Is PF-00835231 a Pan-SARS-CoV-2 Mpro Inhibitor? A Comparative Study
Abstract
:1. Introduction
2. Result and Discussion
3. Materials and Methods
3.1. Protein Structure Preparation
3.2. Ligand Structure Preparation
3.3. Redocking: Co-Crystallized Ligand Pose Validation Study
3.4. Virtual Screening
3.5. Molecular Dynamics Simulation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Hu, B.; Guo, H.; Zhou, P.; Shi, Z.L. Characteristics of SARS-CoV-2 and COVID-19. Nat. Rev. Microbiol. 2021, 19, 141–154. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J. Everything you need to know about the COVID-19 therapy trials. Pharm. J. 2021. [Google Scholar] [CrossRef]
- Clinicaltrials. gov. Available online: https://www.clinicaltrials.gov/ (accessed on 15 January 2021).
- Rubin, D.; Chan-Tack, K.; Farley, J.; Sherwat, A. FDA Approval of Remdesivir—A Step in the Right Direction. N. Engl. J. Med. 2020, 383, 2598–2600. [Google Scholar] [CrossRef]
- Pau, A.K.; Aberg, J.; Baker, J.; Belperio, P.S.; Coopersmith, C.; Crew, P.; Grund, B.; Gulick, R.M.; Harrison, C.; Kim, A.; et al. Convalescent Plasma for the Treatment of COVID-19: Perspectives of the National Institutes of Health COVID-19 Treatment Guidelines Panel. Ann. Intern. Med. 2021, 174, 93–95. [Google Scholar] [CrossRef]
- Artigas, L.; Coma, M.; Matos-Filipe, P.; Aguirre-Plans, J.; Farrés, J.; Valls, R.; Fernandez-Fuentes, N.; De La Haba-Rodriguez, J.; Olvera, A.; Barbera, J.; et al. In-silico drug repurposing study predicts the combination of pirfenidone and melatonin as a promising candidate therapy to reduce SARS-CoV-2 infection progression and respiratory distress caused by cytokine storm. PLoS ONE 2020, 15, e0240149. [Google Scholar] [CrossRef]
- Riva, L.; Yuan, S.; Yin, X.; Martin-Sancho, L.; Matsunaga, N.; Pache, L.; Burgstaller-Muehlbacher, S.; De Jesus, P.D.; Teriete, P.; Hull, M.V.; et al. Discovery of SARS-CoV-2 antiviral drugs through large-scale compound repurposing. Nature 2020, 586, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Krichel, B.; Falke, S.; Hilgenfeld, R.; Redecke, L.; Uetrecht, C. Processing of the SARS-CoV pp1a/ab nsp7–10 region. Biochem. J. 2020, 477, 1009–1019. [Google Scholar] [CrossRef] [Green Version]
- Vlachakis, D.; Papakonstantinou, E.; Mitsis, T.; Pierouli, K.; Diakou, I.; Chrousos, G.; Bacopoulou, F. Molecular mechanisms of the novel coronavirus SARS-CoV-2 and potential anti-COVID19 pharmacological targets since the outbreak of the pandemic. Food Chem. Toxicol. 2020, 146, 111805. [Google Scholar] [CrossRef] [PubMed]
- Xue, X.; Yu, H.; Yang, H.; Xue, F.; Wu, Z.; Shen, W.; Li, J.; Zhou, Z.; Ding, Y.; Zhao, Q.; et al. Structures of Two Coronavirus Main Proteases: Implications for Substrate Binding and Antiviral Drug Design. J. Virol. 2007, 82, 2515–2527. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghahremanpour, M.M.; Tirado-Rives, J.; Deshmukh, M.; Ippolito, J.A.; Zhang, C.-H.; De Vaca, I.C.; Liosi, M.-E.; Anderson, K.S.; Jorgensen, W.L. Identification of 14 Known Drugs as Inhibitors of the Main Protease of SARS-CoV-2. ACS Med. Chem. Lett. 2020, 11, 2526–2533. [Google Scholar] [CrossRef]
- Callaway, E. The coronavirus is mutating—Does it matter? Nat. Cell Biol. 2020, 585, 174–177. [Google Scholar] [CrossRef]
- Analytica, O. New COVID-19 Variants Could Prolong Pandemic: Expert Briefings. 2021. Available online: https://www.emerald.com/insight/content/doi/10.1108/OXAN-ES259153/full/html (accessed on 15 January 2021).
