New Nitro-Laterally Substituted Azomethine Derivatives; Synthesis, Mesomorphic and Computational Characterizations
Abstract
1. Introduction
2. Results and Discussion
2.1. Mesomorphic and Optical Behavior
2.2. DFT Calculations
2.2.1. Geometrical Simulations and Thermal Parameters
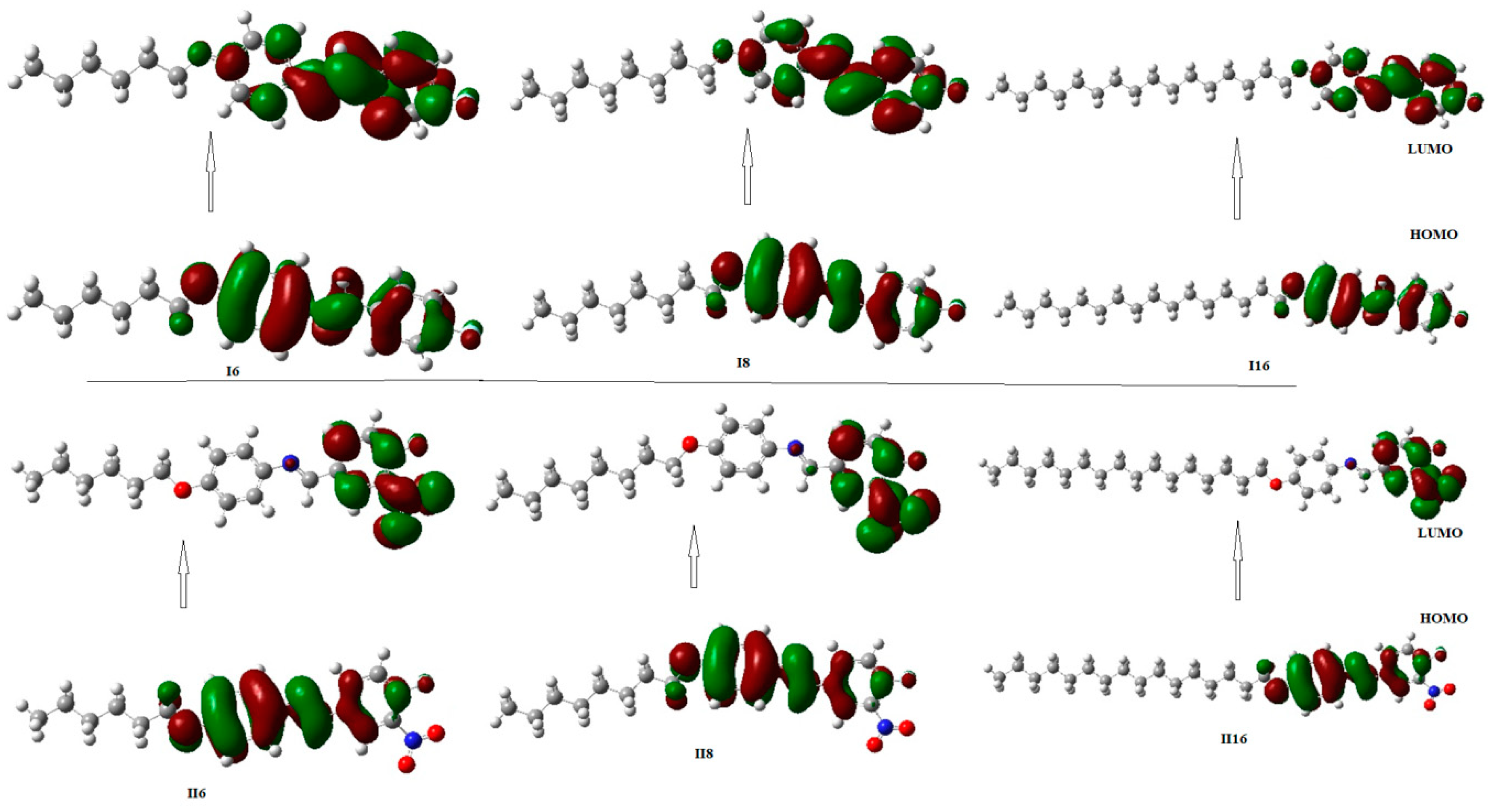
2.2.2. Frontier Molecular Orbitals (FMOs)
2.2.3. Molecular Electrostatic Potential (MEP)
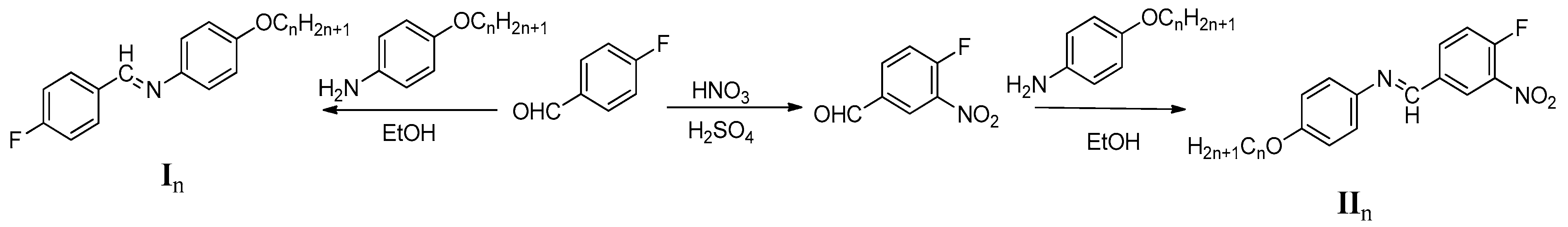
3. Experimental
3.1. Materials
3.2. Synthesis
4. Conclusions
- All compounds of the synthesized laterally neat group (In) were mesomorphic, exhibiting liquid crystalline mesophases monotropically.
- The observed smectic A phase in group In covered all lengths of terminal alkoxy chains.
- The compounds in group IIn were non-mesomorphic, which attributed to the steric hindrance of the large size of the nitro group, which disrupted the SmA phase.
- The molecular geometries of the investigated compounds were highly impacted by the electronic nature, location, and size of the attached NO2 substituent.
- The high dipole moment and steric hindrance of the NO2 moiety explain their non-mesomorphic properties.
- The electronic and geometric structures of the nitro group through intermolecular interactions have an essential role that affects the thermal and physical properties of the present investigated compounds.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Sample Availability
References
- Reyes, C.G.; Sharma, A.; Lagerwall, J.P. Non-electronic gas sensors from electrospun mats of liquid crystal core fibres for detecting volatile organic compounds at room temperature. Liq. Cryst. 2016, 43, 1986–2001. [Google Scholar] [CrossRef]
- Setia, S.; Sidiq, S.; De, J.; Pani, I.; Pal, S.K. Applications of liquid crystals in biosensing and organic light-emitting devices: Future aspects. Liq. Cryst. 2016, 43, 2009–2050. [Google Scholar] [CrossRef]
- Gupta, R.; Manjuladevi, V.; Karthik, C.; Choudhary, K. Thin films of discotic liquid crystals and their applications. Liq. Cryst. 2016, 43, 2079–2091. [Google Scholar] [CrossRef]
- Imrie, C.T.; Henderson, P.A.; Yeap, G.-Y. Liquid crystal oligomers: Going beyond dimers. Liq. Cryst. 2009, 36, 755–777. [Google Scholar] [CrossRef]
