Phytochemical Screening by LC-ESI-MS/MS and Effect of the Ethyl Acetate Fraction from Leaves and Stems of Jatropha macrantha Müll Arg. on Ketamine-Induced Erectile Dysfunction in Rats
Abstract
:1. Introduction
2. Results
2.1. Phytochemical Analysis of the Ethyl Acetate Fraction of Leaves and Stems of J. macrantha
2.2. Total Phenolic Content, Total Flavonoids and Antioxidant Activity of J. macrantha
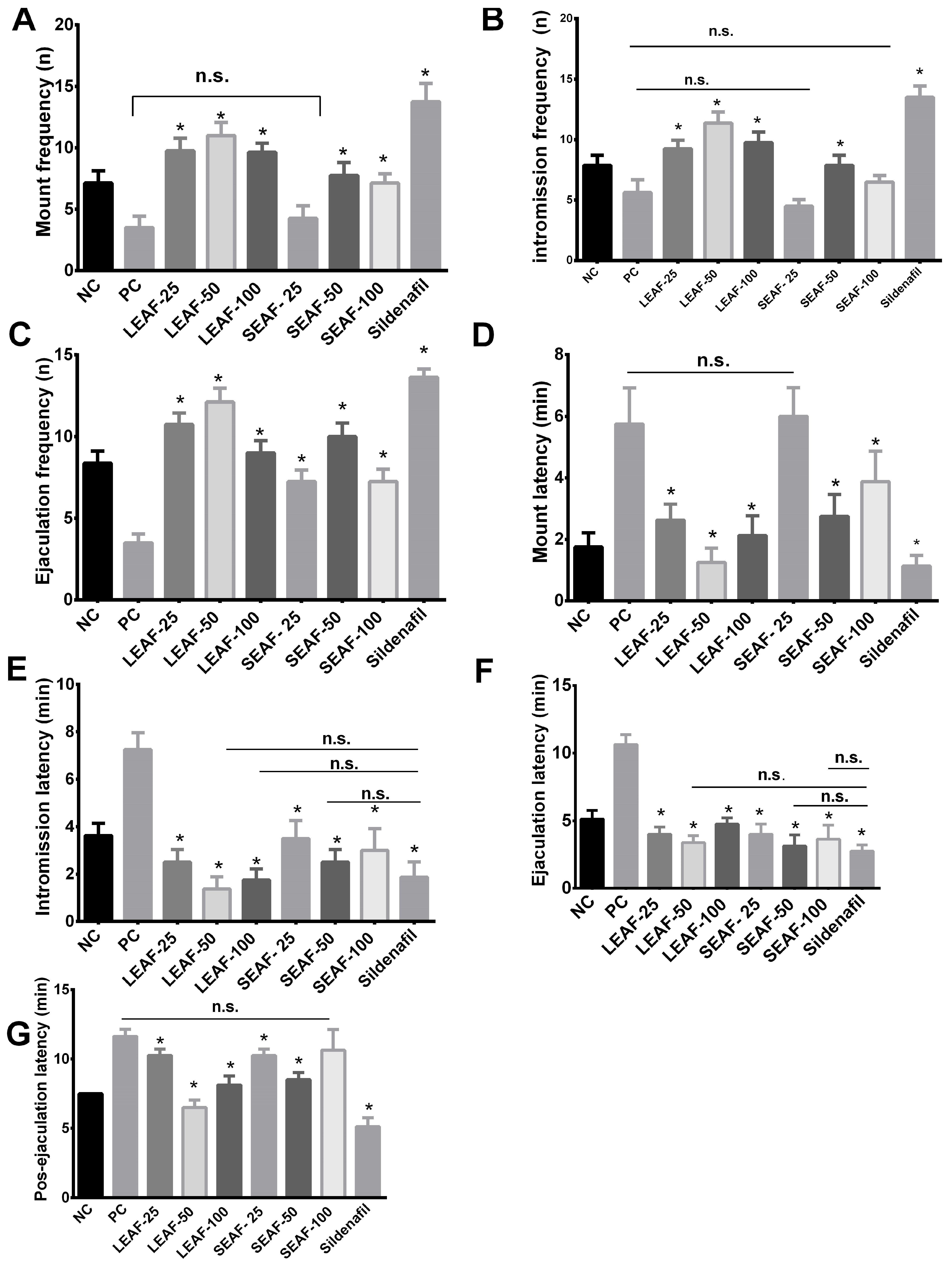
2.3. Effect of the Ethyl Acetate Fraction from Leaves and Stems of J. macrantha on Ketamine-Induced Erectile Dysfunction in Rats
2.4. Evaluation of the Vasodilator Effect in Rats Treated with J. macrantha and Sildenafil
3. Discussion
4. Materials and Methods
4.1. Collection of the Botanical Species
4.2. Preparation of the Ethanolic Extract and Ethyl Acetate Fraction
4.3. Phytochemical Analysis by LC-ESI-MS/MS of the Main Constituents of the Ethyl Acetate Fraction of Leaves and Stems of J. macrantha
4.3.1. Preparation of the Sample
4.3.2. Chromatographic Conditions
4.3.3. Mass Spectrometry Conditions
4.4. Determination of Total Phenolic Compounds (TPC)
4.5. Determination of Total Flavonoids
4.6. Determination of the Antioxidant Capacity by the Free Radical Sequestration Method with 2,2-Diphenyl-1-Picrylhydrazyl
4.7. Determination of the Antioxidant Capacity by the Sequestration Method with the Radical Cation of the 2.2’-Azinobis-(3-Ethylbenzothiazoline)-6-Sulfonic Acid
4.8. Determination of the Antioxidant Capacity by the Ferric Reducing Antioxidant Power (FRAP) Method
4.9. Effect of the Ethyl Acetate Fraction from Leaves and Stems of J. Macrantha on Ketamine-Induced Erectile Dysfunction in Rats
Evaluation of Erectly Dysfunction
- (a)
- Group I: distilled water at doses of 10 mL/kg was administered orally, which served as negative control (NC).
