A Pilot Metabolomic Study on Myocardial Injury Caused by Chronic Alcohol Consumption—Alcoholic Cardiomyopathy
Abstract
:1. Introduction
2. Results
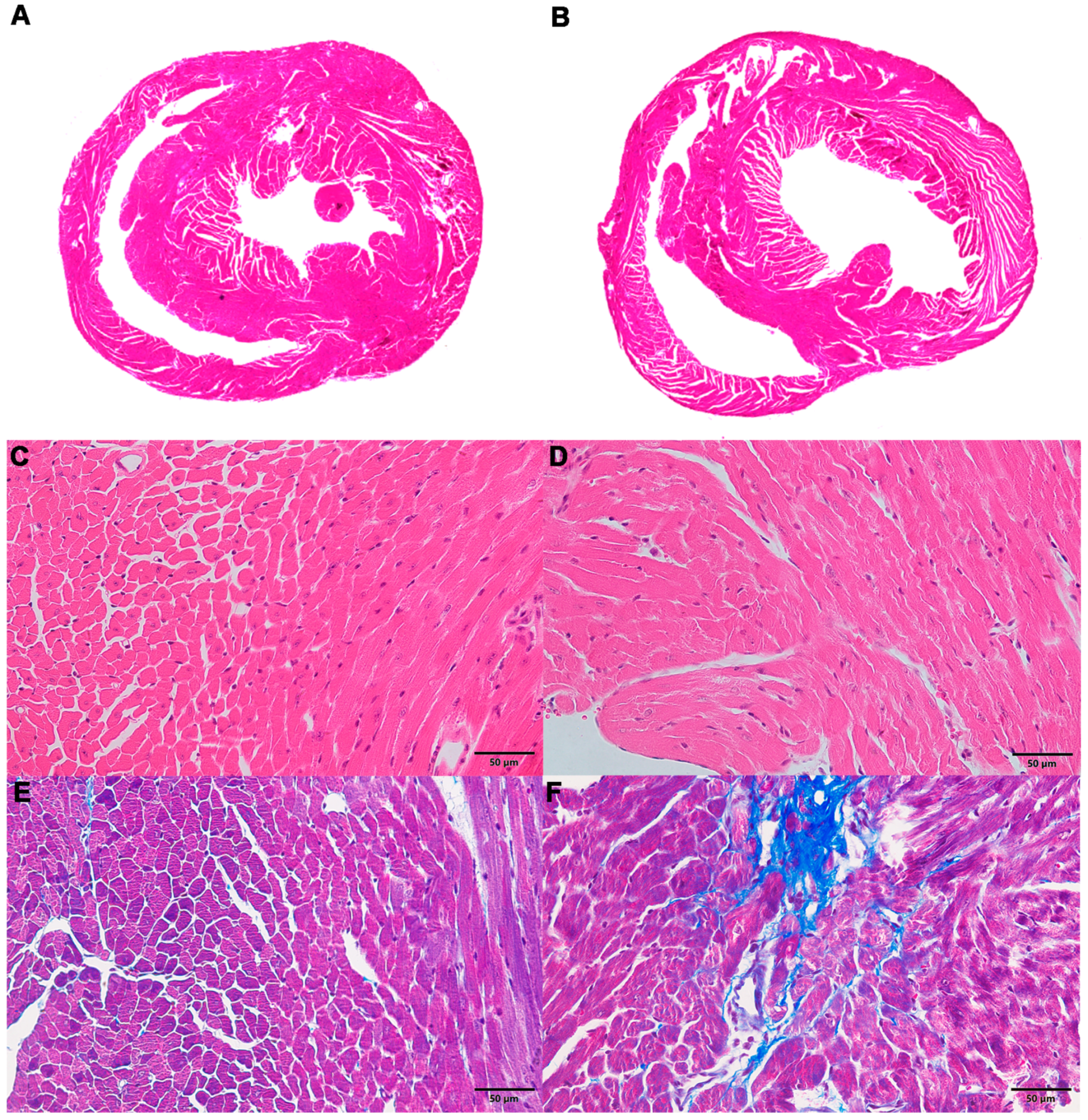
2.1. Morphological Changes in the Heart of ACM Model Mice
2.2. Assessment of Cardiac Function
2.3. Metabolic Pattern in the Myocardium of ACM Model Mice
2.3.1. Sample Quality Control (QC)
2.3.2. PCA and Partial Least Squares Discriminant Analysis (PLS-DA) Results of the ACM and Control Groups
2.3.3. Volcano Plot and Heatmaps of Differentially Expressed Metabolites
2.3.4. Enrichment of KEGG Pathway Analysis
3. Discussion
3.1. Metabolism of Fatty Acids
3.2. Metabolism of Lipids
3.3. Metabolism of B Vitamins
3.4. Metabolism of Pyrimidines and Purines
4. Materials and Methods
4.1. Chemical Reagents
4.2. Animal Model and Sample Collection
4.3. Histological Staining
4.4. Echocardiography
4.5. ELISA
4.6. Real-Time Quantitative PCR (qPCR)
4.7. Metabolite Extraction
4.8. Metabolomic Analysis QC
4.9. Analysis of UHPLC-MS/MS
4.10. Processing of Data and Identification of Metabolite
4.11. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Cao, Z.; Zhang, T.; Xu, C.; Jia, Y.; Wang, T.; Zhu, B. AIN-93 Diet as an alternative model to Lieber-DeCarli diet for alcoholic cardiomyopathy. Alcohol Clin. Exp. Res. 2019, 43, 1452–1461. [Google Scholar] [CrossRef]
- Fernández-Solà, J. The effects of ethanol on the heart: Alcoholic cardiomyopathy. Nutrients 2020, 12, 572. [Google Scholar] [CrossRef] [Green Version]
- Shaaban, A.; Gangwani, M.K.; Pendela, V.S.; Vindhyal, M.R. Alcoholic Cardiomyopathy; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2021. [Google Scholar]
- Wang, W.; Liu, T.; Liu, Y.; Yu, L.; Yan, X.; Weng, W.; Lu, X.; Zhang, C. Astaxanthin attenuates alcoholic cardiomyopathy via inhibition of endoplasmic reticulum stress-mediated cardiac apoptosis. Toxicol. Appl. Pharmacol. 2021, 412, 115378. [Google Scholar] [CrossRef]
- Lai, Q.; Yuan, G.; Wang, H.; Liu, Z.; Kou, J.; Yu, B.; Li, F. Metabolomic profiling of metoprolol-induced cardioprotection in a murine model of acute myocardial ischemia. Biomed. Pharmacother. 2020, 124, 109820. [Google Scholar] [CrossRef] [PubMed]
- Sansbury, B.E.; DeMartino, A.M.; Xie, Z.; Brooks, A.C.; Brainard, R.E.; Watson, L.J.; DeFilippis, A.P.; Cummins, T.D.; Harbeson, M.A.; Brittian, K.R.; et al. Metabolomic analysis of pressure-overloaded and infarcted mouse hearts. Circ. Heart Fail. 2014, 7, 634–642. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Voutilainen, T.; Karkkainen, O. Changes in the human metabolome associated with alcohol use: A review. Alcohol Alcohol. 2019, 54, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Xia, W.; Zhang, X.; Yuan, H.; Guan, D.; Gao, L. Hepatotoxicity of nutmeg: A pilot study based on metabolomics. Biomed. Pharmacother. 2020, 131, 110780. [Google Scholar] [CrossRef]
