Systematic Studies on the Effect of Fluorine Atoms in Fluorinated Tolanes on Their Photophysical Properties
Abstract
:1. Introduction
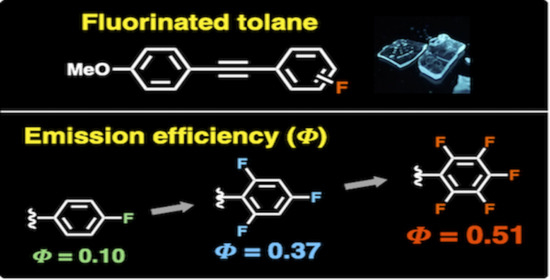
2. Results and Discussion
3. Materials and Methods
3.1. Materials
3.2. General Synthesis Procedure for the Pd(0)-Catalyzed Sonogashira Cross-Coupling Reaction
3.2.1. [2-(4-methoxyphenyl)ethyn-1-yl]benzene (0F)
3.2.2. 1-Fluoro-4-[2-(4-methoxyphenyl)ethyn-1-yl]benzene (1F)
3.2.3. 1,2,3-Trifluoro-4-[2-(4-methoxyphenyl)ethyn-1-yl]benzene (3Fa)
3.2.4. 1,3,5-Trifluoro-4-[2-(4-methoxyphenyl)ethyn-1-yl]benzene (3Fb)
3.2.5. 1,2,6-Trifluoro-4-[2-(4-methoxyphenyl)ethyn-1-yl]benzene (3Fc)
3.2.6. 2,3,5,6-Tetrafluoro-4-[2-(4-methoxyphenyl)ethyn-1-yl]benzene (4F)
3.3. Photophysical Measurements
3.4. Single Crystal X-ray Diffraction
3.5. Cyclic Voltammetry
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Gutmann, M.; Gudipati, M.; Schoenzart, P.F.; Hohlneicher, G. Electronic spectra of matrix-isolated tolan: Site selective one- and two-photon spectra. J. Phys. Chem. 1992, 96, 2433–2442. [Google Scholar] [CrossRef]
- Suzuki, T.; Nakamura, M.; Isozaki, T.; Ikoma, T. “Dark” Excited States of Diphenylacetylene Studied by Nonresonant Two-Photon Excitation Optical-Probing Photoacoustic Spectroscopy. Int. J. Thermophys. 2012, 33, 2046–2054. [Google Scholar] [CrossRef]
- Isozaki, T.; Oba, H.; Ikoma, T.; Suzuki, T. Simultaneous two-photon absorption to gerade excited singlet states of diphe-nylacetylene and diphenylbutadiyne using optical-probing photoacoustic spectroscopy. J. Phys. Chem. A 2016, 120, 6137–6145. [Google Scholar] [CrossRef]
- Young, D.D.; Scharrer, E.; Yoa, M.V. Synthesis and phase behavior of liquid crystalline diphenylacetylene derivatives possessing high clearing temperatures. Mol. Cryst. Liq. Cryst. 2004, 408, 21–31. [Google Scholar] [CrossRef]
- Cheng, Z.; Zang, Y.; Li, Y.; Li, B.; Hu, C.; Li, H.; Yang, Y. A chiral luminescent liquid crystal with a tolane unit. Liq. Cryst. 2016, 43, 777–782. [Google Scholar] [CrossRef]
- Arakawa, Y.; Inui, S.; Tsuji, H. Novel diphenylacetylene-based room-temperature liquid crystalline molecules with alkylthio groups, and investigation of the role for terminal alkyl chains in mesogenic incidence and tendency. Liq. Cryst. 2017, 45, 811–820. [Google Scholar] [CrossRef]
- Yang, W.-Y.; Roy, S.; Phrathep, B.; Rengert, Z.; Kenworthy, R.; Zorio, D.A.R.; Alabugin, I.V. Engineering pH-gated transi-tions for selective and efficient double-strand DNA photocleavage in hypoxic tumors. J. Med. Chem. 2011, 54, 8501–8516. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.-Y.; Marrone, S.A.; Minors, N.; Zorio, D.A.R.; Alabugin, I.V. Fine-tuning alkyne cycloadditions: Insights into photochemistry responsible for the double-strand DNA cleavage via structural perturbations in diaryl alkyne conjugates. Beilstein J. Org. Chem. 2011, 7, 813–823. [Google Scholar] [CrossRef] [PubMed]