- (b)
- Group II: Ketamine at doses of 50 mg/kg was administered intraperitoneally, which served as a positive control (PC).
- (c)
- Group III: Ketamine at 50 mg/kg (IP) plus leaves ethyl acetate fraction at doses of 25 mg/kg (LEAF-25) by oral administration.
- (d)
- Group IV: Ketamine at 50 mg/kg (IP) plus leaves ethyl acetate fraction at doses of 50 mg/kg (LEAF-50) by oral administration.
- (e)
- Group V: Ketamine at 50 mg/kg (IP) plus leaves ethyl acetate fraction at doses of 100 mg/kg (LEAF-100) by oral administration.
- (f)
- Group VI: Ketamine at 50 mg/kg (IP) plus stems ethyl acetate fraction at doses of 25 mg/kg (SEAF-25) by oral administration.
- (g)
- Group VII: Ketamine at 50 mg/kg (IP) plus stems ethyl acetate fraction at doses of 50 mg/kg (SEAF-50) by oral administration.
- (h)
- Group VIII: Ketamine at 50 mg/kg (IP) plus stems ethyl acetate fraction at doses of 100 mg/kg (SEAF-100) by oral administration.
- (i)
- Group XI: Ketamine 50 mg/kg (IP) plus sildenafil citrate at 5 mg/kg by oral administration.
- (a)
- Mount frequency (MF): It is the number of mounts without penetration at the moment that female rat is introduced to the male’s cage until ejaculation.
- (b)
- Intromission frequency (IF): It is the number of penetrations at the moment that female rat is introduced to the male’s cage until ejaculation.
- (c)
- Ejaculation frequency (EF): It is the time interval between the introduction of the female rat and the first mount of the male rat.
- (d)
- Mount latency (ML): It is the time interval that elapses when the female rat is introduced to the cage, until the male performs the first mount of the copulatory series.
- (e)
- Intromission latency (IL): It is the time interval at the moment of introduction of the female rat to the first penetration of male rat. It is usually characterized by pelvic thrust and jump to dismount.
- (f)
- Ejaculatory latency (EL): It is the time interval between the first penetration and ejaculation. It is usually characterized by a prolonged, deep pelvic thrust and slow dismounting followed by a period of inactivity or reduced activity.
- (g)
- Post ejaculatory latency (PEL): It is the time interval between ejaculation and the first penetration of the following series.
4.10. Evaluation of the Vasodilator Effect
4.11. Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Yafi, F.A.; Jenkins, L.; Albersen, M.; Corona, G.; Isidori, A.M.; Goldfarb, S.; Maggi, M.; Nelson, C.J.; Parish, S.; Salonia, A.; et al. Erectile Dysfunction. Nat. Rev. Dis. Primers 2016, 2, 16003. [Google Scholar] [CrossRef]
- Corona, G.; Lee, D.M.; Forti, G.; O’Connor, D.B.; Maggi, M.; O’Neill, T.W.; Pendleton, N.; Bartfai, G.; Boonen, S.; Casanueva, F.F.; et al. Age-Related Changes in General and Sexual Health in Middle-Aged and Older Men: Results from the European Male Ageing Study (EMAS). J. Sex. Med. 2010, 7, 1362–1380. [Google Scholar] [CrossRef] [PubMed]
- Saeed, R.; Amin, F.; Durrani, N.; Ali Saif, S.; Zafar, M. Prevalence of Erectile Dysfunction and Associated Factors among Males Visiting Family Medicine Clinics in a Tertiary Care Hospital in Karachi, Pakistan. J. Fam. Med. Prim. Care 2021, 10, 1294. [Google Scholar] [CrossRef] [PubMed]
- Mulhall, J.P.; Luo, X.; Zou, K.H.; Stecher, V.; Galaznik, A. Relationship between Age and Erectile Dysfunction Diagnosis or Treatment Using Real-World Observational Data in the United States. Int. J. Clin. Pract. 2016, 70, 1012. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Colson, M.H.; Cuzin, B.; Faix, A.; Grellet, L.; Huyghes, E. Current Epidemiology of Erectile Dysfunction, an Update. Sexologies 2018, 27, e7–e13. [Google Scholar] [CrossRef]
