Individualized Dosage of Tacrolimus for Renal Transplantation Patients Based on Pharmacometabonomics
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Sample Collection
2.3. Metabolomics Analysis
2.4. Pharmacodynamic Analysis
2.5. Statistical Analysis
3. Results
3.1. Basic Characteristics of the Study Cohorts
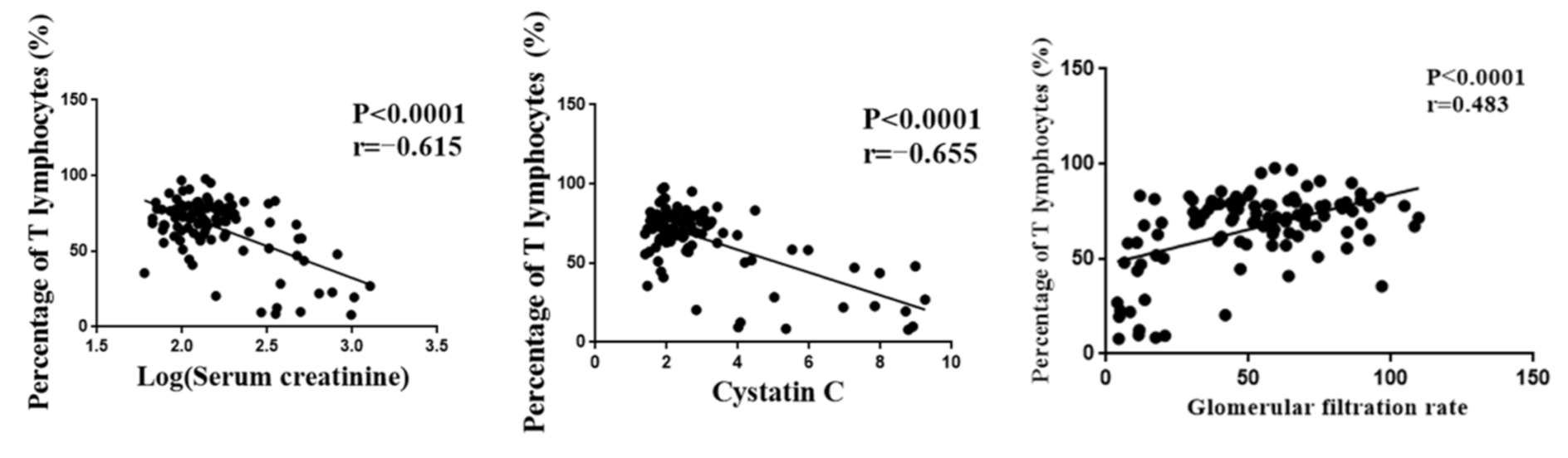
3.2. Correlation of T Lymphocytes with Renal Function Indexes and Outcome of Renal Transplantation
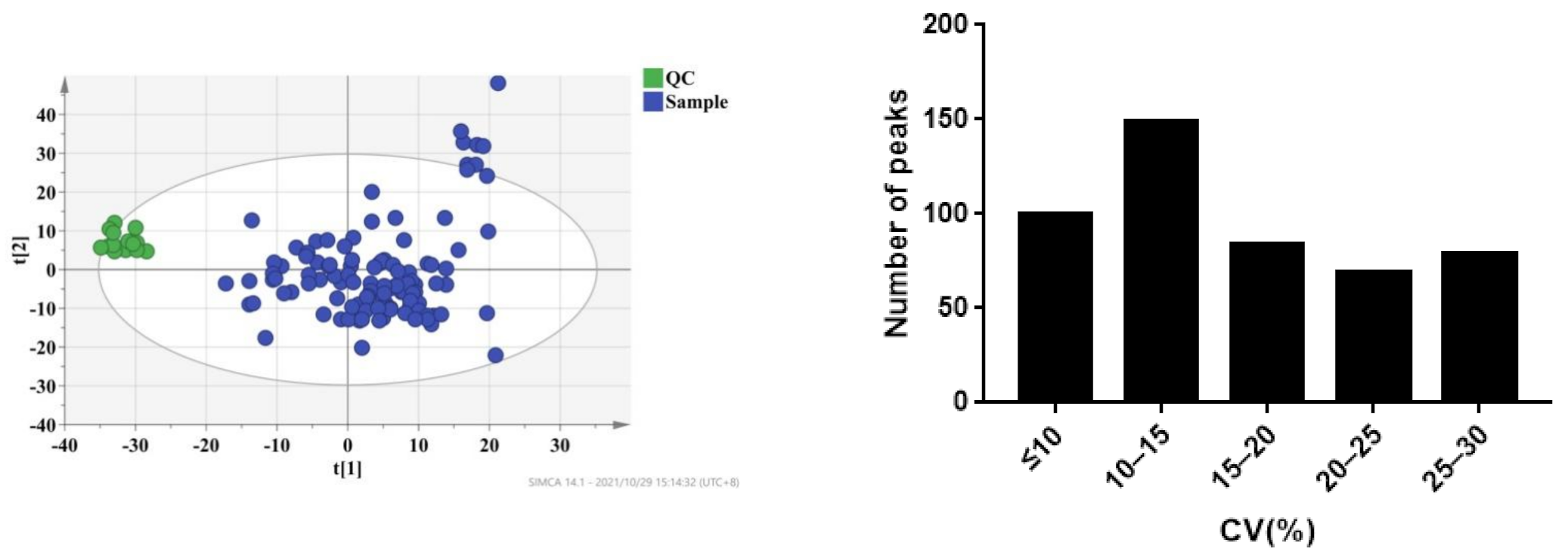
3.3. Metabolic Profiling of Plasma Samples
3.4. Identification of Metabolites Significantly Associated with the Percentage of T Lymphocytes
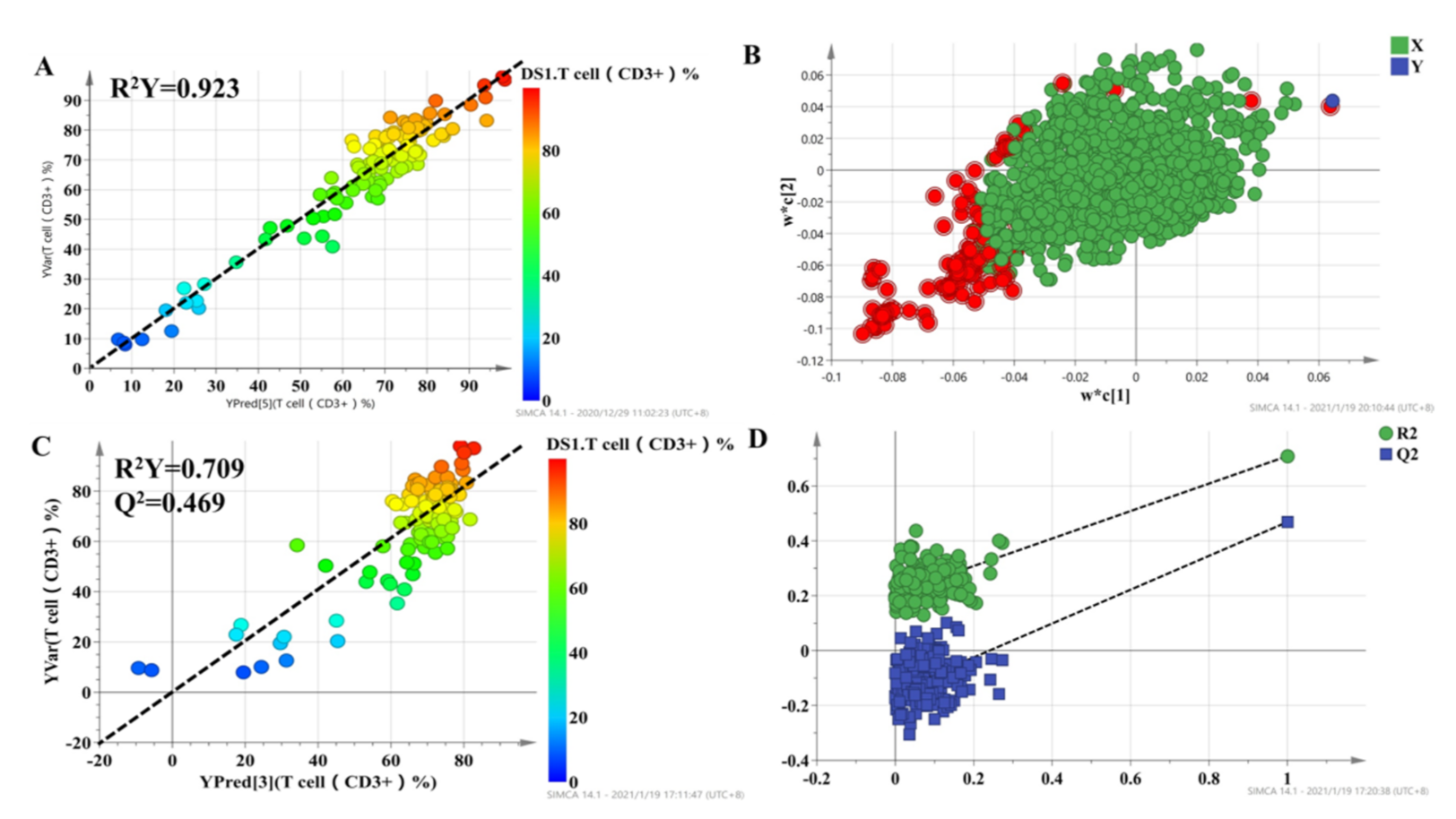
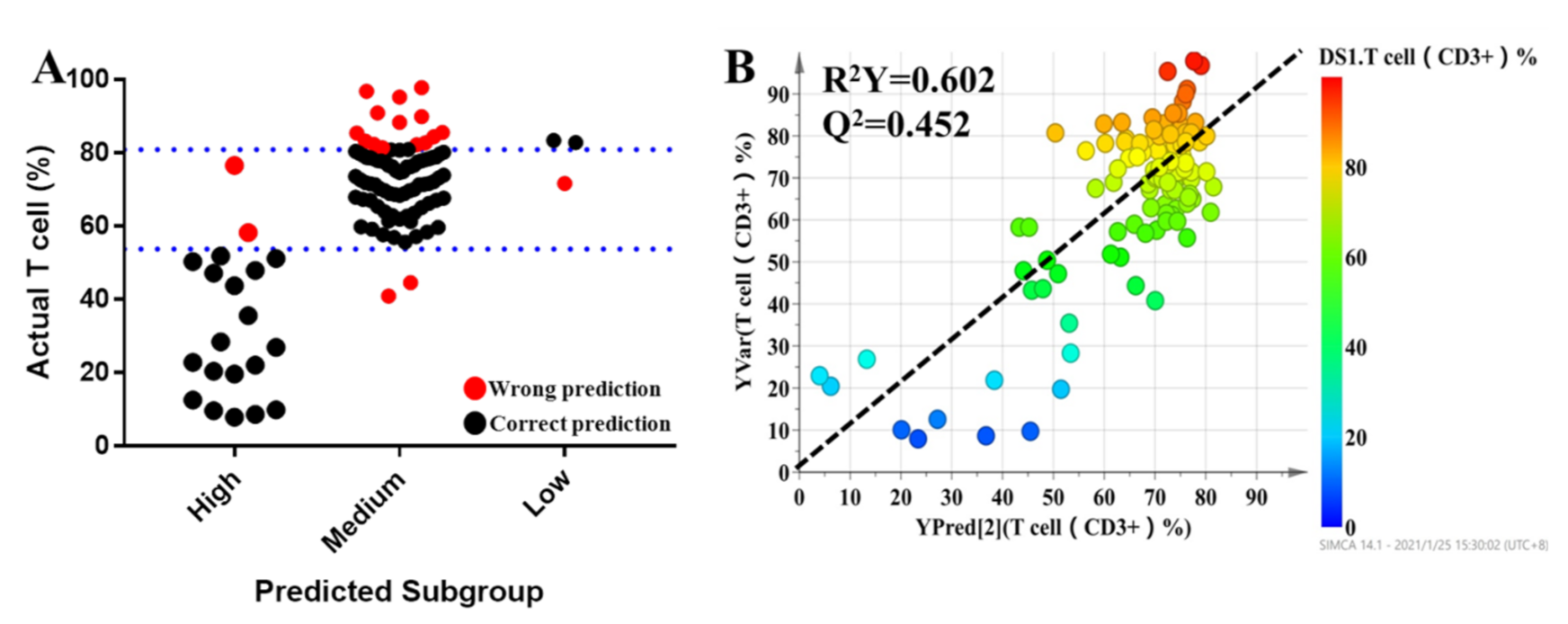
3.5. Prediction of the Percentage of T Lymphocytes Based on Key Metabolites and Clinical Characteristics
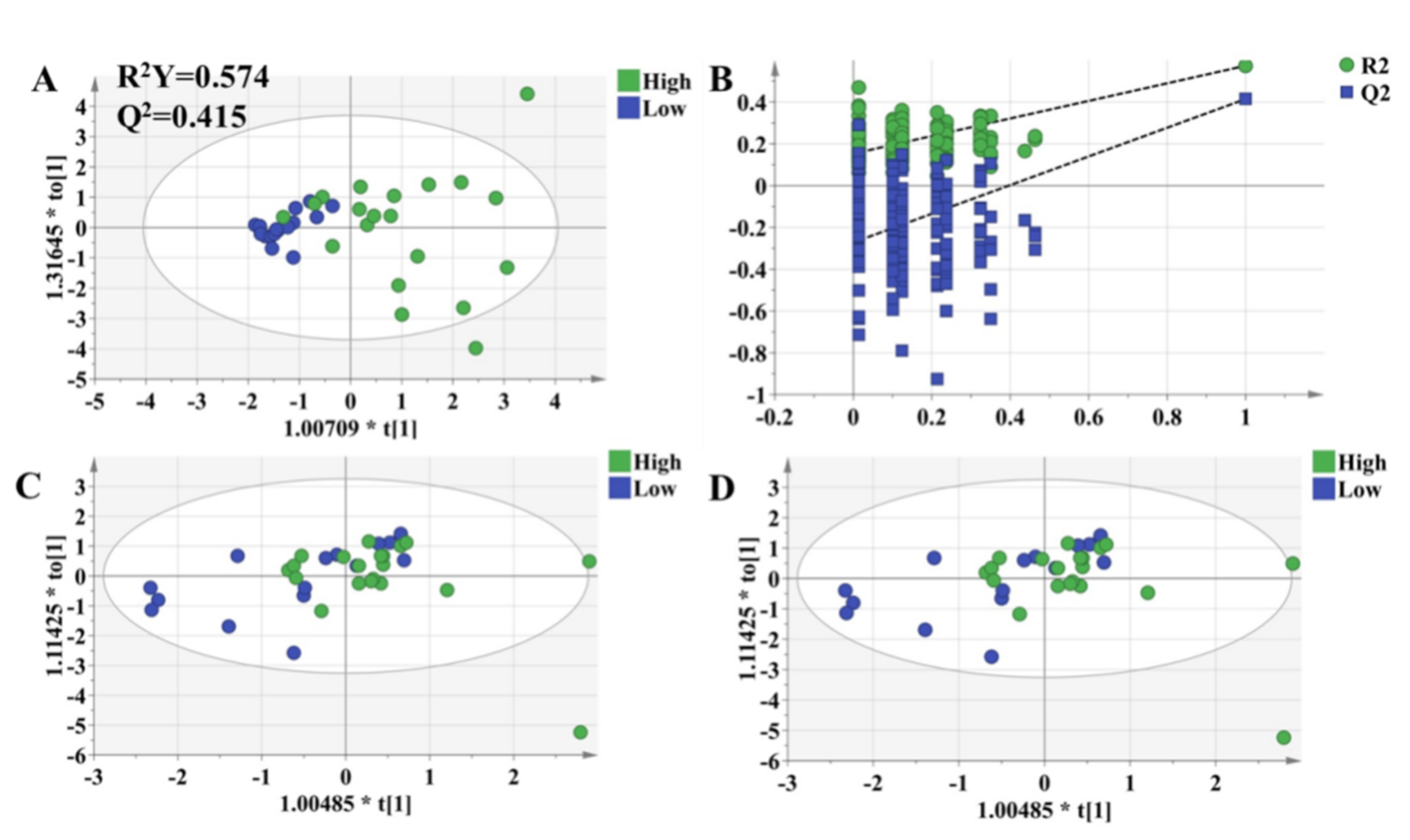
3.6. OPLS-DA Models to Characterize Pharmacodynamic Responses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Nankivell, B.J.; Alexander, S.I. Rejection of the kidney allograft. N. Engl. J. Med. 2010, 363, 1451–1462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, G.; Teixeira-Pinto, A.; Chapman, J.R.; Craig, J.C.; Pleass, H.; McDonald, S.; Lim, W.H. The Impact of Total Ischemic Time, Donor Age and the Pathway of Donor Death on Graft Outcomes After Deceased Donor Kidney Transplantation. Transplantation 2017, 101, 1152–1158. [Google Scholar] [CrossRef] [PubMed]
- Lim, M.A.; Kohli, J.; Bloom, R.D. Immunosuppression for kidney transplantation: Where are we now and where are we going? Transplant. Rev. 2017, 31, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Sikma, M.A.; van Maarseveen, E.M.; van de Graaf, E.A.; Kirkels, J.H.; Verhaar, M.C.; Donker, D.W.; Kesecioglu, J.; Meulenbelt, J. Pharmacokinetics and Toxicity of Tacrolimus Early After Heart and Lung Transplantation. Am. J. Transplant. 2015, 15, 2301–2313. [Google Scholar] [CrossRef]