- Wu, S.L.; Wei, T.Y.; Lin, S.W.; Su, K.Y.; Kuo, C.H. Metabolomics investigation of voriconazole-induced hepatotoxicity in mice. Chem. Res. Toxicol. 2019, 32, 1840–1849. [Google Scholar] [CrossRef] [PubMed]
- Choi, R.Y.; Ji, M.; Lee, M.K.; Paik, M.J. Metabolomics study of serum from a chronic alcohol-fed rat model following administration of defatted Tenebrio molitor larva fermentation extract. Metabolites 2020, 10, 436. [Google Scholar] [CrossRef] [PubMed]
- Mavrelis, P.G.; Ammon, H.V.; Gleysteen, J.J.; Komorowski, R.A.; Charaf, U.K. Hepatic free fatty acids in alcoholic liver disease and morbid obesity. Hepatology 1983, 3, 226–231. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chen, H.; Zhang, W.; Cai, Y.; Shan, P.; Wu, D.; Zhang, B.; Liu, H.; Khan, Z.A.; Liang, G. Arachidonic acid inhibits inflammatory responses by binding to myeloid differentiation factor-2 (MD2) and preventing MD2/toll-like receptor 4 signaling activation. Biochim. Biophys. Acta Mol. Basis Dis. 2020, 1866, 165683. [Google Scholar] [CrossRef]
- Wong, S.W.; Kwon, M.J.; Choi, A.M.; Kim, H.P.; Nakahira, K.; Hwang, D.H. Fatty acids modulate toll-like receptor 4 activation through regulation of receptor dimerization and recruitment into lipid rafts in a reactive oxygen species-dependent manner. J. Biol. Chem. 2009, 284, 27384–27392. [Google Scholar] [CrossRef] [Green Version]
- Shahidi, F.; Ambigaipalan, P. Omega-3 polyunsaturated fatty acids and their health benefits. Annu. Rev. Food Sci. Technol. 2018, 9, 345–381. [Google Scholar] [CrossRef]
- Yamaguchi, A.; Stanger, L.; Freedman, C.J.; Standley, M.; Hoang, T.; Adili, R.; Tsai, W.C.; van Hoorebeke, C.; Holman, T.R.; Holinstat, M. DHA 12-LOX-derived oxylipins regulate platelet activation and thrombus formation through a PKA-dependent signaling pathway. J. Thromb. Haemost. 2020, 19, 839–851. [Google Scholar] [CrossRef] [PubMed]
- Yeung, J.; Adili, R.; Yamaguchi, A.; Freedman, C.J.; Chen, A.; Shami, R.; Das, A.; Holman, T.R.; Holinstat, M. Omega-6 DPA and its 12-lipoxygenase-oxidized lipids regulate platelet reactivity in a nongenomic PPARalpha-dependent manner. Blood Adv. 2020, 4, 4522–4537. [Google Scholar] [CrossRef]
- Drouin, G.; Rioux, V.; Legrand, P. The n-3 docosapentaenoic acid (DPA): A new player in the n-3 long chain polyunsaturated fatty acid family. Biochimie 2019, 159, 36–48. [Google Scholar] [CrossRef]
- Zhu, L.; Zhang, Y.; Guo, Z.; Wang, M. Cardiovascular biology of prostanoids and drug discovery. Arterioscler. Thromb. Vasc. Biol. 2020, 40, 1454–1463. [Google Scholar] [CrossRef]
- Wang, Y.; Qian, Y.; Fang, Q.; Zhong, P.; Li, W.; Wang, L.; Fu, W.; Zhang, Y.; Xu, Z.; Li, X.; et al. Saturated palmitic acid induces myocardial inflammatory injuries through direct binding to TLR4 accessory protein MD2. Nat. Commun. 2017, 8, 13997. [Google Scholar] [CrossRef] [PubMed]
