Synthesis and Biological Evaluation of 2,3,4-Triaryl-1,2,4-oxadiazol-5-ones as p38 MAPK Inhibitors
Abstract
:1. Introduction
2. Results and Discussion
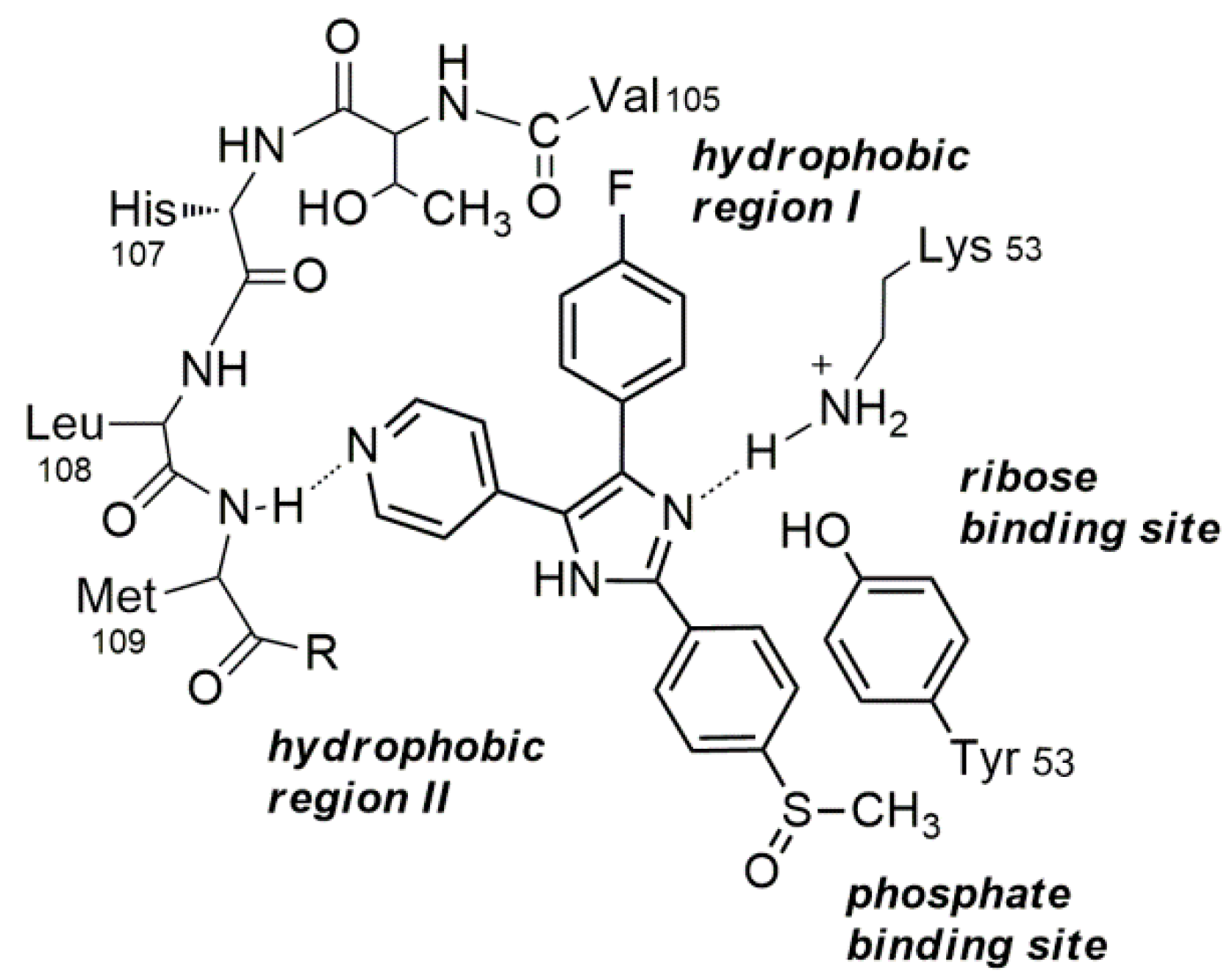
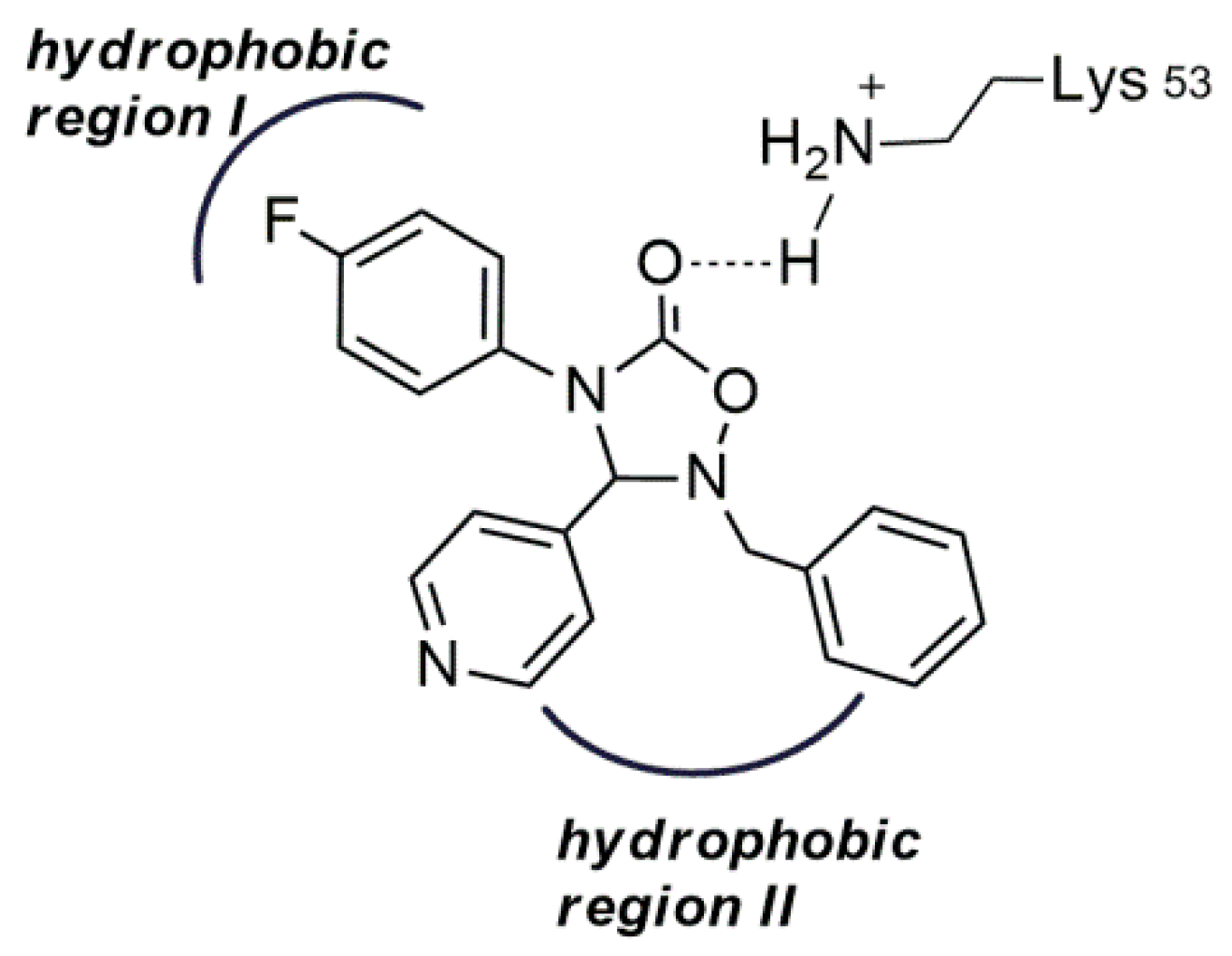
2.1. Design
2.2. Synthesis of 2,4-Oxazolidinyl-5-Ones (Azastilbenes)
2.3. Inhibition of p38α MAPK for Compounds 3a–l
3. Experimental Section
3.1. Materials and Methods
3.2. General Procedure
3.3. p38 MAP Kinase Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Rivière, C.; Pawlus, A.D.; Mérillon, J.-M. Natural stilbenoids: Distribution in the plant kingdom and chemotaxonomic interest in Vitaceae. Nat. Prod. Rep. 2012, 29, 1317–1333. [Google Scholar] [CrossRef] [PubMed]
- Esatbeyoglu, T.; Ewald, P.; Yasui, Y.; Yokokawa, H.; Wagner, A.E.; Matsugo, S.; Winterhalter, P.; Rimbach, G. Chemical characterization, free radical scavenging, and cellular antioxidant and anti-inflammatory properties of a stilbenoid-rich root extract of VitisVinifera. Oxid. Med. Cell. Longev. 2016, 8591286, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Delaunois, B.; Cordelier, S.; Conreux, A.; Clément, C.; Jeandet, P. Molecular engineering of resveratrol in plants. Plant Biotechnol. J. 2009, 7, 2–12. [Google Scholar] [CrossRef]
- Jeandet, P.; Delaunois, B.; Conreux, A.; Donnez, D.; Nuzzo, V.; Cordelier, S.; Clément, C.; Courot, E. Biosynthesis, metabolism, molecular engineering and biological functions of phytoalexins in plants. Bio Factors 2010, 36, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Jeandet, P.; Delaunois, B.; Azi, A.; Donnez, D.; Vasserot, Y.; Cordelier, S.; Courot, E. Molecular engineering of yeast and plants for the production of the biological active hydroxystilbene, resveratrol. J. Biomed. Biotechnol. 2012, 579089, 1–15. [Google Scholar] [CrossRef]
- Jeandet, P.; Clément, C.; Courot, E.; Cordelier, S. Modulation of phytoalexin biosynthesis in engineered plants for disease resistance. Int. J. Mol. Sci. 2013, 14, 14136–14170. [Google Scholar] [CrossRef] [Green Version]
- Jeandet, P.; Hébrard, C.; Deville, M.A.; Cordelier, S.; Dorey, S.; Aziz, A.; Crouzet, J. Deciphering the role of phytoalexins in plant-microorganism interactions and human health. Molecules 2014, 19, 18033–18056. [Google Scholar] [CrossRef] [Green Version]
- Carrizzo, A.; Forte, M.; Damato, A.; Trimarco, V.; Salzano, F.; Bartolo, M.; Maciag, A.; Puca, A.A.; Vecchione, C. Antioxidants effects of resveratrol in cardiovascular, cerebral and metabolic diseases. Food Chem. Toxicol. 2013, 61, 215–226. [Google Scholar] [CrossRef] [PubMed]
