Essential Oil Profile and Yield of Corolla, Calyx, Leaf, and Whole Flowering Top of Cultivated Lavandula angustifolia Mill. (Lamiaceae) from Utah
Abstract
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Bailey, L.H. Manual of Cultivated Plants, revised ed.; Macmillan Publishing Company: New York, NY, USA, 1951; pp. 850–851. [Google Scholar]
- Lis-Balchin, M. Lavender: The Genus Lavandula, 1st ed.; Taylor & Francis: London, UK, 2002; pp. 35–45. [Google Scholar]
- Schmiderer, C.; Grassi, P.; Novak, J.; Weber, M.; Franz, C. Diversity of essential oil glands of clary sage (Salvia sclarea L., Lamiaceae). Plant Biol. 2008, 10, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Kirchoff, B.K.; Liao, J. The capitate and peltate glandular trichomes of Lavandula pinnata L. (Lamiaceae): histochemistry, ultrastructure, and secretion1. J. Torrey Bot. Soc. 2008, 135, 155–167. [Google Scholar] [CrossRef]
- Urwin, N.A.R.; Horsnell, J.; Moon, T. Generation and characterisation of colchicine-induced autotetraploid Lavandula angustifolia. Euphytica 2007, 156, 257–266. [Google Scholar] [CrossRef]
- Iriti, M.; Colnaghi, G.; Chemat, F.; Smadja, J.; Faoro, F.; Visinoni, F.A. Histo-cytochemistry and scanning electron microscopy of lavender glandular trichomes following conventional and microwave-assisted hydrodistillation of essential oils: a comparative study. Flavour Fragr. J. 2006, 21, 704–712. [Google Scholar] [CrossRef]
- Buchbauer, G.; Jirovetz, L.; Jäger, W. Aromatherapy: Evidence for Sedative Effects of the Essential Oil of Lavender after Inhalation. Zeitschrift für Naturforschung C 1991, 46, 1067–1072. [Google Scholar] [CrossRef]
- Kim, S.; Kim, H.-J.; Yeo, J.-S.; Hong, S.-J.; Lee, J.-M.; Jeon, Y. The Effect of Lavender Oil on Stress, Bispectral Index Values, and Needle Insertion Pain in Volunteers. J. Altern. Complement. Med. 2011, 17, 823–826. [Google Scholar] [CrossRef] [Green Version]
- Cardia, G.F.E.; Silva-Filho, S.E.; Silva, E.L.; Uchida, N.S.; Cavalcante, H.A.O.; Cassarotti, L.L.; Salvadego, V.E.C.; Spironello, R.A.; Bersani-Amado, C.A.; Cuman, R.K.N. Effect of Lavender (Lavandula angustifolia) Essential Oil on Acute Inflammatory Response. Evidence-Based Complement. Altern. Med. 2018, 2018, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Huang, M.-Y.; Liao, M.-H.; Wang, Y.-K.; Huang, Y.-S.; Wen, H.-C. Effect of Lavender Essential Oil on LPS-Stimulated Inflammation. Am. J. Chin. Med. 2012, 40, 845–859. [Google Scholar] [CrossRef]
- Donelli, D.; Antonelli, M.; Bellinazzi, C.; Gensini, G.F.; Firenzuoli, F. Effects of lavender on anxiety: A systematic review and meta-analysis. Phytomedicine 2019, 65, 153099. [Google Scholar] [CrossRef]
- Karan, N.B. Influence of lavender oil inhalation on vital signs and anxiety: A randomized clinical trial. Physiol. Behav. 2019, 211, 112676. [Google Scholar] [CrossRef]
- Kang, H.-J.; Nam, E.S.; Lee, Y.; Kim, M. How Strong is the Evidence for the Anxiolytic Efficacy of Lavender?: Systematic Review and Meta-analysis of Randomized Controlled Trials. Asian Nurs. Res. 2019, 13, 295–305. [Google Scholar] [CrossRef] [Green Version]
- Sánchez-Vidaña, D.I.; Po, K.K.-T.; Fung, T.K.-H.; Chow, J.K.-W.; Lau, W.K.-W.; So, P.-K.; Lau, B.W.-M.; Tsang, H.W.-H. Lavender essential oil ameliorates depression-like behavior and increases neurogenesis and dendritic complexity in rats. Neurosci. Lett. 2019, 701, 180–192. [Google Scholar] [CrossRef]
- Jafari-Koulaee, A.; Elyasi, F.; Taraghi, Z.; Ilali, E.S.; Moosazadeh, M. A Systematic Review of the Effects of Aromatherapy with Lavender Essential Oil on Depression. Central Asian J. Glob. Heal. 2020, 9, 14. [Google Scholar] [CrossRef]
- Casabianca, H.; Graff, J.B.; Faugier, V.; Fleig, F.; Grenier, C. Enantiomeric Distribution Studies of Linalool and Linalyl Acetate. A Powerful Tool for Authenticity Control of Essential Oils. J. High Resolut. Chromatogr. 1998, 21, 107–112. [Google Scholar] [CrossRef]
- ISO 3515:2002, Oil of lavender (Lavandula angustifolia Mill.) Standard. Available online: http://www.iso.org (accessed on 4 February 2021).
