Statistical Bioinformatics to Uncover the Underlying Biological Mechanisms That Linked Smoking with Type 2 Diabetes Patients Using Transcritpomic and GWAS Analysis
Abstract
:1. Introduction
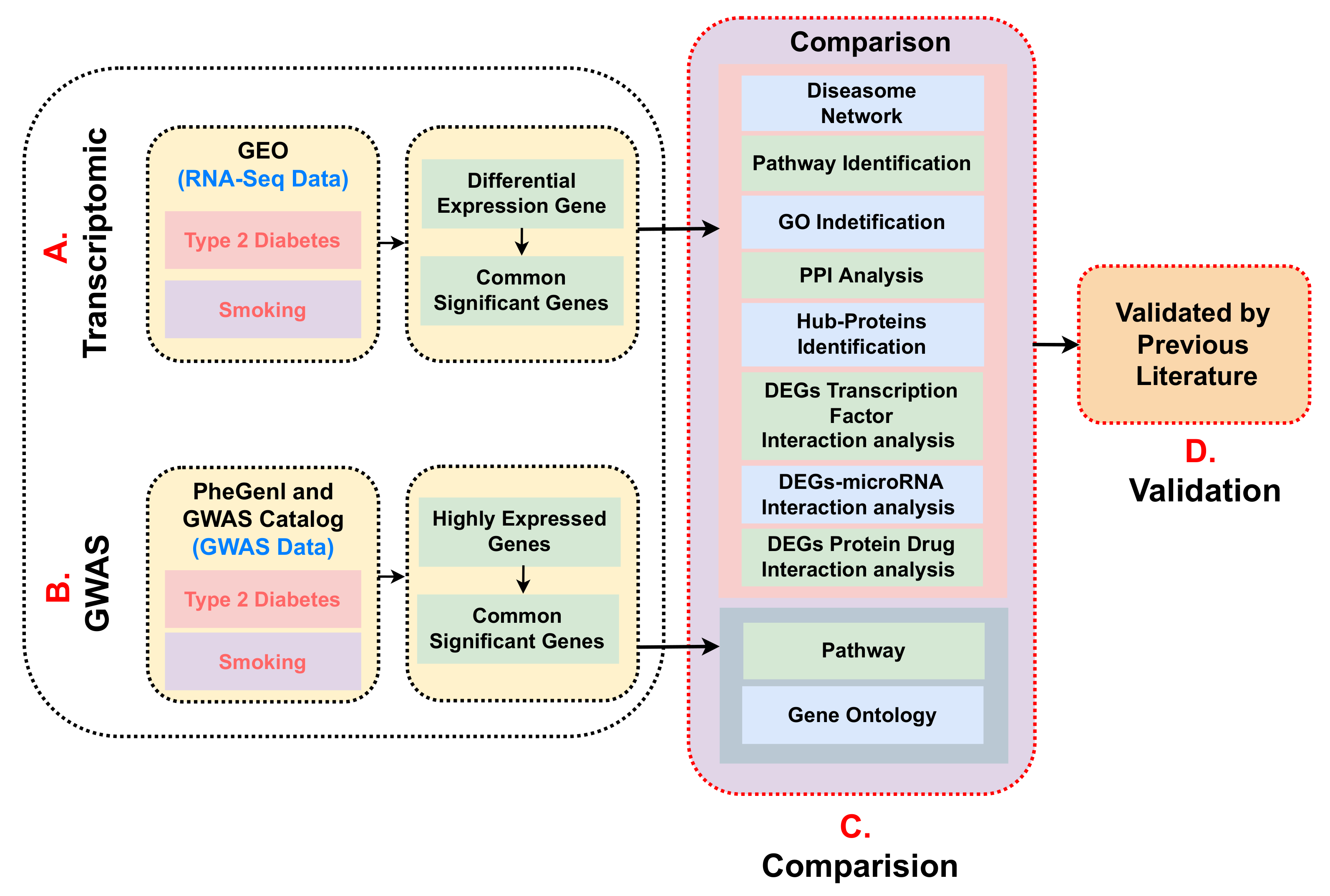
2. Materials and Methods
2.1. Datasets Employed in This Study
2.2. Preprocessing of Raw Counts and Its Differentially Expression Analysis
2.3. Identification of Molecular Pathway and Gene Ontology
2.4. GWAS Data of Type 2 Diabetes Mined to Compare with the GWAS Data of Smoker
3. Results
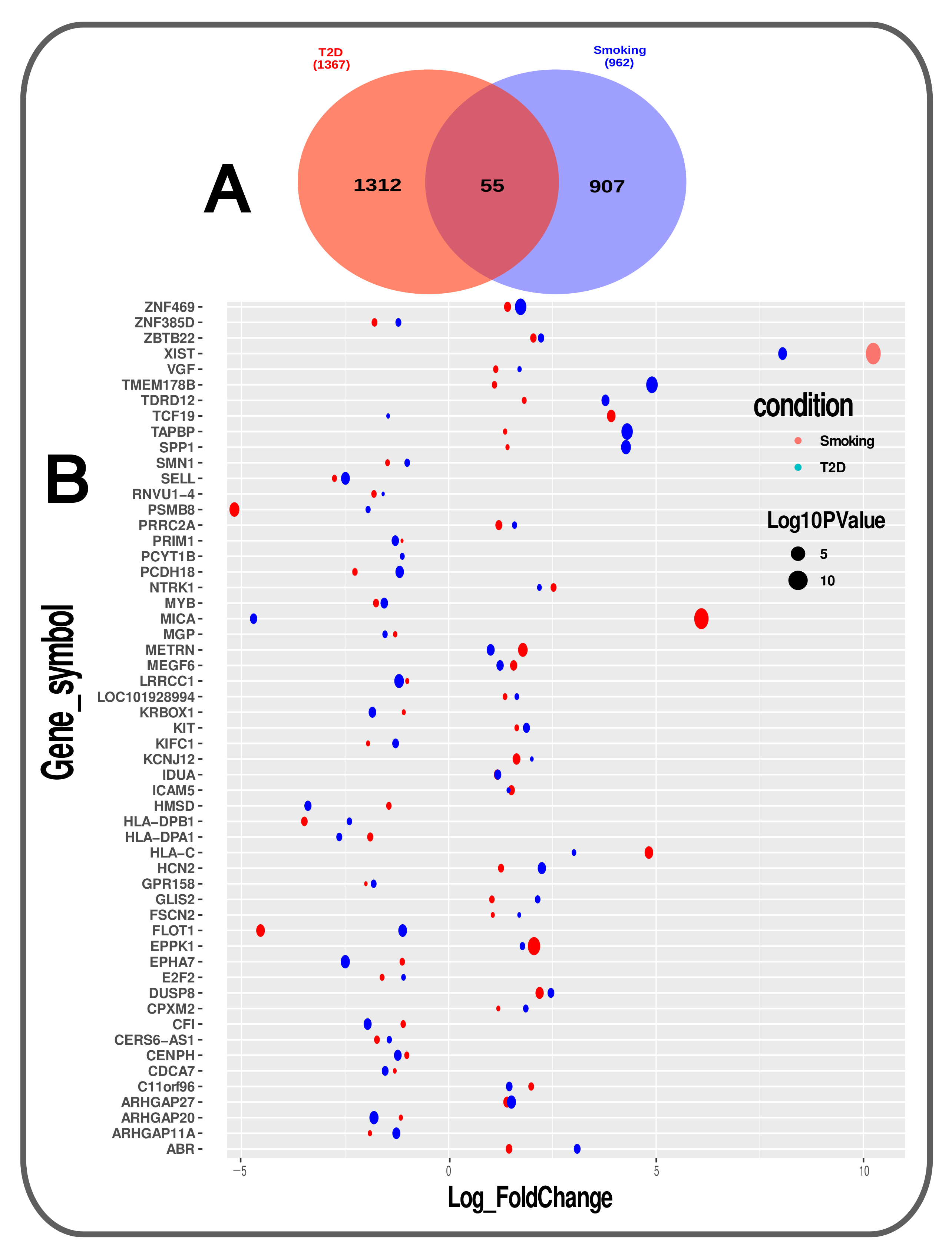
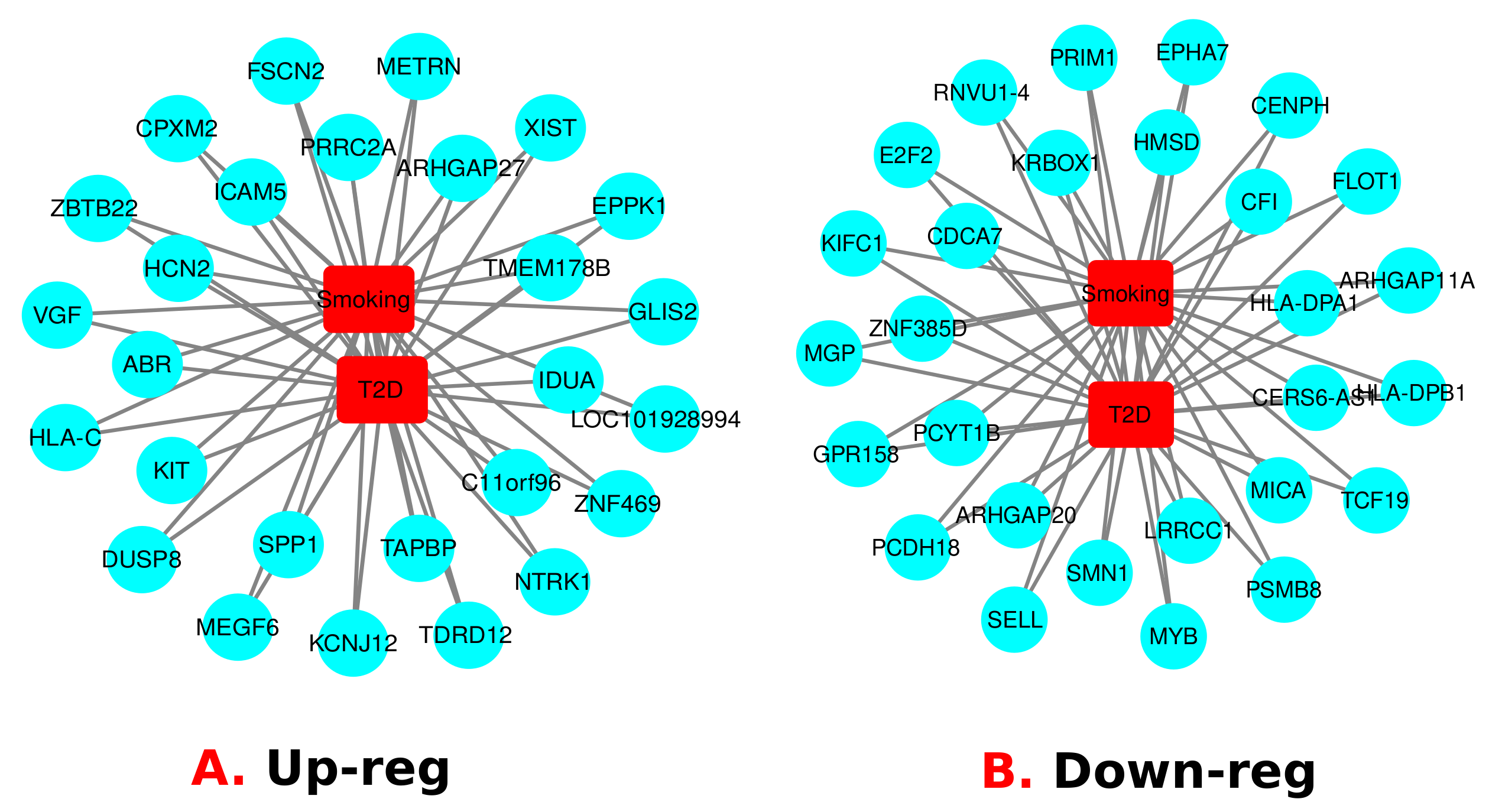
3.1. Gene Expression Analysis of Transcriptomic Data
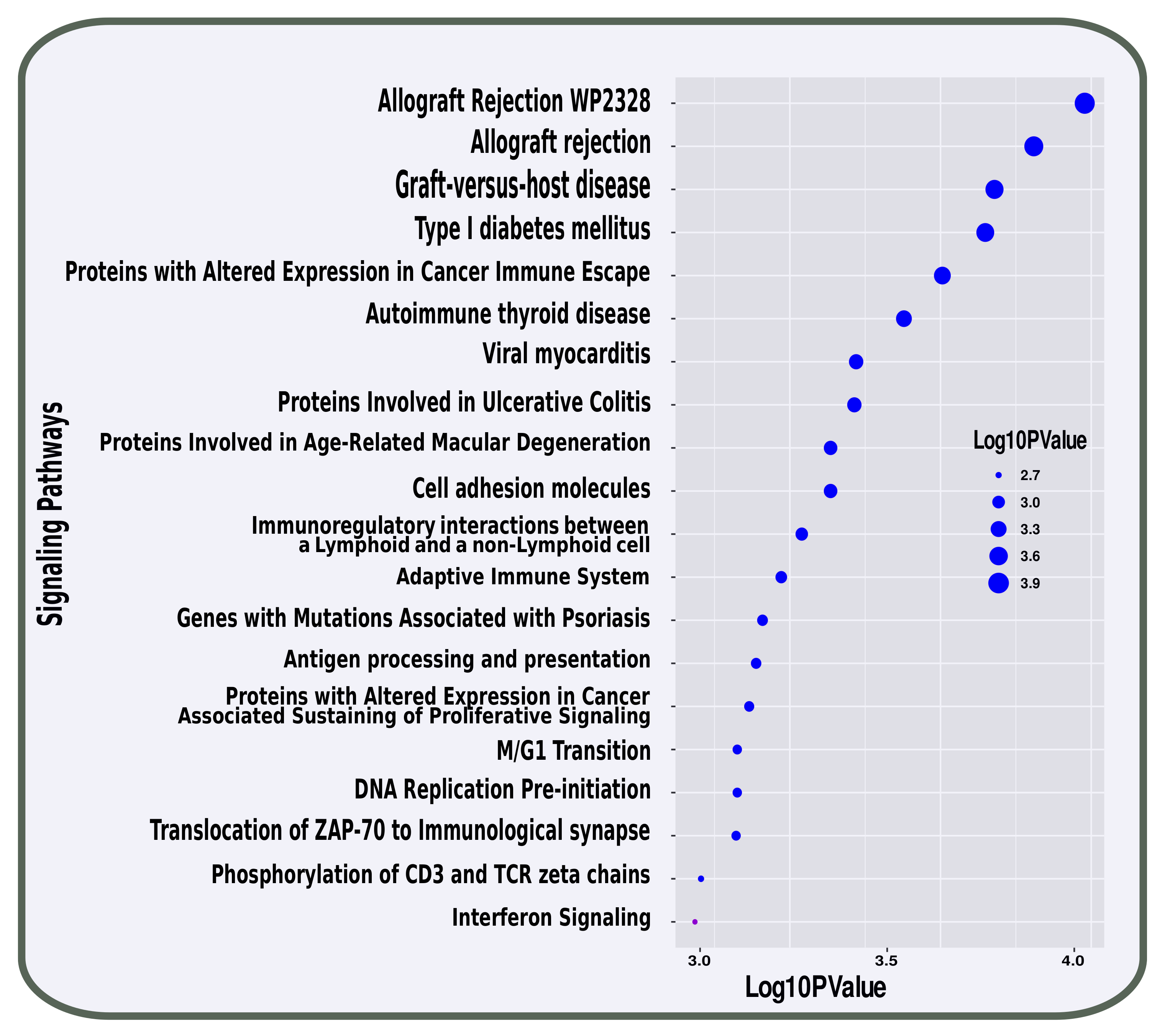
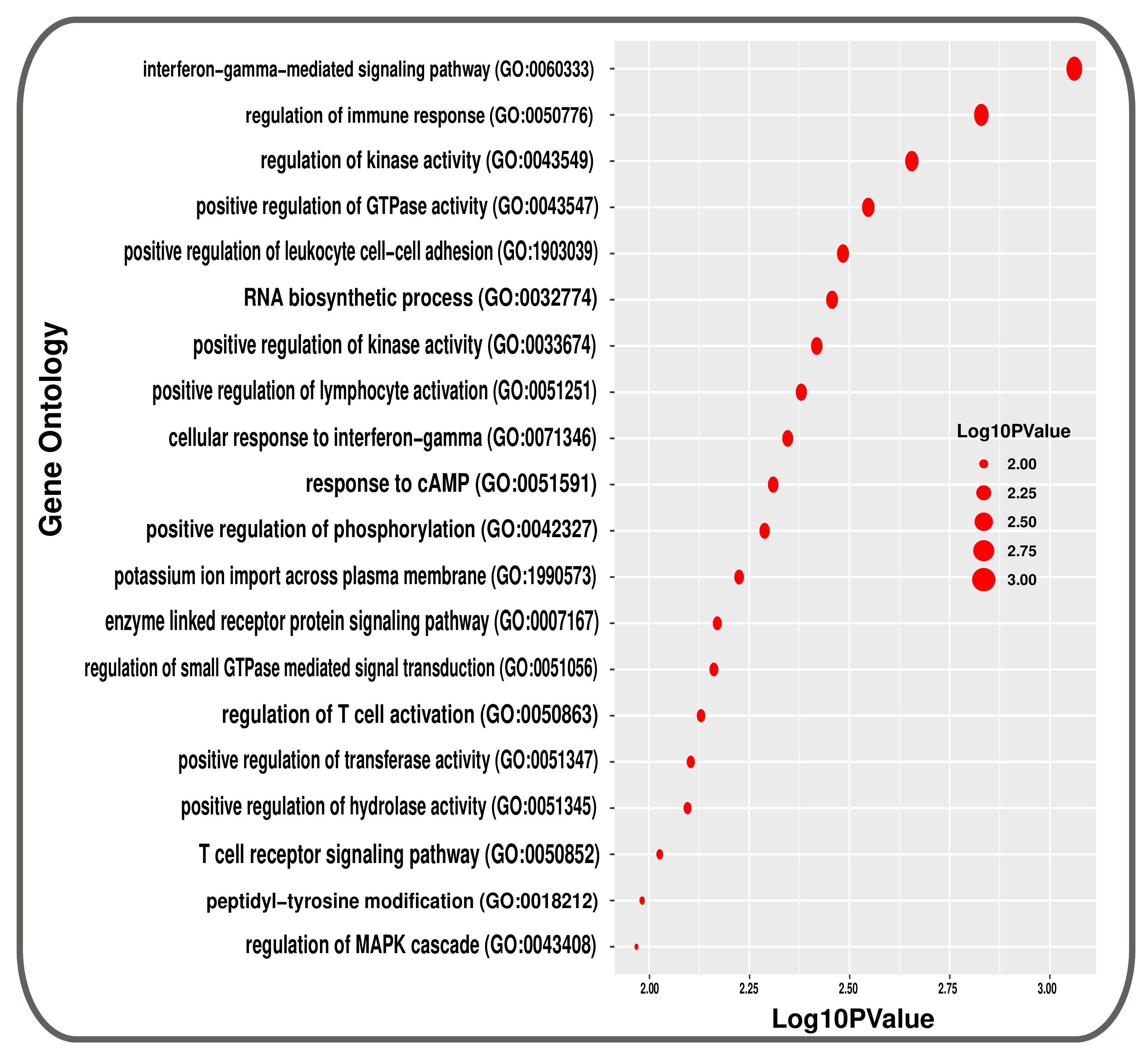
3.2. Pathway and GO Related Functional Association Analysis
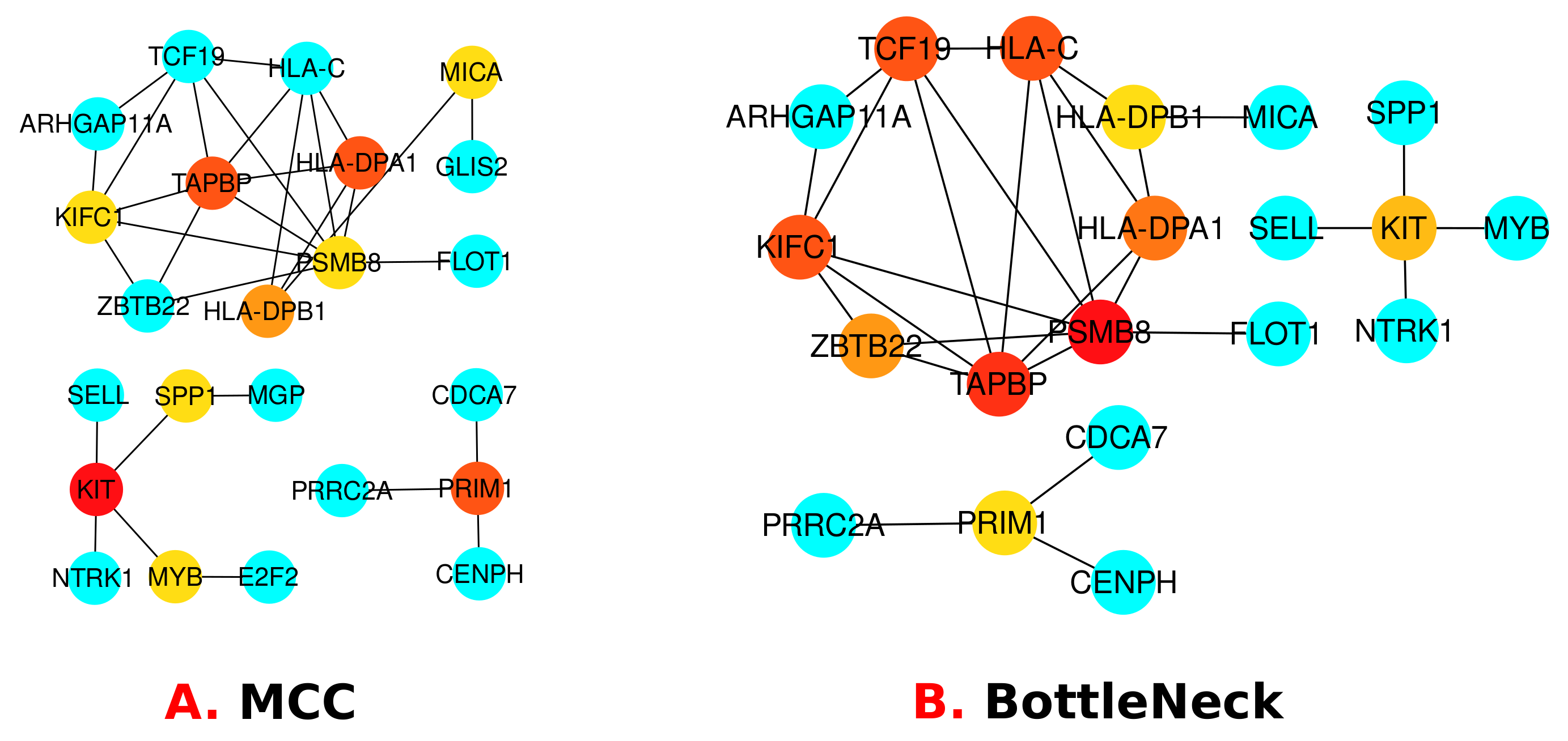
3.3. Protein–Protein Interactions (PPIs) Analysis
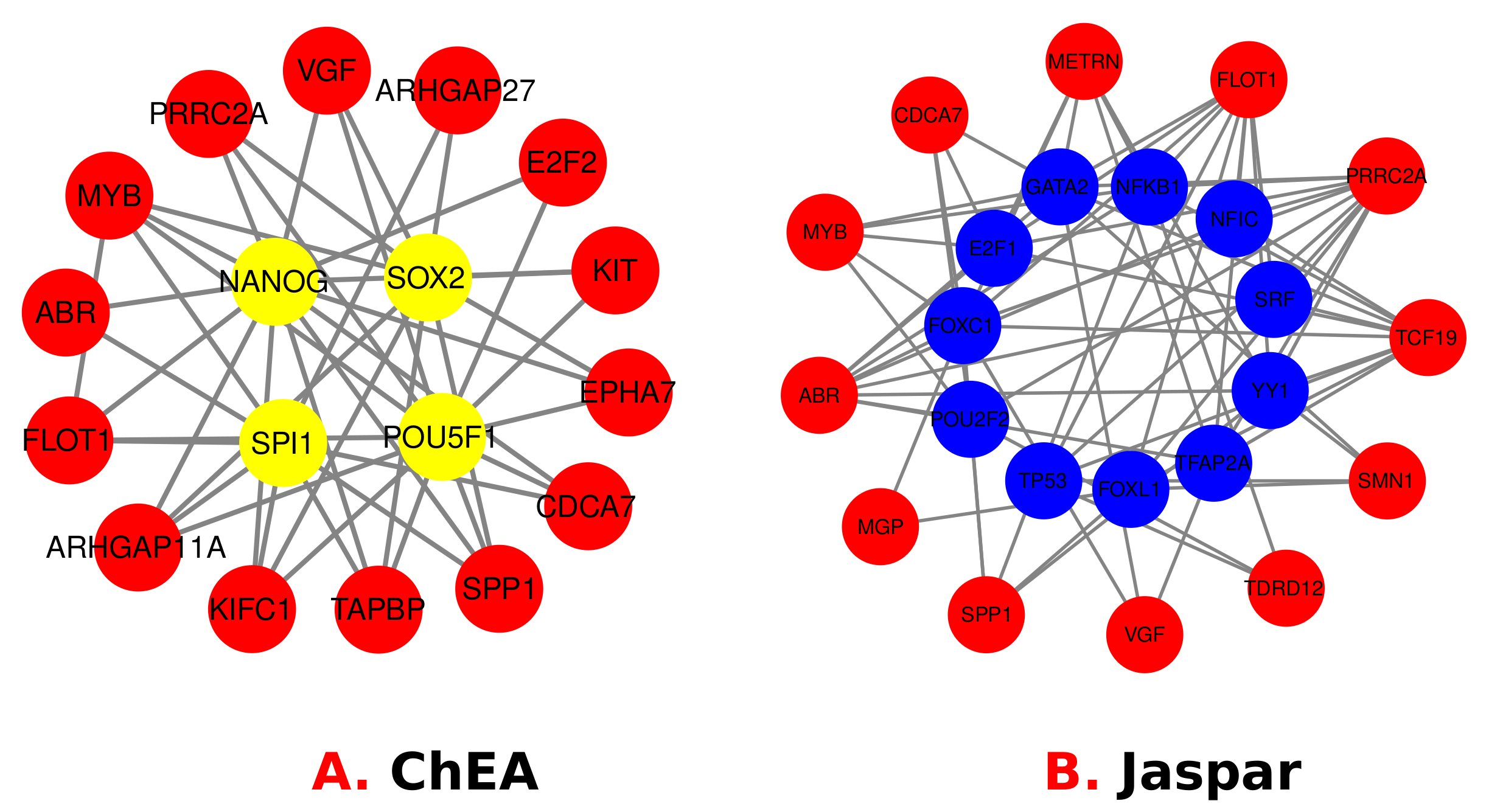
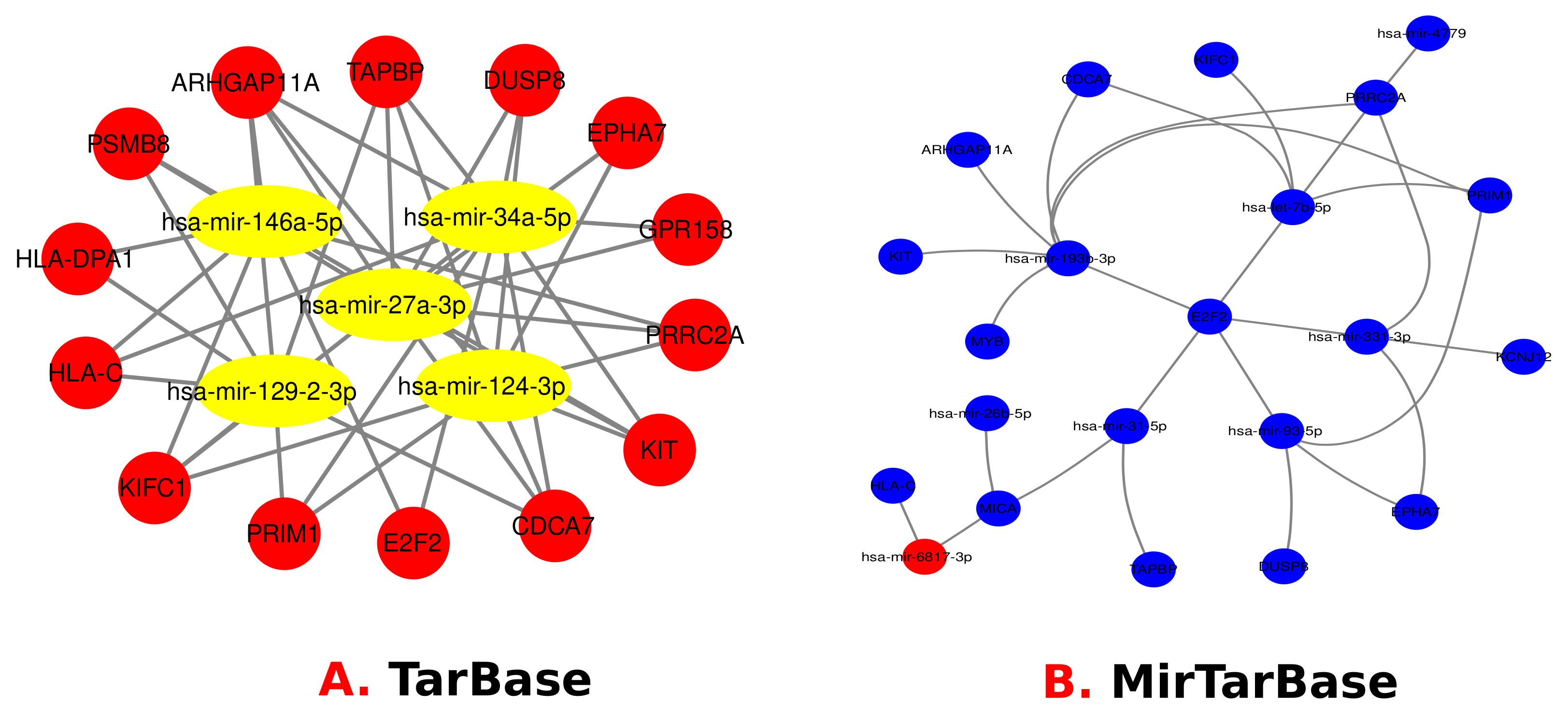
3.4. Identification of Transcriptional and Post-Transcriptional Regulators of the Differentially Expressed Genes
3.5. GWAS Analysis of Type 2 Diabetes with Smoker and Comparison with Transcriptomic Analysis
3.6. Validating Potential Biomarker Targets Using Earlier Literatures
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Olokoba, A.B.; Obateru, O.A.; Olokoba, L.B. Type 2 diabetes mellitus: A review of current trends. Oman Med. J. 2012, 27, 269. [Google Scholar] [CrossRef] [PubMed]
- Beta Cells. 2021. Available online: https://www.medicalnewstoday.com/articles/beta-cells-in-type-2-diabetes (accessed on 14 April 2022).
- Martín-Timón, I.; Sevillano-Collantes, C.; Segura-Galindo, A.; del Cañizo-Gómez, F.J. Type 2 diabetes and cardiovascular disease: Have all risk factors the same strength? World J. Diabetes 2014, 5, 444. [Google Scholar] [CrossRef] [PubMed]