- Muller, C.; Ullmann, K.; Wilkens, A.; Winterhalter, P.; Toyokuni, S.; Steinberg, P. Potent antioxidative activity of Vineatrol 30 Grapevine-shoot extracts. Biosci. Biotechnol. Biochem. 2009, 73, 1831–1836. [Google Scholar] [CrossRef] [Green Version]
- Gülçin, I. Antioxidant properties of resveratrol: A structure-activity insight. Innov. Food Sci. Emerg. Technol. 2010, 11, 210–218. [Google Scholar] [CrossRef]
- Minagawa, T.; Okui, T.; Takahashi, N.; Nakajima, T.; Tabeta, K.; Murakami, S.; Yamazaki, K. Resveratrol suppresses the inflammatory response of human gingival cells in a SIRT1 independent manner. J. Periodontal Res. 2015, 50, 586–593. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.; Schueller, K.; Schaefer, L.M.; Pignitter, M.; Esefelder, L.; Somoza, V. Resveratrol and its metabolites inhibit pro-inflammatory effect of lypopolysaccharides in U-937 macrophages in plasma-representative concentrations. Food Funct. 2014, 5, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Bishayee, A.; Barnes, K.F.; Bhatia, D.; Darvesh, A.S.; Carroll, R.T. Resveratrol suppresses oxidative stress and inflammatory response in diethylnitrosamine-initiated rat hepatocarcinogenesis. Cancer Prev. Res. 2010, 3, 753–763. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, C.; Fang, J.S.; Lian, W.W.; Pang, X.C.; Liu, A.; Du, G. In vitro antiviral effects and 3D QSAR study of resveratrol derivatives as potent inhibitors of influenza H1N1 neuraminidase. Chem. Biol. Drug Des. 2015, 85, 427–438. [Google Scholar] [CrossRef]
- Berrougui, H.; Grenier, G.; Loued, S.; Drouin, G.; Khalil, A. A new insight into resveratrol as an atheroprotective compound: Inhibition of lipid peroxidation and enhancement of cholesterol efflux. Atherosclerosis 2009, 207, 420–427. [Google Scholar] [CrossRef]
- Ma, T.; Tan, M.; Yu, J.; Tan, L. Resveratrol as a therapeutic agent for Alzheimer’s disease. BioMed Res. Int. 2014, 350516, 1–13. [Google Scholar] [CrossRef]
- Wu, J.M.; Hsieh, T. Resveratrol: A cardioprotective substance. Ann. N. Y. Acad. Sci. 2011, 1215, 16–21. [Google Scholar] [CrossRef]
- Szkudelski, T.; Szkudelska, K. Resveratrol and diabetes: From animal to human studies. Biochim. Biophys. Acta 2015, 1852, 1145–1154. [Google Scholar] [CrossRef] [Green Version]
- Fulda, S. Resveratrol and derivatives for the prevention and treatment of cancer. Drug Discov. Today 2010, 15, 757–765. [Google Scholar] [CrossRef]
- Nivelle, L.; Hubert, J.; Courot, E.; Jeandet, P.; Aziz, A.; Nuzillard, J.M.; Renault, J.H.; Clément, C.; Martiny, L.; Delmas, D.; et al. Anti-cancer activity of resveratrol and derivatives produced by Grapevine cell suspension in a 14 l stirred bioreactor. Molecules 2017, 22, 474. [Google Scholar] [CrossRef]