- Sangiorgi, G.; Cereda, A.; Benedetto, D.; Bonanni, M.; Chiricolo, G.; Cota, L.; Martuscelli, E.; Greco, F. Anatomy, Pathophysiology, Molecular Mechanisms, and Clinical Management of Erectile Dysfunction in Patients Affected by Coronary Artery Disease: A Review. Biomedicines 2021, 9, 432. [Google Scholar] [CrossRef]
- Burnett, A.L.; Nehra, A.; Breau, R.H.; Culkin, D.J.; Faraday, M.M.; Hakim, L.S.; Heidelbaugh, J.; Khera, M.; McVary, K.T.; Miner, M.M.; et al. Erectile Dysfunction: AUA Guideline. J. Urol. 2018, 200, 633–641. [Google Scholar] [CrossRef]
- Wu, Y.-N.; Wu, C.-C.; Sheu, M.-T.; Chen, K.-C.; Ho, H.-O.; Chiang, H.-S. Optimization of Platelet-Rich Plasma and Its Effects on the Recovery of Erectile Function after Bilateral Cavernous Nerve Injury in a Rat Model. J. Tissue Eng. Regen. Med. 2016, 10, E294–E304. [Google Scholar] [CrossRef]
- Yiou, R.; Hamidou, L.; Birebent, B.; Bitari, D.; Lecorvoisier, P.; Contremoulins, I.; Khodari, M.; Rodriguez, A.M.; Augustin, D.; Roudot-Thoraval, F.; et al. Safety of Intracavernous Bone Marrow-Mononuclear Cells for Postradical Prostatectomy Erectile Dysfunction: An Open Dose-Escalation Pilot Study. Eur. Urol. 2016, 69, 988–991. [Google Scholar] [CrossRef]
- Garza-Gangemi, A.M.; Sotomayor-de Zavaleta, M. Erectile Dysfunction Therapy in Countries Where Implant Is Economically Not Feasible. Transl. Androl. Urol. 2017, 6, 176. [Google Scholar] [CrossRef] [Green Version]
- Shaaban, M.; Ali, M.; Tala, M.F.; Hamed, A.; Hassan, A.Z. Ecological and Phytochemical Studies on Euphorbia Retusa (Forssk.) from Egyptian Habitat. J. Anal. Methods Chem. 2018, 2018, 9143683. [Google Scholar] [CrossRef] [Green Version]
- Cavalcante, N.B.; Diego da Conceição Santos, A.; Guedes da Silva Almeida, J.R. The Genus Jatropha (Euphorbiaceae): A Review on Secondary Chemical Metabolites and Biological Aspects. Chem.-Biol. Interact. 2020, 318, 108976. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.K.; Singh, H. A Review on Pharmacological Significance of Genus Jatropha (Euphorbiaceae). Chin. J. Integr. Med. 2012, 18, 868–880. [Google Scholar] [CrossRef]
- Crepaldi, C.G.; Campos, J.L.A.; Albuquerque, U.P.; Sales, M.F. Richness and Ethnobotany of the Family Euphorbiaceae in a Tropical Semiarid Landscape of Northeastern Brazil. S. Afr. J. Bot. 2016, 102, 157–165. [Google Scholar] [CrossRef]
- De Almeida, C.; de, F.C.B.R.; Ramos, M.A.; de Amorim, E.L.C.; de Albuquerque, U.P. A Comparison of Knowledge about Medicinal Plants for Three Rural Communities in the Semi-Arid Region of Northeast of Brazil. J. Ethnopharmacol. 2010, 127, 674–684. [Google Scholar] [CrossRef]
- Tinco, A.; Arroyo, J.; Bonilla, P. Efecto Del Extracto Metanólico de Jatropha Macrantha Müll. Arg., En La Disfunción Eréctil Inducida En Ratas. An. Fac. Med. 2011, 72, 161–168. [Google Scholar] [CrossRef] [Green Version]
- Chávez, H.; Palomino, F.; Angelino, J.; Torres, E.; Bendezú, M.R.; García, J.A.; Loja, B.; Muñoz, A.M.; Alvarado, A.T. In Vivobronchodilator Evaluation of the Ethanolic Extract of the Stems of Jatropha MacranthaMüll.Arg. Evaluación Broncodilatadora in Vivo Del Extracto Etanólico de Los Tallos de Jatropha Macrantha Müll.Arg. J. Pharm. Pharmacogn. Res. 2021, 9, 937–946. [Google Scholar]
- Apaza Ticona, L.; Serban, A.M.; Puerto Madorrán, M.J.; Fernández-Grifol, M.; Rumbero Sánchez, Á. Anti-Melanogenic and Anti-Inflammatory Activities of Triterpenoids from Jatropha Macrantha. Rev. Bras. Farmacogn. 2021, 31, 40–50. [Google Scholar] [CrossRef]