- Clipstone, N.A.; Crabtree, G.R. Identification of calcineurin as a key signalling enzyme in T-lymphocyte activation. Nature 1992, 357, 695–697. [Google Scholar] [CrossRef]
- Gaynor, J.J.; Ciancio, G.; Guerra, G.; Sageshima, J.; Roth, D.; Goldstein, M.J.; Chen, L.; Kupin, W.; Mattiazzi, A.; Tueros, L.; et al. Lower tacrolimus trough levels are associated with subsequently higher acute rejection risk during the first 12 months after kidney transplantation. Transpl. Int. 2016, 29, 216–226. [Google Scholar] [CrossRef] [Green Version]
- Laskow, D.A.; Vincenti, F.; Neylan, J.F.; Mendez, R.; Matas, A.J. An open-label, concentration-ranging trial of FK506 in primary kidney transplantation: A report of the United States Multicenter FK506 Kidney Transplant Group. Transplantation 1996, 62, 900–905. [Google Scholar] [CrossRef]
- Dasari, B.V.; Hodson, J.; Nassir, A.; Widmer, J.; Isaac, J.; Mergentel, H.; Muiesan, P.; Mirza, D.F.; Perera, M.T. Variations in Practice to Therapeutic Monitoring of Tacrolimus following Primary Adult Liver Transplantation. Int. J. Organ Transplant. Med. 2016, 7, 1–8. [Google Scholar]
- Borni-Duval, C.; Caillard, S.; Olagne, J.; Perrin, P.; Braun-Parvez, L.; Heibel, F.; Moulin, B. Risk factors for BK virus infection in the era of therapeutic drug monitoring. Transplantation 2013, 95, 1498–1505. [Google Scholar] [CrossRef]
- Bentata, Y. Tacrolimus: 20 years of use in adult kidney transplantation. What we should know about its nephrotoxicity. Artif. Organs 2020, 44, 140–152. [Google Scholar] [CrossRef]
- Andrews, L.M.; Li, Y.; De Winter, B.C.M.; Shi, Y.Y.; Baan, C.C.; Van Gelder, T.; Hesselink, D.A. Pharmacokinetic considerations related to therapeutic drug monitoring of tacrolimus in kidney transplant patients. Expert Opin. Drug Metab. Toxicol. 2017, 13, 1225–1236. [Google Scholar] [CrossRef] [PubMed]