- Yeap, G.-Y.; Lee, H.-C.; Mahmood, W.A.K.; Imrie, C.T.; Takeuchi, D.; Osakada, K. Synthesis, thermal and optical behaviour of non-symmetric liquid crystal dimers α-(4-benzylidene-substituted-aniline-4′-oxy)-ω-[pentyl-4-(4′-phenyl) benzoateoxy] hexane. Ph. Transit. 2011, 84, 29–37. [Google Scholar] [CrossRef]
- Yeap, G.-Y.; Osman, F.; Imrie, C.T. Non-symmetric dimers: Effects of varying the mesogenic linking unit and terminal substituent. Liq. Cryst. 2015, 42, 543–554. [Google Scholar] [CrossRef]
- Yeap, G.-Y.; Hng, T.-C.; Yeap, S.-Y.; Gorecka, E.; Ito, M.M.; Ueno, K.; Okamoto, M.; Mahmood, W.A.K.; Imrie, C.T. Why do non-symmetric dimers intercalate? The synthesis and characterisation of the α-(4-benzylidene-substituted-aniline-4′-oxy)-ω-(2-methylbutyl-4′-(4 ″-phenyl) benzoateoxy) alkanes. Liq. Cryst. 2009, 36, 1431–1441. [Google Scholar] [CrossRef]
- Hagar, M.; Ahmed, H.A.; Alhaddad, O.A. Experimental and theoretical approaches of molecular geometry and mesophase behaviour relationship of laterally substituted azopyridines. Liq. Cryst. 2019, 46, 1440–1451. [Google Scholar] [CrossRef]
- Hagar, M.; Ahmed, H.A.; Saad, G.R. Mesophase stability of new Schiff base ester liquid crystals with different polar substituents. Liq. Cryst. 2018, 45, 1324–1332. [Google Scholar] [CrossRef]
- Ahmed, H.; Hagar, M.; Saad, G. Impact of the proportionation of dialkoxy chain length on the mesophase behaviour of Schiff base/ester liquid crystals; experimental and theoretical study. Liq. Cryst. 2019, 46, 1611–1620. [Google Scholar] [CrossRef]
- Hagar, M.; Ahmed, H.; Saad, G. Synthesis and mesophase behaviour of Schiff base/ester 4-(arylideneamino) phenyl-4 ″-alkoxy benzoates and their binary mixtures. J. Mol. Liq. 2019, 273, 266–273. [Google Scholar] [CrossRef]
- Ahmed, H.; Hagar, M.; El-Sayed, T.; Alnoman, R.B. Schiff base/ester liquid crystals with different lateral substituents: Mesophase behaviour and DFT calculations. Liq. Cryst. 2019, 46, 1–11. [Google Scholar] [CrossRef]
- Ahmed, H.; Hagar, M.; Alhaddad, O. New chair shaped supramolecular complexes-based aryl nicotinate derivative; mesomorphic properties and DFT molecular geometry. RSC Adv. 2019, 9, 16366–16374. [Google Scholar] [CrossRef]
