scholar]
- Ghazi, Z.; Ramdani, M.; Tahri, M.; Rmili, R.; Elmsellem, H.; Mahi, B.E.; Fauconnier, M.L. Chemical Composition and Antioxidant Activity of Seeds Oils and Fruit Juice of Opuntia Ficus Indica and Opuntia Dillenii from Morocco. J. Mater. Environ. Sci 2015, 6, 2338–2345. [Google Scholar]
- Bnouham, M.; Bouhrim, M.; Ouassou, H.; Choukri, M.; Mekhfi, H.; Ziyyat, A.; Legssyer, A.; Aziz, M. Hepatoprotective effect of Opuntia dillenii seed oil on CCl4 induced acute liver damage in rat. Asian Pac. J. Trop. Biomed. 2018, 8, 254. [Google Scholar] [CrossRef]
- Bnouham, M.; Bouhrim, M.; Ouassou, H.; Loukili, E.H.; Ramdani, M.; Mekhfi, H.; Ziyyat, A.; Legssyer, A.; Aziz, M. Antidiabetic effect of Opuntia dillenii seed oil on streptozotocin-induced diabetic rats. Asian Pac. J. Trop. Biomed. 2019, 9, 381–388. [Google Scholar] [CrossRef]
- Bouhrim, M.; Daoudi, N.E.; Ouassou, H.; Benoutman, A.; Loukili, E.H.; Ziyyat, A.; Mekhfi, H.; Legssyer, A.; Aziz, M.; Bnouham, M.; et al. Phenolic Content and Antioxidant, Antihyperlipidemic, and Antidiabetogenic Effects of Opuntia dillenii Seed Oil. Sci. World J. 2020, 2020, 1–8. [Google Scholar] [CrossRef]
- Aragno, M.; Mastrocola, R.; Catalano, M.G.; Brignardello, E.; Danni, O.; Boccuzzi, G. Oxidative Stress Impairs Skeletal Muscle Repair in Diabetic Rats. Diabetes 2004, 53, 1082–1088. [Google Scholar] [CrossRef] [Green Version]
- Yadav, P.; Sarkar, S.; Bhatnagar, D. Action of capparis deciduaagainst alloxan-induced oxidative stress and diabetes in rat tissues. Pharmacol. Res. 1997, 36, 221–228. [Google Scholar] [CrossRef]
- Guillet, C. Implication Des Produits Terminaux de Glycation Dans Les Complications Liées Au Diabète. Nutr. Clin. Métabolisme 2010, 24, 109–114. (In French) [Google Scholar] [CrossRef]
- D’Amico, M.; Marfella, R.; Nappo, F.; DiFilippo, C.; De Angelis, L.; Berrino, L.; Rossi, F.; Giugliano, D. High Glucose Induces Ventricular Instability and Increases Vasomotor Tone in Rats. Diabetologia 2001, 44, 464–470. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tomimatsu, T.; Horie, T. Enhanced Glucose Absorption in the Rat Small Intestine Following Repeated Doses of 5-Fluorouracil. Chemico-Biol. Interact. 2005, 155, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Shai, L.J.; Masoko, P.; Mokgotho, M.P.; Magano, S.R.; Mogale, A.M.; Boaduo, N.; Eloff, J.N. Yeast Alpha Glucosidase Inhibitory and Antioxidant Activities of Six Medicinal Plants Collected in Phalaborwa, South Africa. S. Afr. J. Bot. 2010, 76, 465–470. [Google Scholar] [CrossRef] [Green Version]
- Loizzo, M.R.; Saab, A.M.; Tundis, R.; Menichini, F.; Bonesi, M.; Piccolo, V.; Statti, G.A.; de Cindio, B.; Houghton, P.J.; Menichini, F. In Vitro Inhibitory Activities of Plants Used in Lebanon Traditional Medicine against Angiotensin Converting Enzyme (ACE) and Digestive Enzymes Related to Diabetes. J. Ethnopharmacol. 2008, 119, 109–116. [Google Scholar] [CrossRef]