- Aluyen, J.K.; Ton, Q.N.; Tran, T.; Yang, A.E.; Gottlieb, H.B.; Bellanger, R.A. Resveratrol: Potential as anti-cancer agent. J. Diet. Suppl. 2012, 9, 45–56. [Google Scholar] [CrossRef]
- Metzler, M.; Neumann, H.G. Epoxidation of the stilbene double bond, a major pathway in aminostilbene metabolism. Xenobiotica 1977, 7, 117–132. [Google Scholar] [CrossRef]
- Tron, G.C.; Pirali, T.; Sorba, G.; Pagliai, F.; Busacca, S.; Genazzani, A.A. Medicinal chemistry of Combretastatin A4: Present and future directions. J. Med. Chem. 2006, 49, 3033–3044. [Google Scholar] [CrossRef]
- Ohsumi, K.; Hatanaka, T.; Fujita, K.; Nakagawa, R.; Fukuda, Y.; Nihei, Y.; Suga, Y.; Morinaga, Y.; Akiyama, Y.; Tsuji, T. Synthesis and antitumor activity of cis-restricted combretastatins: 5-membered heterocyclic analogues. Bioorg. Med. Chem. Lett. 1998, 8, 3153–3158. [Google Scholar] [CrossRef]
- Mateo, C.; Perez-Melero, C.; Pelaez, R.; Medarde, M. Stilbenophane analogues of deoxycombretastatin A-4. J. Org. Chem. 2005, 70, 6544–6547. [Google Scholar] [CrossRef]
- Nam, N.H. Combretastatin A-4 analogues as antimitotic antitumor agents. Curr. Med. Chem. 2003, 10, 1697–1722. [Google Scholar] [CrossRef]
- Nam, N.H.; Kim, Y.; You, Y.J.; Hong, D.H.; Kim, H.M.; Ahn, B.Z. Synthesis and anti-tumor activity of novel combretastatins: Combretocyclopentenones and related analogues. Bioorg. Med. Chem. Lett. 2002, 12, 1955–1958. [Google Scholar] [CrossRef]
- Kim, Y.; Nam, N.H.; You, Y.J.; Ahn, B.Z. Synthesis and citotoxicity of 3,4-diaryl- 2(5H)-furanones. Bioorg. Med. Chem. Lett. 2002, 12, 719–722. [Google Scholar] [CrossRef]
- Wang, L.; Woods, K.W.; Li, Q.; Barr, K.J.; McCroskey, R.W.; Hannick, S.M.; Gherke, L.; Credo, R.B.; Hui, Y.H.; Marsh, K.; et al. Potent, orally active heterocyclic-based combretastatin A-4 analogues: Synthesis, structure-activity relationship, pharmacokinetics, and in vivo antitumor activity evaluation. J. Med. Chem. 2002, 45, 1697–1711. [Google Scholar] [CrossRef]
- Simoni, D.; Grisolia, G.; Giannini, G.; Roberti, M.; Rondanin, R.; Piccagli, L.; Baruchello, R.; Rossi, M.; Romagnoli, R.; Invidiata, F.P.; et al. Heterocyclic and phenyl double-bonded-locked combretastyatin analogues possessing potent apotosis inducing activity in HL60 and in MDR cell lines. J. Med. Chem. 2005, 48, 723–736. [Google Scholar] [CrossRef]
- Nam, N.H.; Kim, Y.; You, Y.J.; Hong, D.H.; Kim, H.M.; Ahn, B.Z. Combretoxazsolones: Synthesis, cytotoxicity and antitumor activity. Bioorg. Med. Chem. Lett. 2001, 11, 3073–3076. [Google Scholar] [CrossRef]