- Do, T.T.; Chavhan, S.; Subbiah, J.; Ou, T.-H.; Manzhos, S.; Jones, D.J.; Bell, J.M.; Jou, J.-H.; Sonar, P.M. Naphthalimide end-capped diphenylacetylene: A versatile organic semiconductor for blue light emitting diodes and a donor or an acceptor for solar cells. New J. Chem. 2019, 43, 9243–9254. [Google Scholar] [CrossRef]
- Ferrante, C.; Kensy, U.; Dick, B. Does diphenylacetylene (tolan) fluorescence from its second excited singlet state? Sem-iempirical MO calculations and fluorescence quantum yield measurements. J. Phys. Chem. 1993, 97, 13457–13463. [Google Scholar] [CrossRef] [Green Version]
- Zgierski, M.Z.; Lim, E.C. Nature of the ’dark’ state in diphenylacetylene and related molecules: State switch from the linear ππ* state to the bent πσ*state. Chem. Phys. Lett. 2004, 387, 352–355. [Google Scholar] [CrossRef]
- Saltiel, J.; Kumar, V.K.R. Photophysics of diphenylacetylene: Light from the “dark state”. J. Phys. Chem. A 2012, 116, 10548–10558. [Google Scholar] [CrossRef] [PubMed]
- Karunakaran, V.; Prabhu, D.D.; Das, S. Optical investigation of self-aggregation of a tetrazole-substituted diphenylacety-lene derivative: Steady and excited state dynamics in solid and solution state. J. Phys. Chem. C 2013, 117, 9404–9415. [Google Scholar] [CrossRef]
- Wierzbicka, M.; Bylińska, I.; Czaplewski, C.; Wiczk, W. Experimental and theoretical studies of the spectroscopic properties of simple symmetrically substituted diphenylacetylene derivatives. RSC Adv. 2015, 5, 29294–29303. [Google Scholar] [CrossRef]
- Kozhemyakin, Y.; Krämer, M.; Rominger, F.; Dreuw, A.; Bunz, U.H.F. A Tethered Tolane: Twisting the Excited State. Chem. A Eur. J. 2018, 24, 15219–15222. [Google Scholar] [CrossRef]
- Menning, S.; Krämer, M.; Duckworth, A.; Rominger, F.; Beeby, A.; Dreuw, A.; Bunz, U.H.F. Bridged Tolanes: A Twisted Tale. J. Org. Chem. 2014, 79, 6571–6578. [Google Scholar] [CrossRef] [PubMed]
- Tong, J.; Wang, Y.J.; Wang, Z.; Sun, J.Z.; Tang, B.Z. Crystallization-Induced Emission Enhancement of a Simple Tolane-Based Mesogenic Luminogen. J. Phys. Chem. C 2015, 119, 21875–21881. [Google Scholar] [CrossRef]
- Zang, Y.; Li, Y.; Li, B.; Li, H.; Yang, Y. Light emission properties and self-assembly of a tolane-based luminogen. RSC Adv. 2015, 5, 38690–38695. [Google Scholar] [CrossRef]
- Bu, X.; Zhu, D.; Liu, T.; Li, Y.; Cai, S.; Wang, H.; Zeng, Z. Approach to tuned emitting color of luminescent liquid crystals with substituted fluoropyrrole acceptor unit. Dye. Pigment. 2017, 145, 324–330. [Google Scholar] [CrossRef]
- Kirsch, P. Modern Fluoroorganic Chemistry: Synthesis, Reactivity, Applications, 2nd ed.; Wiley-VCH: Weinheim, Germany, 2013; pp. 7–21. [Google Scholar]
- O’Hagan, D. Understanding organofluorine chemistry. An introduction to the C–F bond. Chem. Soc. Rev. 2007, 37, 308–319. [Google Scholar] [CrossRef]