- Clemens, K.K.; McArthur, E.; Dixon, S.N.; Fleet, J.L.; Hramiak, I.; Garg, A.X. The Hypoglycemic Risk of Glyburide (Glibenclamide) Compared with Modified-Release Gliclazide. Can. J. Diabetes 2015, 39, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Koubaa, M.; Mhemdi, H.; Barba, F.J.; Angelotti, A.; Bouaziz, F.; Chaabouni, S.E.; Vorobiev, E. Seed Oil Extraction from Red Prickly Pear Using Hexane and Supercritical CO2: Assessment of Phenolic Compound Composition, Antioxidant and Antibacterial Activities. J. Sci. Food Agric. 2016, 97, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Ghazi, Z.; Ramdani, M.; Fauconnier, M.L.; Mahi, B.E.; Cheikh, R. Fatty Acids Sterols and Vitamin E Composition of Seed Oil of Opuntia ficus Indica and Opuntia dillenii from Morocco. J. Mater. Environ. Sci. 2013, 4, 967–972. [Google Scholar]
- Mobraten, K.; Haug, T.M.; Kleiveland, C.R.; Lea, T. Omega-3 and Omega-6 PUFAs Induce the Same GPR120-Mediated Signalling Events, but with Different Kinetics and Intensity in Caco-2 Cells. Lipids Health Dis. 2013, 12, 101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holaly, G.E.; Efui Holaly, G.; Kodjovi, A.; Simplice Damintoti, K.; Kokou, A.; Yao, A.; Gérard, T.; Blaise, A.; Jacques, S.; Messanvi, G. Composés Bioactifs Isolés Des Plantes à Propriété Anti-Diabétique: Revue de Littérature [Isolated Bioactive Plant Compounds with Anti-Diabetic Property: Review]. Int. J. Innov. Appl. Stud. 2017, 19, 839–849. [Google Scholar]
- Ganapaty, S.; Nandeesh, R.; Veerapur, V.P.; Thippeswamy, B.; Shivasharan, D. In vivo and In vitro Anti-Diabetic Effects of Madhuca indica Roxb. Alloxan-Induc. Diabet. Rats IJAPBC 2013, 2, 282–290. [Google Scholar]
- Kumar, S.; Kumar, V.; Prakash, O. Enzymes Inhibition and Antidiabetic Effect of Isolated Constituents from dillenia indica. BioMed Res. Int. 2013, 2013, 1–7. [Google Scholar] [CrossRef]
- Somani, R.; Kasture, S.; Singhai, A.K. Antidiabetic Potential of Butea Monosperma in Rats. Fitoterapia 2006, 77, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Panda, S.; Jafri, M.; Kar, A.; Meheta, B.K. Thyroid Inhibitory, Antiperoxidative and Hypoglycemic Effects of Stigmasterol Isolated from Butea Monosperma. Fitoterapia 2009, 80, 123–126. [Google Scholar] [CrossRef]
- Båvenholm, P.N.; Pigon, J.; Östenson, C.G.; Efendic, S. Insulin Sensitivity of Suppression of Endogenous Glucose Production Is the Single Most Important Determinant of Glucose Tolerance. Diabetes 2001, 50, 1449–1454. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Oguzhan, B.; Louchami, K.; Chardigny, J.-M.; Portois, L.; Carpentier, Y.A.; Malaisse, W.J.; Herchuelz, A.; Sener, A. Pancreatic islet function in ω-3 fatty acid-depleted rats: Alteration of calcium fluxes and calcium-dependent insulin release. Am. J. Physiol. Metab. 2006, 291, E441–E448. [Google Scholar] [CrossRef] [Green Version]
- Manco, M.; Calvani, M.; Mingrone, G. Effects of dietary fatty acids on insulin sensitivity and secretion. Diabetes Obes. Metab. 2004, 6, 402–413. [Google Scholar] [CrossRef]
- Nugent, C.; Prins, J.B.; Whitehead, J.P.; Wentworth, J.M.; Chatterjee, V.K.K.; O’Rahilly, S. Arachidonic Acid Stimulates Glucose Uptake in 3T3-L1 Adipocytes by Increasing GLUT1 and GLUT4 Levels at the Plasma Membrane. J. Biol. Chem. 2001, 276, 9149–9157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sampaio, E.M.; Furtado, F.A.S.; Furtado, J.S.; Cavalacante, M.N.N.; Riedel, O.O. Hypoglycemia-Producing Activities of Raw Coffee Beans (Coffea arabica). Rev. Med. Univ. Fed. Ceara 1979, 19, 49–54. [Google Scholar]