- Cho, N.; Shaw, J.; Karuranga, S.; Huang, Y.; da Rocha Fernandes, J.; Ohlrogge, A.; Malanda, B. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res. Clin. Pract. 2018, 138, 271–281. [Google Scholar] [CrossRef]
- Diabetic-WHO. 2021. Available online: https://www.who.int/news-room/fact-sheets/detail/diabetes (accessed on 14 April 2022).
- Diabetes Smoking. 2022. Available online: https://www.cdc.gov/tobacco/campaign/tips/diseases/diabetes.html (accessed on 14 April 2022).
- Xu, H.; Wang, Q.; Sun, Q.; Qin, Y.; Han, A.; Cao, Y.; Yang, Q.; Yang, P.; Lu, J.; Liu, Q.; et al. In type 2 diabetes induced by cigarette smoking, activation of p38 MAPK is involved in pancreatic β-cell apoptosis. Environ. Sci. Pollut. Res. 2018, 25, 9817–9827. [Google Scholar] [CrossRef]
- Consortium, I.; Spijkerman, A.M.; van der A, D.L.; Nilsson, P.M.; Ardanaz, E.; Gavrila, D.; Agudo, A.; Arriola, L.; Balkau, B.; Beulens, J.W.; et al. Smoking and long-term risk of type 2 diabetes: The EPIC-InterAct study in European populations. Diabetes Care 2014, 37, 3164–3171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, S.; Xue, H.L.; Yu, H.J.; Huang, Y.; Tang, B.W.; Yang, X.H.; Li, Q.X.; He, Q.Q. Cigarette smoking as a risk factor for type 2 diabetes in women compared with men: A systematic review and meta-analysis of prospective cohort studies. J. Public Health 2019, 41, e169–e176. [Google Scholar] [CrossRef]