- Tron, G.C.; Pagliai, F.; Del Grosso, E.; Genazzani, A.A.; Sorba, G. Synthesis and cytotoxic evaluation of combretafurazans. J. Med. Chem. 2005, 48, 3260–3268. [Google Scholar] [CrossRef]
- Pati, H.N.; Wicks, M.; Holt, H.L.; LeBlanc, R.; Weisbruch, P.; Forest, L.; Lee, M. Synthesis and biological avaluation of cis-combretastatin analogues and their novel 1,2,3-triazole derivatives. Heterocycl. Commun. 2005, 11, 117–120. [Google Scholar] [CrossRef]
- Flynn, B.L.; Flynn, G.P.; Hamel, E.; Jung, M.K. The synthesis and tubulin binding activity of tiophene-based analogues of combretastatin A–4. Bioorg. Med. Chem. Lett. 2001, 11, 2341–2343. [Google Scholar] [CrossRef]
- Bailly, C.; Bal, C.; Barbier, P.; Combes, S.; Finet, J.P.; Hildebrand, M.P.; Peyrot, N.; Wattez, N. Synthesis and biological evaluation of 4-arylcoumarin analogues of combretastatins. J. Med. Chem. 2003, 46, 5437–5444. [Google Scholar] [CrossRef]
- Sharma, V.M.; AdiSeshu, K.V.; Krishna, C.V.; Prasanna, P.; Sekhar, V.C.; Venkateswarlu, A.; Rajagopal, S.; Ayaykumar, R.; Deevi, D.S.; Rao Mamidi, N.V.S.; et al. Novel 6,7-diphenyl-2,3,8,8a-tetrahydro-1H-indolizin-5-one analogues as cyctotoxic agents. Bioorg. Med. Chem. Lett. 2003, 13, 1679–1682. [Google Scholar] [CrossRef]
- Jonnalagadda, S.S.; Haar, E.; Hamel, E.; Lin, C.M.; Magarian, R.A.; Day, B.W. Synthesis and biological evaluation of 1,1-dichloro-2,3-diarylcyclopropanes as antitubulin and anti-breast cancer agents. Bioorg. Med. Chem. 1997, 5, 715–722. [Google Scholar] [CrossRef]
- Hadfield, J.A.; Gaukroger, K.; Hirst, N.; Weston, A.P.; Lawrence, N.J.; McGown, A.T. Synthesis and evaluation of double bond substituted combretastatins. Eur. J. Med. Chem. 2005, 40, 529–541. [Google Scholar] [CrossRef]
- Sun, L.; Vasilevich, N.I.; Fuselier, J.A.; Hocart, S.J.; Coy, D.H. Examination of the 1,4-disubstituted azetidinone ring system as a template for combretastatin A4 conformationally restricted analogue design. Bioorg. Med. Chem. Lett. 2004, 14, 2041–2046. [Google Scholar] [CrossRef]
- Pearson, G.; Robinson, F.; Beers Gibson, T.; Xu, B.E.; Karandikar, M.; Berman, K.; Cobb, M.H. Mitogen-activated protein (MAP) kinase pathway: Regulation and physiological functions. Endocr. Rev. 2001, 22, 153–183. [Google Scholar]
- Chen, Z.; Gibson, T.B.; Robinson, F.; Silvestro, L.; Pearson, G.; Xu, B.; Wright, A.; Vanderbilt, C.; Cobb, M.H. MAP kinases. Chem. Rev. 2001, 101, 2449–2476. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.D.; Bierhaus, A.; Nawroth, P.P.; Stern, D.M. RAGE and Alzheimer’s disease: A progression factor for amyloid-beta-induced perturbation? J. Alzheimer’s Dis. 2009, 16, 833–843. [Google Scholar] [CrossRef] [Green Version]