- Rocha, D.M.; Caldas, A.P.; Oliveira, L.L.; Bressan, J.; Hermsdorff, H.H. Saturated fatty acids trigger TLR4-mediated inflammatory response. Atherosclerosis 2016, 244, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Lai, H.T.M.; de Oliveira Otto, M.C.; Lemaitre, R.N.; McKnight, B.; King, I.B.; Song, X.; Huggins, G.S.; Vest, A.R.; Siscovick, D.S.; et al. Serial biomarkers of de novo lipogenesis fatty acids and incident heart failure in older adults: The cardiovascular health study. J. Am. Heart Assoc. 2020, 9, e014119. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Wang, S.; Liang, Z.; Zeng, L.; Lai, R.; Ye, Z.; Liao, P. Impacts of a specific cyclooxygenase-2 inhibitor on pressure overload-induced myocardial hypertrophy in rats. Heart Surg. Forum 2019, 22, E432–E437. [Google Scholar] [CrossRef]
- Cao, Z.; Jia, Y.; Zhu, B. BNP and NT-proBNP as diagnostic biomarkers for cardiac dysfunction in both clinical and forensic medicine. Int. J. Mol. Sci. 2019, 20, 1820. [Google Scholar] [CrossRef] [Green Version]
- Tian, M.; Xiao, Y.; Xue, J.; Zhang, Y.; Jia, Y.; Luo, X.; Wang, T.; Zhu, B.; Cao, Z. The expression of BNP, ET-1, and TGF-beta1 in myocardium of rats with ventricular arrhythmias. Int. J. Mol. Sci. 2019, 20, 5845. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Xu, W.; Jiang, L.; Gu, H.; Li, M.; Zhang, J.; Guo, W.; Deng, P.; Long, H.; Bu, Q.; et al. Lipidomic signature of serum from the rats exposed to alcohol for one year. Toxicol. Lett. 2018, 294, 166–176. [Google Scholar] [CrossRef] [PubMed]
- Engelbrecht, E.; MacRae, C.A.; Hla, T. Lysolipids in vascular development, biology, and disease. Arterioscler. Thromb. Vasc. Biol. 2020, 41, 564–584. [Google Scholar] [CrossRef]
- Tan, S.T.; Ramesh, T.; Toh, X.R.; Nguyen, L.N. Emerging roles of lysophospholipids in health and disease. Prog. Lipid Res. 2020, 80, 101068. [Google Scholar] [CrossRef] [PubMed]
- Tomczyk, M.M.; Dolinsky, V.W. The cardiac lipidome in models of cardiovascular disease. Metabolites 2020, 10, 254. [Google Scholar] [CrossRef] [PubMed]
- Jaremek, M.; Yu, Z.; Mangino, M.; Mittelstrass, K.; Prehn, C.; Singmann, P.; Xu, T.; Dahmen, N.; Weinberger, K.M.; Suhre, K.; et al. Alcohol-Induced metabolomic differences in humans. Transl. Psychiatry 2013, 3, e276. [Google Scholar] [CrossRef] [Green Version]
- Lacruz, M.E.; Kluttig, A.; Tiller, D.; Medenwald, D.; Giegling, I.; Rujescu, D.; Prehn, C.; Adamski, J.; Frantz, S.; Greiser, K.H.; et al. Cardiovascular risk factors associated with blood metabolite concentrations and their alterations during a 4-year period in a population-based cohort. Circ. Cardiovasc. Genet. 2016, 9, 487–494. [Google Scholar] [CrossRef] [Green Version]
- Van Roekel, E.H.; Trijsburg, L.; Assi, N.; Carayol, M.; Achaintre, D.; Murphy, N.; Rinaldi, S.; Schmidt, J.A.; Stepien, M.; Kaaks, R.; et al. Circulating metabolites associated with alcohol intake in the European Prospective investigation into cancer and nutrition cohort. Nutrients 2018, 10, 654. [Google Scholar] [CrossRef] [Green Version]