- Östgren, C.J.; Lindblad, U.; Ranstam, J.; Melander, A.; Råstam, L. Associations between smoking and β-cell function in a non-hypertensive and non-diabetic populationSkaraborg Hypertension and Diabetes Project. Diabet. Med. 2000, 17, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Wang, H.; Qiu, Q.; Tan, L.l.; Chen, W.; Yu, X.Q.; Sun, X.L.; Chen, W.Q. Mediation of abdominal obesity on the association between cigarette smoking and β-cell function. Zhonghua Liu Xing Bing Xue Za Zhi 2010, 31, 988–991. [Google Scholar] [PubMed]
- Artese, A.; Stamford, B.A.; Moffatt, R.J. Cigarette smoking: An accessory to the development of insulin resistance. Am. J. Lifestyle Med. 2019, 13, 602–605. [Google Scholar] [CrossRef]
- Mouhamed, D.H.; Ezzaher, A.; Neffati, F.; Douki, W.; Gaha, L.; Najjar, M. Effect of cigarette smoking on insulin resistance risk. In Proceedings of the Annales de Cardiologie et d’Angéiologie; Elsevier: Amsterdam, The Netherlands, 2016; Volume 65, pp. 21–25. [Google Scholar]
- Ren, W.; Gu, Y.; Zhu, L.; Wang, L.; Chang, Y.; Yan, M.; Han, B.; He, J. The effect of cigarette smoking on vitamin D level and depression in male patients with acute ischemic stroke. Compr. Psychiatry 2016, 65, 9–14. [Google Scholar] [CrossRef]
- Piazzolla, G.; Castrovilli, A.; Liotino, V.; Vulpi, M.R.; Fanelli, M.; Mazzocca, A.; Candigliota, M.; Berardi, E.; Resta, O.; Sabbà, C.; et al. Metabolic syndrome and Chronic Obstructive Pulmonary Disease (COPD): The interplay among smoking, insulin resistance and vitamin D. PLoS ONE 2017, 12, e0186708. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lips, P.; Eekhoff, M.; van Schoor, N.; Oosterwerff, M.; de Jongh, R.; Krul-Poel, Y.; Simsek, S. Vitamin D and type 2 diabetes. J. Steroid Biochem. Mol. Biol. 2017, 173, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Haase, C.L.; Tybjærg-Hansen, A.; Nordestgaard, B.G.; Frikke-Schmidt, R. HDL cholesterol and risk of type 2 diabetes: A Mendelian randomization