- Apaza Ticona, L.; Antognoni, F.; Potente, G.; Rumbero Sánchez, Á. Triterpenoids Isolated from Jatropha Macrantha (Müll. Arg.) Inhibit the NF-ΚB and HIF-1α Pathways in Tumour Cells. Nat. Prod. Res. 2020, 35, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Oshima, M.; Gu, Y.; Tsukada, S. Effects of Lepidium Meyenii Walp and Jatropha Macrantha on Blood Levels of Estradiol-17β, Progesterone, Testosterone and the Rate of Embryo Implantation in Mice. J. Vet. Med. Sci. 2003, 65, 1145–1146. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Hwang, S.H.; Guillen Quispe, Y.N.; Gonzales Arce, P.H.; Lim, S.S. Investigation of the Antioxidant and Aldose Reductase Inhibitory Activities of Extracts from Peruvian Tea Plant Infusions. Food Chem. 2017, 231, 222–230. [Google Scholar] [CrossRef]
- Leisegang, K.; Finelli, R. Alternative Medicine and Herbal Remedies in the Treatment of Erectile Dysfunction: A Systematic Review. Arab J. Urol. 2021, 19, 323–339. [Google Scholar] [CrossRef]
- El Shehaby, D.M.; El-Mahdy, R.I.; Ahmed, A.M.; Hosny, A.; Abd el-Rady, N.M. Neurobehavioral, Testicular and Erectile Impairments of Chronic Ketamine Administration: Pathogenesis and Ameliorating Effect of N-Acetyl Cysteine. Reprod. Toxicol. 2020, 96, 57–66. [Google Scholar] [CrossRef]
- Shang, H.S.; Wu, Y.N.; Liao, C.H.; Chiueh, T.S.; Lin, Y.F.; Chiang, H.S. Long-Term Administration of Ketamine Induces Erectile Dysfunction by Decreasing Neuronal Nitric Oxide Synthase on Cavernous Nerve and Increasing Corporal Smooth Muscle Cell Apoptosis in Rats. Oncotarget 2017, 8, 73670. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, B.R.; Kumar, S.K.; Zhao, C.; Zhang, L.T.; Kim, C.Y.; Lee, S.W.; Jeon, J.H.; So, I.; Kim, S.H.; Park, N.C.; et al. Additive Effects of Artemisia Capillaris Extract and Scopoletin on the Relaxation of Penile Corpus Cavernosum Smooth Muscle. Int. J. Impot. Res. 2015, 27, 225–232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Z.M.; Liu, N.; Jiang, Y.P.; Yang, J.M.; Zheng, J.; Sun, M.; Li, Y.X.; Sun, T.; Wu, J.; Yu, J.Q. Vitexin Alleviates Streptozotocin-Induced Sexual Dysfunction and Fertility Impairments in Male Mice via Modulating the Hypothalamus–Pituitary–Gonadal Axis. Chem.-Biol. Interact. 2019, 297, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Sabphon, C.; Temkitthawon, P.; Ingkaninan, K.; Sawasdee, P. Phosphodiesterase Inhibitory Activity of the Flavonoids and Xanthones from Anaxagorea Luzonensis. Nat. Prod. Commun. 2015, 10, 301–303. [Google Scholar] [CrossRef] [Green Version]
- Oboh, G.; Ademiluyi, A.O.; Ademosun, A.O.; Olasehinde, T.A.; Oyeleye, S.I.; Boligon, A.A.; Athayde, M.L. Phenolic Extract from Moringa Oleifera Leaves Inhibits Key Enzymes Linked to Erectile Dysfunction and Oxidative Stress in Rats’ Penile Tissues. Biochem. Res. Int. 2015, 2015, 175950. [Google Scholar] [CrossRef] [Green Version]
- Sheweita, S.; Salama, B.; Hassan, M. Erectile Dysfunction Drugs and Oxidative Stress in the Liver of Male Rats. Toxicol. Rep. 2015, 2, 933–938. [Google Scholar] [CrossRef] [Green Version]
- Adefegha, S.A.; Oyeleye, S.I.; Dada, F.A.; Olasehinde, T.A.; Oboh, G. Modulatory Effect of Quercetin and Its Glycosylated Form on Key Enzymes and Antioxidant Status in Rats Penile Tissue of Paroxetine-Induced Erectile Dysfunction. Biomed. Pharmacother. 2018, 107, 1473–1479. [Google Scholar] [CrossRef]
- Cripps, S.M.; Mattiske, D.M.; Pask, A.J. Erectile Dysfunction in Men on the Rise: Is There a Link with Endocrine Disrupting Chemicals? Sex. Dev. 2021, 15, 187–212. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Xu, Y.; Wang, J.; Yang, X.; Cao, H.; Huang, P. Relaxation Effect of Patchouli Alcohol in Rat Corpus Cavernous and Its Underlying Mechanisms. Evid.-Based Complementary Altern. Med. 2020, 2020, 3109069. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Felices, E.J.; Romero-Viacava, M.; Enciso-Roca, E.; Herrera-Calderon, O.; Común-Ventura, P.; Yuli-Posadas, R.Á.; Chacaltana-Ramos, L.; Pari-Olarte, B. Antioxidant Activity of the Germinated Seed of Four Varieties of Amaranthus Caudatus L. From Peru. Pharmacogn. J. 2019, 11, 588–593. [Google Scholar] [CrossRef] [Green Version]