- Bachstetter, A.D.; Xing, B.; de Almeida, L.; Dimayuga, E.R.; Watterson, D.M.; Van Eldik, L.J. Microglial p38α MAPK is a key regulator of proinflammatory cytokine up-regulation induced by toll-like receptor (TLR) ligands or beta-amyloid (Aβ). J. NeuroInflamm. 2011, 8, 79–83. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Z.; Bachstetter, A.D.; Späni, C.B.; Roy, S.M.; Watterson, D.M.; Van Eldik, L.J. Retention of normal glia function by an isoform-selective protein kinase inhibitor drug candidate that modulates cytokine production and cognitive outcomes. J. NeuroInflamm. 2017, 14, 75–79. [Google Scholar] [CrossRef] [Green Version]
- Wei, S.; Siegal, G.P. Mechanisms modulating inflammatory osteolysis: A review with insights into therapeutic targets. Pathol. Res. Pract. 2008, 204, 695–706. [Google Scholar] [CrossRef] [Green Version]
- Barnes, P.J. Kinases as novel therapeutic targets in asthma and chronic obstructive pulmonary disease. Pharmacol. Rev. 2016, 68, 788–815. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Ding, L.; Ji, H.; Xu, Z.; Liu, Q.; Zheng, Y. The role of MAPK in the development of diabetic cardiomyopahy. Int. J. Mol. Sci. 2016, 17, 1037. [Google Scholar] [CrossRef] [Green Version]
- Segalés, J.; Perdiguero, E.; Muñoz-Cánoves, P. Regulation of muscle stem cell functions: A focus on the p38 MAPK signaling pathway. Front. Cell. Dev. Biol. 2016, 4, 91–97. [Google Scholar] [CrossRef] [Green Version]
- Lang, E.; Bissinger, R.; Qadri, S.M.; Lang, F. Suicidal death of erythrocytes in cancer and its chemotherapy: A potential target in the treatment of tumor-associated anemia. Int. J. Cancer 2017, 141, 1522–1528. [Google Scholar] [CrossRef] [Green Version]
- Bonney, E.A. Mapping out p38 MAPK. Am. J. Reprod. Immunol. 2017, 77, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vlahopoulos, S.A. Aberrant control of permits transcriptional and phentypic NF-kB in cancer plasticity, to curtail dependence on host tissue: Molecular mode. Cancer Biol. Med. 2017, 14, 254–270. [Google Scholar] [PubMed] [Green Version]
- Yong, H.Y.; Koh, M.S.; Moon, A. The p38 MAPK inhibitors for the treatment of inflammatory diseases and cancer. Expert Opin. Invest. Drugs 2009, 18, 1893–1905. [Google Scholar] [CrossRef]
- Chen, L.; Mayer, J.A.; Krisko, T.I.; Speers, C.W.; Wang, T.; Hilsenbeck, S.G.; Brown, P.H. Inhibition of the p38 kinase suppresses the proliferation of human ER-negative breast cancer cells. Cancer Res. 2009, 69, 8853–8861. [Google Scholar] [CrossRef] [Green Version]