- Yore, M.M.; Syed, I.; Moraes-Vieira, P.M.; Zhang, T.; Herman, M.A.; Homan, E.A.; Patel, R.T.; Lee, J.; Chen, S.; Peroni, O.D.; et al. Discovery of a class of endogenous mammalian lipids with anti-diabetic and anti-inflammatory effects. Cell 2014, 159, 318–332. [Google Scholar] [CrossRef] [Green Version]
- Zhu, Q.F.; Yan, J.W.; Gao, Y.; Zhang, J.W.; Yuan, B.F.; Feng, Y.Q. Highly sensitive determination of fatty acid esters of hydroxyl fatty acids by liquid chromatography-mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2017, 1061–1062, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Kuda, O.; Brezinova, M.; Rombaldova, M.; Slavikova, B.; Posta, M.; Beier, P.; Janovska, P.; Veleba, J.; Kopecky, J., Jr.; Kudova, E.; et al. Docosahexaenoic acid-derived fatty acid esters of hydroxy fatty acids (FAHFAs) with anti-inflammatory properties. Diabetes 2016, 65, 2580–2590. [Google Scholar] [CrossRef] [Green Version]
- Dongoran, R.A.; Lin, T.J.; Byekyet, A.; Tang, S.C.; Yang, J.H.; Liu, C.H. Determination of major endogenous FAHFAs in healthy human circulation: The correlations with several circulating cardiovascular-related biomarkers and anti-inflammatory effects on RAW 264.7 cells. Biomolecules 2020, 10, 1689. [Google Scholar] [CrossRef] [PubMed]
- Dastur, D.K.; Santhadevi, N.; Quadros, E.V.; Avari, F.C.; Wadia, N.H.; Desai, M.N.; Bharucha, E.P. The B-vitamins in malnutrition with alcoholism. A model of intervitamin relationships. Br. J. Nutr. 1976, 36, 143–159. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sechi, G.; Sechi, E.; Fois, C.; Kumar, N. Advances in clinical determinants and neurological manifestations of B vitamin deficiency in adults. Nutr. Rev. 2016, 74, 281–300. [Google Scholar] [CrossRef] [Green Version]
- Jiawan, A.; Tandjung, K. New-Onset heart failure. JAMA Cardiol. 2019, 4, 592–593. [Google Scholar] [CrossRef]
- DiNicolantonio, J.J.; Liu, J.; O’Keefe, J.H. Thiamine and cardiovascular disease: A literature review. Prog. Cardiovasc. Dis. 2018, 61, 27–32. [Google Scholar] [CrossRef]
- Page, G.L.; Laight, D.; Cummings, M.H. Thiamine deficiency in diabetes mellitus and the impact of thiamine replacement on glucose metabolism and vascular disease. Int. J. Clin. Pract. 2011, 65, 684–690. [Google Scholar] [CrossRef] [Green Version]
- Subramanian, V.S.; Subramanya, S.B.; Ghosal, A.; Said, H.M. Chronic alcohol feeding inhibits physiological and molecular parameters of intestinal and renal riboflavin transport. Am. J. Physiol. Cell Physiol. 2013, 305, C539–C546. [Google Scholar] [CrossRef] [Green Version]
- Keith, M.E.; Walsh, N.A.; Darling, P.B.; Hanninen, S.A.; Thirugnanam, S.; Leong-Poi, H.; Barr, A.; Sole, M.J. B-Vitamin deficiency in hospitalized patients with heart failure. J. Am. Diet. Assoc. 2009, 109, 1406–1410. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, H.G. The oxidative pentose phosphate pathway in the heart: Regulation, physiological significance, and clinical implications. Basic Res. Cardiol. 1992, 87, 303–316. [Google Scholar] [CrossRef] [PubMed]