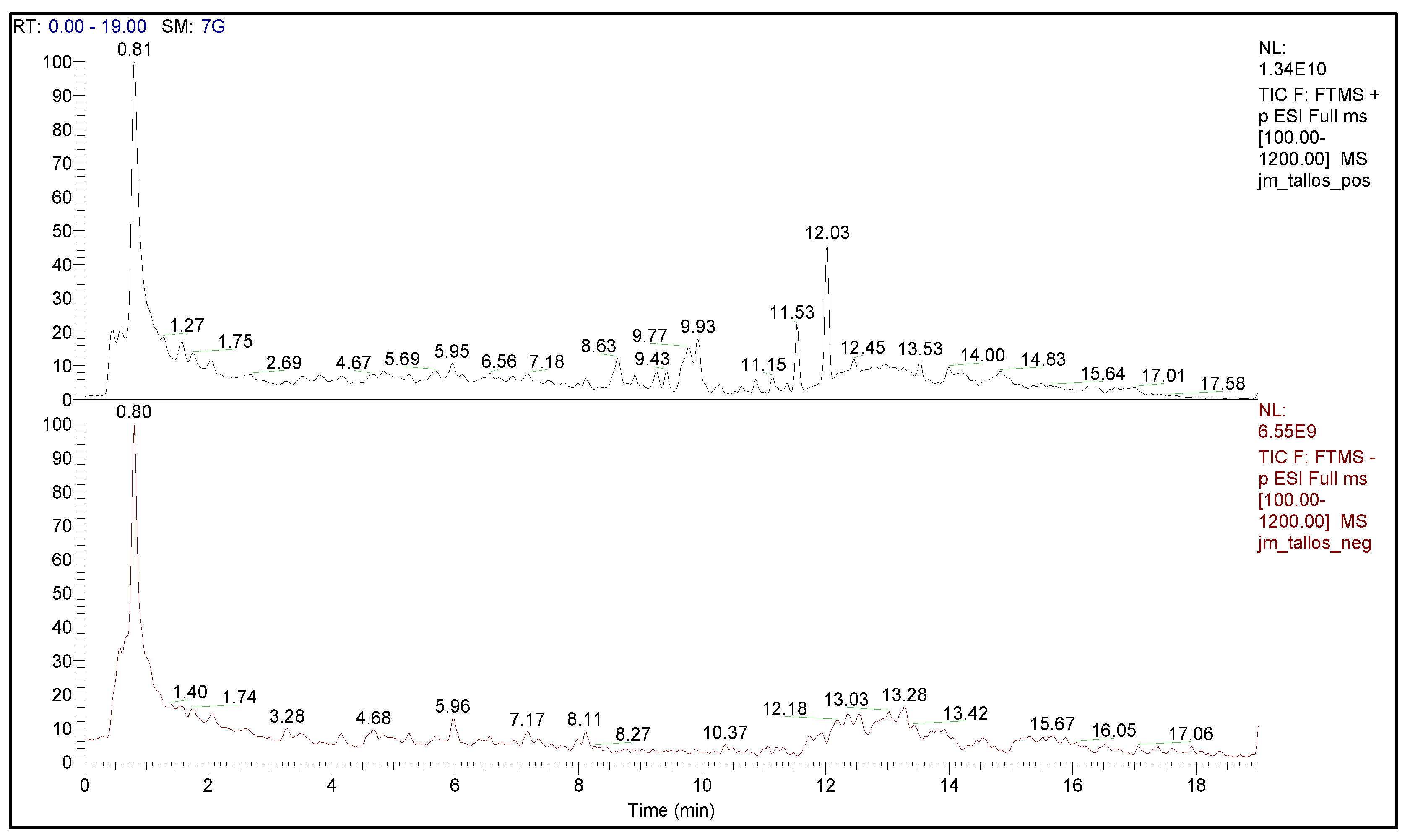


| Ethyl Acetate Fraction | ESI (−) | ESI (+) | ESI (+/−) | Total |
|---|---|---|---|---|
| Leaves | 25 | 42 | 10 | 77 |
| Stems | 18 | 21 | 3 | 42 |
| N° | Retention Time (Min) | Theoretical Mass (Neutral Form) | Molecular Formula (Neutral Form) | Predicted Metabolite | Chemical Group |
|---|---|---|---|---|---|
| 1 | 0.41 | 104.10754 | C5H14NO | Choline | Cholines |
| 2 | 0.41 | 117.07898 | C5H11NO2 | Betaine | Alpha amino acids |
| 3 | 0.45 | 122.04801 | C6H6N2O | Niacinamide | Vitamin B₃ |
| 4 | 0.46 | 118.02661 | C4H6O4 | Succinic acid | Dicarboxylic acids and derivatives |
| 5 | 0.54 | 99.06841 | C5H9NO | 2-Piperidone | Piperidinones |
| 6 | 0.55 | 110.03678 | C6H6O2 | Catechol | Catechols |
| 7 | 0.66 | 184.03717 | C8H8O5 | Methylgallate | Galloyl esters |
| 8 | 0.69 | 138.03169 | C7H6O3 | Salicylic acid | Salicylic acids |
| 9 | 0.70 | 178.02661 | C9H6O4 | Esculetin Syn. 6,7-Dihydroxycoumarin | Dihydroxycoumarins |
| 0.71 | 178.02661 | C9H6O4 | |||
| 10 | 0.73 | 152.04734 | C8H8O3 | 2,4-Dihydroxyacetophenone | Alkyl-phenylketones |
| 11 | 0.74 | 121.05276 | C7H7NO | Benzamide | Benzamides |
| 12 | 0.78 | 564.14791 | C26H28O14 | NP-000004 Syn. Apigenin 6-C-glucoside 8-C-arabinoside | Flavonoid C-glycosides |
| 0.79 | 564.14791 | C26H28O14 | |||
| 13 | 0.79 | 208.03717 | C10H8O5 | Fraxetin | Dihydroxycoumarins |
| 14 | 0.84 | 448.10056 | C21H20O11 | Homoorientin Syn. Luteolin-6-C-glucoside | Flavonoid C-glycosides |
| 15 | 0.84 | 448.10056 | C21H20O11 | Luteolin-8-C-glucoside Syn. Orientin | Flavonoid C-glycosides |
| 0.85 | 448.10056 | C21H20O11 | |||
| 16 | 0.89 | 122.03678 | C7H6O2 | 3-Hydroxybenzaldehyde | Phenolic acids |
| 17 | 0.90 | 147.06841 | C9H9NO | Indole-3-carbinol | 3-alkylindoles |
| 18 | 0.90 | 174.07931 | C10H10N2O | Indole-3-acetamide | 3-alkylindoles |
| 19 | 0.93 | 282.14672 | C15H22O5 | 5,9-dihydroxy-7-(hydroxymethyl)-5,7-dimethyl-4,5a,6,8,8a,9-hexahydro-1H-azuleno[5,6-c]furan-3-onenSyn. Lactarorufin B | Lactarane sesquiterpenes |
| 20 | 1.04 | 192.04226 | C10H8O4 | Scopoletin | Hydroxycoumarins |
| 1.05 | 192.04226 | C10H8O4 | |||
| 21 | 1.07 | 222.05282 | C11H10O5 | Isofraxidin | Hydroxycoumarins |
| 22 | 1.08 | 222.05282 | C11H10O5 | 8-hydroxy-6,7-dimethoxy-2H-chromen-2-oneSyn. Fraxidin | Hydroxycoumarins |
| 23 | 1.10 | 432.10565 | C21H20O10 | Vitexin n (Isomer I) | Flavonoid C-glycosides |
| 1.11 | 432.10565 | C21H20O10 | |||
| 24 | 1.17 | 196.10994 | C11H16O3 | Loliolide | Benzofurans |
| 25 | 1.32 | 432.10565 | C21H20O10 | Vitexin Syn. Flavone, 8-D-glucosyl-4’,5,7-trihydroxy- | Flavonoid C-glycosides |
| 26 | 1.31 | 432.10565 | C21H20O10 | Isovitexin Syn. Homovitexin | Flavonoid C-glycosides |
| 1.32 | 432.10565 | C21H20O10 | |||
| 27 | 1.40 | 448.10056 | C21H20O11 | Kaempferol-7-O-glucoside | Flavonoid O-glycosides |
| 28 | 1.41 | 448.10056 | C21H20O11 | NCGC00385820-01!5,7-dihydroxy-2-[3-hydroxy-4-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxyphenyl]chromen-4-one Syn. Luteolin 4’-O-glucoside (Isomer I) | Flavonoid O-glycosides |
| 29 | 1.48 | 145.05276 | C9H7NO | 2-hydroxyquinoline | Hydroquinolones |
| 30 | 1.49 | 145.05276 | C9H7NO | Indole-3-carboxyaldehyde | Indoles |
| 31 | 1.49 | 196.10994 | C11H16O3 | Loliolide | Benzofurans |