- Sattar, N.A.; Hussain, F.; Iqbal, T.; Sheikh, M.A. Determination of in vitro antidiabetic effects of Zingiber officinale Roscoe. Braz. J. Pharm. Sci. 2012, 48, 601–607. [Google Scholar] [CrossRef] [Green Version]
- Nammi, S.; Sreemantula, S.; Roufogalis, B.D. Protective Effects of Ethanolic Extract of Zingiber officinale Rhizome on the Development of Metabolic Syndrome in High-fat Diet-fed Rats. Basic Clin. Pharmacol. Toxicol. 2009, 104, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Bansal, P.; Paul, P.; Mudgal, J.; Nayak, P.G.; Thomas Pannakal, S.; Priyadarsini, K.I.; Unnikrishnan, M.K. Antidiabetic, Antihyperlipidemic and Antioxidant Effects of the Flavonoid Rich Fraction of Pilea microphylla (L.) in High Fat Diet/Streptozotocin-Induced Diabetes in Mice. Exp. Toxicol. Pathol. 2012, 64, 651–658. [Google Scholar] [CrossRef]
- Abdelmoaty, M.A.; Ibrahim, M.A.; Ahmed, N.S.; Abdelaziz, M.A. Confirmatory Studies on the Antioxidant and Antidiabetic Effect of Quercetin in Rats. Indian J. Clin. Biochem. 2010, 25, 188–192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moorthi, R.V.; Bobby, Z.; Selvaraj, N.; Sridhar, M.G. Vitamin E Protects the Insulin Sensitivity and Redox Balance in Rat L6 Muscle Cells Exposed to Oxidative Stress. Clin. Chim. Acta 2006, 367, 132–136. [Google Scholar] [CrossRef]
- Labuschagne, M.T.; Hugo, A. Oil Content and Fatty Acid Composition of Cactus Pear Seed Compared with Cotton and Grape Seed. J. Food Biochem. 2010, 34, 93–100. [Google Scholar] [CrossRef]
- Chang, S.F.; Hsieh, C.L.; Yen, G.C. The Protective Effect of Opuntia dillenii Haw Fruit against Low-Density Lipoprotein Peroxidation and Its Active Compounds. Food Chem. 2008, 106, 569–575. [Google Scholar] [CrossRef]
- Salvo, F.; Galati, E.M.; Curto, S.L.; Tripodo, M.M. Study on the Chemical Characterization of Lipid Composition of Opuntia Ficus indica L. Seed Oil. Riv. Ital. Delle Sostanze Grasse 2002, 79, 395–398. [Google Scholar]
- El Mannoubi, I.; Barrek, S.; Skanji, T.; Casabianca, H.; Zarrouk, H. Characterization of Opuntia Ficus indica Seed Oil from Tunisia. Chem. Nat. Compd. 2009, 45, 616–620. [Google Scholar] [CrossRef]
- Matthäus, B.; Özcan, M.M. Habitat Effects on Yield, Fatty Acid Composition and Tocopherol Contents of Prickly Pear (Opuntia Ficus indica L.) Seed Oils. Sci. Hortic. 2011, 131, 95–98. [Google Scholar] [CrossRef]
- National Research Council. Guide for the Care and Use of Laboratory Animals, 8th ed.; The National Academies Press: Washington, DC, USA, 2011; ISBN 9780309154000. [Google Scholar]
- Bellahcen, S.; Mekhfi, H.; Ziyyat, A.; Legssyer, A.; Hakkou, A.; Aziz, M.; Bnouham, M. Prevention of Chemically Induced Diabetes Mellitus in Experimental Animals by Virgin Argan Oil. Phytotherapy Res. 2012, 26, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Luzi, L.; Pozza, G. Glibenclamide: An Old Drug with a Novel Mechanism of Action? Acta Diabetol. 1997, 34, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Nishigaki, A.; Noma, H.; Kakizawa, T. The relations between doses of streptozotocin and pathosis in induced diabetes mellitus. Shika gakuho. Dent. Sci. Rep. 1989, 89, 639–662. [Google Scholar]