- Bradham, C.; McClay, D.R. p38 MPKA in development and cancer. Cell Cycle 2006, 5, 824–828. [Google Scholar] [CrossRef]
- Davidson, B.; Givant-Horwitz, V.; Lazarovici, P.; Risberg, B.; Nesland, J.; Trope, C.; Schaefer, E.; Reich, R. Matrix metalloproteinases (MMP), EMMPRIN (extracellular matrix metalloproteinase inducer) and mitogen-activated protein kinases (MAPK): Coexpression in metastatic serous ovarian carcinoma. Clin. Exp. Metastasis 2003, 20, 621–631. [Google Scholar] [CrossRef]
- Peifer, C.; Abadleh, M.; Bischof, J.; Hauser, D.; Schattel, V.; Hirner, H.; Knippschild, U.; Laufer, S. 3,4-Diaryl-isoxazoles and –imidazoles as potent dual inhibitors of p38α mitogen activated protein kinase and casein kinase 1δ. J. Med. Chem. 2009, 52, 7618–7630. [Google Scholar] [CrossRef]
- Laufer, S.A.; Margutti, S.; Fritz, M.D. Substituted isoxazoles as potent inhibitors of p38 MAP kinase. ChemMedChem 2006, 1, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.K.; Selness, S.R.; Devraj, R.V.; Hepperle, M.E.; Naing, W.; Shieh, H.; Kurambail, R.; Yang, S.; Flynn, D.L.; Benson, A.G.; et al. Identification of SD-0006, a potent diarylpyrazole inhibitor of p38 MAP kinase. Bioorg. Med. Chem. Lett. 2010, 20, 2634–2638. [Google Scholar] [CrossRef]
- Graneto, M.J.; Kurumbail, R.G.; Vazquez, M.L.; Shieh, H.S.; Pawlitz, J.L.; Williams, J.M.; Stallings, W.C.; Geng, L.; Naraian, A.S.; Koszyk, F.J.; et al. Synthesis, crystal structure and activity of pyrazole-based inhibitors of p38 kinase. J. Med. Chem. 2007, 50, 5712–5719. [Google Scholar] [CrossRef] [PubMed]
- Dinér, P.; VeideVilg, J.; Kjellén, J.; Migdal, I.; Andersson, T.; Gebbia, M.; Giaever, G.; Nislow, C.; Hohmann, S.; Wysocki, R.; et al. Design, synthesis, and characterization of a highly effective Hog1 inhibitor: A powerful tool for analyzing MAP kinase signaling in yeast. PLoS ONE 2011, 6, 20012. [Google Scholar] [CrossRef] [Green Version]
- Dinér, P.; Andersson, T.; Kjellén, J.; Elbing, K.; Hohmann, S.; Grøtli, M. Short cut to 1,2,3-triazole-based p38 MAP kinase inhibitors via [3+2]-cycloaddition chemistry. New J. Chem. 2009, 33, 1010–1016. [Google Scholar] [CrossRef]
- Tullis, J.S.; Van, R.J.C.; Natchus, M.G.; Clark, M.P.; De, B.; Hsieh, L.C.; Janusz, M.J. The development of new triazole based inhibitors of tumor necrosis factor-alpha (TN-alpha) production. Bioorg. Med. Chem. Lett. 2003, 13, 1665–1668. [Google Scholar] [CrossRef]