- Law, S.; Leung, A.W.; Xu, C. COVID-19 mutation in the United Kingdom. Microbes Infect. Dis. 2021. [Google Scholar] [CrossRef]
- Boras, B.; Jones, R.M.; Anson, B.J.; Arenson, D.; Aschenbrenner, L.; Bakowski, M.A.; Beutler, N.; Binder, J.; Chen, E.; Eng, H.; et al. Discovery of a Novel Inhibitor of Coronavirus 3CL Protease as a Clinical Candidate for the Potential Treatment of COVID-19. BioRxiv 2020. [Google Scholar] [CrossRef]
- Hoffman, R.L.; Kania, R.S.; Brothers, M.A.; Davies, J.F.; Ferre, R.A.; Gajiwala, K.S.; He, M.; Hogan, R.J.; Kozminski, K.; Li, L.Y.; et al. Discovery of Ketone-Based Covalent Inhibitors of Coronavirus 3CL Proteases for the Potential Therapeutic Treatment of COVID-19. J. Med. Chem. 2020, 63, 12725–12747. [Google Scholar] [CrossRef]
- Colovos, C.; Yeates, T.O. Verification of protein structures: Patterns of nonbonded atomic interactions. Protein Sci. 1993, 2, 1511–1519. [Google Scholar] [CrossRef] [Green Version]
- Bowie, J.U.; Luthy, R.; Eisenberg, D. A method to identify protein sequences that fold into a known three-dimensional structure. Science 1991, 253, 164–170. [Google Scholar] [CrossRef] [Green Version]
- Lüthy, R.; Bowie, J.U.; Eisenberg, D. Assessment of protein models with three-dimensional profiles. Nature 1992, 356, 83–85. [Google Scholar] [CrossRef]
- Seeliger, D.; De Groot, B.L. Ligand docking and binding site analysis with PyMOL and Autodock/Vina. J. Comput. Mol. Des. 2010, 24, 417–422. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jin, Z.; Du, X.; Xu, Y.; Deng, Y.; Liu, M.; Zhao, Y.; Zhang, B.; Li, X.; Zhang, L.; Peng, C.; et al. Structure of Mpro from SARS-CoV-2 and discovery of its inhibitors. Nature 2020, 582, 289–293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bello, M.; Martínez-Muñoz, A.; Balbuena-Rebolledo, I. Identification of saquinavir as a potent inhibitor of dimeric SARS-CoV2 main protease through MM/GBSA. J. Mol. Model. 2020, 26, 340. [Google Scholar] [CrossRef] [PubMed]
- Tejera, E.; Munteanu, C.; López-Cortés, A.; Cabrera-Andrade, A.; Pérez-Castillo, Y. Drugs Repurposing Using QSAR, Docking and Molecular Dynamics for Possible Inhibitors of the SARS-CoV-2 Mpro Protease. Molecules 2020, 25, 5172. [Google Scholar] [CrossRef]
- Oerlemans, R.; Ruiz-Moreno, A.J.; Cong, Y.; Kumar, N.D.; Velasco-Velazquez, M.A.; Neochoritis, C.G.; Smith, J.; Reggiori, F.; Groves, M.R.; Dömling, A. Repurposing the HCV NS3–4A protease drug boceprevir as COVID-19 therapeutics. RSC Med. Chem. 2020. [Google Scholar] [CrossRef]