| 32 | 1.59 | 206.05791 | C11H10O4 | (4S,5Z,6S)-4-(2-methoxy-2-oxoethyl)-5-[2-[(E)-3-phenylprop-2-enoyl]oxyethylidene]-6-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-4H-pyran-3-carboxylic acid Syn. Jasminoside | Coumarins and derivatives |
| 33 | 1.60 | 288.06339 | C15H12O6 | (2S,3S)-3,5,7-trihydroxy-2-(4-hydroxyphenyl)-2,3-dihydrochromen-4-one Syn. (-)-dihydrokaempferol | Flavonoids |
| 34 | 1.71 | 175.06333 | C10H9NO2 | 3-Indoleacetic acid | Indole-3-acetic acid derivatives |
| 35 | 1.91 | 164.04734 | C9H8O3 | Coumaric acid (Isomer I) | Coumaric acid and derivatives |
| 36 | 2.00 | 416.11073 | C21H20O9 | Puerarin Syn. Daidzein-8-C-glucoside | Isoflavonoid C-glycosides |
| 2.01 | 416.11073 | C21H20O9 | |||
| 37 | 2.26 | 432.10565 | C21H20O10 | Apigetrin Syn. Apigenin 7-O-glucoside | Flavonoid O-glycosides |
| 38 | 2.27 | 432.10565 | C21H20O10 | Aloenin | Anthraquinones |
| 39 | 2.31 | 164.04734 | C9H8O3 | Coumaric acid (Isomer II) | Coumaric acid and derivatives |
| 40 | 2.31 | 178.06299 | C10H10O3 | Coniferylaldehyde | Methoxyphenols |
| 41 | 2.54 | 448.10056 | C21H20O11 | Luteolin-7-glucoside (Isomer I) | Flavonoid O-glycosides |
| 42 | 2.54 | 448.10056 | C21H20O11 | NCGC00385820-01!5,7-dihydroxy-2-[3-hydroxy-4-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxyphenyl]chromen-4-oneSyn. Luteolin 4’-O-glucoside (Isomer II) | Flavonoid O-glycosides |
| 43 | 3.11 | 332.18350 | C16H28O7 | 2-(hydroxymethyl)-6-(6-hydroxy-6-methyl-3-propan-2-ylcyclohex-3-en-1-yl)oxyoxane-3,4,5-triol Syn. MCULE-9958171223 | Fatty acyl glycosides of mono- and disaccharides |
| 44 | 3.21 | 188.10486 | C9H16O4 | Azelaic acid | Organic acids |
| 45 | 3.56 | 448.10056 | C21H20O11 | Luteolin-7-glucoside | Flavonoid O-glycosides |
| 3.57 | 448.10056 | C21H20O11 | |||
| 46 | 3.96 | 164.04734 | C9H8O3 | Coumaric acid (Isomer III) | Coumaric acid and derivatives |
| 47 | 4.02 | 264.13616 | C15H20O4 | Abscisic acid | Abscisic acids and derivatives |
| 48 | 4.40 | 432.10565 | C21H20O10 | Vitexin (Isomer II) | Flavonoid C-glycosides |
| 49 | 4.62 | 272.06847 | C15H12O5 | Naringenin | Flavanone |
| 50 | 4.68 | 286.04774 | C15H10O6 | Luteolin (Isomer I) | Flavones |
| 4.69 | 286.04774 | C15H10O6 | |||
| 51 | 5.06 | 262.12051 | C15H18O4 | Dihydro-8-deoxy-lactucin | Gamma butyrolactones |
| 52 | 5.21 | 164.04734 | C9H8O3 | Coumaric acid (Isomer VI) | Coumaric acid and derivatives |
| 53 | 5.23 | 210.12559 | C12H18O3 | Jasmonic Acid | Jasmonic acids |
| 54 | 5.32 | 286.04774 | C15H10O6 | Luteolin(Isomer II) | Flavones |
| 55 | 5.42 | 594.13734 | C30H26O13 | Kaempferol-3-O-glucoside-2’’-p-coumaroyl | Flavonoid O-glycosides |
| 56 | 5.60 | 164.04734 | C9H8O3 | Coumaric acid (Isomer V) | Coumaric acid and derivatives |
| 5.60 | 164.04734 | C9H8O3 | |||
| 57 | 5.70 | 270.05282 | C15H10O5 | Apigenin | Flavones |
| 58 | 5.70 | 270.05282 | C15H10O5 | Aloe-emodin | Anthraquinones |
| 59 | 5.83 | 300.06339 | C16H12O6 | Diosmetin | Flavones |
| 5.84 | 300.06339 | C16H12O6 | |||
| 60 | 6.03 | 252.17254 | C15H24O3 | NCGC00169905-02_C15H24O3_2-Pentenoic acid, 5-(3-hydroxy-2,3-dimethylbicyclo[2.2.1]hept-2-yl)-2-methyl-, (2E)- | Sesquiterpenoids |
| 61 | 6.10 | 334.17802 | C19H26O5 | Arnicolide C | Sesquiterpene lactones |
| 62 | 6.25 | 232.14633 | C15H20O2 | Alantolactone | Eudesmanolides, secoeudesmanolides, and derivatives |
| 63 | 6.63 | 282.14672 | C15H22O5 | Artemisinin | Terpene lactones |
| 64 | 7.12 | 292.20384 | C18H28O3 | NCGC00386020-01_C18H28O3_8-{(1S,5R)-4-Oxo-5-[(2Z)-2-penten-1-yl]-2-cyclopenten-1-yl}octanoic acidSyn. Chromomoric acid B (Isomer I) | Phytoprostane |
| 65 | 8.28 | 316.20384 | C20H28O3 | Cafestol | Naphthofurans |