study. Diabetes 2015, 64, 3328–3333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abbasi, A.; Corpeleijn, E.; Gansevoort, R.T.; Gans, R.O.; Hillege, H.L.; Stolk, R.P.; Navis, G.; Bakker, S.J.; Dullaart, R.P. Role of HDL cholesterol and estimates of HDL particle composition in future development of type 2 diabetes in the general population: The PREVEND study. J. Clin. Endocrinol. Metab. 2013, 98, E1352–E1359. [Google Scholar] [CrossRef] [Green Version]
- Rader, D.J.; Hobbs, H.H. Disorders of lipoprotein metabolism. Harrisons Princ. Intern. Med. 2005, 16, 2286. [Google Scholar]
- He, B.M.; Zhao, S.P.; Peng, Z.Y. Effects of cigarette smoking on HDL quantity and function: Implications for atherosclerosis. J. Cell. Biochem. 2013, 114, 2431–2436. [Google Scholar] [CrossRef]
- Zhao, J.; Zhang, Y.; Wei, F.; Song, J.; Cao, Z.; Chen, C.; Zhang, K.; Feng, S.; Wang, Y.; Li, W.D. Triglyceride is an independent predictor of type 2 diabetes among middle-aged and older adults: A prospective study with 8-year follow-ups in two cohorts. J. Transl. Med. 2019, 17, 1–7. [Google Scholar] [CrossRef]
- Bading-Taïka, B.; Souza, A.; Bourobou Bourobou, H.P.; Lione, L.A. Hypoglycaemic and anti-hyperglycaemic activity of Tabernanthe iboga aqueous extract in fructose-fed streptozotocin type 2 diabetic rats. Adv. Tradit. Med. 2021, 21, 281–295. [Google Scholar] [CrossRef]
- Koda, M.; Kitamura, I.; Okura, T.; Otsuka, R.; Ando, F.; Shimokata, H. The associations between smoking habits and serum triglyceride or hemoglobin A1c levels differ according to visceral fat accumulation. J. Epidemiol. 2016, 26, JE20150064. [Google Scholar] [CrossRef] [Green Version]
- Endocrinology. 2022. Available online: https://rb.gy/eptoy1 (accessed on 13 June 2022).
