Supramolecular Control of Singlet Oxygen Generation
Abstract
:1. Introduction
2. Singlet Oxygen Background
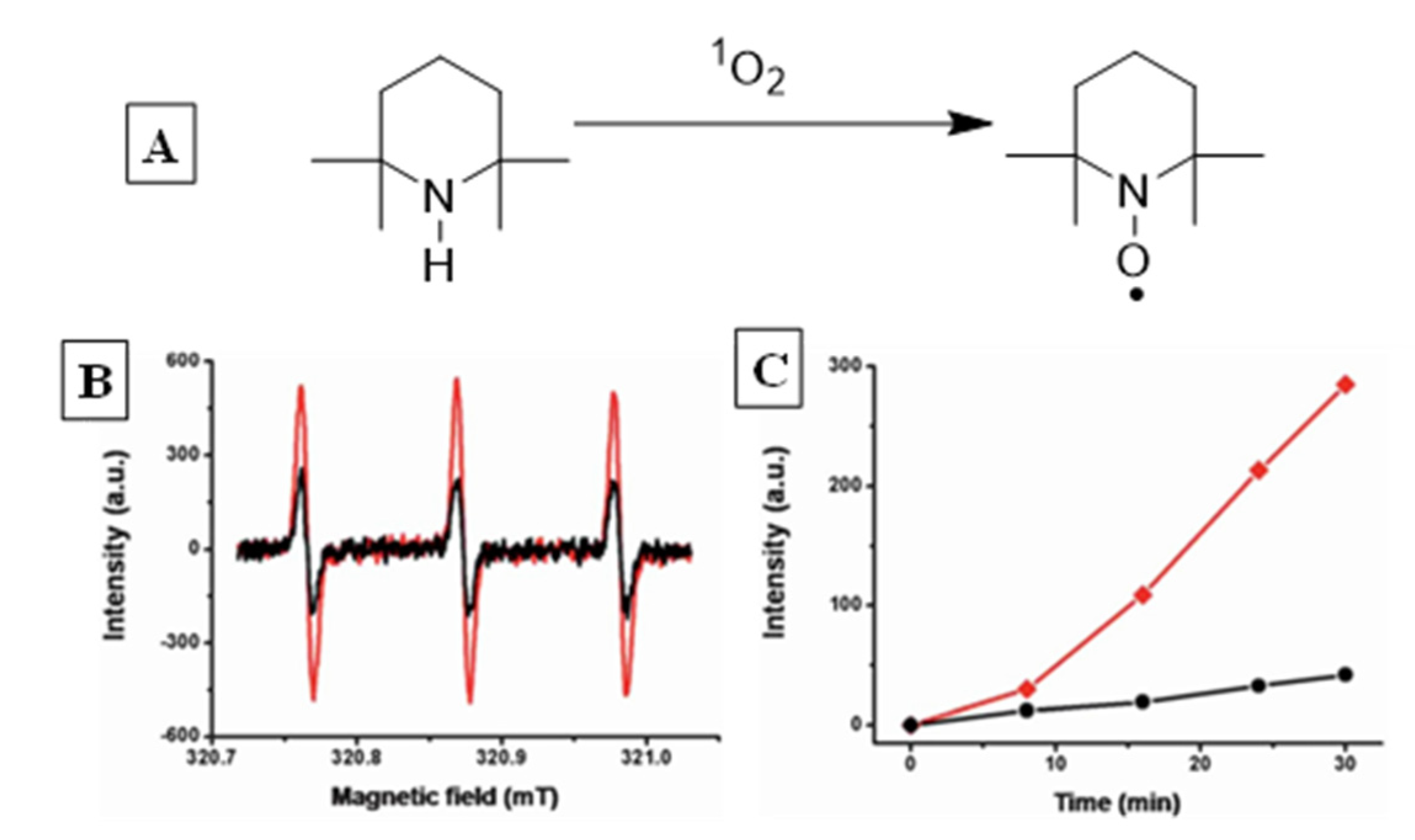
2.1. Stability, Detection and Quantification of Singlet Oxygen
2.2. Sensitized Singlet Oxygen Generation
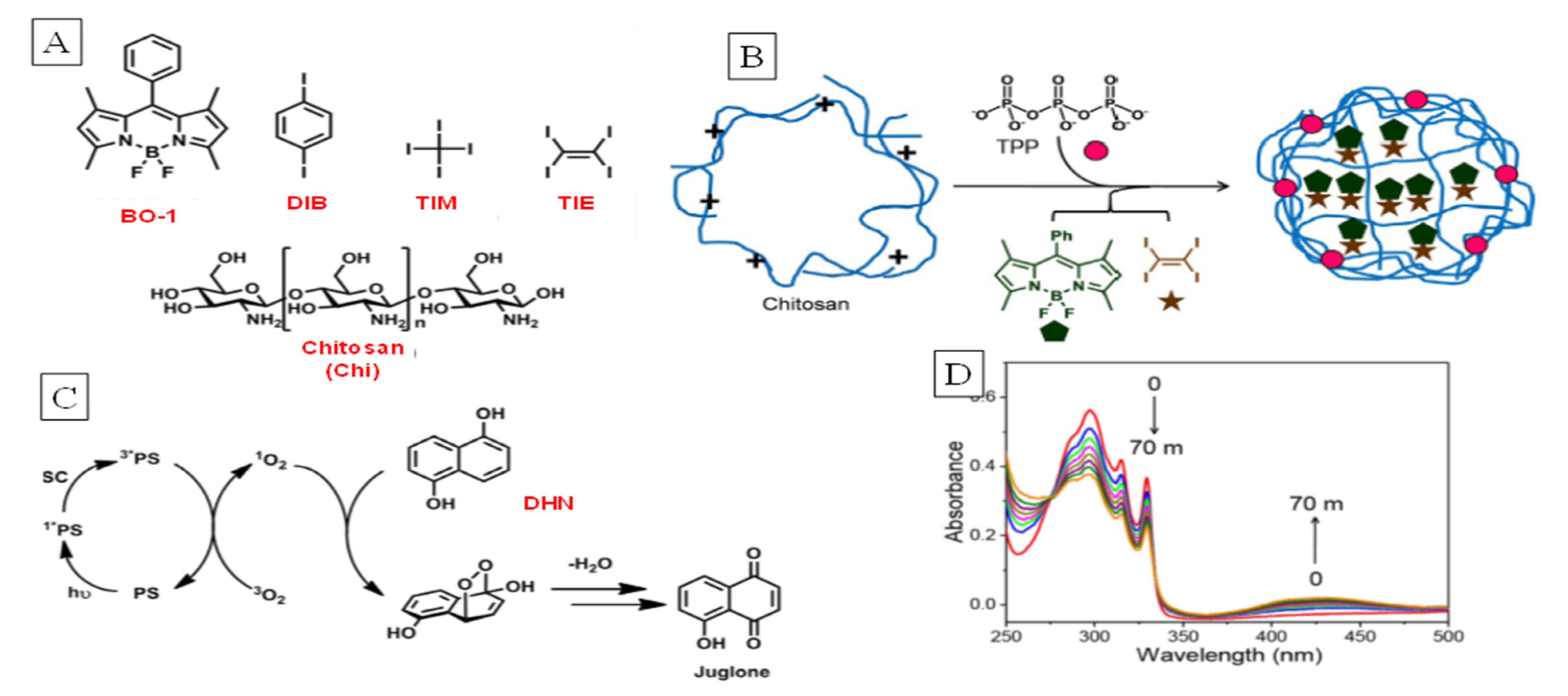
2.3. Host–Guest Chemistry and Nano-Containers
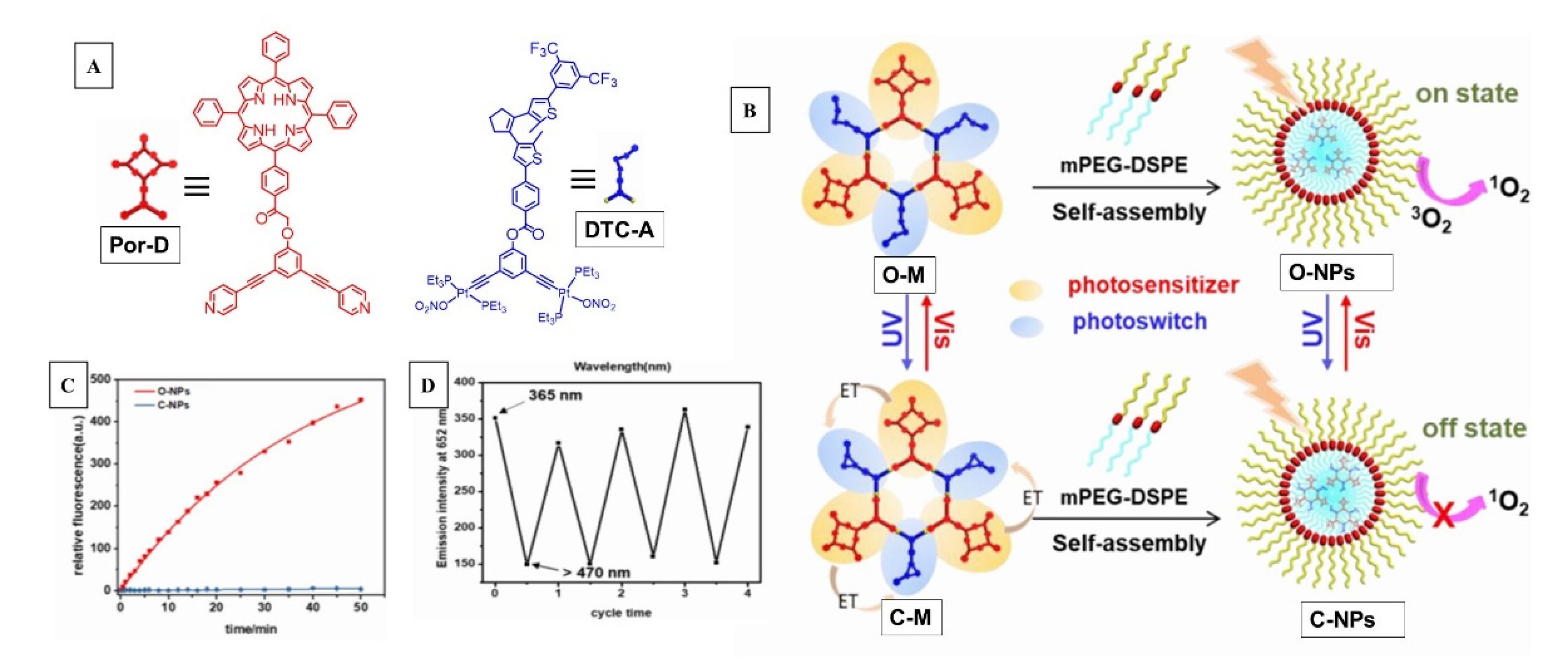
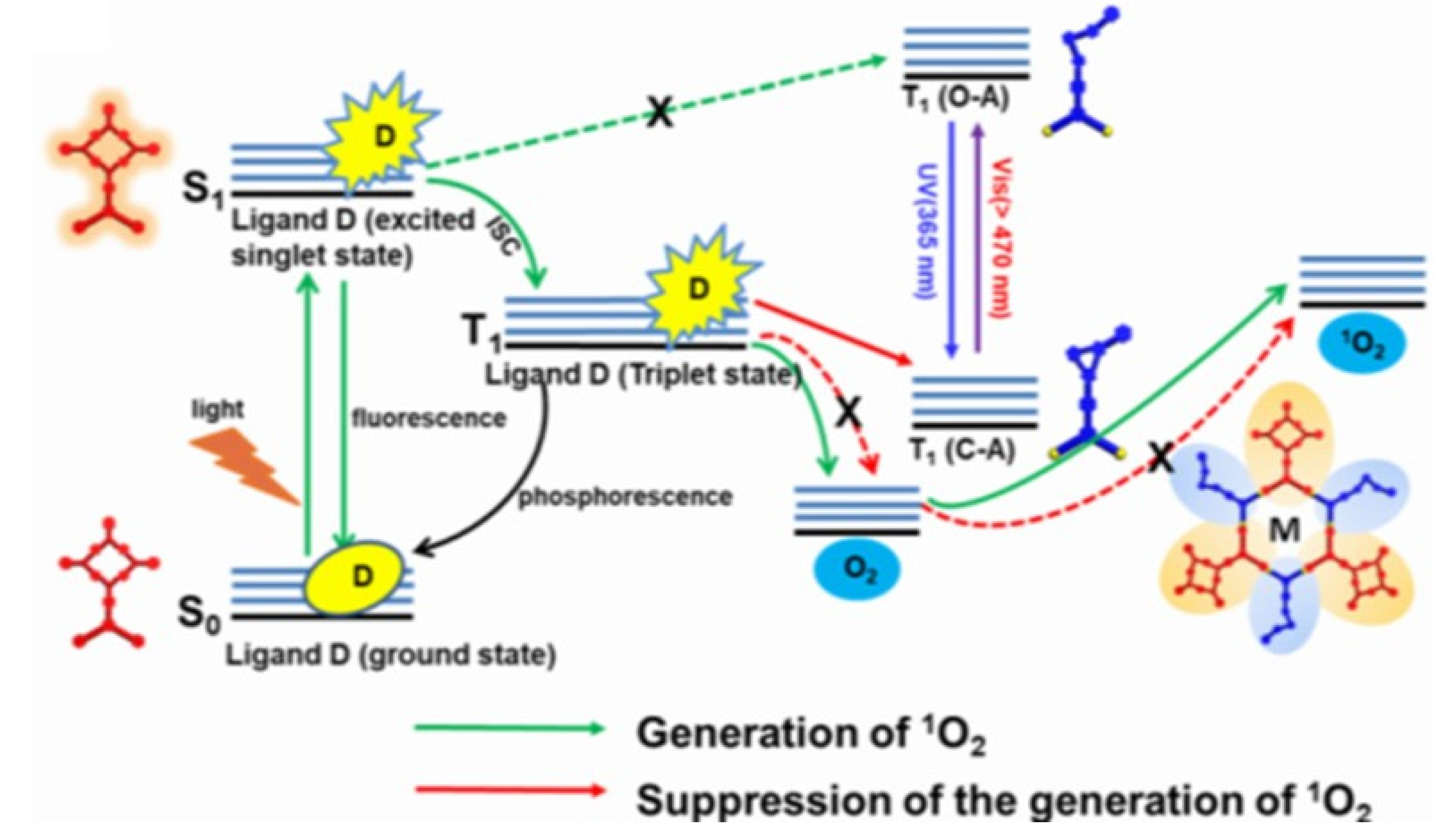
3. Supramolecular Approaches to Singlet Oxygen Generation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Turro, N.J. Modern Molecular Photochemistry; Addison-Wesley Publishing Co.: Boston, MA, USA, 1978; p. 628. [Google Scholar]
- Ogilby, P.R. Symposium-in-print: Singlet oxygen singlet oxygen-introduction. Photochem. Photobiol. 2006, 82, 1133–1135. [Google Scholar] [CrossRef]
- Wayne, R.P. Singlet molecular oxygen. Advan. Photochem. 1969, 7, 311–371. [Google Scholar]
- Greer, A. Christopher Foote’s Discovery of the Role of Singlet Oxygen [1O2 (1Δg)] in Photosensitized Oxidation Reactions. Acc. Chem. Res. 2006, 39, 797–804. [Google Scholar] [CrossRef] [PubMed]
- Krumova, K.; Cosa, G. Overview of reactive oxygen species. Compr. Ser. Photochem. Photobiol. Sci. 2016, 13, 3–21. [Google Scholar]
- Khan, A.U.; Kasha, M. Singlet molecular oxygen evolution upon simple acidification of aqueous hypochlorite: Application to studies on the deleterious health effects of chlorinated drinking water. Proc. Natl. Acad. Sci. USA 1994, 91, 12362–12364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blazquez-Castro, A. Direct 1O2 optical excitation: A tool for redox biology. Redox Biol. 2017, 13, 39–59. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, R. Photosensitized generation of singlet oxygen. Photochem. Photobiol. 2006, 82, 1161–1177. [Google Scholar] [CrossRef]
- Triantaphylides, C.; Havaux, M. Singlet oxygen in plants: Production, detoxification and signaling. Trends Plant Sci. 2009, 14, 219–228. [Google Scholar] [CrossRef]