- Yamada, S.; Uto, E.; Agou, T.; Kubota, T.; Konno, T. Fluorinated tolane dyads with alkylene linkage: Synthesis and eval-uation of photophysical characteristics. Crystals 2020, 10, 711. [Google Scholar] [CrossRef]
- Morita, M.; Yamada, S.; Konno, T. Fluorine-induced emission enhancement of tolanes via formation of tight molecular aggregates. New J. Chem. 2020, 44, 6704–6708. [Google Scholar] [CrossRef]
- Yamada, S.; Mitsuda, A.; Miyano, K.; Tanaka, T.; Morita, M.; Agou, T.; Kubota, T.; Konno, T. Development of novel sol-id-state light-emitting materials based on pentafluorinated tolane fluorophores. ACS Omega 2018, 3, 9105–9113. [Google Scholar] [CrossRef] [PubMed]
- Melnikov, A.R.; Davydova, M.P.; Sherin, P.S.; Korolev, V.V.; Stepanov, A.A.; Kalneus, E.V.; Benassi, E.; Vasilevsky, S.F.; Stass, D.V. X-Ray Generated Recombination Exciplexes of Substituted Diphenylacetylenes with Tertiary Amines: A Ver-satile Experimental Vehicle for Targeted Creation of Deep-Blue Electroluminescent Systems. J. Phys. Chem. A 2018, 122, 1235–1252. [Google Scholar] [CrossRef]
- Hirata, Y.; Okada, T.; Nomoto, T. Higher excited singlet state of diphenylacetylene in solution phase. Chem. Phys. Lett. 1993, 209, 397–402. [Google Scholar] [CrossRef]
- Khundkar, L.R.; Stiegman, A.E.; Perry, J.W. Solvent-tuned intramolecular charge-recombination rates in a conjugated donor-acceptor molecule. J. Phys. Chem. 1990, 94, 1224–1226. [Google Scholar] [CrossRef]
- Szyszkowska, M.; Bylińska, I.; Wiczk, W. Influence of an electron-acceptor substituent type on the photophysical properties of unsymmetrically substituted diphenylacetylene. J. Photochem. Photobiol. A Chem. 2016, 326, 76–88. [Google Scholar] [CrossRef]
- Soloshonok, V.A.; Hayashi, T. Gold(I)-catalyzed asymmetric aldol reaction of methyl isocyanoacetate with fluorinated benzaldehydes. Tetrahedron Lett. 1994, 35, 2713–2716. [Google Scholar] [CrossRef]
- Soloshonok, V.A.; Hayashi, T. Gold(I)-catalyzed asymmetric aldol reactions of fluorinated benzaldehydes with an α-isocyanoacetamide. Tetrahedron Asymmetry 1994, 5, 1091–1094. [Google Scholar] [CrossRef]
- Zhang, H.; Zhao, Z.; Turley, A.T.; Wang, L.; McGonigal, P.R.; Tu, Y.; Li, Y.; Wang, Z.; Kwok, R.T.K.; Lam, J.W.Y.; et al. Aggregate Science: From Structures to Properties. Adv. Mater. 2020, 32, 2001457. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.-T.; Liu, C.; Li, Q.; Zhang, H.; Lam, J.W.Y.; Tang, B.Z. Structure, Assembly, and Function of (Latent)-Chiral AIEgens. ACS Mater. Lett. 2019, 1, 192–202. [Google Scholar] [CrossRef] [Green Version]
- He, Z.; Ke, C.; Tang, B.Z. Journey of Aggregation-Induced Emission Research. ACS Omega 2018, 3, 3267–3277. [Google Scholar] [CrossRef] [Green Version]