- Revesz, L.; Di Padova, F.E.; Buhl, T.; Feifel, R.; Gram, H.; Hiestand, P.; Manning, U.; Wolf, R.; Zimmerlin, A.G. SAR of 2,6-diamino-3,5-difluoropyridinyl substituted heterocycles as novel p38 MAP kinase inhibitors. Bioorg. Med. Chem. Lett. 2002, 12, 2109–2112. [Google Scholar] [CrossRef]
- Peifer, C.; Urich, R.; Schattel, V.; Abadleh, M.; Roettig, M.; Kohlbacher, O.; Laufer, S. Implication for selectivity of 3,4-diarylquinolinones as p38 alpha MAP kinase inhibitors. Bioorg. Med. Chem. Lett. 2008, 18, 1431–1435. [Google Scholar] [CrossRef] [PubMed]
- Peifer, C.; Kinkel, K.; Abadleh, M.; Schollmeyer, D.; Laufer, S. From five- to six-membered rings: 3,4-diarylquinolinone as lead for novel p38MAP kinase inhibitors. J. Med. Chem. 2007, 50, 1213–1221. [Google Scholar] [CrossRef] [PubMed]
- Laufer, S.A.; Wagner, G.K. From imidazoles to pyrimidines: New inhibitors of cytokine release. J. Med. Chem. 2002, 45, 2733–2740. [Google Scholar] [CrossRef]
- Dyrager, C.; Möllers, L.N.; Kjäll, L.K.; Alao, J.P.; Dinér, P.; Wallner, F.K.; Sunnerhagen, P.; Grøtli, M. Design, synthesis and biological evaluation of chromone-based p38MAP kinase inhibitors. J. Med. Chem. 2011, 54, 7427–7431. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gum, R.J.; Mclaughlin, M.M.; Kumar, S.; Wang, Z.; Bower, M.J.; Lee, J.C.; Adams, J.L.; Livi, G.P.; Goldsmith, E.J.; Young, P.R. Acquisition of sensitivity of stress- activated protein kinases to the p38 inhibitor, SB 203580, by alteration of one or more amino acids within the ATP binding pocket. J. Biol. Chem. 1998, 273, 15605–15610. [Google Scholar] [CrossRef] [Green Version]
- Chiacchio, M.A.; Legnani, L.; Campisi, A.; Bottino, P.; Iannazzo, D.; Veltri, L.; Giofrè, S.; Romeo, R. 1,2,4-oxadiazole-5-ones as analogues of tamoxifen: Synthesia and biological evaluation. Org. Biomol. Chem. 2019, 17, 4892–4905. [Google Scholar] [CrossRef]
- Romeo, R.; Carnovale, C.; Giofrè, S.V.; Chiacchio, M.A.; Garozzo, A.; Amata, E.; Romeo, G. C-5′-triazolyl-2′-oxa-3′-aza-4′a-carbanucleosides: Synthesis and biological evaluation. Beilstein, J. Org. Chem. 2015, 11, 328–334. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Romeo, R.; Giofrè, S.V.; Garozzo, A.; Bisignano, B.; Corsaro, A.; Chiacchio, M.A. Synthesis and biological evaluation of furopyrimidineN.,O.-nucleosides. Bioorg. Med. Chem. 2013, 21, 5688–5693. [Google Scholar] [CrossRef]
- Rescifina, A.; Chiacchio, M.A.; Corsaro, A.; DeClercq, E.; Iannazzo, D.; Mastino, A.; Piperno, A.; Romeo, R.; Valveri, V. Synthesis and biological activity of isoxazolidinyl polycyclic aromatic hydrocarbons: Potential DNA intercalators. J. Med. Chem. 2006, 49, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Piperno, A.; Rescifina, A.; Corsaro, A.; Chiacchio, M.A.; Procopio, A.; Romeo, R. A novel class of modified nucleosides: Synthesis of alkylidenes isoxazolidinyl nucleosides containing thymine. Eur. J. Org. Chem. 2007, 1517–1521. [Google Scholar] [CrossRef]