- Khan, A.U.; Pitts, J.N.; Smith, E.B. Singlet oxygen in the environmental sciences. Role of singlet molecular oxygen in the production of photochemical air pollution. Environ. Sci. Technol. 1967, 1, 656–657. [Google Scholar] [CrossRef]
- Dmitrieva, V.A.; Tyutereva, E.V.; Voitsekhovskaja, O.V. Singlet Oxygen in Plants: Generation, Detection, and Signaling Roles. Int. J. Mol. Sci. 2020, 21, 3237. [Google Scholar] [CrossRef]
- Stief, T.W. The physiology and pharmacology of singlet oxygen. Med. Hypotheses 2003, 60, 567–572. [Google Scholar] [CrossRef]
- Chilakamarthi, U.; Giribabu, L. Photodynamic Therapy: Past, Present and Future. Chem. Rec. 2017, 17, 775–802. [Google Scholar] [CrossRef]
- Darlenski, R.; Fluhr, J.W. Photodynamic therapy in dermatology: Past, present, and future. J. Biomed. Opt. 2013, 18, 061208. [Google Scholar] [CrossRef]
- Tandon, Y.K.; Yang, M.F.; Baron, E.D. Role of photodynamic therapy in psoriasis: A brief review. Photodermatol. Photoimmunol. Photomed. 2008, 24, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Bruschi, M.L.; Bassi da Silva, J.; Rosseto, H.C. Photodynamic Therapy of Psoriasis Using Photosensitizers of Vegetable Origin. Curr. Pharm. Des. 2019, 25, 2279–2291. [Google Scholar] [CrossRef]
- Agostinis, P.; Berg, K.; Cengel, K.A.; Foster, T.H.; Girotti, A.W.; Gollnick, S.O.; Hahn, S.M.; Hamblin, M.R.; Juzeniene, A.; Kessel, D.; et al. Photodynamic therapy of cancer: An update. CA: Cancer J. Clin. 2011, 61, 250–281. [Google Scholar] [CrossRef] [PubMed]
- El-Hussein, A.; Manoto, S.L.; Ombinda-Lemboumba, S.; Alrowaili, Z.A.; Mthunzi-Kufa, P. A Review of Chemotherapy and Photodynamic Therapy for Lung Cancer Treatment. AntiCancer Agents Med. Chem. 2021, 21, 149–161. [Google Scholar] [CrossRef]
- Nseyo, U.O. Photodynamic therapy in the management of bladder cancer. J. Clin. Laser Med. Surg 1996, 14, 271–280. [Google Scholar] [CrossRef]
- van den Bergh, H. Photodynamic therapy of age-related macular degeneration: History and principles. Semin. Ophthalmol. 2001, 16, 181–200. [Google Scholar] [CrossRef]