- Birdwell, K.A.; Decker, B.; Barbarino, J.M.; Peterson, J.F.; Stein, C.M.; Sadee, W.; Wang, D.; Vinks, A.A.; He, Y.; Swen, J.J.; et al. Clinical Pharmacogenetics Implementation Consortium (CPIC) Guidelines for CYP3A5 Genotype and Tacrolimus Dosing. Clin. Pharmacol. Ther. 2015, 98, 19–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hesselink, D.A.; Bouamar, R.; Elens, L.; van Schaik, R.H.; van Gelder, T. The role of pharmacogenetics in the disposition of and response to tacrolimus in solid organ transplantation. Clin. Pharmacokinet. 2014, 53, 123–139. [Google Scholar] [CrossRef] [PubMed]
- Kitsios, G.D.; Kent, D.M. Personalised medicine: Not just in our genes. BMJ 2012, 344, e2161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Corona, G.; Rizzolio, F.; Giordano, A.; Toffoli, G. Pharmaco-metabolomics: An emerging “omics” tool for the personalization of anticancer treatments and identification of new valuable therapeutic targets. J. Cell. Physiol. 2012, 227, 2827–2831. [Google Scholar] [CrossRef] [PubMed]
- Phapale, P.B.; Kim, S.D.; Lee, H.W.; Lim, M.; Kale, D.D.; Kim, Y.L.; Cho, J.H.; Hwang, D.; Yoon, Y.R. An integrative approach for identifying a metabolic phenotype predictive of individualized pharmacokinetics of tacrolimus. Clin. Pharmacol. Ther. 2010, 87, 426–436. [Google Scholar] [CrossRef]
- Huang, Q.; Aa, J.; Jia, H.; Xin, X.; Tao, C.; Liu, L.; Zou, B.; Song, Q.; Shi, J.; Cao, B.; et al. A Pharmacometabonomic Approach To Predicting Metabolic Phenotypes and Pharmacokinetic Parameters of Atorvastatin in Healthy Volunteers. J. Proteome Res. 2015, 14, 3970–3981. [Google Scholar] [CrossRef]
- Gao, Y.; Li, W.; Chen, J.; Wang, X.; Lv, Y.; Huang, Y.; Zhang, Z.; Xu, F. Pharmacometabolomic prediction of individual differences of gastrointestinal toxicity complicating myelosuppression in rats induced by irinotecan. Acta Pharm. Sin. B 2019, 9, 157–166. [Google Scholar] [CrossRef]
- Xing, X.; Ma, P.; Huang, Q.; Qi, X.; Zou, B.; Wei, J.; Tao, L.; Li, L.; Zhou, G.; Song, Q. Predicting Pharmacokinetics Variation of Faropenem Using a Pharmacometabonomic Approach. J. Proteome Res. 2020, 19, 119–128. [Google Scholar] [CrossRef]