- Darù, A.; Roca-Lopez, D.; Tejero, T.; Merino, P. Revealing stepwise mechanisms in dipolar cycloaddition reaction: Computational study of the reaction between nitrones and isocyanates. J. Org. Chem. 2016, 81, 673–680. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Laufer, S.; Thuma, S.; Peifer, C.; Greim, C.; Herweh, Y.; Albrecht, A.; Dehner, F. An immunosorbent, nonradioactive p38 MAP kinase assay comparable to standard radioactive liquid-phase assays. Anal. Biochem. 2005, 344, 135–137. [Google Scholar] [CrossRef] [PubMed]




| Entry | R1 a | R2 a | Ar | Product (Yield%) |
|---|---|---|---|---|
| 1 | Ph | Py | 4-NO2-C6H4 | 3a (76%) |
| 2 | Ph | Py | 4-F-C6H4 | 3b (77%) |
| 3 | Ph | Py | 4-CF3-C6H4 | 3c (75%) |
| 4 | Ph | Py | 4-Cl-C6H4 | 3d (78%) |
| 5 | PhCH2 | Py | 4-F-C6H4 | 3e (62%) |
| 6 | PhCH2 | Py | 4-NO2-C6H4 | 3f (58%) |
| 7 | PhCH2 | Ph | 4-F-C6H4 | 3g (60%) |
| 8 | PhCH2 | Ph | 4-NO2-C6H4 | 3h (55%) |
| 9 | PhCH2 | Me | 4-F-C6H4 | 3i (75%) |
| 10 | Ph | Me | 4-F-C6H4 | 3l (78%) |
| Entry | Compound | R1 | R2 | Ar | IC50-Value (μM) a |
|---|---|---|---|---|---|
| 1 | 3a | Ph | Py | 4-NO2-C6H4 | 2.1 |
| 2 | 3b | Ph | Py | 4-F-C6H4 | 0.1 |
| 3 | 3c | Ph | Py | 4-CF3-C6H4 | 2.0 |
| 4 | 3d | Ph | Py | 4-Cl-C6H4 | 1.5 |
| 5 | 3e | PhCH2 | Py | 4-F-C6H4 | 0.08 |
| 6 | 3f | PhCH2 | Py | 4-NO2-C6H4 | 0.15 |
| 7 | 3g | PhCH2 | Ph | 4-F-C6H4 | 15.1 |
| 8 | 3h | PhCH2 | Ph | 4-NO2-C6H4 | 60.5 |
| 9 | 3i | PhCH2 | Me | 4-F-C6H4 | 18.5 |
| 10 | 3l | Ph | Me | 4-F-C6H4 | 22.8 |
| SB 203580 | 0.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romeo, R.; Giofrè, S.V.; Chiacchio, M.A.; Veltri, L.; Celesti, C.; Iannazzo, D. Synthesis and Biological Evaluation of 2,3,4-Triaryl-1,2,4-oxadiazol-5-ones as p38 MAPK Inhibitors. Molecules 2021, 26, 1745. https://doi.org/10.3390/molecules26061745
Romeo R, Giofrè SV, Chiacchio MA, Veltri L, Celesti C, Iannazzo D. Synthesis and Biological Evaluation of 2,3,4-Triaryl-1,2,4-oxadiazol-5-ones as p38 MAPK Inhibitors. Molecules. 2021; 26(6):1745. https://doi.org/10.3390/molecules26061745
Chicago/Turabian StyleRomeo, Roberto, Salvatore V. Giofrè, Maria A. Chiacchio, Lucia Veltri, Consuelo Celesti, and Daniela Iannazzo. 2021. "Synthesis and Biological Evaluation of 2,3,4-Triaryl-1,2,4-oxadiazol-5-ones as p38 MAPK Inhibitors" Molecules 26, no. 6: 1745. https://doi.org/10.3390/molecules26061745