- Hagar, M.; Ahmed, H.; Alhaddad, O. New azobenzene-based natural fatty acid liquid crystals with low melting point: Synthesis, DFT calculations and binary mixtures. Liq. Cryst. 2019, 46, 2223–2234. [Google Scholar] [CrossRef]
- Hagar, M.; Ahmed, H.; El-Sayed, T.; Alnoman, R. Mesophase behavior and DFT conformational analysis of new symmetrical diester chalcone liquid crystals. J. Mol. Liq. 2019, 285, 96–105. [Google Scholar] [CrossRef]
- Hammett, L.P. Physical Organic Chemistry, 1st ed.; Wiley Online Library: New York, NY, USA, 1940. [Google Scholar]
- MarekáKrygowski, T. The nitro group as substituent. Chem. Soc. Rev. 1996, 25, 71–75. [Google Scholar]
- Irle, S.; Krygowski, T.; Niu, J.; Schwarz, W. Substituent Effects of-NO and-NO2, Groups in Aromatic Systems. J. Org. Chem. 1995, 60, 6744–6755. [Google Scholar] [CrossRef]
- Yumatov, V.D.; Murakhtanov, V.; Salakhutdinov, N.; Okotrub, A.; Mazalov, L.; Logunova, L.; Koptyug, V.; Furin, G. Electronic structure of molecules of substituted benzenes by X-ray spectroscopy. I. Nitrobenzene. J. Struct. Chem. 1988, 28, 696–703. [Google Scholar] [CrossRef]
- Domenicano, A.; Schultz, G.; Hargittai, I.; Colapietro, M.; Portalone, G.; George, P.; Bock, C.W. Molecular structure of nitrobenzene in the planar and orthogonal conformations. J. Struct. Chem. 1990, 1, 107–122. [Google Scholar] [CrossRef]
- Boese, R.; Bläser, D.; Nussbaumer, M.; Krygowski, T.M. Low temperature crystal and molecular structure of nitrobenzene. J. Struct. Chem. 1992, 3, 363–368. [Google Scholar] [CrossRef]
- Politzer, P.; Lane, P.; Jayasuriya, K.; Domelsmith, L.N. Examination of some effects of NO2 rotation in nitrobenzene. J. Am. Chem. Soc. 1987, 109, 1899–1901. [Google Scholar] [CrossRef]
- Ritchie, J.P. A look at mesomerism in nitrobenzene. Tetrahedron 1988, 44, 7465–7478. [Google Scholar] [CrossRef]
- Gill, P.M.; Head-Gordon, M.; Pople, J.A. Efficient computation of two-electron-repulsion integrals and their nth-order derivatives using contracted Gaussian basis sets. J. Phys. Chem. 1990, 94, 5564–5572. [Google Scholar] [CrossRef]
- Minkin, V.I. Dipole Moments in Organic Chemistry; Springer Science & Business Media: Berlin, Germany, 2012. [Google Scholar]