- Asnani, A.; Shi, X.; Farrell, L.; Lall, R.; Sebag, I.A.; Plana, J.C.; Gerszten, R.E.; Scherrer-Crosbie, M. Changes in citric acid cycle and nucleoside metabolism are associated with anthracycline cardiotoxicity in patients with breast cancer. J. Cardiovasc. Transl. Res. 2020, 13, 349–356. [Google Scholar] [CrossRef]
- Lian, J.S.; Liu, W.; Hao, S.R.; Guo, Y.Z.; Huang, H.J.; Chen, D.Y.; Xie, Q.; Pan, X.P.; Xu, W.; Yuan, W.X.; et al. A serum metabonomic study on the difference between alcohol-and HBV-induced liver cirrhosis by ultraperformance liquid chromatography coupled to mass spectrometry plus quadrupole time-of-flight mass spectrometry. Chin. Med. J. 2011, 124, 1367–1373. [Google Scholar] [PubMed]
- Mittal, A.; Dabur, R. Detection of new human metabolic urinary markers in chronic alcoholism and their reversal by aqueous extract of Tinospora cordifolia stem. Alcohol Alcohol. 2015, 50, 271–281. [Google Scholar] [CrossRef]
- Irwin, C.; van Reenen, M.; Mason, S.; Mienie, L.J.; Wevers, R.A.; Westerhuis, J.A.; Reinecke, C.J. The 1H-NMR-based metabolite profile of acute alcohol consumption: A metabolomics intervention study. PLoS ONE 2018, 13, e0196850. [Google Scholar] [CrossRef] [Green Version]
- Vikhert, A.M.; Tsiplenkova, V.G.; Cherpachenko, N.M. Alcoholic cardiomyopathy and sudden cardiac death. J. Am. Coll. Cardiol. 1986, 8, 3A–11A. [Google Scholar] [CrossRef] [Green Version]
- Silvestri, F.; Bussani, R. Hypoxic right ventricular cardiomyopathy. A morphological and pathogenetic study on the myocardial atrophy and fatty infiltration. Pathologica 1990, 82, 593–616. [Google Scholar]
- Want, E.J.; Masson, P.; Michopoulos, F.; Wilson, I.D.; Theodoridis, G.; Plumb, R.S.; Shockcor, J.; Loftus, N.; Holmes, E.; Nicholson, J.K. Global metabolic profiling of animal and human tissues via UPLC-MS. Nat. Protoc. 2013, 8, 17–32. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, M.; Ma, J.; Zhang, Y.; Hong, G.; Sun, F.; Lin, G.; Hu, L. Metabolic changes in paraquat poisoned patients and support vector machine model of discrimination. Biol. Pharm. Bull. 2015, 38, 470–475. [Google Scholar] [CrossRef] [Green Version]
- Broadhurst, D.I.; Kell, D.B. Statistical strategies for avoiding false discoveries in metabolomics and related experiments. Metabolomics 2006, 2, 171–196. [Google Scholar] [CrossRef] [Green Version]
- Musharraf, S.G.; Siddiqui, A.J.; Shamsi, T.; Choudhary, M.I.; Rahman, A.U. Serum metabonomics of acute leukemia using nuclear magnetic resonance spectroscopy. Sci. Rep. 2016, 6, 30693. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sedghipour, M.R.; Sadeghi-Bazargani, H. Applicability of supervised discriminant analysis models to analyze astigmatism clinical trial data. Clin. Ophthalmol. 2012, 6, 1499–1506. [Google Scholar] [CrossRef] [PubMed] [Green Version]







| Parameters | Control Group | ACM Group |