- Demmers, M.W.; Korevaar, S.S.; Roemeling-van Rhijn, M.; van den Bosch, T.P.; Hoogduijn, M.J.; Betjes, M.G.; Weimar, W.; Baan, C.C.; Rowshani, A.T. Human renal tubular epithelial cells suppress alloreactive T cell proliferation. Clin. Exp. Immunol. 2015, 179, 509–519. [Google Scholar] [CrossRef] [Green Version]
- Randhawa, P. T-cell-mediated rejection of the kidney in the era of donor-specific antibodies: Diagnostic challenges and clinical significance. Curr. Opin. Organ Transplant. 2015, 20, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Linfert, D.; Chowdhry, T.; Rabb, H. Lymphocytes and ischemia-reperfusion injury. Transplant. Rev. 2009, 23, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vafadari, R.; Kraaijeveld, R.; Weimar, W.; Baan, C.C. Tacrolimus inhibits NF-kappaB activation in peripheral human T cells. PLoS ONE 2013, 8, e60784. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.K.; Lu, Q.B.; Chen, W.W.; Xu, W.; Liu, R.; Zhang, S.F.; Du, J.; Li, H.; Yao, K.; Zhai, D.; et al. Arginine deficiency is involved in thrombocytopenia and immunosuppression in severe fever with thrombocytopenia syndrome. Sci. Transl. Med. 2018, 10, eaat4162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, X.; Sun, G.; Feng, T.; Zhang, J.; Huang, X.; Wang, T.; Xie, Z.; Chu, X.; Yang, J.; Wang, H.; et al. Sodium oligomannate therapeutically remodels gut microbiota and suppresses gut bacterial amino acids-shaped neuroinflammation to inhibit Alzheimer’s disease progression. Cell Res. 2019, 29, 787–803. [Google Scholar] [CrossRef]
- Ding, Y.; Qin, L.; Kotenko, S.V.; Pestka, S.; Bromberg, J.S. A single amino acid determines the immunostimulatory activity of interleukin 10. J. Exp. Med. 2000, 191, 213–224. [Google Scholar] [CrossRef]