- Anjum, A.; Jaggi, S.; Varghese, E.; Lall, S.; Bhowmik, A.; Rai, A. Identification of differentially expressed genes in rna-seq data of arabidopsis thaliana: A compound distribution approach. J. Comput. Biol. 2016, 23, 239–247. [Google Scholar] [CrossRef] [Green Version]
- Mahi, N.A.; Najafabadi, M.F.; Pilarczyk, M.; Kouril, M.; Medvedovic, M. GREIN: An interactive web platform for re-analyzing GEO RNA-seq data. Sci. Rep. 2019, 9, 7580. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kovács, S.A.; Gyorffy, B. Transcriptomic datasets of cancer patients treated with immune-checkpoint inhibitors: A systematic review. J. Transl. Med. 2022, 20, 249. [Google Scholar] [CrossRef] [PubMed]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vandereyken, K.; Van Leene, J.; De Coninck, B.; Cammue, B. Hub protein controversy: Taking a closer look at plant stress response hubs. Front. Plant Sci. 2018, 9, 694. [Google Scholar] [CrossRef] [Green Version]
- Ernst, J.; Beg, Q.K.; Kay, K.A.; Balázsi, G.; Oltvai, Z.N.; Bar-Joseph, Z. A semi-supervised method for predicting transcription factor–gene interactions in Escherichia coli. PLoS Comput. Biol. 2008, 4, e1000044. [Google Scholar] [CrossRef] [Green Version]
- Hausser, J.; Zavolan, M. Identification and consequences of miRNA–target interactions—Beyond repression of gene expression. Nat. Rev. Genet. 2014, 15, 599–612. [Google Scholar] [CrossRef]
- Miyagi, M.; Tanaka, K.; Watanabe, S.; Kondo, J.; Kishimoto, T. Identifying Protein–Drug Interactions in Cell Lysates Using Histidine Hydrogen Deuterium Exchange. Anal. Chem. 2021, 93, 14985–14995. [Google Scholar] [CrossRef]
- Jia, P.; Wang, L.; Meltzer, H.Y.; Zhao, Z. Common variants conferring risk of schizophrenia: A pathway analysis of GWAS data. Schizophr. Res. 2010, 122, 38–42. [Google Scholar] [CrossRef] [Green Version]
- Barrett, T.; Wilhite, S.E.; Ledoux, P.; Evangelista, C.; Kim, I.F.; Tomashevsky, M.; Marshall, K.A.; Phillippy, K.H.; Sherman, P.M.; Holko, M.; et al. NCBI GEO: Archive for functional genomics data sets—Update. Nucleic Acids Res. 2012, 41, D991–D995. [Google Scholar] [CrossRef] [Green Version]
- Islam, M.K.; Rahman, M.H.; Islam, M.R.; Islam, M.Z.; Mamun, M.M.I.; Azad, A.; Moni, M.A. Network based systems biology approach to identify diseasome and comorbidity associations of Systemic Sclerosis with cancers. Heliyon 2022, 8, e08892. [Google Scholar] [CrossRef]
- Spallotta, F.; Cencioni, C.; Atlante, S.; Garella, D.; Cocco, M.; Mori, M.; Mastrocola, R.; Kuenne, C.; Guenther, S.; Nanni, S.; et al. Stable oxidative cytosine modifications accumulate in cardiac mesenchymal cells from type2 diabetes patients: Rescue by α-ketoglutarate and TET-TDG functional reactivation. Circ. Res. 2018, 122, 31–46. [Google Scholar] [CrossRef] [PubMed]
- Ryan, D.M.; Vincent, T.L.; Salit, J.; Walters, M.S.; Agosto-Perez, F.; Shaykhiev, R.; Strulovici-Barel, Y.; Downey, R.J.; Buro-Auriemma, L.J.; Staudt, M.R.; et al. Smoking dysregulates the human airway basal cell transcriptome at COPD risk locus 19q13. 2. PLoS ONE 2014, 9, e88051. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rahman, M.H.; Peng, S.; Hu, X.; Chen, C.; Rahman, M.R.; Uddin, S.; Quinn, J.M.; Moni, M.A. A network-based bioinformatics approach to identify molecular biomarkers for type 2 diabetes that are linked to the progression of neurological diseases. Int. J. Environ. Res. Public Health 2020, 17, 1035. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuleshov, M.V.; Jones, M.R.; Rouillard, A.D.; Fernandez, N.F.; Duan, Q.; Wang, Z.; Koplev, S.; Jenkins, S.L.; Jagodnik, K.M.; Lachmann, A.; et al. Enrichr: A comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 2016, 44, W90–W97. [Google Scholar] [CrossRef] [Green Version]
- Gene Ontology Consortium. Creating the gene ontology resource: Design and implementation. Genome Res. 2001, 11, 1425–1433. [Google Scholar] [CrossRef] [Green Version]
- García-Campos, M.A.; Espinal-Enríquez, J.; Hernández-Lemus, E. Pathway analysis: State of the art. Front. Physiol. 2015, 6, 383. [Google Scholar] [CrossRef] [Green Version]
- Ramos, E.M.; Hoffman, D.; Junkins, H.A.; Maglott, D.; Phan, L.; Sherry, S.T.; Feolo, M.; Hindorff, L.A. Phenotype–Genotype Integrator (PheGenI): Synthesizing genome-wide association study (GWAS) data with existing genomic resources. Eur. J. Hum. Genet. 2014, 22, 144–147. [Google Scholar] [CrossRef]
- Buniello, A.; MacArthur, J.A.L.; Cerezo, M.; Harris, L.W.; Hayhurst, J.; Malangone, C.; McMahon, A.; Morales, J.; Mountjoy, E.; Sollis, E.; et al. The NHGRI-EBI GWAS Catalog of published genome-wide association studies, targeted arrays and summary statistics 2019. Nucleic Acids Res. 2019, 47, D1005–D1012. [Google Scholar] [CrossRef] [Green Version]
- Szklarczyk, D.; Morris, J.H.; Cook, H.; Kuhn, M.; Wyder, S.; Simonovic, M.; Santos, A.; Doncheva, N.T.; Roth, A.; Bork, P.; et al. The STRING database in 2017: Quality-controlled protein–protein association networks, made broadly accessible. Nucleic Acids Res. 2016, 45, gkw937. [Google Scholar] [CrossRef]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef]