| 66 | 8.52 | 250.15689 | C15H22O3 | NCGC00169029-02_C15H22O3_Naphtho[2,3-b]furan-2(4H)-one, 4a,5,6,7,8,8a,9,9a-octahydro-9a-hydroxy-3,4a,5-trimethyl-, (4aR,5S,8aS,9aR)- | Terpene lactones |
| 67 | 8.54 | 250.15689 | C15H22O3 | 2-[(2S,4aR,8aS)-2-hydroxy-4a-methyl-8-methylidene-3,4,5,6,7,8a-hexahydro-1H-naphthalen-2-yl]prop-2-enoic acid | Eudesmane, isoeudesmane or cycloeudesmane sesquiterpenoids |
| 68 | 8.76 | 470.33961 | C30H46O4 | NCGC00169801-02_C30H46O4_Lanosta-8,24-dien-26-oic acid, 21-hydroxy-3-oxo-, (5xi,13alpha,14beta,17alpha,20S,24E)- Syn. MEGxp0_001112 | Triterpenoids |
| 69 | 8.98 | 292.20384 | C18H28O3 | NCGC00386020-01_C18H28O3_8-{(1S,5R)-4-Oxo-5-[(2Z)-2-penten-1-yl]-2-cyclopenten-1-yl}octanoic acidSyn. Chromomoric acid B (Isomer II) | Phytoprostane |
| 70 | 9.16 | 292.20384 | C18H28O3 | NCGC00386020-01_C18H28O3_8-{(1S,5R)-4-Oxo-5-[(2Z)-2-penten-1-yl]-2-cyclopenten-1-yl} octanoic acid Syn. Chromomoric acid B (Isomer III) | Phytoprostane |
| 71 | 9.53 | 472.35526 | C30H48O4 | NCGC00385237-01_C30H48O4 | Cucurbitacins |
| 72 | 10.42 | 676.36701 | C33H56O14 | DGMG 18:3 | Lipids |
| 73 | 10.65 | 514.31418 | C27H46O9 | MGMG 18:3 | Lipids |
| 74 | 10.66 | 514.31418 | C27H46O9 | NCGC00380867-01_C27H46O9_9,12,15-Octadecatrienoic acid, 3-(hexopyranosyloxy)-2-hydroxypropyl ester, (9Z,12Z,15Z)- | Glycosylmonoacylglycerols |
| 10.66 | 514.31418 | C27H46O9 | |||
| 75 | 11.18 | 304.24023 | C20H32O2 | NCGC00384643-01_C20H32O2_(2E)-3-Methyl-5-[(1S,8aS)-5,5,8a-trimethyl-2-methylenedecahydro-1-naphthalenyl] -2-pentenoic acid | Diterpenoids |
| 76 | 11.69 | 356.29266 | C21H40O4 | Monoolein | 1-Monoglyceride |
| 77 | 11.92 | 592.26857 | C35H36N4O5 | Pheophorbide A | Chlorins |
| N° | Retention time (min) | Theoretical mass (neutral form) | Molecular Formula (neutral form) | Predicted metabolite | Chemical group |
|---|---|---|---|---|---|
| 1 | 0.41 | 117.07898 | C5H11NO2 | Betaine | Alpha amino acids |
| 2 | 0.42 | 135.05450 | C5H5N5 | Adenine | 6-Aminopurines |
| 3 | 0.42 | 169.07389 | C8H11NO3 | Pyridoxine | Vitamin B6 |
| 4 | 0.45 | 118.02661 | C4H6O4 | Succinic acid | Dicarboxylic acids and derivatives |
| 5 | 0.46 | 122.04801 | C6H6N2O | Niacinamide | Nicotinamides |
| 6 | 0.51 | 104.04734 | C4H8O3 | 2-Hydroxybutyric acid | Alpha hydroxy acids and derivatives |
| 7 | 0.55 | 99.06841 | C5H9NO | 2-Piperidone | Piperidinones |
| 8 | 0.56 | 110.03678 | C6H6O2 | Catechol | Catechols |
| 9 | 0.56 | 132.04226 | C5H8O4 | Glutaric acid | Dicarboxylic acids and derivatives |
| 10 | 0.67 | 138.03169 | C7H6O3 | Salicylic acid | Salicylic acids |
| 11 | 0.67 | 145.05276 | C9H7NO | 2-hydroxyquinoline | Hydroquinolones |
| 12 | 0.70 | 178.02661 | C9H6O4 | 6,7-Dihydroxycoumarin Syn. Esculetin | Dihydroxycoumarins |
| 0.71 | 178.02661 | C9H6O4 | |||
| 13 | 0.70 | 196.07356 | C10H12O4 | 1-(2-hydroxy-4,6-dimethoxyphenyl)ethanone Syn. Xanthoxylin | Alkyl-phenylketones |
| 14 | 0.74 | 152.04734 | C8H8O3 | 2,4-Dihydroxyacetophenone | Alkyl-phenylketones |
| 15 | 0.81 | 208.03717 | C10H8O5 | NCGC00017270-07!7,8-dihydroxy-6-methoxychromen-2-oneSyn. Fraxetin | Dihydroxycoumarins |
| 0.81 | 208.03717 | C10H8O5 | |||
| 16 | 0.91 | 122.03678 | C7H6O2 | 3-Hydroxybenzaldehyde | Phenolic acids |
| 17 | 0.93 | 282.14672 | C15H22O5 | 5,9-Dihydroxy-7-(hydroxymethyl)-5,7-dimethyl-4,5a,6,8,8a,9-hexahydro-1H-azuleno[5,6-c]furan-3-one Syn. Lactarorufin B | Lactarane sesquiterpenes |
| 18 | 1.05 | 192.04226 | C10H8O4 | Scopoletin | Hydroxycoumarins |
| 1.06 | 192.04226 | C10H8O4 | |||
| 19 | 1.08 | 178.06299 | C10H10O3 | 2-Methoxycinnamic acid | Cinnamic acids |
| 20 | 1.09 | 222.05282 | C11H10O5 | 8-Hydroxy-6,7-dimethoxy-2H-chromen-2-one Syn. Fraxidin | Hydroxycoumarins |