- Jin, U.H.; Lee, S.O.; Sridharan, G.; Lee, K.; Davidson, L.A.; Jayaraman, A.; Chapkin, R.S.; Alaniz, R.; Safe, S. Microbiome-derived tryptophan metabolites and their aryl hydrocarbon receptor-dependent agonist and antagonist activities. Mol. Pharmacol. 2014, 85, 777–788. [Google Scholar] [CrossRef] [Green Version]
- Lamas, B.; Richard, M.L.; Leducq, V.; Pham, H.P.; Michel, M.L.; Da Costa, G.; Bridonneau, C.; Jegou, S.; Hoffmann, T.W.; Natividad, J.M.; et al. CARD9 impacts colitis by altering gut microbiota metabolism of tryptophan into aryl hydrocarbon receptor ligands. Nat. Med. 2016, 22, 598–605. [Google Scholar] [CrossRef]
- Moffett, J.R.; Namboodiri, M.A. Tryptophan and the immune response. Immunol. Cell Biol. 2003, 81, 247–265. [Google Scholar] [CrossRef]
- Domanski, L.; Safranow, K.; Dolegowska, B.; Rozanski, J.; Myslak, M.; Ciechanowski, K.; Jakubowska, K.; Dziedziejko, V.; Romanowski, M.; Sulikowski, T.; et al. Hypoxanthine as a graft ischemia marker stimulates catalase activity in the renal vein during reperfusion in humans. Transplant. Proc. 2006, 38, 35–38. [Google Scholar] [CrossRef]
- Jouve, T.; Noble, J.; Rostaing, L.; Malvezzi, P. An update on the safety of tacrolimus in kidney transplant recipients, with a focus on tacrolimus minimization. Expert Opin. Drug Saf. 2019, 18, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Li, S.S.; Niu, M.; Jing, J.; Huang, Y.; Zhang, Z.T.; Chen, S.S.; Shi, G.Z.; He, X.; Zhang, H.Z.; Xiao, X.H.; et al. Metabolomic Signatures of Autoimmune Hepatitis in the Development of Cirrhosis. Front. Med. 2021, 8, 644376. [Google Scholar] [CrossRef] [PubMed]
- Quan, W.; Jiao, Y.; Xue, C.; Li, Y.; Liu, G.; He, Z.; Qin, F.; Zeng, M.; Chen, J. The Effect of Exogenous Free N(epsilon)-(Carboxymethyl)Lysine on Diabetic-Model Goto-Kakizaki Rats: Metabolomics Analysis in Serum and Urine. J. Agric. Food Chem. 2021, 69, 783–793. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Wang, Q.; Xia, T.; Fu, S.; Tao, X.; Wen, Y.; Chan, S.; Gao, S.; Xiong, X.; Chen, W. Diagnostic value of plasma tryptophan and symmetric dimethylarginine levels for acute kidney injury among tacrolimus-treated kidney transplant patients by targeted metabolomics analysis. Sci. Rep. 2018, 8, 14688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Homan van der Heide, J.J.; Bilo, H.J.; Tegzess, A.M.; Donker, A.J. The effects of dietary supplementation with fish oil on renal function in cyclosporine-treated renal transplant recipients. Transplantation 1990, 49, 523–527. [Google Scholar] [CrossRef] [PubMed]
- Culp, B.R.; Titus, B.G.; Lands, W.E. Inhibition of prostaglandin biosynthesis by eicosapentaenoic acid. Prostaglandins Med. 1979, 3, 269–278. [Google Scholar] [CrossRef] [Green Version]
- Jiao, W.; Zhang, Z.; Xu, Y.; Gong, L.; Zhang, W.; Tang, H.; Zeng, S.; Zhang, Q.; Sun, Z.; Liu, L.; et al. Butyric acid normalizes hyperglycemia caused by the tacrolimus-induced gut microbiota. Am. J. Transplant. 2020, 20, 2413–2424. [Google Scholar] [CrossRef]

| Clinical Characteristics | Normal Group (n = 73) | Low-Response Group (n = 16) | High-Response Group (n = 20) | p |
|---|---|---|---|---|
| Ages (years) | 40.73 ± 11.29 | 40.44 ± 11.14 | 48.8 ± 8.03 | 0.093 |
| Female (%) | 24 (32.9%) | 4 (25%) | 5 (25%) | 0.741 |
| Tacrolimus dose | 3.03 ± 0.75 | 3.19 ± 0.66 | 2.83 ± 0.90 | 0.369 |
| BMI (kg/m2) | 21.46 ± 3.14 | 21.70 ± 3.18 | 22.60 ± 3.04 | 0.254 |
| Cr (μmol/L) | 156.43 ± 110.56 | 154.06 ± 79.36 | 484.1 ± 338.47 | <0.001 |
| CYC (mg/mL) | 2.43 ± 0.81 | 2.38 ± 0.75 | 5.58 ± 2.71 | <0.001 |
| Ccr (ml/min) | 57.03 ± 22.62 | 56.66 ± 23.72 | 24.13 ± 25.62 | <0.001 |
| Albumin (g/L) | 36.05 ± 3.04 | 35.83 ± 3.60 | 34.99 ± 2.78 | 0.405 |
| Hemoglobin (g/L) | 751.81 ± 538.32 | 97.35 ± 87.39 | 1017.98 ± 619.39 | <0.001 |
| Hematocrit (%) | 30.70 ± 5.77 | 27.96 ± 5.76 | 29.53 ± 6.32 | 0.233 |
| ALT (U/L) | 30.23 ± 37.84 | 35.81 ± 22.31 | 31.15 ± 26.94 | 0.843 |
| AST (U/L) | 19.22 ± 13.47 | 18.25 ± 6.23 | 22.70 ± 16.40 | 0.532 |
| Total bilirubin (μmol/L) | 7.29 ± 3.49 | 6.73 ± 2.05 | 8.33 ± 4.94 | 0.398 |