- Jung, J.; Sewenig, S.; Hener, U.; Mosandl, A. Comprehensive authenticity assessment of lavender oils using multielement/multicomponent isotope ratio mass spectrometry analysis and enantioselective multidimensional gas chromatography-mass spectrometry. Eur. Food Res. Technol. 2005, 220, 232–237. [Google Scholar] [CrossRef]
- ISO 8902:2009, Oil of lavandin Grosso (Lavandula angustifolia Mill. x Lavandula latifolia Medik.), French type Standard. Available online: http://www.iso.org (accessed on 2 April 2021).
- ISO 4719:2012, Essential oil of spike lavender (Lavandula latifolia Medikus), Spanish type Standard. Available online: http://www.iso.org (accessed on 2 April 2021).
- Burfield, T. The Adulteration of Essential oils-and the Consequences to Aromatherapy and Natural Perfumery Practice. A presentation to the IFA Annual AGM London. 2003. Available online: https://issuu.com/achs/docs/adulterationupdate08/8; (accessed on 16 April 2021).
- Chakopoulou, P.S.; Goliaris, A.H.; Katsiotis, S.T. Contribution to the Analysis of the Volatile Constituents from some Lavender and Lavandin Cultivars Grown in Greece. Sci. Pharm. 2003, 71, 229–234. [Google Scholar] [CrossRef] [Green Version]
- Détár, E.; Németh, É.Z.; Gosztola, B.; Demján, I.; Pluhár, Z. Effects of variety and growth year on the essential oil properties of lavender (Lavandula angustifolia Mill.) and lavandin (Lavandula x intermedia Emeric ex Loisel.). Biochem. Syst. Ecol. 2020, 90, 104020. [Google Scholar] [CrossRef]
- Zagorcheva, T.; Stanev, S.; Rusanov, K.; Atanassov, I. Comparative GC/MS analysis of lavender (Lavandula angustifolia Mill.) inflorescence and essential oil volatiles. J. Agric. Sci. Technol. 2013, 5, 459–462. [Google Scholar]
- Hassiotis, C.; Ntana, F.; Lazari, D.; Poulios, S.; Vlachonasios, K. Environmental and developmental factors affect essential oil production and quality of Lavandula angustifolia during flowering period. Ind. Crop. Prod. 2014, 62, 359–366. [Google Scholar] [CrossRef]
- Zheljazkov, V.D.; Astatkie, T.; Hristov, A.N. Lavender and hyssop productivity, oil content, and bioactivity as a function of harvest time and drying. Ind. Crop. Prod. 2012, 36, 222–228. [Google Scholar] [CrossRef]