| 21 | 1.15 | 432.10565 | C21H20O10 | Vitexin | Flavonoid C-glycosides |
| 22 | 1.18 | 196.10994 | C11H16O3 | Loliolide | Benzofurans |
| 23 | 1.22 | 132.07864 | C6H12O3 | 2-Hydroxy-4-methylpentanoic acid | Hydroxy fatty acids |
| 24 | 1.51 | 145.05276 | C9H7NO | Indole-3-carboxyaldehyde | Indoles |
| 25 | 1.94 | 150.10447 | C10H14O | Cuminyl alcohol (Isomer I) | Benzyl alcohols |
| 26 | 2.59 | 262.14164 | C12H22O6 | 9-(2,3-dihydroxypropoxy)-9-oxononanoic acid | Organic acids |
| 27 | 2.65 | 372.21480 | C19H32O7 | NCGC00384741-01_C19H32O7_2-Cyclohexen-1-one, 3-[3-(beta-D-glucopyranosyloxy)butyl]-2,4,4-trimethyl-Syn. Megastigm-5-En-4-One 9-Glucoside | Fatty acyl glycosides of mono- and disaccharides |
| 28 | 2.91 | 316.20384 | C20H28O3 | Cafestol | Naphthofurans |
| 29 | 3.25 | 150.10447 | C10H14O | Cuminyl alcohol (Isomer II) | Benzyl alcohols |
| 30 | 3.28 | 188.10486 | C9H16O4 | Azelaic acid | Organic acids |
| 31 | 4.63 | 150.10447 | C10H14O | Thymol (Isomer I) | Aromatic monoterpenoids |
| 32 | 4.73 | 286.04774 | C15H10O6 | Kaempferol / 3’,4’,5,7-tetrahydroxyflavone | Flavonols |
| 33 | 5.09 | 262.12051 | C15H18O4 | Dihydro-8-deoxy-lactucin | Gamma butyrolactones |
| 34 | 5.88 | 300.06339 | C16H12O6 | Diosmetin / tectorigenin | Flavonoids |
| 35 | 5.71 | 150.10447 | C10H14O | Thymol(Isomer II) | Aromatic monoterpenoids |
| 36 | 7.99 | 348.19367 | C20H28O5 | Ingenol | Tigliane and ingenane diterpenoids |
| 37 | 8.30 | 392.21989 | C22H32O6 | NCGC00180384-03_C22H32O6_(1S,2R,4aR,8aR)-1-Acetoxy-7-isopropylidene-1,4a-dimethyl-6-oxodecahydro-2-naphthalenyl 2,3-dimethyl-2-oxiranecarboxylate (Isomer I) | Sesquiterpenoids |
| 38 | 8.80 | 229.24056 | C14H31NO | N,N-Dimethyldodecylamine N-oxide | Long-chain alkyl amine oxides |
| 39 | 8.97 | 392.21989 | C22H32O6 | NCGC00180384-03_C22H32O6_(1S,2R,4aR,8aR)-1-Acetoxy-7-isopropylidene-1,4a-dimethyl-6-oxodecahydro-2-naphthalenyl 2,3-dimethyl-2-oxiranecarboxylate (Isomer II) | Sesquiterpenoids |
| 40 | 9.88 | 148.05243 | C9H8O2 | 3,4-Dihydrocoumarin | Dihydrocoumarins |
| 41 | 12.03 | 162.03169 | C9H6O3 | Umbelliferone | Hydroxycoumarins |
| 42 | 12.05 | 358.30831 | C21H42O4 | 1-Monostearin | 1-monoacylglycerols |
| Samples | Total Phenolic Compounds (mg GAE/g extract) | Total Flavonoids (mg QE/g) | DPPH (µmol TE/g) | ABTS (µmol TE/ g) | FRAP (µmol TE/g) |
|---|---|---|---|---|---|
| Leaves | 359 ± 5.21 | 101 ± 1.42 | 796 ± 3.15 | 679 ± 0.85 | 806 ± 3.42 |
| Stems | 306 ± 1.93 * | 23.7 ± 0.80 * | 647 ± 3.27 * | 668 ± 2.30 | 575 ± 2.86 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tinco-Jayo, J.A.; Aguilar-Felices, E.J.; Enciso-Roca, E.C.; Arroyo-Acevedo, J.L.; Herrera-Calderon, O. Phytochemical Screening by LC-ESI-MS/MS and Effect of the Ethyl Acetate Fraction from Leaves and Stems of Jatropha macrantha Müll Arg. on Ketamine-Induced Erectile Dysfunction in Rats. Molecules 2022, 27, 115. https://doi.org/10.3390/molecules27010115
Tinco-Jayo JA, Aguilar-Felices EJ, Enciso-Roca EC, Arroyo-Acevedo JL, Herrera-Calderon O. Phytochemical Screening by LC-ESI-MS/MS and Effect of the Ethyl Acetate Fraction from Leaves and Stems of Jatropha macrantha Müll Arg. on Ketamine-Induced Erectile Dysfunction in Rats. Molecules. 2022; 27(1):115. https://doi.org/10.3390/molecules27010115
Chicago/Turabian StyleTinco-Jayo, Johnny Aldo, Enrique Javier Aguilar-Felices, Edwin Carlos Enciso-Roca, Jorge Luis Arroyo-Acevedo, and Oscar Herrera-Calderon. 2022. "Phytochemical Screening by LC-ESI-MS/MS and Effect of the Ethyl Acetate Fraction from Leaves and Stems of Jatropha macrantha Müll Arg. on Ketamine-Induced Erectile Dysfunction in Rats" Molecules 27, no. 1: 115. https://doi.org/10.3390/molecules27010115