- Ma, C.; Sacco, M.D.; Hurst, B.; Townsend, J.A.; Hu, Y.; Szeto, T.; Zhang, X.; Tarbet, B.; Marty, M.T.; Chen, Y.; et al. Boceprevir, GC-376, and calpain inhibitors II, XII inhibit SARS-CoV-2 viral replication by targeting the viral main protease. Cell Res. 2020, 30, 678–692. [Google Scholar] [CrossRef]
- Fu, L.; Ye, F.; Feng, Y.; Yu, F.; Wang, Q.; Wu, Y.; Zhao, C.; Sun, H.; Huang, B.; Niu, P.; et al. Both Boceprevir and GC376 efficaciously inhibit SARS-CoV-2 by targeting its main protease. Nat. Commun. 2020, 11, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ogihara, T.; Nakagawa, M.; Ishikawa, H.; Mikami, H.; Takeda, K.; Nonaka, H.; Nagano, M.; Sasaki, S.; Kagoshima, T.; Higashimori, K. Effect of manidipine, a novel calcium channel blocker, on quality of life in hypertensive patients. Blood Press. Suppl. 1992, 3, 135–139. [Google Scholar]
- De Wilde, A.H.; Jochmans, D.; Posthuma, C.C.; Zevenhoven-Dobbe, J.C.; Van Nieuwkoop, S.; Bestebroer, T.M.; van den Hoogen, B.G.; Neyts, J.; Snijder, E.J. Screening of an FDA-Approved Compound Library Identifies Four Small-Molecule Inhibitors of Middle East Respiratory Syndrome Coronavirus Replication in Cell Culture. Antimicrob. Agents Chemother. 2014, 58, 4875–4884. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maffucci, I.; Contini, A. In Silico Drug Repurposing for SARS-CoV-2 Main Proteinase and Spike Proteins. J. Proteome Res. 2020, 19, 4637–4648. [Google Scholar] [CrossRef] [PubMed]
- McClellan, K.J.; Jarvis, B. Lercanidipine: A review of its use in hypertension. Drugs 2000, 60, 1123–1140. [Google Scholar] [CrossRef]
- Mahajan, R. Bedaquiline: First FDA-approved tuberculosis drug in 40 years. Int. J. Appl. Basic Med. Res. 2013, 3, 1–2. [Google Scholar] [CrossRef]
- Rose, P.W.; Bi, C.; Bluhm, W.F.; Christie, C.H.; Dimitropoulos, D.; Dutta, S.; Green, R.K.; Goodsell, D.S.; Prlić, A.; Quesada, M.; et al. The RCSB Protein Data Bank: New resources for research and education. Nucleic Acids Res. 2012, 41, D475–D482. [Google Scholar] [CrossRef] [PubMed]
- Sacco, M.D.; Ma, C.; Lagarias, P.; Gao, A.; Townsend, J.A.; Meng, X.; Dube, P.; Zhang, X.; Hu, Y.; Kitamura, N.; et al. Structure and inhibition of the SARS-CoV-2 main protease reveal strategy for developing dual inhibitors against Mpro and cathepsin L. Sci. Adv. 2020, 6, eabe0751. [Google Scholar] [CrossRef]