- Fraser, R.R.; Ragauskas, A.J.; Stothers, J. Nitrobenzene valence bond structures: Evidence in support of through-resonance from oxygen-17 shieldings. J. Am. Chem. Soc. 1982, 104, 6475–6476. [Google Scholar] [CrossRef]
- Craik, D.J.; Levy, G.C.; Brownlee, R.T. Substituent effects on nitrogen-15 and oxygen-17 chemical shifts in nitrobenzenes: Correlations with electron densities. J. Org. Chem. 1983, 48, 1601–1606. [Google Scholar] [CrossRef]
- Petrov, V.F.; Shimizu, Y. Nitro substitution in achiral calamitic liquid crystals. Liq. Cryst. 2001, 28, 1627–1647. [Google Scholar] [CrossRef]
- Hird, M.; Toyne, K.J.; Goodby, J.W.; Gray, G.W.; Minter, V.; Tuffin, R.P.; McDonnell, D.G. Synthesis, mesomorphic behaviour and optical anisotropy of some novel materials for nematic mixtures of high birefringence. J. Mater. Chem. 2004, 14, 1731–1743. [Google Scholar] [CrossRef]
- Da Rosa, R.R.; Tariq, M.; Weber, C.S.; Hameed, S.; Silva, S.; Merlo, A.A. Hybrid liquid crystals tetrazolyl and isoxazolyl cinnamates. Liq. Cryst. 2016, 43, 1659–1670. [Google Scholar] [CrossRef]
- Chen, B.-Q.; Wen, J.-X. Synthesis and mesophase behaviour of mesogens bearing omega, alpha, alpha-trihydroperfluoroalkoxy end tails. Liq. Cryst. 1999, 26, 1135–1140. [Google Scholar] [CrossRef]
- Nafee, S.S.; Hagar, M.; Ahmed, H.A.; Alhaddad, O.; El-Shishtawy, R.M.; Raffah, B.M. New two rings Schiff base liquid crystals; ball mill synthesis, mesomorphic, Hammett and DFT studies. J. Mol. Liq. 2020, 299, 112161. [Google Scholar] [CrossRef]
- Ahmed, N.H.; Saad, G.R.; Ahmed, H.A.; Hagar, M. New wide-stability four-ring azo/ester/Schiff base liquid crystals: Synthesis, mesomorphic, photophysical, and DFT approaches. RSC Adv. 2020, 10, 9643–9656. [Google Scholar] [CrossRef]
- Ahmed, H.; Hagar, M.; Alhaddad, O. Mesomorphic and geometrical orientation study of the relative position of fluorine atom in some thermotropic liquid crystal systems. Liq. Cryst. 2020, 47, 404–413. [Google Scholar] [CrossRef]
- Alnoman, R.B.; Hagar, M.; Ahmed, H.A.; Naoum, M.M.; Sobaih, H.A.; Almshaly, J.S.; Haddad, M.M.; Alhaisoni, R.A.; Alsobhi, T.A. Binary Liquid Crystal Mixtures Based on Schiff Base Derivatives with Oriented Lateral Substituents. Crystals 2020, 10, 319. [Google Scholar] [CrossRef]