- Chen, S.H.; Chin, C.H.; Wu, H.H.; Ho, C.W.; Ko, M.T.; Lin, C.Y. cyto-Hubba: A Cytoscape plug-in for hub object analysis in network biology. In Proceedings of the 20th International Conference on Genome Informatics, Yokohama, Japan, 14–16 December 2009. [Google Scholar]
- Xia, J.; Gill, E.E.; Hancock, R.E. NetworkAnalyst for statistical, visual and network-based meta-analysis of gene expression data. Nat. Protoc. 2015, 10, 823–844. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; Alexander, R.; Min, R.; Leng, J.; Yip, K.Y.; Rozowsky, J.; Yan, K.K.; Dong, X.; Djebali, S.; Ruan, Y.; et al. Understanding transcriptional regulation by integrative analysis of transcription factor binding data. Genome Res. 2012, 22, 1658–1667. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jonker, N.; Kool, J.; Irth, H.; Niessen, W. Recent developments in protein–ligand affinity mass spectrometry. Anal. Bioanal. Chem. 2011, 399, 2669–2681. [Google Scholar] [CrossRef] [Green Version]
- de Azevedo, J.; Walter, F.; Caceres, R.A.; Pauli, I.; Timmers, L.F.S.; Barcellos, G.B.; Rocha, K.B.; Soares, M.B. Protein-drug interaction studies for development of drugs against Plasmodium falciparum. Curr. Drug Targets 2009, 10, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Lachmann, A.; Xu, H.; Krishnan, J.; Berger, S.I.; Mazloom, A.R.; Ma’ayan, A. ChEA: Transcription factor regulation inferred from integrating genome-wide ChIP-X experiments. Bioinformatics 2010, 26, 2438–2444. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Fornes, O.; Stigliani, A.; Gheorghe, M.; Castro-Mondragon, J.A.; Van Der Lee, R.; Bessy, A.; Cheneby, J.; Kulkarni, S.R.; Tan, G.; et al. JASPAR 2018: Update of the open-access database of transcription factor binding profiles and its web framework. Nucleic Acids Res. 2018, 46, D260–D266. [Google Scholar] [CrossRef] [Green Version]
- Sethupathy, P.; Corda, B.; Hatzigeorgiou, A.G. TarBase: A comprehensive database of experimentally supported animal microRNA targets. Rna 2006, 12, 192–197. [Google Scholar] [CrossRef] [Green Version]
- Hsu, S.D.; Lin, F.M.; Wu, W.Y.; Liang, C.; Huang, W.C.; Chan, W.L.; Tsai, W.T.; Chen, G.Z.; Lee, C.J.; Chiu, C.M.; et al. miRTarBase: A database curates experimentally validated microRNA–target interactions. Nucleic Acids Res. 2011, 39, D163–D169. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Hu, G.; Wang, K.; Brylinski, M.; Xie, L.; Kurgan, L. PDID: Database of molecular-level putative protein–drug interactions in the structural human proteome. Bioinformatics 2016, 32, 579–586. [Google Scholar] [CrossRef] [Green Version]
- Miao, T.W.; Xiao, W.; Du, L.y.; Mao, B.; Huang, W.; Chen, X.m.; Li, C.; Wang, Y.; Fu, J.j. High expression of SPP1 in patients with chronic obstructive pulmonary disease (COPD) is correlated with increased risk of lung cancer. FEBS Open Bio 2021, 11, 1237–1249. [Google Scholar] [CrossRef]
- Stoynev, N.; Dimova, I.; Rukova, B.; Hadjidekova, S.; Nikolova, D.; Toncheva, D.; Tankova, T. Gene expression in peripheral blood of patients with hypertension and patients with type 2 diabetes. J. Cardiovasc. Med. 2014, 15, 702–709. [Google Scholar] [CrossRef] [PubMed]
- Tichelaar, J.W.; Borchers, M.T.; Wesselkamper, S.C.; Curull, V.; Ramirez-Sarmiento, A.; Sanchez-Font, A.; Garcia-Aymerich, J.; Coronell, C.; Lloreta, J.; Agusti, A.G.; et al. Sustained CTL activation by murine pulmonary epithelial cells promotes the development of COPD-like disease. J. Clin. Investig. 2009, 119, 636–649. [Google Scholar]
- Prince, C.; Hammerton, G.; Taylor, A.E.; Anderson, E.L.; Timpson, N.J.; Davey Smith, G.; Munafò, M.R.; Relton, C.L.; Richmond, R.C. Investigating the impact of cigarette smoking behaviours on DNA methylation patterns in adolescence. Hum. Mol. Genet. 2019, 28, 155–165. [Google Scholar] [CrossRef] [Green Version]
- Rai, A.; Pawar, A.K.; Jalan, S. Prognostic interaction patterns in diabetes mellitus II: A random-matrix-theory relation. Phys. Rev. E 2015, 92, 022806. [Google Scholar] [CrossRef]
- Li, Y.; Yuan, X.; Shi, Z.; Wang, H.; Ren, D.; Zhang, Y.; Fan, Y.; Liu, Y.; Cui, Z. LncRNA XIST serves as a diagnostic biomarker in gestational diabetes mellitus and its regulatory effect on trophoblast cell via miR-497-5p/FOXO1 axis. Cardiovasc. Diagn. Ther. 2021, 11, 716. [Google Scholar] [CrossRef]
- Kaneko, K.; Katagiri, H. Dual-specificity phosphatase 8: A gatekeeper in hypothalamic control of glucose metabolism in males. J. Diabetes Investig. 2021, 12, 1138. [Google Scholar] [CrossRef] [PubMed]
- Abo-Elfetoh, N.M.; Mohamed, E.S.; Tag, L.M.; Gamal, R.M.; Gandour, A.M.; Razek, A.E.; Mohamed, R.; El-Baz, M.A.; Ez Eldeen, M.E. The relationship between auditory brainstem response, nerve conduction studies, and metabolic risk factors in type II diabetes mellitus. Egypt. Rheumatol. Rehabil. 2016, 43, 163–171. [Google Scholar] [CrossRef]