- Dušková, E.; Dušek, K.; Indrák, P.; Smékalová, K. Postharvest changes in essential oil content and quality of lavender flowers. Ind. Crop. Prod. 2016, 79, 225–231. [Google Scholar] [CrossRef]
- Smigielski, K.; Prusinowska, R.; Raj, A.; Sikora, M.; Woliñska, K.; Gruska, R. Effect of Drying on the Composition of Essential Oil from Lavandula angustifolia. J. Essent. Oil Bear. Plants 2011, 14, 532–542. [Google Scholar] [CrossRef]
- Zheljazkov, V.D.; Cantrell, C.L.; Astatkie, T.; Jeliazkova, E. Distillation Time Effect on Lavender Essential Oil Yield and Composition. J. Oleo Sci. 2013, 62, 195–199. [Google Scholar] [CrossRef]
- Babu, G.K.; Singh, B.I. Characteristics variation of lavender oil produced by different hydrodistillation techniques. In Comprehensive Bioactive Natural Products: Quality Control & Standardization; Studium Press: New Delhi, India, 2010; Volume 8, pp. 122–136. [Google Scholar]
- Babu, G.D.K.; Thakur, V.; Singh, B. Variability in the Composition of Lavandula angustifolia Extracts due to Extraction Methods. J. Herbs, Spices Med. Plants 2016, 22, 173–182. [Google Scholar] [CrossRef]
- Da Porto, C.; Decorti, D.; Kikic, I. Flavour compounds of Lavandula angustifolia L. to use in food manufacturing: Comparison of three different extraction methods. Food Chem. 2009, 112, 1072–1078. [Google Scholar] [CrossRef]
- Kim, N.-S.; Lee, D.-S. Comparison of different extraction methods for the analysis of fragrances from Lavandula species by gas chromatography–mass spectrometry. J. Chromatogr. A 2002, 982, 31–47. [Google Scholar] [CrossRef]
- Adaszynska-Skwirzynska, M.; Dzieciol, M. Comparison of Chemical Composition and Antimicrobial Activity of Essential Oils Obtained from Different Cultivars and Morphological Parts of Lavandula angustifolia. J. Essent. Oil Bear. Plants 2018, 21, 1532–1541. [Google Scholar] [CrossRef]
- Venskutonis, P.R.; Dapkevicius, A.; Baranauskiene, M. Composition of the Essential Oil of Lavender (Lavandula angustifolia Mill.) from Lithuania. J. Essent. Oil Res. 1997, 9, 107–110. [Google Scholar] [CrossRef]
- Özel, A. Determining leaf yield, some plant characters and leaf essential oil components of different cultivars of lavender and lavandin (Lavandula spp.) on the Harran Plain ecological conditions. Appl. Ecol. Environ. Res. 2019, 17, 14087–14094. [Google Scholar] [CrossRef]