| Subgroups | Delayed Graft Function (%) | Acute Rejection (%) |
|---|---|---|
| Normal group a (n = 73) | 3 (4.11%) | 7 (9.59%) |
| Low-response group b (n = 16) | 0 | 4 (25%) |
| High-response group c (n = 20) | 11 (68.75%) | 0 |
| p | <0.0001 | 0.105 |
| Metabolites | ID | Adduct (Observed) | Retention Time | m/z (Observed) | VIP | Correlation Coefficient | p-Value |
|---|---|---|---|---|---|---|---|
| Mesobilirubinogen | HMDB01898 | [M + H]+ | 4.69 | 593.33 | 1.25 | −0.337 | <0.001 |
| Cinnamoside | HMDB38923 | [M + NH4]+ | 4.78 | 536.27 | 1.23 | −0.455 | <0.001 |
| L-Isoleucine | HMDB00172 | [M + H]+ | 1.05 | 132.10 | 1.22 | −0.271 | 0.003 |
| 5-Methoxyindoleacetate | HMDB04096 | [M − H]− | 1.04 | 204.07 | 1.20 | −0.413 | <0.001 |
| DG(18:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)/0:0) | HMDB07179 | [M − H]− | 11.13 | 667.53 | 1.19 | −0.337 | <0.001 |
| PI(16:1(9Z)/0:0) | LMGP06050009 | [M + H − H2O]+ | 4.71 | 553.28 | 1.17 | −0.483 | <0.001 |
| Isoleucyl-Proline | HMDB03141 | [M + H]+ | 1.05 | 229.15 | 1.16 | −0.500 | <0.001 |
| Retinoyl b-glucuronide | HMDB28915 | [M + H − H2O]+ | 4.64 | 459.24 | 1.16 | −0.462 | <0.001 |
| Tryptophyl-Arginine | HMDB29077 | [2M + H]+ | 4.65 | 721.39 | 1.15 | −0.489 | <0.001 |
| Butyric acid | HMDB00039 | [2M + H]+ | 4.65 | 177.11 | 1.13 | −0.480 | <0.001 |
| Norepinephrine | HMDB00216 | [M + H]+ | 1.05 | 170.08 | 1.12 | −0.452 | <0.001 |
| Eicosapentaenoic acid | HMDB01999 | [M − H]− | 9.72 | 301.22 | 1.08 | −0.303 | 0.001 |
| Gamma glutamyl ornithine | HMDB02248 | [2M + H]+ | 4.47 | 523.27 | 1.05 | −0.446 | <0.001 |
| Methionyl-Methionine | HMDB28979 | [M − H2O − H]- | 3.19 | 261.07 | 1.05 | −0.309 | 0.001 |
| Hydroxybutyrylcarnitine | HMDB13127 | [M + NH4]+ | 4.94 | 265.18 | 1.05 | −0.359 | <0.001 |
| N2-Succinoylarginine | HMDB32764 | [2M + H]+ | 4.84 | 549.26 | 1.05 | −0.299 | 0.001 |
| L-Aspartyl-L-phenylalanine | HMDB00706 | [M - H]- | 0.98 | 279.10 | 1.04 | −0.301 | 0.001 |