- Nafee, S.S.; Ahmed, H.; Hagar, M. Theoretical, experimental and optical study of new thiophene-based liquid crystals and their positional isomers. Liq. Cryst. 2020, 47, 1291–1302. [Google Scholar] [CrossRef]
- Kelker, H.; Scheurle, B. Eine flüssig-kristalline (nematische) Phase mit besonders niedrigem Erstarrungspunkt. Angew. Chem. 1969, 81, 903–904. [Google Scholar] [CrossRef]
- Nesrullajev, A.; Bilgin-Eran, B. Mesomorphic, morphologic and thermotropic properties of 4-hexyl-N-(4-hexadecyloxysalicylidene) aniline. Mater. Chem. Phys. 2005, 93, 21–25. [Google Scholar] [CrossRef]
- Yasa-Sahin, O.; Yazici, O.; Karaagac, B.; Sakar, D.; Cankurtaran, O.; Bilgin-Eran, B.; Karaman, F. A new liquid crystal of considerable value for the separation of closely related solvents by gas chromatography. Liq. Cryst. 2010, 37, 1111–1118. [Google Scholar] [CrossRef]
- Canli, N.Y.; Günes, S.; Pivrikas, A.; Fuchsbauer, A.; Sinwel, D.; Sariciftci, N.; Yasa, Ö.; Bilgin-Eran, B. Chiral (S)-5-octyloxy-2-[{4-(2-methylbuthoxy)-phenylimino}-methyl]-phenol liquid crystalline compound as additive into polymer solar cells. Sol. Energy Mater. Sol. Cells 2010, 94, 1089–1099. [Google Scholar] [CrossRef]
- Ge, L.-N.; Xian, S.-W.; Huang, Y.; Min, Y.; Lv, J.-M.; Tian, M.; Yao, D.-S. Synthesis and mesomorphism of novel multi-arm liquid crystals with cholic acid as chiral centre linking Schiff base moieties as mesogens. Liq. Cryst. 2017, 45, 1055–1067. [Google Scholar] [CrossRef]
- Huang, C.-C.; Chen, Y.-H.; Chen, S.-Y.; Sun, Y.-Z.; Wu, Z.-Y.; Yu, M.-C.; Chen, B.-H.; Hsu, I.-J.; Wu, L.-C.; Lee, J.-J. Broad temperature range of cubic blue phase present in simple binary mixture systems containing rodlike Schiff base mesogens with tolane moiety. Soft Matter 2016, 12, 3110–3120. [Google Scholar] [CrossRef]
- Veerabhadraswamy, B.N.; Rao, D.S.S.; Yelamaggad, C.V. Ferroelectric Liquid Crystals: Synthesis and Thermal Behavior of Optically Active, Three-Ring Schiff Bases and Salicylaldimines. Chem. Asian J. 2018, 13, 1012–1023. [Google Scholar] [CrossRef]