- Liu, M.; Xu, K.; Saaoud, F.; Shao, Y.; Zhang, R.; Lu, Y.; Sun, Y.; Drummer, C.; Li, L.; Wu, S.; et al. 29 m6A-RNA Methylation (Epitranscriptomic) Regulators Are Regulated in 41 Diseases including Atherosclerosis and Tumors Potentially via ROS Regulation–102 Transcriptomic Dataset Analyses. J. Immunol. Res. 2022, 2022, 1433323. [Google Scholar] [CrossRef]
- Laqqan, M.M.; Yassin, M.M. Influence of tobacco cigarette heavy smoking on DNA methylation patterns and transcription levels of MAPK8IP3, GAA, ANXA2, PRRC2A, and PDE11A genes in human spermatozoa. Middle East Fertil. Soc. J. 2021, 26, 41. [Google Scholar] [CrossRef]
- Ma, L.; Hanson, R.L.; Que, L.N.; Cali, A.M.; Fu, M.; Mack, J.L.; Infante, A.M.; Kobes, S.; The International Type 2 Diabetes 1q Consortium; Bogardus, C.; et al. Variants in ARHGEF11, a candidate gene for the linkage to type 2 diabetes on chromosome 1q, are nominally associated with insulin resistance and type 2 diabetes in Pima Indians. Diabetes 2007, 56, 1454–1459. [Google Scholar] [CrossRef]
- Wang, J.; Kataoka, H.; Suzuki, M.; Sato, N.; Nakamura, R.; Tao, H.; Maruyama, K.; Isogaki, J.; Kanaoka, S.; Ihara, M.; et al. Downregulation of EphA7 by hypermethylation in colorectal cancer. Oncogene 2005, 24, 5637–5647. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stavarachi, M.; Panduru, N.; Serafinceanu, C.; Moţa, E.; Moţa, M.; Cimponeriu, D.; Ion, D.A. Investigation of P213S SELL gene polymorphism in type 2 diabetes mellitus and related end stage renal disease. A case-control study. Rom J. Morphol. Embryol. 2011, 52, 995–998. [Google Scholar] [PubMed]
- Zhu, M.; Wu, M.; Bian, S.; Song, Q.; Xiao, M.; Huang, H.; You, L.; Zhang, J.; Zhang, J.; Cheng, C.; et al. DNA primase subunit 1 deteriorated progression of hepatocellular carcinoma by activating AKT/mTOR signaling and UBE2C-mediated P53 ubiquitination. Cell Biosci. 2021, 11, 42. [Google Scholar] [CrossRef]
- Lee, W.H.; Chen, L.C.; Lee, C.J.; Huang, C.C.; Ho, Y.S.; Yang, P.S.; Ho, C.T.; Chang, H.L.; Lin, I.H.; Chang, H.W.; et al. DNA primase polypeptide 1 (PRIM1) involves in estrogen-induced breast cancer formation through activation of the G2/M cell cycle checkpoint. Int. J. Cancer 2019, 144, 615–630. [Google Scholar] [CrossRef]
- Hammouz, R.Y.; Kostanek, J.K.; Dudzisz, A.; Witas, P.; Orzechowska, M.; Bednarek, A.K. Differential expression of lung adenocarcinoma transcriptome with signature of tobacco exposure. J. Appl. Genet. 2020, 61, 421–437. [Google Scholar] [CrossRef]
- Ambatipudi, S.; Cuenin, C.; Hernandez-Vargas, H.; Ghantous, A.; Le Calvez-Kelm, F.; Kaaks, R.; Barrdahl, M.; Boeing, H.; Aleksandrova, K.; Trichopoulou, A.; et al. Tobacco smoking-associated genome-wide DNA methylation changes in the EPIC study. Epigenomics 2016, 8, 599–618. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, C.; Xu, G.; Tsai, S.Y.A.; Freed, W.J.; Lee, C.T. Transcriptional profiles of type 2 diabetes in human skeletal muscle reveal insulin resistance, metabolic defects, apoptosis, and molecular signatures of immune activation in response to infections. Biochem. Biophys. Res. Commun. 2017, 482, 282–288. [Google Scholar] [CrossRef]
- Lin, F.; Yang, D.; Huang, Y.; Zhao, Y.; Ye, J.; Xiao, M. The potential of neoagaro-oligosaccharides as a treatment of type II diabetes in mice. Mar. Drugs 2019, 17, 541. [Google Scholar] [CrossRef] [Green Version]
- Sandhu, H.; Xu, C.B.; Edvinsson, L. Upregulation of contractile endothelin type B receptors by lipid-soluble cigarette smoking particles in rat cerebral arteries via activation of MAPK. Toxicol. Appl. Pharmacol. 2010, 249, 25–32. [Google Scholar] [CrossRef]
- Thomas, M.C.; Mathew, T.; Russ, G.; Rao, M.M.; Moran, J. Early peri-operative glycaemic control and allograft rejection in patients with diabetes mellitus: A pilot study. Transplantation 2001, 72, 1321–1324. [Google Scholar] [CrossRef]
- Ohiomoba, R.; Youmans, Q.R.; Ezema, A.; Anderson, A.S.; Jackson, K.; Mandieka, E.; Pham, D.T.; Rich, J.D.; Yancy, C.W.; Okwuosa, I.S. History of Cigarette Smoking and Heart Transplant Outcomes. J. Card. Fail. 2019, 25, S182. [Google Scholar] [CrossRef] [Green Version]
- Khanna, A.K.; Xu, J.; Uber, P.A.; Burke, A.P.; Baquet, C.; Mehra, M.R. Tobacco smoke exposure in either the donor or recipient before transplantation accelerates cardiac allograft rejection, vascular inflammation, and graft loss. Circulation 2009, 120, 1814–1821. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, X.; Liu, G.; Guo, J.; Su, Z. The PI3K/AKT pathway in obesity and type 2 diabetes. Int. J. Biol. Sci. 2018, 14, 1483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, X.; Chen, L.; He, Z. PI3K/Akt-Nrf2 and anti-inflammation effect of macrolides in chronic obstructive pulmonary disease. Curr. Drug Metab. 2019, 20, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Wei, K.W. Identification of Shared Molecular Pathways and Networks between Alzheimer’s Disease and Type 2 Diabetes; University of California: Los Angeles, CA, USA, 2016. [Google Scholar]