- Floyd, R.A.; Schneider, J.E.; Dittmer, D.P. Methylene blue photoinactivation of RNA viruses. Antivir. Res. 2004, 61, 141–151. [Google Scholar] [CrossRef]
- Garcia-Fresnadillo, D. Singlet oxygen photosensitizing materials for point-of-use water disinfection with solar reactors. ChemPhotoChem 2018, 2, 512–534. [Google Scholar] [CrossRef]
- Ghogare, A.A.; Greer, A. Using Singlet Oxygen to Synthesize Natural Products and Drugs. Chem. Rev. 2016, 116, 9994–10034. [Google Scholar] [CrossRef]
- Isomura, M. Singlet oxygen for organic syntheses: Novel approaches for scale-up. Yuki Gosei Kagaku Kyokaishi 2010, 68, 262–263. [Google Scholar] [CrossRef] [Green Version]
- Nardello-Rataj, V.; Alsters, P.L.; Aubry, J.-M. Industrial Prospects for the Chemical and Photochemical Singlet Oxygenation of Organic Compounds; Wiley-VCH GmbH & Co. KGaA: Weinheim, Germany, 2016; pp. 369–395. [Google Scholar]
- Stephenson, L.M. Recent Experimental Developments in the Singlet Oxygen Ene Reaction with Olefins; Academic Press: Cambridge, MA, USA, 1981; pp. 371–378. [Google Scholar]
- Stephenson, L.M.; Grdina, M.J.; Orfanopoulos, M. Mechanism of the ene reaction between singlet oxygen and olefins. Acc. Chem. Res. 1980, 13, 419–425. [Google Scholar] [CrossRef]
- Adam, W.; Saha-Moller, C.R.; Schambony, S.B.; Schmid, K.S.; Wirth, T. Stereocontrolled photooxygenations-a valuable synthetic tool. Photochem. Photobiol. 1999, 70, 476–483. [Google Scholar] [CrossRef]
- Clennan, E.L. Overview of the chemical reactions of singlet oxygen. Compr. Ser. Photochem. Photobiol. Sci. 2016, 13, 353–367. [Google Scholar]
- Clennan, E.L.; Pace, A. Advances in singlet oxygen chemistry. Tetrahedron 2005, 61, 6665–6691. [Google Scholar] [CrossRef]
- Velez-Pena, E.; Perez-Obando, J.; Pais-Ospina, D.; Marin-Silva, D.A.; Pinotti, A.; Canneva, A.; Donadelli, J.A.; Damonte, L.; Pizzio, L.R.; Osorio-Vargas, P.; et al. Self-cleaning and antimicrobial photo-induced properties under indoor lighting irradiation of chitosan films containing Melon/TiO2 composites. Appl. Surf. Sci. 2020, 508, 144895. [Google Scholar] [CrossRef]