- Dubey, R.; Yerrasani, R.; Karunakar, M.; Singh, A.K.; Kumar, S.; Rao, T. Synthesis, structural and mesophase characterization of isoindoline-1, 3-dione based mesogenic Schiff bases. J. Mol. Liq. 2018, 251, 142–149. [Google Scholar] [CrossRef]
- Lei, Z.-Y.; Lee, G.-H.; Lai, C.K. Luminescent mesogenic borondifluoride complexes with the Schiff bases containing salicylideneamines and β-enaminoketones core systems. J. Mol. Liq. 2018, 260, 44–56. [Google Scholar] [CrossRef]
- Nagaveni, N.; Roy, A.; Prasad, V. Achiral bent-core azo compounds: Effect of different types of linkage groups and their direction of linking on liquid crystalline properties. J. Mater. Chem. 2012, 22, 8948–8959. [Google Scholar] [CrossRef]
- Ahmed, H.; Mansour, E.; Hagar, M. Mesomorphic study and DFT simulation of calamitic Schiff base liquid crystals with electronically different terminal groups and their binary mixtures. Liq. Cryst. 2020, 47, 2292–2304. [Google Scholar] [CrossRef]
- Hagar, M.; Ahmed, H.; Alhaddadd, O. DFT Calculations and Mesophase Study of Coumarin Esters and Its Azoesters. Crystals 2018, 8, 359. [Google Scholar] [CrossRef]
- Hagar, M.; Soliman, S.M.; Ibid, F.; El Sayed, H. Quinazolin-4-yl-sulfanylacetyl-hydrazone derivatives; Synthesis, molecular structure and electronic properties. J. Mol. Struct. 2013, 1049, 177–188. [Google Scholar] [CrossRef]
- Soliman, S.M.; Hagar, M.; Ibid, F.; El Sayed, H. Experimental and theoretical spectroscopic studies, HOMO–LUMO, NBO analyses and thione–thiol tautomerism of a new hybrid of 1, 3, 4-oxadiazole-thione with quinazolin-4-one. Spectrochim. Acta A Mol. Biol. Spectrosc. 2015, 145, 270–279. [Google Scholar] [CrossRef]
- Hagar, M.; Soliman, S.M.; Ibid, F.; El Sayed, H. Synthesis, molecular structure and spectroscopic studies of some new quinazolin-4 (3H)-one derivatives; an account on the N-versus S-Alkylation. J. Mol. Struct. 2016, 1108, 667–679. [Google Scholar] [CrossRef]
- Aboelnaga, A.; Hagar, M.; Soliman, S.M. Ultrasonic Synthesis, Molecular Structure and Mechanistic Study of 1, 3-Dipolar Cycloaddition Reaction of 1-Alkynylpyridinium-3-olate and Acetylene Derivatives. Molecules 2016, 21, 848. [Google Scholar] [CrossRef] [PubMed]