- Žemeckienė, Ž.; Vitkauskienė, A.; Sjakste, T.; Šitkauskienė, B.; Sakalauskas, R. Proteasomes and proteasomal gene polymorphism in association with inflammation and various diseases. Medicina 2013, 49, 33. [Google Scholar] [CrossRef]
- Fjukstad, K.; Athanasiu, L.; Dieset, I.; Steen, N.; Djurovic, S.; Spigset, O.; Andreassen, O. Metabolic Abnormalities Related to Treatment with Selective Serotonin Reuptake Inhibitors in Patients with Schizophrenia or Bipolar Disorder: A Genome Wide Association Study. Eur. Neuropsychopharmacol. 2019, 29, S998–S999. [Google Scholar] [CrossRef]
- Zhang, C.; Shen, L.; Zhu, Y.; Xu, R.; Deng, Z.; Liu, X.; Ding, Y.; Wang, C.; Shi, Y.; Bei, L.; et al. KDM6A promotes imatinib resistance through YY1-mediated transcriptional upregulation of TRKA independently of its demethylase activity in chronic myelogenous leukemia. Theranostics 2021, 11, 2691. [Google Scholar] [CrossRef]
- Dingli, D.; Wolf, R.C.; Vella, A. Imatinib and type 2 diabetes. Endocr. Pract. 2007, 13, 126–130. [Google Scholar] [CrossRef]
- Imarisio, I.; Paglino, C.; Ganini, C.; Magnani, L.; Caccialanza, R.; Porta, C. The effect of sorafenib treatment on the diabetic status of patients with renal cell or hepatocellular carcinoma. Future Oncol. 2012, 8, 1051–1057. [Google Scholar] [CrossRef]
- Guo, T.; Agaram, N.P.; Wong, G.C.; Hom, G.; D’Adamo, D.; Maki, R.G.; Schwartz, G.K.; Veach, D.; Clarkson, B.D.; Singer, S.; et al. Sorafenib inhibits the imatinib-resistant KIT T670I gatekeeper mutation in gastrointestinal stromal tumor. Clin. Cancer Res. 2007, 13, 4874–4881. [Google Scholar] [CrossRef] [Green Version]
- Tyrrell, H.; Pwint, T. Sunitinib and improved diabetes control. BMJ Case Rep. 2014, 2014, bcr2014207521. [Google Scholar] [CrossRef] [Green Version]
- Huda, M.S.; Amiel, S.A.; Ross, P.; Aylwin, S.J. Tyrosine kinase inhibitor sunitinib allows insulin independence in long-standing type 1 diabetes. Diabetes Care 2014, 37, e87–e88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Minor, D.R.; Kashani-Sabet, M.; Garrido, M.; O’Day, S.J.; Hamid, O.; Bastian, B.C. Sunitinib Therapy for Melanoma Patients with KIT MutationsSunitinib Therapy for Melanoma with KIT Mutations. Clin. Cancer Res. 2012, 18, 1457–1463. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tran, A.; Tawbi, H.A. A potential role for nilotinib in KIT-mutated melanoma. Expert Opin. Investig. Drugs 2012, 21, 861–869. [Google Scholar] [CrossRef] [PubMed]
- Ono, K.; Suzushima, H.; Watanabe, Y.; Kikukawa, Y.; Shimomura, T.; Furukawa, N.; Kawaguchi, T.; Araki, E. Rapid amelioration of hyperglycemia facilitated by dasatinib in a chronic myeloid leukemia patient with type 2 diabetes mellitus. Intern. Med. 2012, 51, 2763–2766. [Google Scholar] [CrossRef] [Green Version]


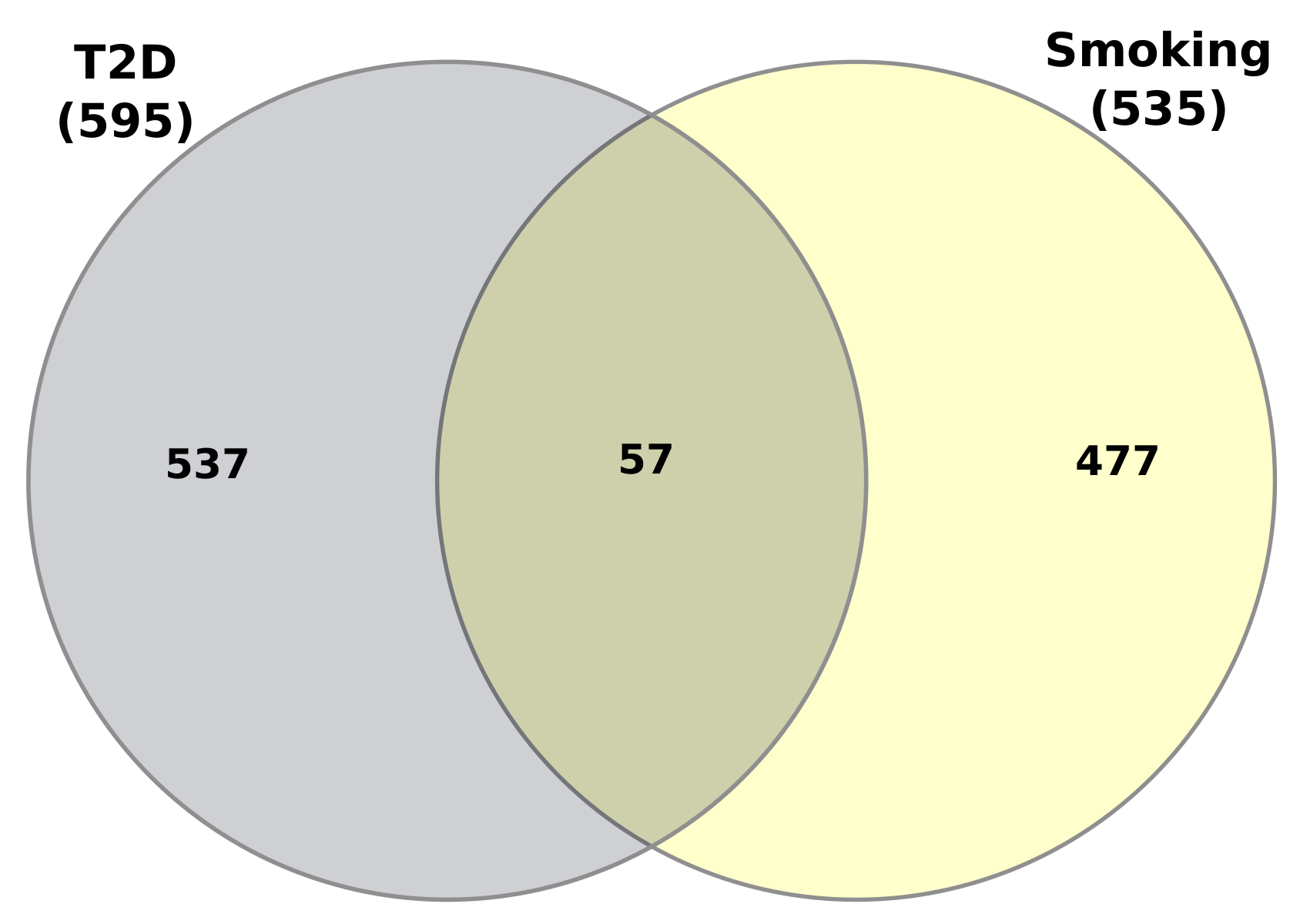
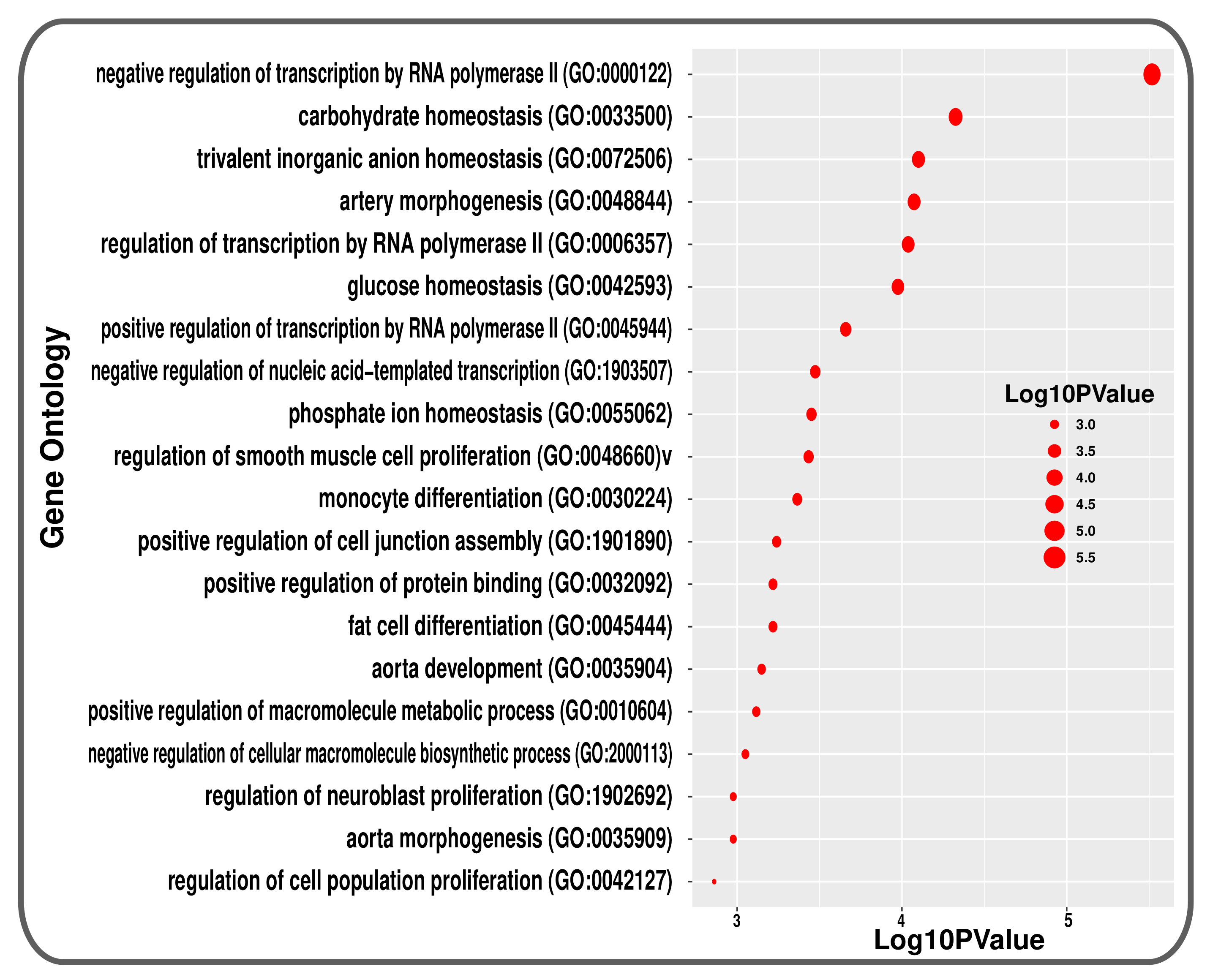
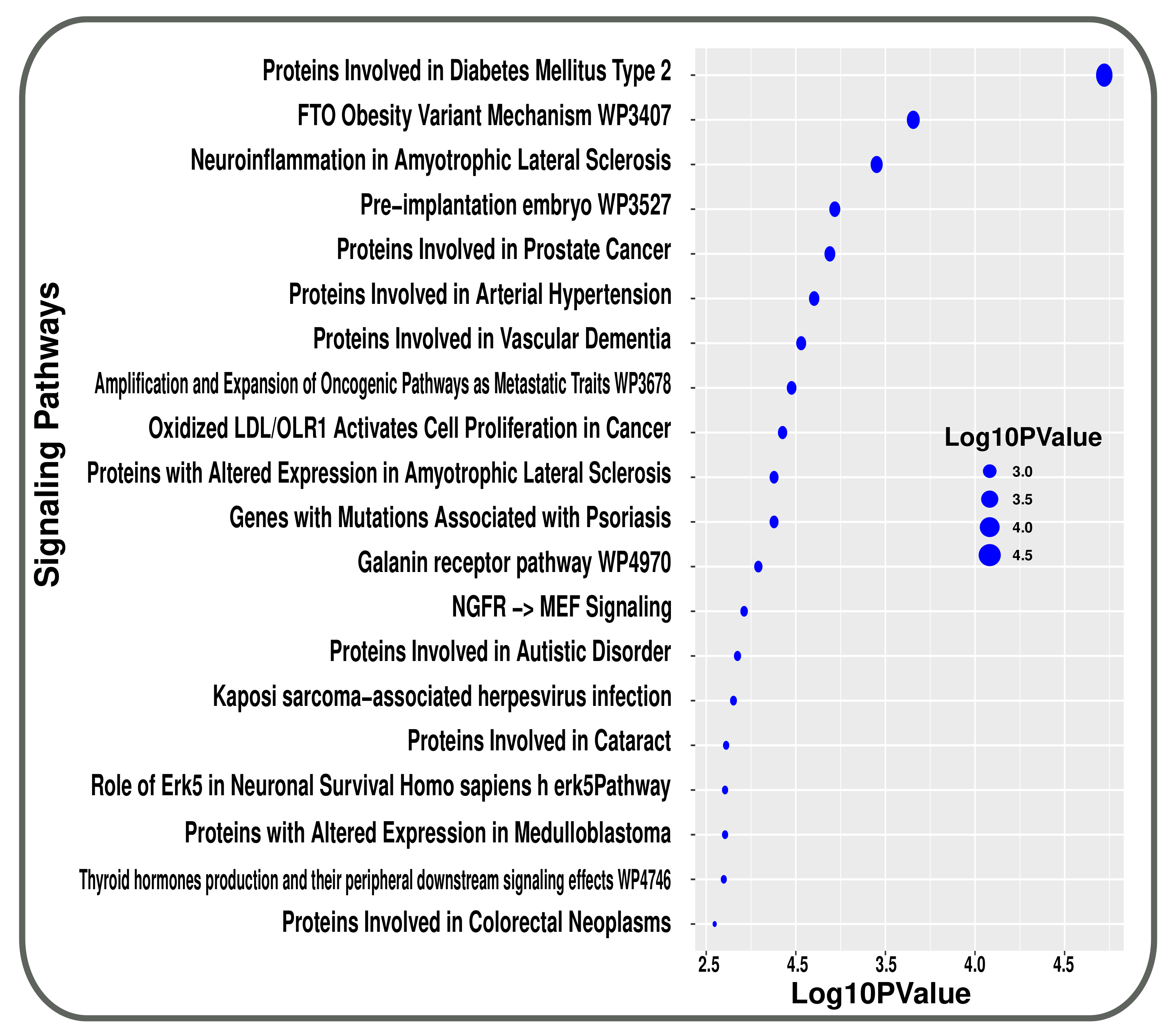
| Disease Name | GEO Platform | Tissues/Cells | GEO Accession | RAW Genes | Case Samples | Control Samples | Significant | Up Reg. Genes | Down Reg. Genes |
|---|---|---|---|---|---|---|---|---|---|
| Type-2 Diabetes (T2D) | Illumina NextSeq 500 (Homo sapiens) | Human cardiac mesenchymal cells | GSE-106177 | 18,619 | 7 | 7 | 1367 | 768 | 599 |
| Smoking | Illumina HiSeq 2000 (Homo sapiens) | Airway Basal Cells | GSE-47718 | 21,178 | 7 | 10 | 962 | 682 | 280 |
| Gene | Smoking | T2D |
|---|---|---|
| SPP1 | [56] | [57] |
| MICA | [58] | - |
| PSMB8 | [59] | [60] |
| XIST | - | [61] |
| DUSP8 | - | [62] |
| ABR | - | [63] |
| PRRC2A | [65] | [64] |
| NTRK1 | - | [66] |
| EPHA7 | [67] | - |
| SELL | - | [68] |
| PRIM1 | [70] | [69] |
| KIFC1 | [71] | - |
| ZNF385D | [72] | - |
| HLA-DPB1 | - | [73] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ripon Rouf, A.S.M.; Amin, M.A.; Islam, M.K.; Haque, F.; Ahmed, K.R.; Rahman, M.A.; Islam, M.Z.; Kim, B. Statistical Bioinformatics to Uncover the Underlying Biological Mechanisms That Linked Smoking with Type 2 Diabetes Patients Using Transcritpomic and GWAS Analysis. Molecules 2022, 27, 4390. https://doi.org/10.3390/molecules27144390
Ripon Rouf ASM, Amin MA, Islam MK, Haque F, Ahmed KR, Rahman MA, Islam MZ, Kim B. Statistical Bioinformatics to Uncover the Underlying Biological Mechanisms That Linked Smoking with Type 2 Diabetes Patients Using Transcritpomic and GWAS Analysis. Molecules. 2022; 27(14):4390. https://doi.org/10.3390/molecules27144390
Chicago/Turabian StyleRipon Rouf, Abu Sayeed Md., Md. Al Amin, Md. Khairul Islam, Farzana Haque, Kazi Rejvee Ahmed, Md. Ataur Rahman, Md. Zahidul Islam, and Bonglee Kim. 2022. "Statistical Bioinformatics to Uncover the Underlying Biological Mechanisms That Linked Smoking with Type 2 Diabetes Patients Using Transcritpomic and GWAS Analysis" Molecules 27, no. 14: 4390. https://doi.org/10.3390/molecules27144390