- Sunday, M.O.; Sakugawa, H. A simple, inexpensive method for gas-phase singlet oxygen generation from sensitizer-impregnated filters: Potential application to bacteria/virus inactivation and pollutant degradation. Sci. Total Environ. 2020, 746, 141186. [Google Scholar] [CrossRef]
- Belousova, I.M.; Danilov, O.B.; Kiselev, V.M.; Mak, A.A. Conversion of solar energy to laser beam by fullerene-oxygen-iodine laser. Proc. SPIE 2011, 7822, 78220N/1–78220N/5. [Google Scholar]
- Flors, C.; Griesbeck, A.G.; Vassilikogiannakis, G. Singlet Oxygen: Chemistry, Applications and Challenges Ahead. ChemPhotoChem 2018, 2, 510–511. [Google Scholar] [CrossRef]
- Torring, T.; Helmig, S.; Ogilby, P.R.; Gothelf, K.V. Singlet oxygen in DNA nanotechnology. Acc. Chem. Res. 2014, 47, 1799–1806. [Google Scholar] [CrossRef] [PubMed]
- Ogilby, P.R. Singlet oxygen: There is indeed something new under the sun. Chem. Soc. Rev. 2010, 39, 3181–3209. [Google Scholar] [CrossRef]
- Shen, Y.; Shuhendler, A.J.; Ye, D.; Xu, J.-J.; Chen, H.-Y. Two-photon excitation nanoparticles for photodynamic therapy. Chem. Soc. Rev. 2016, 45, 6725–6741. [Google Scholar] [CrossRef]
- Ogilby, P.R. Solvent effects on the radiative transitions of singlet oxygen. Acc. Chem. Res. 1999, 32, 512–519. [Google Scholar] [CrossRef]
- Bregnhoej, M.; Westberg, M.; Minaev, B.F.; Ogilby, P.R. Singlet Oxygen Photophysics in Liquid Solvents: Converging on a Unified Picture. Acc. Chem. Res. 2017, 50, 1920–1927. [Google Scholar] [CrossRef]
- Wang, K.-K.; Song, S.; Jung, S.-J.; Hwang, J.-W.; Kim, M.-G.; Kim, J.-H.; Sung, J.; Lee, J.-K.; Kim, Y.-R. Lifetime and diffusion distance of singlet oxygen in air under everyday atmospheric conditions. Phys. Chem. Chem. Phys. 2020, 22, 21664–21671. [Google Scholar] [CrossRef]
- You, Y. Chemical tools for the generation and detection of singlet oxygen. Org. Biomol. Chem. 2018, 16, 4044–4060. [Google Scholar] [CrossRef] [PubMed]
- Ramamurthy, V.; Inoue, Y. (Eds.) Supramolecular Photochemistry: Controlling Photochemical Processes; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2011; p. 623. [Google Scholar]