- Galewski, Z. Liquid-crystalline properties of 4-halogenobenzylidene-4’-alkoxy-anilines. In Proceedings of Liquid and Solid State Crystals: Physics. Technol. Appl. 1992, 1845, 452–458. [Google Scholar]
- Naoum, M.M.; Metwally, N.H.; Abd Eltawab, M.M.; Ahmed, H.A. Polarity and steric effect of the lateral substituent on the mesophase behaviour of some newly prepared liquid crystals. Liq. Cryst. 2015, 42, 1351–1369. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09, Revision A. 01; Gaussian Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Elshakre, M.E.; Alalawy, H.H.; Awad, M.I.; El-Anadouli, B.E. On the role of the electronic states of corrosion inhibitors: Quantum chemical-electrochemical correlation study on urea derivatives. Corros. Sci. 2017, 124, 121–130. [Google Scholar] [CrossRef]
- Naoum, M.M.; Fahmi, A.A.; Ahmed, N.H.; Saad, G.R. The effect of lateral methyl substitution on the mesophase behaviour of aryl 4-alkoxyphenylazo benzoates. Liq. Cryst. 2015, 42, 1627–1637. [Google Scholar] [CrossRef]
- Thaker, B.; Kanojiya, J.; Tandel, R. Effects of different terminal substituents on the mesomorphic behavior of some azo-schiff base and azo-ester-based liquid crystals. Mol. Cryst. Liq. Cryst. 2010, 528, 120–137. [Google Scholar] [CrossRef]
- Khoo, I.-C.; Wu, S.-T. Optics and Nonlinear Optics of Crystals; World Scientific: Singapore, 1993. [Google Scholar]
- Chemla, D.S. Nonlinear Optical Properties of Organic Molecules and Crystals; Elsevier: New York, NY, USA, 2012; Volume 1. [Google Scholar]
- Ahmed, H.A.; Hagar, M.; Alhaddad, O.A. Phase behavior and DFT calculations of laterally methyl supramolecular hydrogen-bonding complexes. Crystals 2019, 9, 133. [Google Scholar] [CrossRef]




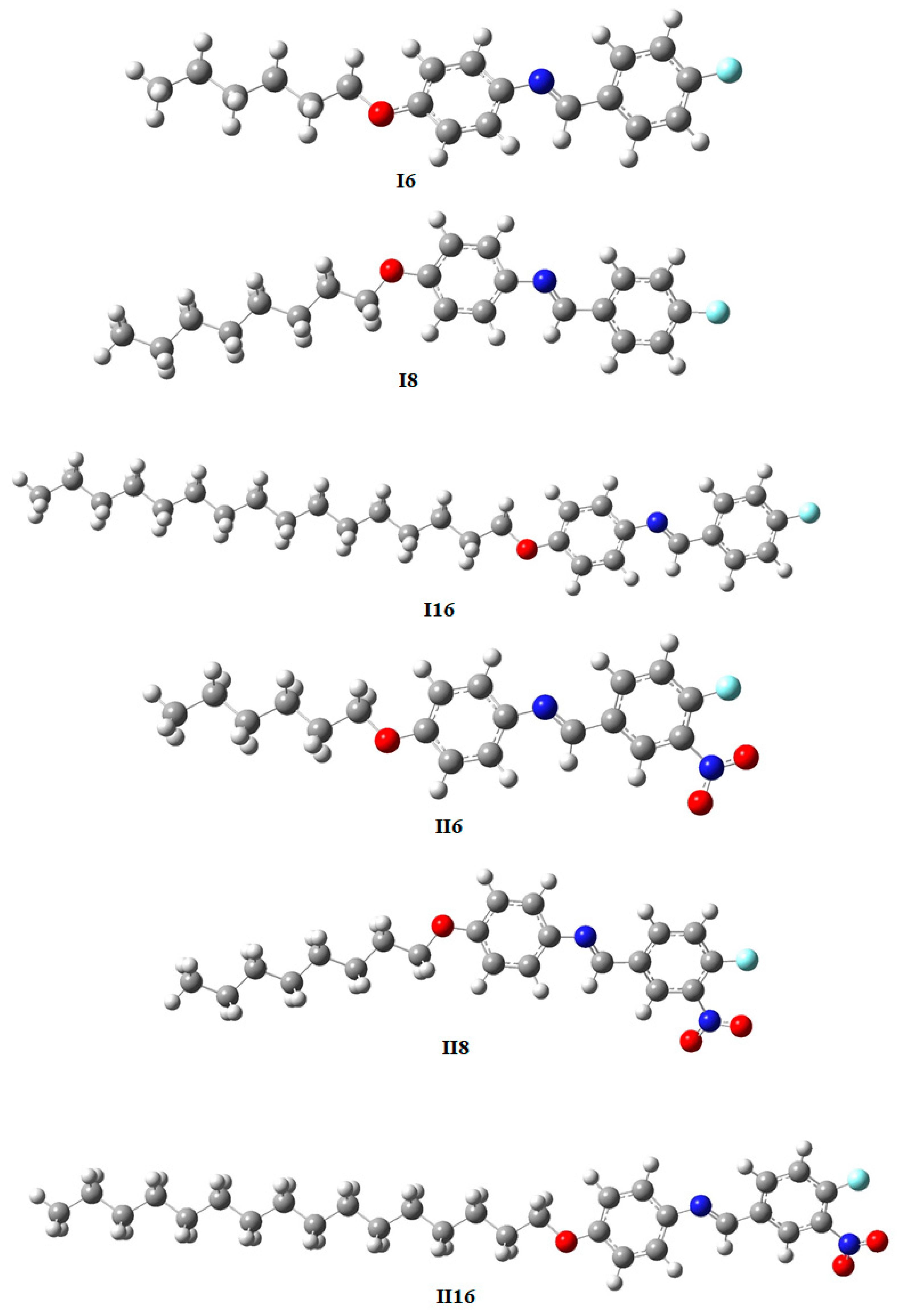

| Cycle | Upon Heating | Upon Cooling | |||||
|---|---|---|---|---|---|---|---|
| Comp. | TCr-I | ΔHCr-I | TI-SmA | ΔHI-SmA | TSmA-Cr | ΔH SmA-Cr | ΔT |
| I6 | 84.2 | 35.64 | 66.9 | 2.15 | 61.5 | 27.6 | 5.4 |
| I8 | 88.2 | 39.35 | 71.0 | 2.05 | 54.7 | 32.4 | 16.3 |
| I16 | 79.1 | 36.72 | 76.3 | 1.98 | <20.0 | - | >56.3 |
| II6 | 98.2 | 40.74 | - | - | - | - | - |
| II8 | 88.7 | 38.27 | - | - | - | - | - |
| II16 | 74.8 | 50.61 | - | - | - | - | - |