- Guitton, Y.; Nicolã, F.; Moja, S.; Valot, N.; Legrand, S.; Jullien, F.; Legendre, L.; Nicolè, F.; Jullien, F. Differential accumulation of volatile terpene and terpene synthase mRNAs during lavender (Lavandula angustifolia and L. x intermedia) inflorescence development. Physiol. Plant. 2010, 138, 150–163. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th ed.; Allured Publ.: Carol Stream, IL, USA, 2007. [Google Scholar]

| KI | Compound | Corolla | Calyx | Leaf | Flowering Top |
|---|---|---|---|---|---|
| 921 | tricyclene | nd | nd | t | 0.1 |
| 924 | α-thujene | nd | 0.1 | t | 0.2 |
| 932 | α-pinene | t | 0.2 | 0.1 | 0.3 |
| 946 | camphene | 0.1 | 0.1 | 0.6 | 0.4 |
| 969 | sabinene | t | t | 0.1 | 0.1 |
| 974 | 1-octen-3-ol | 0.1 | 0.2 | nd | nd |
| 974 | β-pinene | nd | nd | 0.2 | 0.2 |
| 979 | 3-octanone | 0.1 | 0.7 | 0.2 | 1.1 |
| 988 | myrcene | 0.1 | 0.4 | 0.6 | 0.6 |
| 988 | 3-octanol | t | nd | nd | nd |
| 993 | butyl butanoate | nd | 0.2 | nd | 0.2 |
| 1002 | α-phellandrene | t | t | 0.2 | t |
| 1007 | hexyl acetate | t | 0.3 | nd | nd |
| 1008 | δ-3-carene | nd | nd | 1.8 | 0.5 |
| 1014 | α-terpinene | t | t | t | 0.2 |
| 1020 | p-cymene | t | t | 0.7 | 0.1 |
| 1022 | ο-cymene | 0.2 | 0.1 | 1.5 | 0.4 |
| 1024 | limonene | 0.3 | 0.2 | 1.8 | 0.4 |
| 1025 | β-phellandrene | t | 0.1 | 2.3 | 0.3 |
| 1026 | 1,8-cineole | 0.9 | 0.3 | 1.2 | 1.5 |
| 1032 | (Z)-β-ocimene | 0.5 | 5.4 | 0.3 | 4.5 |
| 1044 | (E)-β-ocimene | 0.3 | 1.6 | 0.2 | 2.9 |
| 1054 | γ-terpinene | t | 0.2 | 0.1 | 0.1 |
| 1065 | cis-sabinene hydrate | 0.1 | 0.2 | 0.1 | 0.2 |
| 1067 | cis-linalool oxide | t | 0.1 | nd | t |
| 1084 | trans-linalool oxide | t | t | nd | t |
| 1085 | p-mentha-2,4(8)-diene | nd | nd | 0.3 | nd |
| 1086 | terpinolene | t | t | 0.2 | t |
| 1095 | linalool | 10.8 | 32.9 | 1.1 | 26.5 |
| 1100 | n-nonanal | 0.2 | nd | nd | nd |
| 1101 | hexyl propanoate | nd | nd | nd | 0.1 |
| 1110 | 1-octen-3-yl acetate | 0.4 | 0.5 | 1.5 | 0.7 |
| 1118 | cis-p-menth-2-en-1-ol | 0.2 | nd | 0.1 | nd |
| 1120 | 3-octanol acetate | nd | 0.1 | nd | 0.1 |
| 1122 | α-campholenal | 0.1 | nd | 0.1 | nd |
| 1128 | allo-ocimene | nd | 0.4 | nd | 0.3 |