- Balzani, V.; Credi, A. Supramolecular photochemistry: Recent advances. Nato. Asi. Ser. 1996, 485, 163–177. [Google Scholar]
- Dodziuk, H. (Ed.) Cyclodextrins and Their Complexes; Wiley-VCH GmbH & Co. KGaA: Weinheim, Germany, 2008; p. 489. [Google Scholar]
- Szejtli, J.; Osa, T. (Eds.) Comprehensive Supramolecular Chemistry. In Cyclodextrins; Pergamon: Oxford, UK, 1996; Volume 3, p. 693. [Google Scholar]
- Kim, K.; Murray, J.; Selvapalam, N.; Ko, Y.H.; Hwang, I. Cucurbiturils: Chemistry, Supramolecular Chemistry and Applications; World Scientific Publishing Europe Ltd.: Singapore, 2018; p. 264. [Google Scholar]
- Nau, W.M.; Scherman, O.A. (Eds.) Cucurbiturils; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2011; p. 186. [Google Scholar]
- Sliwa, W.; Kozlowski, C. (Eds.) Calixarenes and Resorcinarenes Synthesis, Properties and Applications; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2009; p. 316. [Google Scholar]
- Ramamurthy, V. Photochemistry within a Water-Soluble Organic Capsule. Acc. Chem. Res. 2015, 48, 2904–2917. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.Q.; Lei, Q.; Zhu, J.Y.; Wang, W.J.; Cheng, Q.; Gao, F.; Sun, Y.X.; Zhang, X.Z. Cucurbit[8]uril Regulated Activatable Supramolecular Photosensitizer for Targeted Cancer Imaging and Photodynamic Therapy. ACS Appl. Mater. Interfaces 2016, 8, 22892–22899. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Huang, Z.; Liu, K.; Kelgtermans, H.; Dehaen, W.; Wang, Z.; Zhang, X. Porphyrin-containing hyperbranched supramolecular polymers: Enhancing 1O2-generation efficiency by supramolecular polymerization. Polym. Chem. 2014, 5, 53–56. [Google Scholar] [CrossRef]
- Leng, X.; Choi, C.-F.; Luo, H.-B.; Cheng, Y.-K.; Ng, D.K.P. Host-Guest Interactions of 4-Carboxyphenoxy Phthalocyanines and β-Cyclodextrins in Aqueous Media. Org. Lett. 2007, 9, 2497–2500. [Google Scholar] [CrossRef]