- Nielsen, A.; Kuzmanich, G.; Garcia-Garibay, M.A. Quantum Chain Reaction of Tethered Diarylcyclopropenones in the Solid State and Their Distance-Dependence in Solution Reveal a Dexter S2–S2Energy-Transfer Mechanism. J. Phys. Chem. A 2014, 118, 1858–1863. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.L.N.; Jadhav, D.N.; Dasgupta, P. Pd-Catalyzed Domino Synthesis of Internal Alkynes Using Triarylbismuths as Multicoupling Organometallic Nucleophiles. Org. Lett. 2010, 12, 2048–2051. [Google Scholar] [CrossRef] [PubMed]
- Severin, R.; Reimer, J.; Doye, S. One-Pot Procedure for the Synthesis of Unsymmetrical Diarylalkynes. J. Org. Chem. 2010, 75, 3518–3521. [Google Scholar] [CrossRef]
- CrysAlisPro 1.171.39.43a. Rigaku Oxford Diffraction; Rigaku Corporation: Akishima, Japan, 2015. [Google Scholar]
- Sheldrick, G.M. SHELXT– Integrated space-group and crystal-structure determination. Acta Cryst. 2015, 71, 3–8. [Google Scholar] [CrossRef] [Green Version]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]







| Compound | EHOMO (eV) a | ELUMO (eV) a | ΔE (eV) a | λabs (nm) [ε, 103 L·mol–1·cm–1] | λPL (nm) b | ФPL c | τave (ns) d | τ1 (ns) d | τ2 (ns) d |
|---|---|---|---|---|---|---|---|---|---|
| 0F | −5.81 | –2.09 | 3.72 | 290 [29.5], 299 [24.6], 307 [25.0] | 328, 447 | <0.01 | 2.63 | 0.82 | 5.08 |
| 1F | –5.81 | –2.06 | 3.75 | 288 [29.6], 297 [24.5], 305 [24.2] | 330, 469 | <0.01 | 2.86 | 0.78 | 5.23 |
| 3Fa | –5.96 | –2.33 | 3.63 | 291 [28.2], 307 [25.0] | 343, 431 | 0.01 | 2.55 | 0.86 | 4.84 |
| 3Fb | –5.96 | –2.27 | 3.69 | 290 [28.5], 307 [25.3] | 340, 433 | 0.01 | 2.12 | 0.75 | 4.92 |
| 3Fc | –5.96 | –2.38 | 3.58 | 293 [27.7], 309 [25.2] | 349, 436 | 0.01 | 2.37 | 0.86 | 5.93 |
| 4F | –6.07 | –2.50 | 3.57 | 300 [28.5], 314 [27.9] | 369, 432 | 0.04 | 3.38 | 1.12 | 5.68 |
| 5F | –6.02 | –2.61 | 3.41 | 297 [26.7], 311 [26.1] | 406 | 0.08 | 2.82 | 1.41 | 5.42 |
| Compound | λPL (nm) a | ФPL b | τPL (ns) c | kr (108 s−1) d | knr (108 s−1) e |
|---|---|---|---|---|---|
| 0F | 359 | 0.04 | 0.76 | 0.53 | 12.6 |
| 1F | 375 | 0.10 | 2.21 | 0.45 | 4.07 |
| 3Fa | 368 | 0.31 | 2.12 | 1.46 | 3.25 |
| 3Fb | 381 | 0.37 | 3.81 | 0.97 | 1.65 |
| 3Fc | 374 | 0.14 | 1.13 | 1.24 | 7.61 |
| 4F | 375 | 0.04 | 0.77 | 0.52 | 12.5 |
| 5F | 465 | 0.51 | 2.37 | 2.15 | 2.07 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morita, M.; Yamada, S.; Konno, T. Systematic Studies on the Effect of Fluorine Atoms in Fluorinated Tolanes on Their Photophysical Properties. Molecules 2021, 26, 2274. https://doi.org/10.3390/molecules26082274
Morita M, Yamada S, Konno T. Systematic Studies on the Effect of Fluorine Atoms in Fluorinated Tolanes on Their Photophysical Properties. Molecules. 2021; 26(8):2274. https://doi.org/10.3390/molecules26082274
Chicago/Turabian StyleMorita, Masato, Shigeyuki Yamada, and Tsutomu Konno. 2021. "Systematic Studies on the Effect of Fluorine Atoms in Fluorinated Tolanes on Their Photophysical Properties" Molecules 26, no. 8: 2274. https://doi.org/10.3390/molecules26082274