| 1132 | cis-limonene oxide | 0.1 | nd | t | nd |
| 1135 | trans-pinocarveol | 0.2 | nd | t | nd |
| 1136 | trans-p-menth-2-en-1-ol | 0.1 | nd | t | nd |
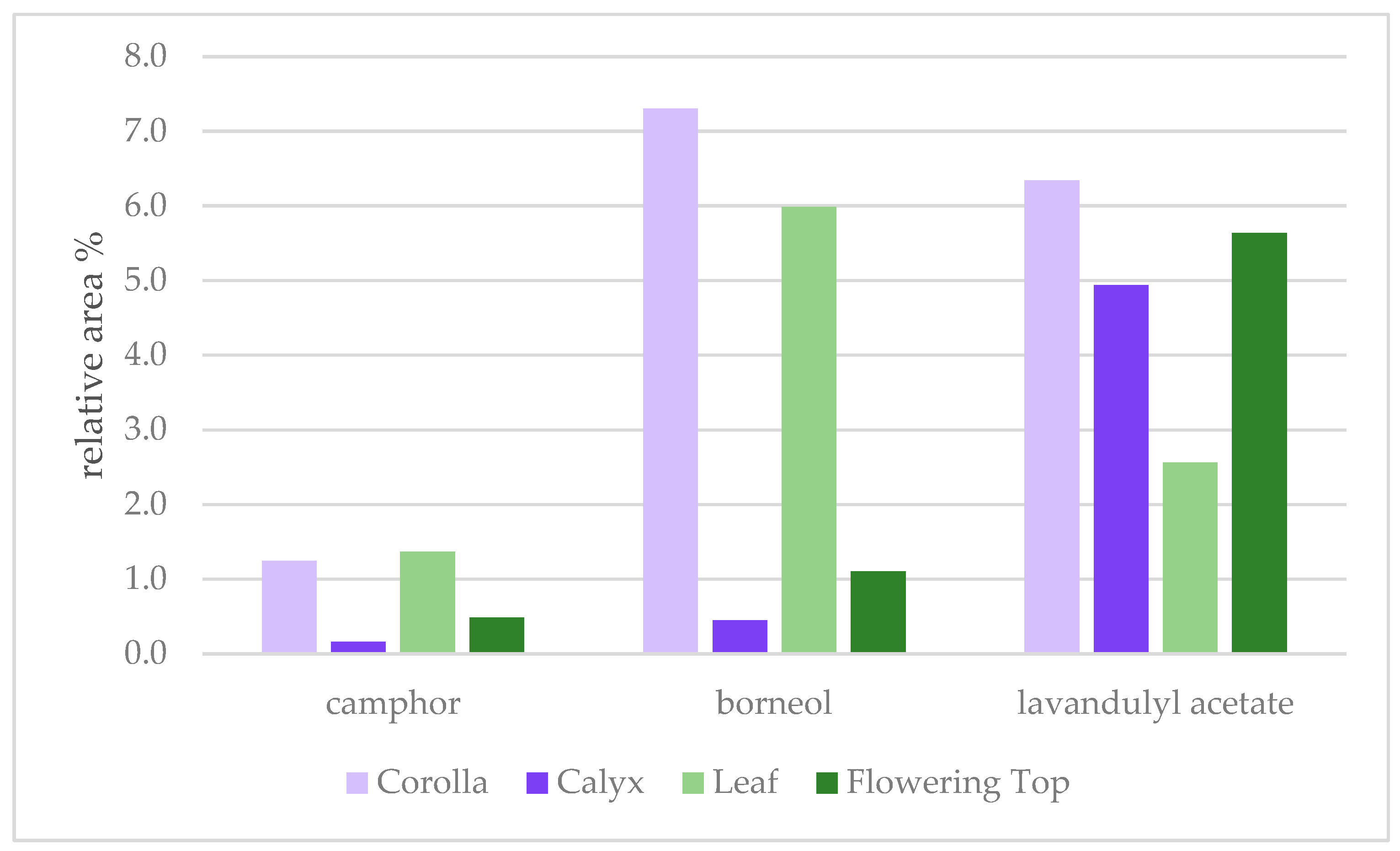
| 1141 | camphor | 1.2 | 0.2 | 1.4 | 0.5 |
| 1145 | camphene hydrate | nd | nd | t | nd |
| 1155 | isoborneol | 0.1 | nd | 0.1 | nd |
| 1165 | lavandulol | 0.8 | 1.0 | 0.3 | 0.6 |
| 1165 | borneol | 7.3 | 0.4 | 6.0 | 1.1 |
| 1174 | terpinen-4-ol | 1.9 | 7.0 | 0.4 | 5.3 |
| 1179 | p-cymen-8-ol | 0.2 | t | 0.1 | 0.1 |
| 1183 | cryptone | 1.7 | nd | 0.3 | 0.2 |
| 1186 | α-terpineol | 0.3 | 0.4 | 0.1 | 0.3 |
| 1191 | hexyl butanoate | nd | 0.4 | nd | 0.4 |
| 1194 | myrtenol | 0.3 | nd | 0.2 | t |
| 1207 | trans-piperitol | 0.3 | nd | 0.1 | nd |
| 1215 | trans-carveol | 0.2 | nd | 0.1 | nd |
| 1227 | nerol | nd | 0.1 | nd | t |
| 1235 | isobornyl formate | 1.0 | 0.1 | 1.6 | 0.2 |
| 1238 | cumin aldehyde | 2.0 | nd | 1.3 | 0.2 |
| 1239 | carvone | 0.5 | nd | 0.3 | t |
| 1254 | linalool acetate | 18.4 | 32.0 | 3.0 | 34.3 |
| 1284 | bornyl acetate | 2.3 | nd | 4.8 | nd |
| 1288 | lavandulyl acetate | 6.3 | 4.9 | 2.6 | 5.6 |
| 1289 | p-cymen-7-ol | 0.5 | nd | nd | t |
| 1298 | carvacrol | nd | nd | t | nd |
| 1330 | hexyl tiglate | t | t | nd | t |
| 1330 | 3-oxo-p-menth-1-en-7-al | 0.1 | nd | nd | nd |
| 1343 | benzyl butanoate | nd | t | nd | nd |
| 1359 | neryl acetate | 0.6 | 0.2 | 0.6 | 0.2 |
| 1379 | geranyl acetate | 0.9 | 0.4 | 2.5 | 0.3 |
| 1382 | hexyl hexanoate | nd | 0.1 | nd | nd |
| 1389 | β-elemene | nd | nd | 0.1 | nd |
| 1410 | α-cedrene | 0.1 | nd | 0.3 | nd |
| 1411 | α-cis-bergamotene | nd | nd | 0.1 | nd |
| 1416 | α-santalene | 0.8 | 0.2 | 1.1 | 0.2 |
| 1417 | (E)-caryophyllene | 1.7 | 2.5 | 2.2 | 2.3 |
| 1419 | β-cedrene | t | nd | 0.1 | nd |
| 1432 | α-trans-bergamotene | 0.2 | 0.1 | 0.3 | 0.1 |
| 1432 | coumarin | nd | nd | 0.5 | 0.2 |
| 1440 | (Z)-β-farnesene | 1.4 | 1.8 | nd | 1.6 |
| 1452 | α-humulene | nd | 0.1 | 0.1 | 0.1 |
| 1465 | cis-muurola-4(14),5-diene | nd | 0.1 | 0.8 | 0.1 |
| 1471 | dauca-5,8-diene | 0.1 | nd | nd | nd |
| 1474 | 10-epi-β-acoradiene | t | nd | 0.2 | nd |
| 1480 | germacrene D | 0.3 | 0.4 | 0.3 | 0.3 |
| 1505 | β-bisabolene | 0.1 | 0.1 | t | t |