- Liu, K.; Liu, Y.; Yao, Y.; Yuan, H.; Wang, S.; Wang, Z.; Zhang, X. Supramolecular photosensitizers with enhanced antibacterial efficiency. Angew. Chem. Int. Ed. Engl. 2013, 52, 8285–8289. [Google Scholar] [CrossRef] [PubMed]
- Ozkan, M.; Keser, Y.; Hadi, S.E.; Tuncel, D. A [5]rotaxane-based photosensitizer for photodynamic therapy. Eur. J. Org. Chem. 2019, 2019, 3534–3541. [Google Scholar] [CrossRef]
- Gonzalez-Bejar, M.; Montes-Navajas, P.; Garcia, H.; Scaiano, J.C. Methylene Blue Encapsulation in Cucurbit[7]uril: Laser Flash Photolysis and Near-IR Luminescence Studies of the Interaction with Oxygen. Langmuir 2009, 25, 10490–10494. [Google Scholar] [CrossRef] [PubMed]
- Voskuhl, J.; Kauscher, U.; Gruener, M.; Frisch, H.; Wibbeling, B.; Strassert, C.A.; Ravoo, B.J. A soft supramolecular carrier with enhanced singlet oxygen photosensitizing properties. Soft Matter. 2013, 9, 2453–2457. [Google Scholar] [CrossRef]
- Xiong, H.; Zhou, D.; Zheng, X.; Qi, Y.; Wang, Y.; Jing, X.; Huang, Y. Stable amphiphilic supramolecular self-assembly based on cyclodextrin and carborane for the efficient photodynamic therapy. Chem. Commun. 2017, 53, 3422–3425. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Chen, L.-J.; Dong, F.; Jiang, S.-T.; Yin, G.-Q.; Li, X.; Tian, Y.; Yang, H.-B. Light-Controlled Generation of Singlet Oxygen within a Discrete Dual-Stage Metallacycle for Cancer Therapy. J. Am. Chem. Soc. 2019, 141, 8943–8950. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Xu, X.; Chen, Y.; Wu, X.; Wu, H.; Liu, Y. A highly efficient supramolecular photoswitch for singlet oxygen generation in water. Chem. Commun. 2016, 52, 7966–7969. [Google Scholar] [CrossRef]
- Robinson-Duggon, J.; Perez-Mora, F.; Valverde-Vasquez, L.; Cortes-Arriagada, D.; De la Fuente, J.R.; Gunther, G.; Fuentealba, D. Supramolecular Reversible On-Off Switch for Singlet Oxygen Using Cucurbit[n]uril Inclusion Complexes. J. Phys. Chem. C 2017, 121, 21782–21789. [Google Scholar] [CrossRef]