- Wheeler, D.L.; Barrett, T.; Benson, D.A.; Bryant, S.H.; Canese, K.; Chetvernin, V.; Church, D.M.; DiCuccio, M.; Edgar, R.; Federhen, S.; et al. Database resources of the National Center for Biotechnology Information. Nucleic Acids Res. 2006, 35, D5–D12. [Google Scholar] [CrossRef] [PubMed]
- Webb, B.; Sali, A. Comparative Protein Structure Modeling Using MODELLER. Curr. Protoc. Bioinform. 2016, 54, 5–6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pontius, J.; Richelle, J.; Wodak, S.J. Deviations from Standard Atomic Volumes as a Quality Measure for Protein Crystal Structures. J. Mol. Biol. 1996, 264, 121–136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eisenberg, D.; Lüthy, R.; Bowie, J.U. [20] VERIFY3D: Assessment of protein models with three-dimensional profiles. Meth. Enzymol. 1997, 277, 396–404. [Google Scholar] [CrossRef]
- Laskowski, R.A.; MacArthur, M.W.; Moss, D.S.; Thornton, J.M. PROCHECK: A program to check the stereochemical quality of protein structures. J. Appl. Crystallogr. 1993, 26, 283–291. [Google Scholar] [CrossRef]
- Kim, S.; Thiessen, P.A.; Bolton, E.E.; Chen, J.; Fu, G.; Gindulyte, A.; Han, L.; He, J.; He, S.; Shoemaker, B.A.; et al. PubChem substance and compound databases. Nucleic Acids Res. 2016, 44, D1202–D1213. [Google Scholar] [CrossRef]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef] [Green Version]
- Jones, G.; Willett, P.; Glen, R.C.; Leach, A.R.; Taylor, R. Development and validation of a genetic algorithm for flexible docking. J. Mol. Biol. 1997, 267, 727–748. [Google Scholar] [CrossRef] [Green Version]
- Schrodinger Inc. PyMOL, Molecular Visualization System, Version 2.4; Schrodinger Inc.: New York, NY, USA, 2002. [Google Scholar]
- Pronk, S.; Páll, S.; Schulz, R.; Larsson, P.; Bjelkmar, P.; Apostolov, R.; Shirts, M.R.; Smith, J.C.; Kasson, P.M.; Van Der Spoel, D.; et al. GROMACS 4.5: A high-throughput and highly parallel open source molecular simulation toolkit. Bioinformatics 2013, 29, 845–854. [Google Scholar] [CrossRef]
- Hess, B.; Kutzner, C.; Van Der Spoel, D.; Lindahl, E. GROMACS 4: Algorithms for Highly Efficient, Load-Balanced, and Scalable Molecular Simulation. J. Chem. Theory Comput. 2008, 4, 435–447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Christen, M.; Hünenberger, P.H.; Bakowies, D.; Baron, R.; Bürgi, R.; Geerke, D.P.; Heinz, T.N.; Kastenholz, M.A.; Kräutler, V.; Oostenbrink, C.; et al. The GROMOS software for biomolecular simulation: GROMOS05. J. Comput. Chem. 2005, 26, 1719–1751. [Google Scholar] [CrossRef]