|---|---|---|
| LVAW; d (mm) | 0.844 ± 0.044 | 0.595 ± 0.085 # |
| LVAW; s (mm) | 0.877 ± 0.016 | 0.616 ± 0.042 # |
| LVPW; d (mm) | 0.805 ± 0.085 | 0.601 ± 0.041 # |
| LVPW; s (mm) | 0.999 ± 0.060 | 0.758 ± 0.044 * |
| LVID; d (mm) | 4.165 ± 0.223 | 4.480 ± 0.226 |
| LVID; s (mm) | 3.164 ± 0.200 | 3.588 ± 0.142 * |
| LV Vol; d (uL) | 77.290 ± 9.664 | 91.756 ± 11.157 |
| LV Vol; s (uL) | 40.032 ± 6.160 | 54.098 ± 5.234 * |
| EF% | 48.244 ± 3.833 | 40.915 ± 1.713 * |
| FS% | 24.049 ± 2.331 | 19.883 ± 1.041 * |
| KEGG Pathway | Screened Metabolite | FC | p Value | ROC | VIP | Trend | Mode |
|---|---|---|---|---|---|---|---|
| Biosynthesis of unsaturated fatty acids | Docosapentaenoic acid | 0.451 | 0.000 | 1.000 | 2.054 | down | negative |
| Adrenic acid | 0.465 | 0.000 | 1.000 | 1.936 | down | positive | |
| Docosahexaenoic acid | 0.514 | 0.000 | 1.000 | 1.910 | down | negative | |
| Arachidonic acid | 0.603 | 0.000 | 1.000 | 1.807 | down | negative | |
| Stearic acid | 1.225 | 0.000 | 0.970 | 1.646 | up | negative | |
| Arachidic acid | 1.454 | 0.001 | 0.900 | 1.461 | up | negative | |
| α-Linolenic acid | 1.493 | 0.001 | 0.880 | 1.587 | up | positive | |
| Palmitic Acid | 1.529 | 0.048 | 0.760 | 1.046 | up | negative | |
| Vitamin digestion and absorption | Thiamine monophosphate | 0.519 | 0.000 | 1.000 | 1.883 | down | positive |
| Pantothenic acid | 0.585 | 0.000 | 0.990 | 2.015 | down | positive | |
| Nicotinamide | 0.753 | 0.000 | 0.950 | 1.768 | down | positive | |
| Riboflavin | 0.788 | 0.001 | 0.920 | 1.588 | down | positive | |
| Metabolism of pyrimidines and purines | Xanthine | 0.595 | 0.000 | 0.960 | 1.799 | down | positive |
| Thymine | 0.708 | 0.032 | 0.940 | 1.194 | down | positive | |
| Cytidine | 0.756 | 0.000 | 0.990 | 1.819 | down | positive | |
| Adenylosuccinic acid | 0.820 | 0.017 | 0.790 | 1.238 | down | negative | |
| d-Ribulose 5-phosphate | 0.786 | 0.001 | 0.890 | 1.574 | down | negative | |
| d-Sedoheptulose 7-phosphate | 0.787 | 0.003 | 0.880 | 1.382 | down | negative | |
| Uridine diphosphate | 1.470 | 0.001 | 0.920 | 1.605 | up | positive |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, Z.; Wang, T.; Xia, W.; Zhu, B.; Tian, M.; Zhao, R.; Guan, D. A Pilot Metabolomic Study on Myocardial Injury Caused by Chronic Alcohol Consumption—Alcoholic Cardiomyopathy. Molecules 2021, 26, 2177. https://doi.org/10.3390/molecules26082177
Cao Z, Wang T, Xia W, Zhu B, Tian M, Zhao R, Guan D. A Pilot Metabolomic Study on Myocardial Injury Caused by Chronic Alcohol Consumption—Alcoholic Cardiomyopathy. Molecules. 2021; 26(8):2177. https://doi.org/10.3390/molecules26082177
Chicago/Turabian StyleCao, Zhipeng, Tianqi Wang, Wei Xia, Baoli Zhu, Meihui Tian, Rui Zhao, and Dawei Guan. 2021. "A Pilot Metabolomic Study on Myocardial Injury Caused by Chronic Alcohol Consumption—Alcoholic Cardiomyopathy" Molecules 26, no. 8: 2177. https://doi.org/10.3390/molecules26082177