| 24-Keto-25dehydrocholesterol | LMST01010299 | [M + H − H2O]+ | 4.93 | 381.31 | 1.03 | −0.287 | 0.002 |
| 7-Methylhypoxanthine | HMDB03162 | [M + H]+ | 1.05 | 151.06 | 1.00 | −0.373 | <0.001 |
| Variables | Low-Response Group | High-Response Group | ||||||
|---|---|---|---|---|---|---|---|---|
| 95% CI | 95% CI | |||||||
| p-Value | OR | Lower | Upper | p-Value | OR | Lower | Upper | |
| Dosages | 0.783 | 0.888 | 0.383 | 2.060 | 0.376 | 0.555 | 0.151 | 2.040 |
| Serum creatinine | 0.693 | 0.471 | 0.011 | 19.876 | 0.104 | 86.550 | 0.401 | 18,686.260 |
| Mesobilirubinogen | 0.179 | 1.135 | 0.944 | 1.364 | 0.010 | 1.351 | 1.074 | 1.699 |
| L-Isoleucine | 0.062 | 0.649 | 0.412 | 1.022 | 0.001 | 0.256 | 0.114 | 0.574 |
| 5-Methoxyindoleacetate | 0.836 | 0.934 | 0.490 | 1.782 | 0.040 | 3.176 | 1.054 | 9.568 |
| Eicosapentaenoic acid | 0.638 | 1.082 | 0.778 | 1.505 | 0.019 | 1.686 | 1.089 | 2.608 |
| N2-succinoylarginine | 0.114 | 1.014 | 0.997 | 1.031 | 0.108 | 1.014 | 0.997 | 1.031 |
| Tryptophyl-arginine | 0.038 | 1.158 | 1.008 | 1.329 | 0.009 | 1.357 | 1.079 | 1.707 |
| Butyric acid | 0.022 | 0.896 | 0.816 | 0.984 | 0.111 | 0.889 | 0.770 | 1.027 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, X.; Yang, X.; Yan, X.; Huang, M.; Xiang, Z.; Lou, Y. Individualized Dosage of Tacrolimus for Renal Transplantation Patients Based on Pharmacometabonomics. Molecules 2022, 27, 3517. https://doi.org/10.3390/molecules27113517
He X, Yang X, Yan X, Huang M, Xiang Z, Lou Y. Individualized Dosage of Tacrolimus for Renal Transplantation Patients Based on Pharmacometabonomics. Molecules. 2022; 27(11):3517. https://doi.org/10.3390/molecules27113517
Chicago/Turabian StyleHe, Xiaoying, Xi Yang, Xiaoting Yan, Mingzhu Huang, Zheng Xiang, and Yan Lou. 2022. "Individualized Dosage of Tacrolimus for Renal Transplantation Patients Based on Pharmacometabonomics" Molecules 27, no. 11: 3517. https://doi.org/10.3390/molecules27113517