- Liu, X.; Shi, D.; Zhou, S.; Liu, H.; Liu, H.; Yao, X. Molecular dynamics simulations and novel drug discovery. Expert Opin. Drug Discov. 2018, 13, 23–37. [Google Scholar] [CrossRef]
- Bao, Y.; Zhou, L.; Dai, D.; Zhu, X.; Hu, Y.; Qiu, Y. Discover potential inhibitors for PFKFB3 using 3D-QSAR, virtual screening, molecular docking and molecular dynamics simulation. J. Recept. Signal Transduct. 2018, 38, 413–431. [Google Scholar] [CrossRef] [PubMed]
- Mark, P.; Nilsson, L. A Molecular Dynamics Study of Tryptophan in Water. J. Phys. Chem. B 2002, 106, 9440–9445. [Google Scholar] [CrossRef]
- Mark, P.; Nilsson, L. Structure and Dynamics of the TIP3P, SPC, and SPC/E Water Models at 298 K. J. Phys. Chem. A 2001, 105, 9954–9960. [Google Scholar] [CrossRef]
- Darden, T.; York, D.; Pedersen, L. Particle mesh Ewald: An N⋅log(N) method for Ewald sums in large systems. J. Chem. Phys. 1993, 98, 10089–10092. [Google Scholar] [CrossRef] [Green Version]
- Hess, B.; Bekker, H.; Berendsen, H.J.; Fraaije, J.G. LINCS: A linear constraint solver for molecular simulations. J. Comput. Chem. 1997, 18, 1463–1472. [Google Scholar] [CrossRef]
- Hess, B. P-LINCS: A Parallel Linear Constraint Solver for Molecular Simulation. J. Chem. Theory Comput. 2007, 4, 116–122. [Google Scholar] [CrossRef]
- Izaguirre, J.A.; Catarello, D.P.; Wozniak, J.M.; Skeel, R.D. Langevin stabilization of molecular dynamics. J. Chem. Phys. 2001, 114, 2090–2098. [Google Scholar] [CrossRef]
- Berendsen, H.J.C.; Postma, J.P.M.; Van Gunsteren, W.F.; DiNola, A.; Haak, J.R. Molecular dynamics with coupling to an external bath. J. Chem. Phys. 1984, 81, 3684–3690. [Google Scholar] [CrossRef] [Green Version]
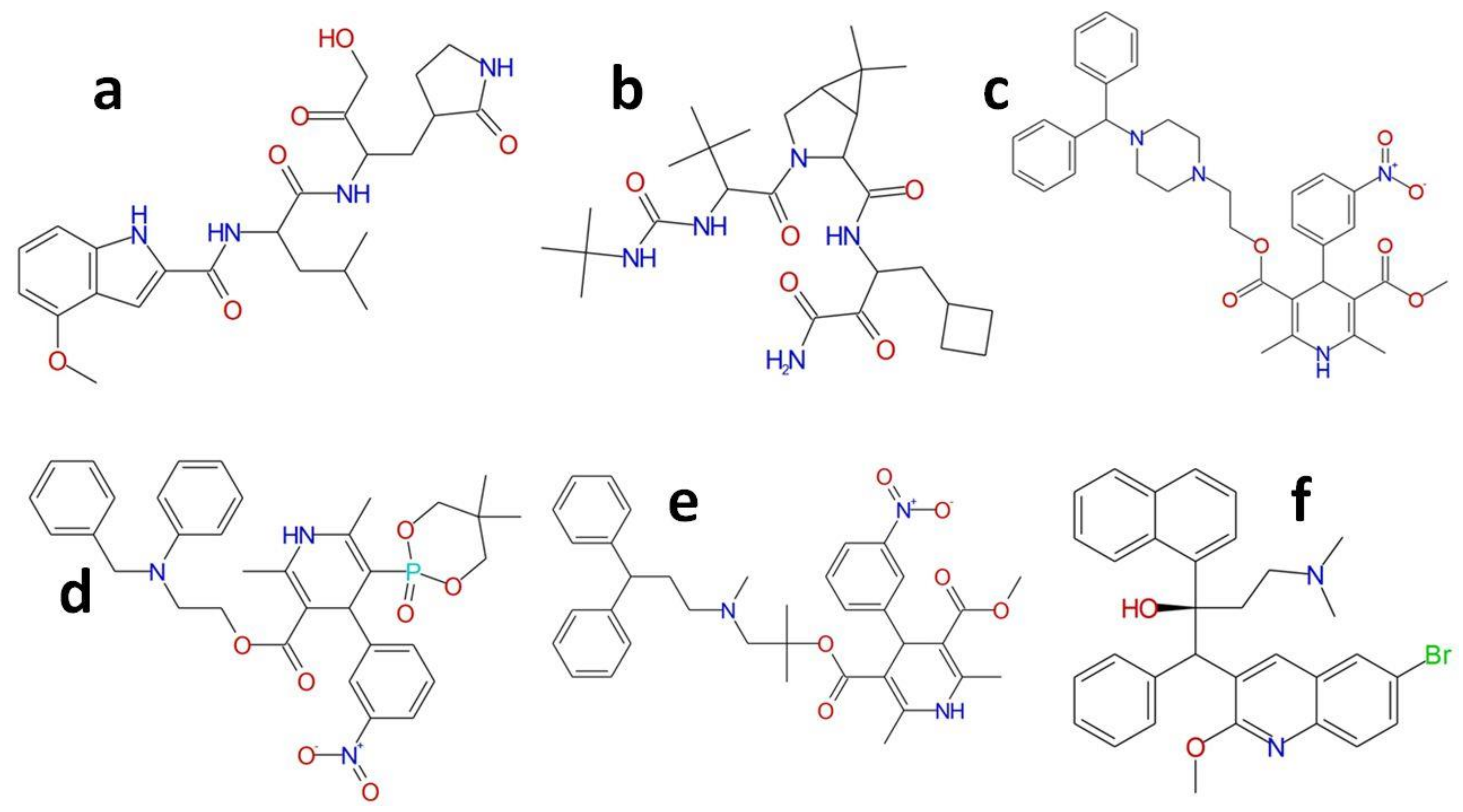




| Protein | Inhibitor | Score | Residues | |
|---|---|---|---|---|
| Hydrogen Bond | Pi Interactions | |||
| WT | PF-00835231 | 83.13 | G143, C145, H164, E166, Q189 | H41, P168 |
| Manidipine | 72.93 | F140, N142, E166 | H41, L141, M165 | |
| Boceprevir | 67.42 | N142, E166, Q189 | H41, M49, C145 | |
| Lercanidipine | 69.73 | Q189, Q192 | C145, M165, L167, P168 | |
| Efonidipine | 71.23 | H41, C145, M165, P168, A191 | ||
| Bedaquiline | 66.92 | N142 | H41 | |
| Protein | Inhibitor | Score | Residues | |
|---|---|---|---|---|
| Hydrogen Bond | Pi Interactions | |||
| Mutant 1 (Y54C) | PF-00835231 | 73.17 | L141, G143, S144, C145, E166, Q189 | M49, P168 |
| Manidipine | 66.68 | T26, S46 | C145, M165 | |