| Compound | ZPE (Kcal/Mol) | Thermal Energy (Kcal/Mol) | Enthalpy (Kcal/Mol) | Gibbs Free Energy (Kcal/Mol) | Entropy (Cal mol.k) | Total Energy (Hartree) |
|---|---|---|---|---|---|---|
| I6 | 232.110 | 245.514 | 246.107 | 198.082 | 161.077 | −966.673 |
| II6 | 233.080 | 248.147 | 248.739 | 196.560 | 175.010 | −1171.136 |
| I8 | 268.005 | 283.060 | 283.652 | 231.671 | 174.347 | −1045.237 |
| II8 | 268.850 | 285.632 | 286.224 | 229.633 | 189.806 | −1249.700 |
| I16 | 411.206 | 433.130 | 433.723 | 364.161 | 233.314 | −1359.495 |
| II16 | 412.130 | 435.744 | 436.336 | 362.232 | 248.547 | −1563.957 |
| Compound | EHOMO (ev) | EluMO (ev) | ∆E (ev) | IE (ev) | EA (ev) | Dipole Moment (Debye) | Polarizability (Bohr3) |
|---|---|---|---|---|---|---|---|
| I6 | −5.820 | −1.871 | 3.949 | 5.820 | 1.871 | 3.4343 | 253.23 |
| II6 | −6.185 | −3.207 | 2.978 | 6.185 | 3.207 | 8.3129 | 277.41 |
| I8 | −5.826 | −1.878 | 3.948 | 5.826 | 1.878 | 3.2523 | 275.53 |
| II8 | −6.178 | −3.224 | 2.954 | 6.178 | 3.224 | 7.2100 | 299.49 |
| I16 | −5.822 | −1.872 | 3.950 | 5.822 | 1.872 | 3.5361 | 372.25 |
| II16 | −6.177 | −3.207 | 2.970 | 6.177 | 3.207 | 8.3918 | 396.18 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-atawy, M.A.; Naoum, M.M.; Al-Zahrani, S.A.; Ahmed, H.A. New Nitro-Laterally Substituted Azomethine Derivatives; Synthesis, Mesomorphic and Computational Characterizations. Molecules 2021, 26, 1927. https://doi.org/10.3390/molecules26071927
El-atawy MA, Naoum MM, Al-Zahrani SA, Ahmed HA. New Nitro-Laterally Substituted Azomethine Derivatives; Synthesis, Mesomorphic and Computational Characterizations. Molecules. 2021; 26(7):1927. https://doi.org/10.3390/molecules26071927
Chicago/Turabian StyleEl-atawy, Mohamed A., Magdi M. Naoum, Salma A. Al-Zahrani, and Hoda A. Ahmed. 2021. "New Nitro-Laterally Substituted Azomethine Derivatives; Synthesis, Mesomorphic and Computational Characterizations" Molecules 26, no. 7: 1927. https://doi.org/10.3390/molecules26071927
APA StyleEl-atawy, M. A., Naoum, M. M., Al-Zahrani, S. A., & Ahmed, H. A. (2021). New Nitro-Laterally Substituted Azomethine Derivatives; Synthesis, Mesomorphic and Computational Characterizations. Molecules, 26(7), 1927. https://doi.org/10.3390/molecules26071927