| 1513 | γ-cadinene | 5.1 | 0.4 | 11.0 | 0.7 |
| 1528 | cis-calamenene | 0.2 | t | 0.6 | t |
| 1537 | α-cadinene | nd | nd | 0.1 | nd |
| 1582 | caryophyllene oxide | 4.9 | 0.1 | 4.9 | 0.3 |
| 1627 | 1-epi-cubenol | 1.0 | 0.1 | 1.8 | 0.1 |
| 1638 | epi-α-cadinol | 10.0 | 1.1 | 19.8 | 1.3 |
| 1685 | germacra-4(15),5,10(14)-trien-1-α-ol | 0.4 | nd | nd | nd |
| 1688 | cis-14-nor-muurol-5-en-4-one | 1.2 | 0.1 | 1.2 | 0.1 |
| 1 1744 | unknown compound | 1.4 | t | 1.5 | t |
| 1759 | benzyl benzoate | t | t | t | t |
| 1 1847 | unknown compound | 1.3 | nd | 0.2 | nd |
| Mass Distilled (g) | Yield EO (g) | Yield EO (%) | ||
|---|---|---|---|---|
| Flowering Top | 1 | 134.60 | 1.12 | 0.83 |
| 2 | 129.48 | 0.86 | 0.66 | |
| 3 | 129.32 | 0.65 | 0.50 | |
| Avg: | 131.13 | 0.88 | 0.66 | |
| %RSD (n = 3) | 24.9 | |||
| Corolla | 1 | 48.44 | 0.04 | 0.08 |
| 2 | 43.04 | 0.05 | 0.12 | |
| 3 | 50.57 | 0.05 | 0.10 | |
| Avg: | 47.35 | 0.05 | 0.10 | |
| %RSD (n = 3) | 20.0 | |||
| Calyx | 1 | 87.05 | 1.09 | 1.25 |
| 2 | 107.02 | 1.42 | 1.33 | |
| 3 | 102.67 | 1.33 | 1.30 | |
| Avg: | 98.91 | 1.28 | 1.29 | |
| %RSD (n = 3) | 3.1 | |||
| Leaf | 1 | 118.53 | 0.06 | 0.05 |
| 2 | 100.03 | 0.05 | 0.05 | |
| 3 | 123.36 | 0.06 | 0.05 | |
| Avg: | 113.97 | 0.06 | 0.05 | |
| %RSD (n = 3) | 0.0 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wilson, T.M.; Poulson, A.; Packer, C.; Carlson, R.E.; Buch, R.M. Essential Oil Profile and Yield of Corolla, Calyx, Leaf, and Whole Flowering Top of Cultivated Lavandula angustifolia Mill. (Lamiaceae) from Utah. Molecules 2021, 26, 2343. https://doi.org/10.3390/molecules26082343
Wilson TM, Poulson A, Packer C, Carlson RE, Buch RM. Essential Oil Profile and Yield of Corolla, Calyx, Leaf, and Whole Flowering Top of Cultivated Lavandula angustifolia Mill. (Lamiaceae) from Utah. Molecules. 2021; 26(8):2343. https://doi.org/10.3390/molecules26082343
Chicago/Turabian StyleWilson, Tyler M., Ariel Poulson, Chris Packer, Richard E. Carlson, and R. Michael Buch. 2021. "Essential Oil Profile and Yield of Corolla, Calyx, Leaf, and Whole Flowering Top of Cultivated Lavandula angustifolia Mill. (Lamiaceae) from Utah" Molecules 26, no. 8: 2343. https://doi.org/10.3390/molecules26082343