| Boceprevir | 65.96 | T26, G143, C145, E166, Q189 | H41, M49, H163, M165 | |
| Lercanidipine | 65.28 | T26, G143, C145 | L27, H41, M165 | |
| Efonidipine | 68.23 | L141, G143, S144, Q189 | L27, C145, H163, P168, H172 | |
| Bedaquiline | 63.27 | H164 | L27, H41, M49, C145 | |
| Protein | Inhibitor | Score | Residues | |
|---|---|---|---|---|
| Hydrogen Bond | Pi Interaction | |||
| Mutant 2 N142S | PF-00835231 | 76.53 | L141, S142, G143, E166, Q189 | S144, C145, L167, P168 |
| Manidipine | 68.64 | F140, E166 | L141, M165 | |
| Boceprevir | 65.33 | Q189 | C44, M49, C145, H163, M165 | |
| Lercanidipine | 69.05 | L27, M49, C145, H163 | ||
| Efonidipine | 87.05 | S142, C145 | L27, M49, M165, L167 | |
| Bedaquiline | 68.83 | H164 | H41, M49, C145, M165 | |
| Protein | Inhibitor | Score | Residues | |
|---|---|---|---|---|
| Hydrogen Bond | Pi Interactions | |||
| Mutant 3 T190I | PF-00835231 | 70.66 | E166, D187, Q189 | C44, M49, C145, H163, M165 |
| Manidipine | 63.42 | F140, N142 | M49, L141, M165 | |
| Boceprevir | 60.36 | N142 | C44, T45, M165 | |
| Lercanidipine | 66.96 | T26 | L27, H41, C44, M49, C145, M165 | |
| Efonidipine | 72.25 | F140, E166 | L141, H163, M165, H172 | |
| Bedaquiline | 67.87 | E166 | H41, C145 | |
| Protein | Inhibitor | Score | Residues | |
|---|---|---|---|---|
| Hydrogen Bond | Pi Interaction | |||
| Mutant 4 A191V | PF-00835231 | 73.61 | L141, G143, S144, C145, Q189 | M165, P168, H172, V191 |
| Manidipine | 65.95 | T25, S46, N142 | M165 | |
| Boceprevir | 62.08 | S46, Q189 | C44, M49, M165 | |
| Lercanidipine | 67.93 | C44, S46, N142 | H41, M49, C145, M165 | |
| Efonidipine | 71.01 | F140 | L141, H163, M165, P168 | |
| Bedaquiline | 59.99 | N142 | H41, C145, M165 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baig, M.H.; Sharma, T.; Ahmad, I.; Abohashrh, M.; Alam, M.M.; Dong, J.-J. Is PF-00835231 a Pan-SARS-CoV-2 Mpro Inhibitor? A Comparative Study. Molecules 2021, 26, 1678. https://doi.org/10.3390/molecules26061678
Baig MH, Sharma T, Ahmad I, Abohashrh M, Alam MM, Dong J-J. Is PF-00835231 a Pan-SARS-CoV-2 Mpro Inhibitor? A Comparative Study. Molecules. 2021; 26(6):1678. https://doi.org/10.3390/molecules26061678
Chicago/Turabian StyleBaig, Mohammad Hassan, Tanuj Sharma, Irfan Ahmad, Mohammed Abohashrh, Mohammad Mahtab Alam, and Jae-June Dong. 2021. "Is PF-00835231 a Pan-SARS-CoV-2 Mpro Inhibitor? A Comparative Study" Molecules 26, no. 6: 1678. https://doi.org/10.3390/molecules26061678