- Yan, H.; Pan, X.; Chua, M.H.; Wang, X.; Song, J.; Ye, Q.; Zhou, H.; Xuan, A.T.Y.; Liu, Y.; Xu, J. Self-assembled supramolecular nanoparticles mediated by host-guest interactions for photodynamic therapy. RSC Adv. 2014, 4, 10708–10717. [Google Scholar] [CrossRef]
- Yuan, B.; Wu, H.; Wang, H.; Tang, B.; Xu, J.F.; Zhang, X. A Self-Degradable Supramolecular Photosensitizer with High Photodynamic Therapeutic Efficiency and Improved Safety. Angew Chem. Int. Ed. 2021, 60, 706–710. [Google Scholar] [CrossRef] [PubMed]
- Naim, K.; Nair, S.T.; Yadav, P.; Shanavas, A.; Neelakandan, P.P. Supramolecular Confinement within Chitosan Nanocomposites Enhances Singlet Oxygen Generation. ChemPlusChem 2018, 83, 418–422. [Google Scholar] [CrossRef]
- Swaminathan, S.; Fowley, C.; Thapaliya, E.R.; McCaughan, B.; Tang, S.; Fraix, A.; Captain, B.; Sortino, S.; Callan, J.F.; Raymo, F.M. Supramolecular nanoreactors for intracellular singlet-oxygen sensitization. Nanoscale 2015, 7, 14071–14079. [Google Scholar] [CrossRef] [PubMed]
- Ozturk, E.; Eserci, H.; Okutan, E. Perylenebisimide-fullerene dyads as heavy atom free triplet photosensitizers with unique singlet oxygen generation efficiencies. J. Photochem. Photobiol. A 2019, 385, 112022. [Google Scholar] [CrossRef]
- Huang, L.; Yu, X.; Wu, W.; Zhao, J. Styryl Bodipy-C60 Dyads as Efficient Heavy-Atom-Free Organic Triplet Photosensitizers. Org. Lett. 2012, 14, 2594–2597. [Google Scholar] [CrossRef]
- Blacha-Grzechnik, A.; Krzywiecki, M.; Motyka, R.; Czichy, M. Electrochemically polymerized terthiopehene-C60 dyads for the photochemical generation of singlet oxygen. J. Phys. Chem. C 2019, 123, 25915–25924. [Google Scholar] [CrossRef]
- Agazzi, M.L.; Durantini, J.E.; Gsponer, N.S.; Durantini, A.M.; Bertolotti, S.G.; Durantini, E.N. Light-harvesting antenna and proton-activated photodynamic effect of a novel BODIPY-fullerene C60 dyad as potential antimicrobial agent. ChemPhysChem 2019, 20, 1110–1125. [Google Scholar] [CrossRef]