- Rossetti, L.; Smith, D.; Shulman, G.; Papachristou, D.; DeFronzo, R. Correction of Hyperglycemia with Phlorizin Normalizes Tissue Sensitivity to Insulin in Diabetic Rats. J. Clin. Investig. 1987, 79, 1510–1515. [Google Scholar] [CrossRef]
- Bnouham, M.; Merhfour, F.Z.; Ziyyat, A.; Mekhfi, H.; Aziz, M.; Legssyer, A. Antihyperglycemic Activity of the Aqueous Extract of Urtica dioica. Fitoterapia 2003, 74, 677–681. [Google Scholar] [CrossRef]
- Eto, B.; Boisset, M.; Griesmar, B.; Desjeux, J.F. Effect of Sorbin on Electrolyte Transport in Rat and Human Intestine. Am. J. Physiol. Liver Physiol. 1999, 276, G107–G114. [Google Scholar] [CrossRef] [PubMed]
- Daoudi, N.E.; Bouhrim, M.; Ouassou, H.; Legssyer, A.; Mekhfi, H.; Ziyyat, A.; Aziz, M.; Bnouham, M. Inhibitory Effect of Roasted/ Unroasted Argania spinosa Seeds Oil on α- Glucosidase, α-Amylase and Intestinal Glucose Absorption Activities. S. Afr. J. Bot. 2020, 135, 413–420. [Google Scholar] [CrossRef]
- Mechchate, H.; Es-safi, I.; Louba, A.; Alqahtani, A.S.; Nasr, F.A.; Noman, O.M.; Farooq, M.; Alharbi, M.S.; Alqahtani, A.; Bari, A.; et al. In Vitro Alpha-Amylase and Alpha-Glucosidase Inhibitory Activity and In Vivo Antidiabetic Activity of Withania frutescens L. Foliar Extract. Molecules 2021, 26, 293. [Google Scholar] [CrossRef]
- Ouassou, H.; Zahidi, T.; Bouknana, S.; Bouhrim, M.; Mekhfi, H.; Ziyyat, A.; Legssyer, A.; Aziz, M.; Bnouham, M. Inhibition of α -Glucosidase, Intestinal Glucose Absorption, and Antidiabetic Properties by Caralluma europaea. Evid. Based Complement. Altern. Med. 2018, 2018, 1–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gholamhoseinian, A.; Fallah, H. Inhibitory Effect of Methanol Extract of Rosa damascena Mill. Flowers on α-Glucosidase Activity and Postprandial Hyperglycemia in Normal and Diabetic Rats. Phytomedicine 2009, 16, 935–941. [Google Scholar] [CrossRef] [PubMed]












| ODSO Concentration (μg/mL) | Km (mM) | Vmax (μM/min) |
|---|---|---|
| 0 | 71.42 | 166.66 |
| 165 | 71.42 | 166.66 |
| 325 | 71.42 | 83.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bouhrim, M.; Ouassou, H.; Boutahiri, S.; Daoudi, N.E.; Mechchate, H.; Gressier, B.; Eto, B.; Imtara, H.; A. Alotaibi, A.; Al-zharani, M.; et al. Opuntia dillenii (Ker Gawl.) Haw., Seeds Oil Antidiabetic Potential Using In Vivo, In Vitro, In Situ, and Ex Vivo Approaches to Reveal Its Underlying Mechanism of Action. Molecules 2021, 26, 1677. https://doi.org/10.3390/molecules26061677
Bouhrim M, Ouassou H, Boutahiri S, Daoudi NE, Mechchate H, Gressier B, Eto B, Imtara H, A. Alotaibi A, Al-zharani M, et al. Opuntia dillenii (Ker Gawl.) Haw., Seeds Oil Antidiabetic Potential Using In Vivo, In Vitro, In Situ, and Ex Vivo Approaches to Reveal Its Underlying Mechanism of Action. Molecules. 2021; 26(6):1677. https://doi.org/10.3390/molecules26061677
Chicago/Turabian StyleBouhrim, Mohamed, Hayat Ouassou, Salima Boutahiri, Nour Elhouda Daoudi, Hamza Mechchate, Bernard Gressier, Bruno Eto, Hamada Imtara, Amal A. Alotaibi, Mohammed Al-zharani, and et al. 2021. "Opuntia dillenii (Ker Gawl.) Haw., Seeds Oil Antidiabetic Potential Using In Vivo, In Vitro, In Situ, and Ex Vivo Approaches to Reveal Its Underlying Mechanism of Action" Molecules 26, no. 6: 1677. https://doi.org/10.3390/molecules26061677