- Meyer, G.J. Antenna molecule drives solar hydrogen generation. Proc. Natl. Acad. Sci. USA 2015, 112, 9146–9147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Milanesio, M.E.; Alvarez, M.G.; Rivarola, V.; Silber, J.J.; Durantini, E.N. Porphyrin-fullerene C60 dyads with high ability to form photoinduced charge-separated state as novel sensitizers for photodynamic therapy. Photochem. Photobiol. 2005, 81, 891–897. [Google Scholar] [CrossRef]
- Kamkaew, A.; Lim, S.H.; Lee, H.B.; Kiew, L.V.; Chung, L.Y.; Burgess, K. BODIPY dyes in photodynamic therapy. Chem. Soc. Rev. 2013, 42, 77–88. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Cui, X.; Therrien, B.; Zhao, J. Energy-Funneling-Based Broadband Visible-Light-Absorbing Bodipy-C60 Triads and Tetrads as Dual Functional Heavy-Atom-Free Organic Triplet Photosensitizers for Photocatalytic Organic Reactions. Chem. Eur. J. 2013, 19, 17472–17482. [Google Scholar] [CrossRef] [PubMed]
- Ooyama, Y.; Enoki, T.; Ohshita, J.; Kamimura, T.; Ozako, S.; Koide, T.; Tani, F. Singlet oxygen generation properties of an inclusion complex of cyclic free-base porphyrin dimer and fullerene C60. RSC Adv. 2017, 7, 18690–18695. [Google Scholar] [CrossRef] [Green Version]
- Natarajan, A.; Kaanumalle, L.S.; Jockusch, S.; Gibb, C.L.; Gibb, B.C.; Turro, N.J.; Ramamurthy, V. Controlling photoreactions with restricted spaces and weak intermolecular forces: Exquisite selectivity during oxidation of olefins by singlet oxygen. J. Am. Chem. Soc. 2007, 129, 4132–4133. [Google Scholar] [CrossRef] [PubMed]























Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kashyap, A.; Ramasamy, E.; Ramalingam, V.; Pattabiraman, M. Supramolecular Control of Singlet Oxygen Generation. Molecules 2021, 26, 2673. https://doi.org/10.3390/molecules26092673
Kashyap A, Ramasamy E, Ramalingam V, Pattabiraman M. Supramolecular Control of Singlet Oxygen Generation. Molecules. 2021; 26(9):2673. https://doi.org/10.3390/molecules26092673
Chicago/Turabian StyleKashyap, Akshay, Elamparuthi Ramasamy, Vijayakumar Ramalingam, and Mahesh Pattabiraman. 2021. "Supramolecular Control of Singlet Oxygen Generation" Molecules 26, no. 9: 2673. https://doi.org/10.3390/molecules26092673




